Abstract
Background:
Inflammatory bowel disease (IBD) presents unique challenges in elderly patients due to comorbidities and treatment-related risks.
Objectives:
This study evaluates ustekinumab (UST) and vedolizumab (VDZ) efficacy and safety in elderly Crohn’s disease (CD) patients.
Design:
A retrospective cohort study at a tertiary medical center.
Methods:
CD patients aged ⩾60 years (elderly) treated with UST, compared to non-elderly (<60 years) patients treated with UST and elderly patients treated with VDZ. Clinical response was evaluated using the Harvey–Bradshaw index (HBI) and clinical biomarkers, alongside monitoring steroid use, hospitalization rates, treatment persistence, and surgical interventions.
Results:
The study included 166 CD patients: 32 elderly and 65 non-elderly patients treated with UST, and 69 elderly patients treated with VDZ. The mean duration of follow-up was 10.8 ± 2.8 months in the non-elderly group, 9.97 ± 3.28 months in the elderly UST group, and 10.0 ± 3.29 months in the VDZ group. Elderly UST patients were more likely to receive corticosteroids at initiation than non-elderly UST patients (44% vs 14%, p = 0.001). At 12 months, clinical response rates did not significantly differ between elderly and non-elderly UST groups, respectively (48% vs 40%, p = 0.5). However, elderly UST patients exhibited higher hospitalization rates over time compared to non-elderly UST patients (6-month: 19% vs 6.2%, p = 0.077; 12-month: 19% vs 4.6%, p = 0.055; log-rank p = 0.004). No significant differences were observed in clinical response and remission rates between elderly UST and elderly VDZ patients at 6 and 12 months. At 6 months, a higher hospitalization rate was observed in the UST group (19% vs 4.3% p = 0.027), but this difference did not persist over time.
Conclusion:
UST and VDZ are effective and safe treatments for elderly CD patients, despite higher hospitalization rates compared to non-elderly patients, likely due to age-related complications.
Plain language summary
In older adults with Crohn’s disease, managing treatment can be complex due to age-related health issues. This study examined how two medications, ustekinumab (UST) and vedolizumab (VDZ), perform in patients aged 60 and older. Researchers compared UST’s effectiveness in elderly patients to its use in younger adults and VDZ’s performance in the elderly. They found that while elderly patients starting UST were more likely to need corticosteroids initially, their 12-month clinical response rates were similar to younger patients. However, elderly UST users had higher hospitalization rates over time. Results also showed no significant difference in outcomes between elderly patients using UST versus VDZ, despite initial concerns about hospitalizations. Overall, both UST and VDZ appear safe and effective for elderly Crohn’s disease patients, although hospitalization risks remain higher compared to younger adults, reflecting age-related health challenges.
Introduction
The aging population profoundly influences medicine by creating a greater demand for specialized healthcare tailored to age-related conditions and chronic diseases. The burden of inflammatory bowel disease (IBD) is rising globally, with substantial variation in levels and trends of disease in different countries and regions. 1 As the prevalence of IBD grows, there is a concurrent rise in the occurrence among the elderly population. The escalating management complexities associated with elderly patients diagnosed with IBD underscore the critical need to acknowledge the substantial influence of comorbidities, polypharmacy, and the criteria determining their surgical eligibility. Additionally, it is crucial to have an understanding of the distinct disease characteristics among age groups, such as the more aggressive disease phenotype typically found in younger individuals and a greater prevalence of immunosuppressive therapy utilization. 2 Older individuals diagnosed with IBD appear to experience higher rates of malignancy.3,4 Frequently, older patients are excluded from clinical trials, leading treatment decisions to rely heavily on concerns regarding adverse effects and clinical experience. Anti-tumor necrosis factor (TNF) agents are commonly utilized in patients with moderate-severe IBD yet their implementation in senior care is not of consensus. 5 Research on anti-TNF agents in older individuals has produced inconsistent findings regarding both effectiveness and safety concerns.6 –8 These studies showed contradictory outcomes regarding whether the increased rate of infections or any significant adverse events observed in the elderly population could be attributed to therapy or to the age characteristics of the population. As treatment with anti-TNF agents raises some safety concerns in the elderly, alternative biologics may be preferred.
Studies on vedolizumab (VDZ), including the GEMINI trials, found no significant differences in adverse reactions leading to hospitalization between elderly and non-elderly patients with IBD.9 –12 Moreover, efficacy levels were comparable in patients with IBD after 12 and 24 months of treatment.13,14 The evaluation of ustekinumab’s (UST) efficacy and safety in CD has progressively accumulated through comprehensive clinical trials and real-world clinical practice through numerous retrospective and prospective series.15 –21 Regarding elderly individuals with IBD undergoing treatment with UST, prior data indicate that the clinical outcomes and safety profiles generally exhibit similarities between elderly and non-elderly IBD patients. However, it is noteworthy that the evidence remains somewhat constrained, with certain findings suggesting a higher incidence of complicated disease phenotypes among elderly patients. 22 Additionally, elevated rates of de novo neoplasm development and hospitalization within the elderly cohort have been observed, necessitating further investigation into these trends.22 –24
There is a need for age-specific data to optimize treatment strategies for elderly IBD patients. Yet, the quantity of studies focusing on IBD in the elderly remains limited, and there is a growing need for real-data comparisons and filling the knowledge gap on improving clinical outcomes in elderly IBD patients.
Hence, this study aimed to assess and compare the safety and efficacy of UST in both elderly and non-elderly patients with Crohn’s disease (CD) and to compare these between UST and VDZ-treated elderly patients.
Materials and methods
Data collection and analysis
For this study, we delineated a cohort of adult CD patients (aged over 18 years) who were treated with UST or VDZ and completed at least 14 weeks of therapy between 2017 and 2023. We excluded patients who lacked follow-up visits in our gastroenterology department, as their files contained insufficient data for utilization (at least one follow-up visit after treatment initiation was required).
This was a retrospective cohort study. The cohort consisted of elderly (aged 60 years and above) patients who were treated with UST compared with two separate groups: non-elderly (age <60 years) UST-treated patients and elderly patients treated with VDZ. The primary outcomes of the two comparisons were clinical response/remission rates and the need for hospitalization.
The age threshold of 60 was selected based on its frequent application as a demarcation point in existing literature.22,24,25 The follow-up period started concurrently with the induction of UST or VDZ based on the respective study group assignments.
Retrieved demographic CD details included year of diagnosis, age at diagnosis, Montreal classification, perianal disease, EIM (extra-intestinal manifestation), and past abdominal surgery. Patient characteristics also included age, gender, past or present smoking, and comorbidities. Additionally, data were collected on patients who had previously undergone any form of biologic therapy before the initiation of UST, including any anti-TNF agents, VDZ. We tracked clinical parameters upon treatment commencement, as well as at intervals of 3, 6, and 12 months thereafter. Clinical variables encompassed C-reactive protein levels, with levels below 5 mg/l considered normal in our medical center, Harvey–Bradshaw Index (HBI) score, previous biological and immunomodulatory treatments, corticosteroid usage during the study’s follow-up periods, discontinuation of treatment (including cessation of medication and reasons), and requirement for hospitalization or surgery. Furthermore, we monitored any notable adverse events reported by the patient and severe infections (infections necessitating hospitalization). The HBI score was either taken directly from the clinical records (where it was completed by the physician during the visit) or calculated based on clinical parameters reviewed during the visit. A clinical response was described as either an enhancement of over 3 points in the HBI score or achieving an HBI score below 5, which defines clinical remission. Clinical remission was classified as having an HBI score below 5.
Statistical analysis
In this retrospective cohort study, we assessed the determinants of 6- and 12-month remission in elderly and non-elderly CD patients initiating UST treatment. Data extraction and cleaning were followed by preprocessing tailored for logistic regression analyses. Baseline characteristics of the participants were presented using descriptive statistics, with continuous variables described by medians and interquartile ranges (IQR), and categorical variables by frequencies and percentages.
Comparative analyses utilized the Mann–Whitney U test for continuous variables with non-normal distributions and the chi-square test for categorical variables.
Time-to-event outcomes, such as hospitalization rates, were analyzed using Kaplan–Meier curves, with differences evaluated using the log-rank test.
Statistical significance was determined at a two-sided alpha level of 0.05. All analyses, from data processing to exploratory and inferential statistics, were conducted in R software (version 4.2.0, R Core Team, 2022) and RStudio (RStudio Team, 2020).
This article was prepared in accordance with the STROBE statement.26,27
Results
Study cohort characteristics
Of the 134 patients with documented CD aged ⩾60 years who received care in our department, 39 were treated with UST, and 95 received VDZ. Within this age group, 33 patients (7 in the UST and 26 in the VDZ group) were excluded from the study due to insufficient follow-up data. Additionally, among 91 CD patients aged <60 years who were treated with UST, 26 were excluded from the analysis due to inconsistent follow-up (Figure 1).

Flow chart showing number of patients screened, included, and excluded.
Consequently, the final cohort comprised 166 patients, including 32 elderly and 65 non-elderly individuals treated with UST, respectively, and 69 elderly patients treated with VDZ.
Elderly UST versus non-elderly UST CD patients
Cohort characteristics
The mean duration of follow-up was 10.8 ± 2.8 months in the non-elderly group (median of 12 months, IQR 12–12), and 9.97 ± 3.28 months in the elderly group (median of 12 months, IQR 6–12). The median age of UST initiation in the elderly group was 67 years (IQR 64–74), and in the non-elderly group 34 years (IQR 28–42). Regarding disease phenotype, no statistically significant differences were found between the groups (Table 1). Both groups had predominantly an isolated ileal disease (68% in the elderly and 56% in non-elderly), however, ileo-colonic involvement was higher in non-elderly patients (36% vs 13%, p = 0.031). Similar HBI scores were noted at treatment initiation in both groups (5 in the elderly group vs 6 in the non-elderly group). Additionally, elderly individuals received corticosteroids more often upon initiation of UST (44% vs 14%, p = 0.001). No statistically significant differences were found in the rates of patients previously treated with biologic therapy compared to those who were biologic-naïve between the study groups (100% vs 97%, p = 0.3). Among elderly patients treated with UST, 84% had prior exposure to anti-TNF agents: 59% to infliximab, 62.5% to adalimumab, and 37.5% to both. Of those treated with infliximab, 47% discontinued due to loss of response or nonresponse, often linked to immunogenicity, while the remainder stopped due to intolerance or adverse events. For adalimumab, 90% discontinued due to ineffectiveness, with others ceasing treatment for similar reasons.
Baseline demographics, disease characteristics, and treatment history of elderly versus non-elderly patients treated with ustekinumab (primary analysis).

p-Value < 0.05.
Previous biologic treatment refer to any biologic treatment previous to the initiation of ustekinumab (any anti-TNF agents, vedolizumab); disease duration- from disease onset to treatment initiation.
HBI, Harvey–Bradshaw index; TNF, anti-tumor necrosis factor.
In the non-elderly UST group, 97% had previously been treated with anti-TNF agents (63% infliximab, 80% adalimumab, and 46% both). Among infliximab users, 80% discontinued treatment due to loss of response or nonresponse, while others stopped for intolerance or adverse events. In the adalimumab group, 71% stopped treatment due to loss of response or nonresponse, while others discontinued for intolerance or adverse effects. The most common adverse event associated with anti-TNF treatment was rash or hypersensitivity.
Additionally, previous VDZ therapy was more prevalent in the elderly group, with over 90% of patients in both age groups discontinuing VDZ due to lack of response.
The overall occurrence of EIMs was comparable between both groups (43% vs 47%). A statistically significant distinction was noted in the rates of prior abdominal surgeries, with the elderly cohort exhibiting a higher frequency of abdominal surgical interventions prior to the initiation of treatment (26% vs 47% p = 0.041). Additionally, as expected, a significant difference was found in the mean Charlson comorbidity index, which was higher in the elderly group (0.25 ± 0.09 vs 2.03 ± 0.43, p < 0.001).
Effectiveness
After 6 months, clinical response rates were 29% in the elderly group compared to 39% in the non-elderly group (p = 0.4), and clinical remission rates were 13% in the elderly group compared to 16% in the non-elderly group (p > 0.9, Figure 2(a) and (b)). After 12 months of treatment, clinical remission rates between the elderly group and the non-elderly group were 22% vs 24%, respectively (p = 0.8), with no significant disparities observed. Similarly, no significant differences were seen between the groups in clinical response rates (48% vs 40%, p = 0.5). Moreover, there was no significant difference regarding the use of steroids at the 3, 6, and 12 months follow-up periods (22% vs 11%, p = 0.2; 19% vs 11%, p = 0.3; and 13% vs 4.6%, p = 0.2, respectively).

(a) Clinical outcomes comparing elderly and non-elderly patients treated with UST after 6 months of treatment. (b) Clinical outcomes comparing elderly and non-elderly patients treated with UST after 12 months of treatment.
Safety
At 6 and 12 months of treatment, the groups showed no difference regarding the need for surgery, or treatment cessation (Table 2). Throughout the entire study follow-up, the rates of treatment cessation remained consistent across both the elderly and non-elderly groups, with 31% of individuals discontinuing treatment in each cohort. The primary reason for treatment cessation in both groups was the inability to achieve a satisfactory clinical response, accounting for 90% of discontinuations in the elderly group and 80% in the non-elderly group. There was one case of discontinuation due to worsening preexisting enthesitis, which necessitated exploring alternative therapies. Additionally, one patient in the non-elderly group discontinued treatment after being diagnosed with aseptic meningitis within a 3-month period, as the differential diagnosis considered this condition an adverse event related to UST. In terms of hospitalizations, a trend toward higher rates was observed among the elderly group at the 6-month time point (6.2% vs 19%, p = 0.077) and at the 12-month follow-up (4.6% vs 19%, p = 0.055). On Kaplan–Meier analysis, the higher hospitalization rate among elderly patients was statistically significant throughout the study’s follow-up period (log-rank p = 0.004; Figure 3).
Safety profile of ustekinumab among elderly and non-elderly CD patients during the study’s follow-up.


Kaplan–Meier plot illustrating the incidence of hospitalization throughout the study’s follow-up period, comparing the elderly and non-elderly groups receiving UST treatment. A significant difference in hospitalization rates between the groups was observed (p = 0.004).
Notably, no infectious-related hospitalizations were reported in the non-elderly group during either the 6-month or 12-month periods. In contrast, 33% (2/6) of hospitalized elderly patients within the 6-month period and 50% (3/6) within the 12-month period were due to infectious causes (specifically, 2 cases of pneumonia, 2 cases of cellulitis, and 1 case of esophageal candidiasis). Importantly, these infections were not attributed to either the treatment or the disease and therefore did not necessitate treatment cessation. The remaining hospitalizations in the elderly group were related to CD. In the non-elderly group, hospitalizations were primarily due to disease exacerbation, with one additional case resulting from severe iron deficiency anemia. There was one non-CD-related mortality case in the elderly group, attributed to gross hematuria with urologic complications.
Elderly UST versus elderly VDZ CD patients
Cohort characteristics
This analysis comprised the 32 elderly patients receiving UST and an additional 69 elderly CD patients treated with VDZ. The mean duration of follow-up was 10 ± 3.29 months in the VDZ group (median of 12 months, IQR 6–12) and 9.97 ± 3.28 months in the UST group (median of 12 months, IQR 6–12). Age at diagnosis was similar between the groups, however, a statistically significant difference in the median age at treatment initiation was evident between elderly patients receiving UST (67, IQR 64–74) and those receiving VDZ (64, IQR 60–72), p = 0.033. Regarding disease phenotype, no statistically significant difference was found between the groups (Table 3). Comparisons between the groups showed no differences in baseline HBI scores, Montreal classification, or the presence of perianal disease. Additionally, there was no significant difference in the mean Charlson Comorbidity Index (2.03 ± 0.43 vs 1.79 ± 0.25, p = 0.6). In terms of prior biologic treatments, the elderly UST group had a significantly higher rate compared to the VDZ group (97% vs 60% p < 0.001). 58% of the patients had previously received anti-TNF agents. Among those treated with infliximab, 64% discontinued therapy due to loss of response or nonresponse, including issues related to immunogenicity. Similarly, 75% of patients who received adalimumab stopped treatment for the same reasons. In the VDZ group, 10% of patients had previously received UST (a total of seven patients), with six of them discontinuing therapy due to lack of effectiveness.
Baseline demographics, disease characteristics, and treatment history for elderly patients treated with ustekinumab versus vedolizumab.
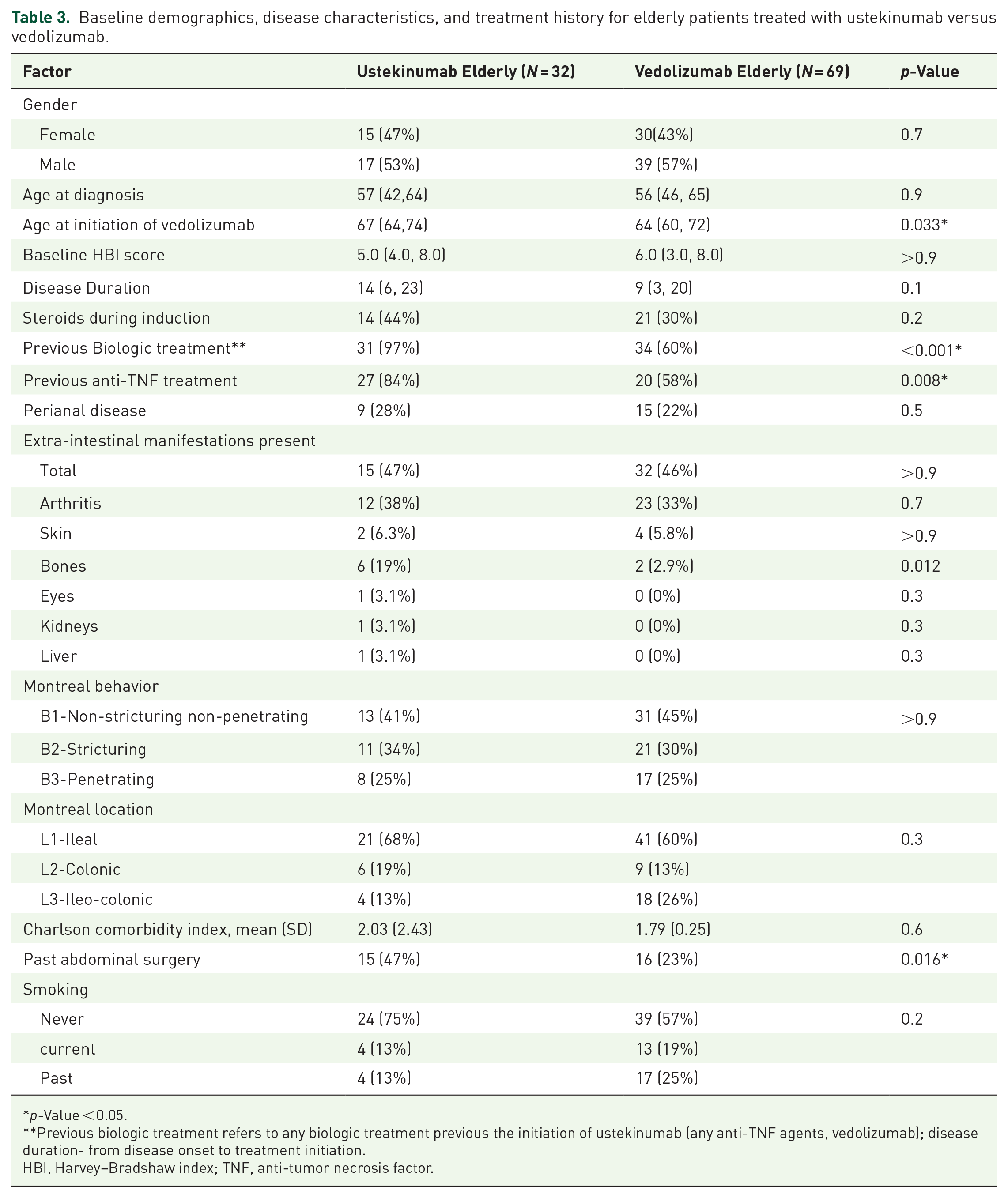
p-Value < 0.05.
Previous biologic treatment refers to any biologic treatment previous the initiation of ustekinumab (any anti-TNF agents, vedolizumab); disease duration- from disease onset to treatment initiation.
HBI, Harvey–Bradshaw index; TNF, anti-tumor necrosis factor.
The overall occurrence of EIMs was comparable between both groups (47% vs 46%). A statistically significant distinction was noted in the rates of prior abdominal surgeries, with the UST cohort exhibiting a higher frequency of abdominal surgical interventions prior to the initiation of treatment (23% vs 47% p = 0.016).
Effectiveness
After 6 months, clinical response rates were 29% in the UST group compared to 23% in the VDZ group (p = 0.6), and clinical remission rates were 13% in the UST group compared to 6.7% in the VDZ group (p = 0.7, Figure 4(a) and (b)). After 12 months of treatment, clinical remission rates between the UST group and the VDZ group were 22% versus 20%, respectively (p = 0.6). No significant differences were seen between the groups in clinical response rates as well (48% vs 37%, p = 0.4). Moreover, there was no significant difference regarding the use of steroids at the 3, 6, and 12 months follow-up periods (22% vs 22%, p > 0.9, 19% vs 17%, p = 0.9, and 13% vs 14%, p > 0.9, respectively).

(a) Clinical outcomes comparing elderly patients treated with UST versus VDZ after 6 months of treatment. (b) Clinical outcomes comparing elderly patients treated with UST versus VDZ after 12 months of treatment.
Safety
At both the 6 and 12-month marks of treatment, no disparity was observed between the groups concerning the necessity for surgery or treatment cessation, as outlined in Table 4. The primary reason for discontinuing VDZ treatment was similar to that in the UST group, with 68% of patients ceasing treatment due to the inability to achieve a satisfactory clinical response. The second most common reason for discontinuation was the emergence or worsening of preexisting arthralgia/arthritis, necessitating the exploration of alternative treatment; it should be noted that the arthralgia was not attributed to VDZ itself. Additionally, one patient in the VDZ group discontinued treatment due to the development of a rash as a reaction to the therapy. In terms of hospitalizations, although there was a significant difference observed at the 6-month mark, with higher rates among the UST group compared to the VDZ group (19% vs 4.3%, p = 0.027), the hospitalization rate over the 12-month period did not show a statistically significant difference. Additionally, survival analysis indicated no statistically significant difference in hospitalization rates over time (log-rank p = 0.063, see Figure 5).
Safety profile of ustekinumab and vedolizumab among elderly CD patients during the study’s follow-up.

p-Value < 0.05.
CD, Crohn’s disease; UST, ustekinumab; VDZ, vedolizumab.

Kaplan–Meier plot illustrating the incidence of hospitalization throughout the study’s follow-up period, comparing the elderly patients receiving UST or VDZ treatment. There was no significant difference in hospitalization rates between the groups (p = 0.063).
In total, within the VDZ group, 9% (1/11) of hospitalizations during the 6- and 12-month periods were attributed to an infectious disease (pneumonia). In contrast, in the UST group, 41.7% (5/12) of hospitalizations during the same timeframe were due to infectious diseases (p = 0.075). One case of venous thromboembolism was identified across the entire cohort, specifically pulmonary embolism in the VDZ group. Importantly, neither the infections nor the venous thromboembolism were attributed to the disease or the treatment, so no changes were made to the management of CD.
Discussion
In this retrospective study including patients with CD across two age groups, we observed comparable clinical responses to treatment between elderly and non-elderly individuals treated with UST. Similar trends were evident when examining elderly patients treated with UST compared to elderly patients treated with VDZ. In addition, elderly patients treated with UST exhibited higher rates of hospitalizations throughout the study period than non-elderly patients undergoing the same treatment. In the analysis comparing elderly patients treated with UST and elderly patients receiving VDZ, the rates of hospitalizations were found to be higher in the former after 6 months period, however, this significance was not seen over time. No difference regarding treatment cessation or need for surgery was observed.
IBD represents a group of chronic diseases posing a significant therapeutic challenge in routine clinical practice. Over the past few years, new therapeutic tools, particularly biological treatments, have become more common, and their effectiveness has been demonstrated in both clinical trials and real-world studies. However, a consistent under-representation of elderly patients was evident. A recent systematic review demonstrated that merely 5% of IBD patients participating in phase III clinical studies were 65 years or older. Moreover, in almost 60% of these studies age was considered an exclusion criterion. 28 The disparity in the representation of elderly patients across various IBD trials, as opposed to their actual proportion in the general population, is likely to be exacerbated by the inherent complexity of multimorbidity and polypharmacy of this group.
Despite its growing relevance in daily clinical practice, there is limited data on the safety and effectiveness of UST in the elderly population. Our evidence on this matter is mainly based on small cohorts and retrospective studies. In addition, clinical trial data is limited due to the restricted inclusion of predominantly healthy elderly patients without significant comorbidities.18,23 In contrast, studies in the non-elderly population have shown that this medication does not increase the risk of serious infections and malignancies compared to anti-TNF treatments.29 –32
In our study, we found that UST is equally effective for both elderly and non-elderly patients. Similar findings were reported by Casas-Deza et al. in the ENEIDA registry, which showed comparable clinical response rates over time for young and elderly patients with CD. 22 Their reported clinical response and remission rates ranged from 60% to 67% and 51% to 58%, respectively. Gebeyehu et al. also highlighted high efficacy in elderly CD patients, reporting clinical response and remission rates of 43.5% and 49% after 6 months. 33 These efficacy rates were higher than those observed in our study. It is important to note that our cohort had a higher percentage of biologic-experienced patients, with 84%–97% having previously received anti-TNF therapy, compared to only 66% in the study by Gebeyehu and colleagues. This aligns with the established understanding that anti-TNF experience is linked to lower clinical efficacy across all age groups in CD patients.34,35 Furthermore, the ENEIDA study was a multicenter national study, while our analysis was limited to a single IBD-referral tertiary center, which may reflect a more complex IBD cohort with more challenging cases.
Despite these differences, our findings of similar clinical response rates across age groups are consistent with other real-world studies, which demonstrate that elderly CD patients receiving UST and VDZ achieve comparable clinical outcomes.24,33,36 These results reinforce the idea that both UST and VDZ are effective treatment options for CD across different age groups.
In the context of safety, our study found no significant difference in hospitalization rates between elderly and non-elderly patients; however, Kaplan–Meier analysis revealed a statistically significant difference between the age groups in terms of hospitalization throughout the study’s follow-up period (log-rank p = 0.004).
Prior data on hospitalizations for various causes among elderly CD patients treated with UST remains limited, with existing studies primarily focusing on hospitalizations related to infections. Nonetheless, when analyzing the incidence of severe infections requiring hospitalization, the available data presents a significant variability, ranging from 2.6% to 12% of hospitalizations over a one-year treatment period.22,23,25,33 Our findings are consistent with this range and as indicated in the results, hospitalizations within the non-elderly cohort were primarily linked to disease exacerbation. In addition, there was no discernible discrepancy in hospitalization rates related to disease activity observed between the two UST age groups. Holvoet and colleagues recent study illustrated that elderly patients treated with VDZ or UST faced a higher risk of hospitalization due to reasons unrelated to IBD, which aligns with our findings. 24
These findings regarding hospitalization rates among elderly patients primarily suggest that the observed differences are attributable to age and its associated comorbidities, as well as the immunosuppressive effects on this population, perhaps rendering them more susceptible to infections. To assess the role of immunosuppression in infection risk within this age group, the safety profile of targeted therapy has been previously validated through studies. In a meta-analysis from 2019, Borren and colleagues demonstrated a higher pooled prevalence of 13% infections rate in older patients treated with biologics compared to 6% in younger individuals (OR 2.28). 37 On the other hand, other studies demonstrated similar outcomes regarding hospitalization and rates of serious infections in elderly populations receiving immunosuppressive treatment compared to those who are treatment-naïve.38 –40 For example, the UNIT IM study revealed a 2.3% incidence of serious infections over a 44-week follow-up period with UST, which was comparable to the 2.3% observed in the placebo group. These results suggest that the elevated rates of serious infections are associated with age rather than treatment with immunosuppression.
Moreover, in our study, a considerable number of elderly patients received steroid therapy during UST induction. This trend may stem from a certain degree of reluctance or hesitation toward initiating biologic therapy. There was no noticeable discrepancy observed between the groups in the analyses regarding rates or causes of treatment discontinuation. In cases where arthralgia was present, preference was given to an anti-TNF agent to address both conditions simultaneously.
All in all, UST and VDZ appear to be effective and safe, offering a feasible and reliable solution for this population, especially after prior anti-TNF failure or nonresponse.
We are aware of the limitations of our study. The major limitation of this study is the retrospective nature of data collection, with potential biases including confounding co-variants. In addition, some data were missing from the clinical follow-ups, including assessment scores and biomarkers. Second, although the cohort included 166 patients, in losing patients to year-long follow-up, the sample size was limited, thereby potentially impacting the generalizability of findings and the robustness of statistical analyses. Moreover, the small sample size did not allow for multivariate analysis such as the prediction of clinical remission. It is also important to note that this is a single-center study, and the follow-up period for the cohorts was relatively short, lasting a median of 12 months. Additionally, the study did not include a non-elderly group treated with vedolizumab, as the primary focus was on evaluating treatment in elderly CD patients. Therefore, the results of the study should be interpreted with caution, and further large-scale studies are required to validate our findings.
In conclusion, our study presents UST and VDZ as effective and safe treatment options for elderly patients with CD, both serving as a considerable therapeutic option for this population.
