Abstract
Background:
The standardized diagnostic categories defined by the World Health Organization (WHO) reporting system support the interdisciplinary interpretation of cytological findings in the management of pancreatic cancer.
Objective:
To compare this classification to the Papanicolaou Society of Cytopathology (PSC) system in terms of predictive value and risk of malignancy (ROM) in solid pancreatic lesions.
Design:
Retrospective cohort study.
Methods:
All consecutive patients with solid pancreatic lesions who underwent endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) sampling at the University of Szeged from 2014 to 2021 were retrospectively enrolled. The predictive value and ROM of cytological findings were determined with comparison to histologic outcome and/or clinical follow-up.
Results:
A total of 521 EUS-FNAs were performed with a malignancy rate of 81.76%. In both classification systems, the absolute ROM of “non-diagnostic,” “negative for malignancy,” “atypical,” “suspicious for malignancy,” and “malignant” categories were 48.2%, 2.3%, 78.1%, 100.0%, and 99.4%, respectively. Despite the heterogeneous nature of the “neoplastic: other” category of the PSC system, the absolute ROM for solid lesions was 100%. Pancreatic neoplasm: high-risk/grade category including only two endosonographically solid cases of high-grade intraductal papillary mucinous neoplasms showed 100% ROM. There were no differences between PSC and WHO systems in sensitivity, specificity, and negative and positive predictive values: excluding the “atypical” category, these were 99.7%, 95.6%, 97.7%, and 99.5%, respectively. The “atypical” category considered benign resulted in a higher decrease in validity and negative predictive value, compared to “atypical” considered true malignant (93.6% vs 97.7% and 65.8% vs 97.7%).
Conclusion:
For solid pancreatic lesions, the WHO system was identical to the PSC system in terms of ROM and predictive values.
Introduction
Solid pancreatic lesions are a group of heterogeneous disease entities that can be generally classified as either neoplastic (benign, premalignant, and malignant) or non-neoplastic. 1 Most solid neoplasms are ductal adenocarcinomas and their subtypes, however, neuroendocrine tumors (NETs), solid pseudopapillary neoplasms (SPNs), and other rare primary and metastatic tumors can also show pancreatic involvement. 2 Non-neoplastic solid masses, like acute and chronic pancreatitis, autoimmune pancreatitis, and intrapancreatic accessory spleen, may mimic invasive cancer3–5; therefore the diagnosis may be difficult and requires a multidisciplinary approach, including clinical, laboratory, imaging, cytopathologic and histopathologic, and other ancillary studies. Endoscopic ultrasound-guided tissue acquisition by fine-needle aspiration (FNA) and fine-needle biopsy has become the recommended sampling method for solid pancreatic lesions, 6 with high sensitivity, specificity, accuracy, and safety, even in small lesions.7,8 The growing number of cytological or minimal tissue samples has greatly challenged pathologists to provide accurate and reproducible diagnoses or interpretations, and clearly communicate their findings to the multidisciplinary team involved in the management of pancreatic cancer. We are witnessing a steady evolution of standardized reporting systems of pancreatic cytology: the Papanicolaou Society of Cytopathology (PSC) system guidelines 9 and the atlas 10 published in 2014 and 2015 classified the cytological diagnoses of the solid and cystic pancreaticobiliary lesions into the “non-diagnostic” (PSC I), “negative for malignancy” (PSC II), “atypical” (PSC III), “neoplastic: benign” (PSC IVa), “neoplastic: other” (PSC IVb), “suspicious for malignancy” (PSC V), and “malignant” (PSC VI) categories. It also provided a guide to the definitions, terminology, diagnostic criteria, corresponding risk of malignancy (ROM), and a suggested therapeutic algorithm for each of the categories, strongly emphasizing the incorporation of radiological, biochemical, immunocytochemical, and molecular-based findings into the final cytopathology report. The newly published World Health Organization reporting system for pancreaticobiliary cytopathology (WHO system) has been updated and refined with the PSC system, predominantly by reorganizing the heterogeneous tumors from the neoplastic (IV) category into already established and newly created categories. The benign neoplasms (predominantly serous cystadenomas, SCAs) have been transferred from the “neoplastic: benign” to the “negative for malignancy” (WHO II) category, which also includes the non-neoplastic lesions. Intraductal papillary mucinous neoplasms (IPMNs) and mucinous cystic neoplasms with low-grade dysplasia are shifted from PSC “neoplastic: other” to a newly formed “pancreatic neoplasm: low-risk/grade” category (PaN-low, WHO IV); however, the same entities with high-grade dysplasia now belong to the “pancreatic neoplasm: high-risk/grade” category (PaN-high, WHO V). The need for clear subdivision of the precursor intraductal/cystic neoplasms based on the severity of epithelial atypia (low grade vs high grade) is strongly supported by some prospective studies,11,12 which indicated an increased ROM of 90%–95.2% with high-grade atypia and a ROM of 4.3%–19% with low-grade atypia, respectively. All low-grade malignancies (well-differentiated NETs and SPNs), previously classified into “neoplastic: other” of the PSC system, are now included in the “positive for malignancy” (WHO VII) category. With these modifications, the WHO system has seven interpretation categories: “insufficient/inadequate/nondiagnostic” (WHO I); “negative for malignancy” (WHO II); “atypical” (WHO III); “PaN-Low” (WHO IV); “PaN-High” (WHO V); “suspicious for malignancy” (WHO VI); and “positive for malignancy” (WHO VII). 13 In the current literature, only a few recent studies are available that provide information on the ROM of each category that can be well translated into the new WHO system.12,14,15 Therefore, the objective of our retrospective single-center study was to evaluate and compare the predictive value and ROM associated with cytological categories of the WHO classification system, contrasting them with the previously widely used PSC system in the diagnosis of solid pancreatic lesions.
Methods
Our retrospective cohort study enrolled all consecutive patients who underwent EUS-FNA sampling for solid pancreatic lesions at the University of Szeged, Hungary from January 2014 to December 2021. The exclusion criteria were as follows: (1) entirely or predominantly cystic pancreatic lesions confirmed during EUS examination; (2) EUS-FNA sampling of extrapancreatic lesions; and (3) patients who refused to allow the scientific use of their clinical data. The two primary aims of the study were to separately determine and compare the predictive values and ROM of diagnostic categories defined by the PSC system and WHO system specified by the histologic outcome and/or clinical follow-up. Medical documentation of patients was collected using a MedSolutions medical recorder. The reporting of this study conforms to the STROBE statement (Supplemental Material). 16 The study was approved by the Regional and Institutional Human Medical Biological Research Ethics Committee of the University of Szeged, Hungary (ethics approval number: 182/2015 SZTE). All the included patients have signed an informed consent form for the scientific use of their medical data. The study was carried out in accordance with the Declaration of Helsinki.
Endoscopic ultrasound procedure and cytological evaluation
EUS-FNA procedures were performed by two experienced endoscopists using linear echoendoscope (Olympus GF-UCT 140; Olympus GF-UCT 160; Olympus Optical, Tokyo, Japan) and 19G, 22G, and 25G FNA needles (Echotip Ultra; Cook Ireland Ltd., Limerick, Ireland; EZ Shot 2 and 3, Olympus Optical, Tokyo, Japan) were applied for the sampling. The samples taken were used to prepare alcohol-fixed direct smears, formalin-fixed, paraffin-embedded (FFPE) cell blocks/small tissue fragments, and needle-rinsing fluid cytospins. The obtained material from the needle was pushed on the slides with the reinsertion of the stylet, from which the grossly visible coherent pieces of tissue were removed and placed in a tube filled with 10% buffered formalin, and it was processed according to the protocol for biopsy samples. Direct smears were made from the remaining specimen and fixed in 96% methanol at least for 10 min. The residual aspirated tissue was flushed out from the needle to a native sampling tube and processed as cytospin preparations and/or cell blocks. Samples were prepared by EUS nurses or gastroenterologists assisting the endosonographer. No rapid on-site evaluation (ROSE) was done. The FFPE tissues, cytospin preparations, and all direct smears were stained with hematoxylin–eosin. Immunohistochemical testing was performed in most FFPE tissues and selected cases of smears with high cellularity. Pathological diagnosis was based on the assessment of direct smears, cytospins, and FFPE cell blocks which together were considered a single EUS-FNA sample. No additional molecular studies for KRAS or GNAS mutations were performed. The smears were assessed by at least one of the three experienced cytopathologists involved in the study, and in the questionable cases, the diagnoses were based on the consensus of two pathologists. During the whole study period, the PSC system was routinely used for the classification of pancreaticobiliary cytopathology findings to facilitate interdisciplinary communication. Consequently, the PSC categories were determined prospectively, while the reclassification of cytological results into the WHO system was performed retrospectively in 2022.
ROM of cytological categories
Each cytological finding was compared with pathological findings and/or clinical data obtained during follow-up to determine the absolute ROM. It was expressed as the absolute proportion of cases with a malignant final diagnosis within each category. In support of a malignant diagnosis, histological samples obtained by other modalities (repeated biopsy, surgical specimen, autopsy) or in the absence of them, the obvious clinical (weight loss, signs of local progression on endoscopy, gastric outlet or duodenal obstruction, rising tumor marker values) and radiologic evidence of neoplasm (radiologic progression of disease, metastasis formation) were used. In the calculation of ROM, malignant histologic follow-up findings included primary and metastatic carcinomas, NETs, neuroendocrine carcinomas, SPNs, sarcomas, and hematolymphoid malignancies, as well as IPMN with high-grade dysplasia, even without obvious invasion because these lesions have high risk for malignant transformation.17,18 The absence of clinical and/or radiologic evidence of disease or lack of disease progression during the follow-up period was considered a benign lesion. The relative ROM was determined as the ratio of the absolute ROM of each diagnostic category to the absolute ROM of the negative for malignancy (PSC II and WHO II) category.
Predictive value of cytological categories
The diagnostic predictive value of cytological categories was determined based on the sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). The “nondiagnostic” categories were excluded from the analysis because these cases were not suitable for pathological evaluation. The cytological finding was considered a false positive if a benign neoplasm or a non-neoplastic lesion (i.e. chronic pancreatitis) was incorrectly diagnosed as malignant. Those cases were regarded as false negatives which were classified into non-neoplastic, benign neoplastic, or low-grade precursor neoplastic categories of both PSC and WHO system, but the definitive diagnosis at the end of follow-up was malignant. The interpretation of the atypical category (PSC III and WHO III) in terms of neoplastic origin is still challenging, so three different assessment methods for this category were used in the analysis: (1) assessment as negative for malignancy; (2) assessment as positive for malignancy; and (3) exclusion from the evaluation as diagnostically inconclusive cases.
Statistical analysis
Statistical analysis was performed using R statistical software version 3.6.0 (R Foundation). Descriptive statistics were used in the demonstration of the cytological diagnostic categories. Categorical variables were reported as event rates and relative frequencies, and continuous variables as means with standard deviation and medians with ranges. Fisher’s exact test was used to assess the statistical significance of the difference between the ROM values for each category and the negative for the malignancy (PSC II and WHO II) category. Values of p < 0.05 were considered statistically significant.
Results
Study population and cytological diagnosis of pancreatic lesions
A total of 473 patients with solid pancreatic lesions had undergone EUS-FNA biopsy with 521 specimens during the 8-year study period. The male:female ratio was 229:244. The mean age at the time of sampling was 66.61 ± 11.81 years (range: 18–95; median: 68). Lesions were located most frequently in the pancreatic head and uncinate process region (68.71%) and their mean diameter was 33.63 ± 14.02 mm. In 482 cases, EUS-FNA sampling obtained both FFPE tissues and cytological smears, while in 38 cases only cytological specimens and in one case only FFPE tissue were forwarded for histological examination. The final diagnosis after an average follow-up of 13.77 months (range: 0.1–106.4 months, median: 5.67 months) was benign disease in 95 cases (18.43%) and malignant in 426 cases (81.76%) (Figure 1). The histological classification and/or final clinical diagnosis of pancreatic lesions are shown in Table 1. Follow-up histologic reports were available for 205 cases (39.35%), and clinical follow-up data were used for 316 cases (60.65%). The histologic specimens included 40 small biopsy samples with other modalities, like transabdominal core needle biopsies and endoscopic biopsies from tumors involving the stomach/duodenum (19.51%), 11 repeated EUS-FNA cell block samples (5.37%), 121 surgical excision or resection specimens (59.02%), and 33 autopsy samples (16.10%). In 60 patients with follow-up histology reports, the EUS-FNA sample was not suitable for diagnosis and was classified as “non-diagnostic” (PSC I and WHO I) or “atypical” (PSC III and WHO III). In 145 patients, the diagnosis was successfully established based on the EUS-FNA samples, which was confirmed by repeat histological examination in 140 cases. In five patients, diagnostic mistakes of histopathological assessment were revealed during follow-up by repeated sampling. Two cases of ductal adenocarcinoma were misdiagnosed as NETs due to a misinterpretation of the initial technical difficulty in assessing immunohistochemistry. In the other two cases, severe reactive abnormalities accompanying chronic pancreatitis complicated by acute inflammation mimicked the morphology of ductal adenocarcinoma (false positive). In one case, peritumoral EUS-FNA sampling was presumed in the background of histological underestimation of the lesion, which was reported initially as chronic pancreatitis (false negative); the definitive diagnosis of ductal adenocarcinoma was confirmed by repeated transabdominal ultrasound-guided biopsy within 1 month.

Samples representative of the most common pancreatic malignancies, including ductal adenocarcinoma (a), solid pseudopapillary neoplasm (b), neuroendocrine tumor (c), and metastatic small cell neuroendocrine carcinoma (d). Direct smears, H&E staining, 400×.
Histological classification and/or final clinical diagnosis of pancreatic lesions.
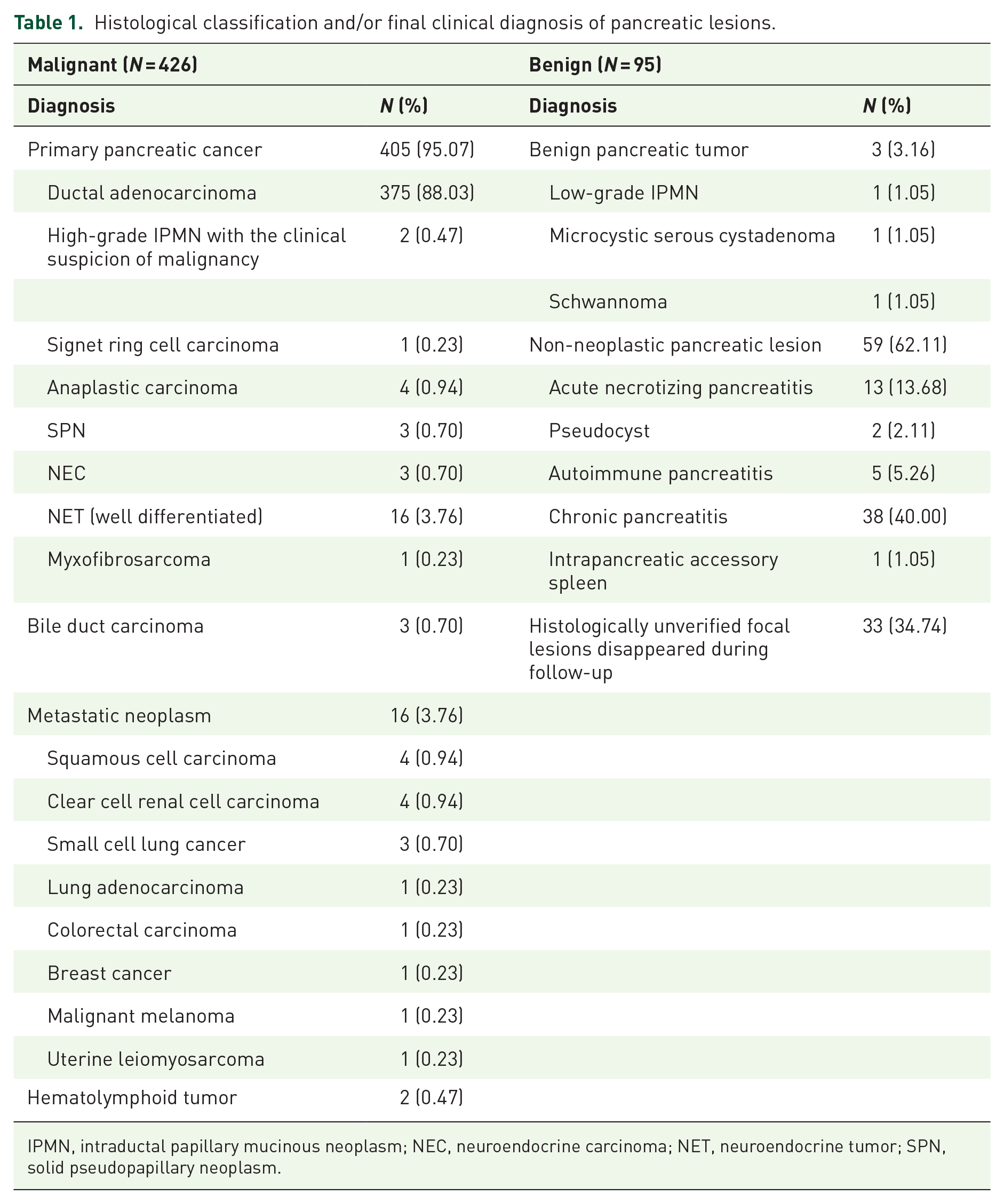
IPMN, intraductal papillary mucinous neoplasm; NEC, neuroendocrine carcinoma; NET, neuroendocrine tumor; SPN, solid pseudopapillary neoplasm.
Reclassification of cytological samples according to the WHO system
The PSC and WHO classification systems show a complete overlap in the definition of “non-diagnostic” (PSC I and WHO I), “negative for malignancy” (PSC II and WHO II), “atypical” (PSC III and WHO III), and “suspicious for malignancy” (PSC V and WHO VI) categories. Our study cohort had no cases in the “neoplastic: benign” (PSC IVa) category but included 20 cases in the “neoplastic: other” (PSC IVb) category. Of these 20 cases, 3 SPN and 15 well-differentiated NETs were reclassified to the “positive for malignancy” (WHO VII) category. There were no cases in the “PaN-low” (WHO IV) category in our cohort, as this category by definition includes intraductal and/or cystic neoplasms with low-grade atypia. Despite the predominantly solid endosonographic features, the cytopathologist strongly suggested the diagnosis of high-grade IPMN in two cases; therefore, these were transferred to the “PaN-high” (WHO V) category. All components of the “malignant” (PSC VI) category were also shifted to the “positive for malignancy” (WHO VII) category.
Risk of malignancy
In 40 of the 83 cases in the “non-diagnostic” (PSC I and WHO I) category, neoplastic lesions were confirmed at the end of follow-up, which was ductal adenocarcinoma in 34 cases, primary bile duct carcinoma in 2 cases, well-differentiated NET in 2 cases, and metastatic clear cell renal cell carcinoma in 2 cases. The unidentified benign lesions were most commonly chronic pancreatitis (n = 19) and acute necrotizing pancreatitis (n = 4), with one case of autoimmune pancreatitis, pseudocyst, microcystic serous cystadenoma, and schwannoma (Table 2). In addition, in 16 cases, the disappearance of the lesion was noted by cross-sectional imaging and/or EUS during follow-up. Corresponding to these, the absolute and relative ROM of the “non-diagnostic” (PSC I and WHO I) category were 48.19% and 21.23%, which is significantly higher compared to the “negative for malignancy” (PSC II and WHO II) category (p < 0.0001) (Table 3).
Distribution of cytological categories proposed by the WHO international system and by PSC in the study cohort and their correlation with the definitive diagnosis of patients.
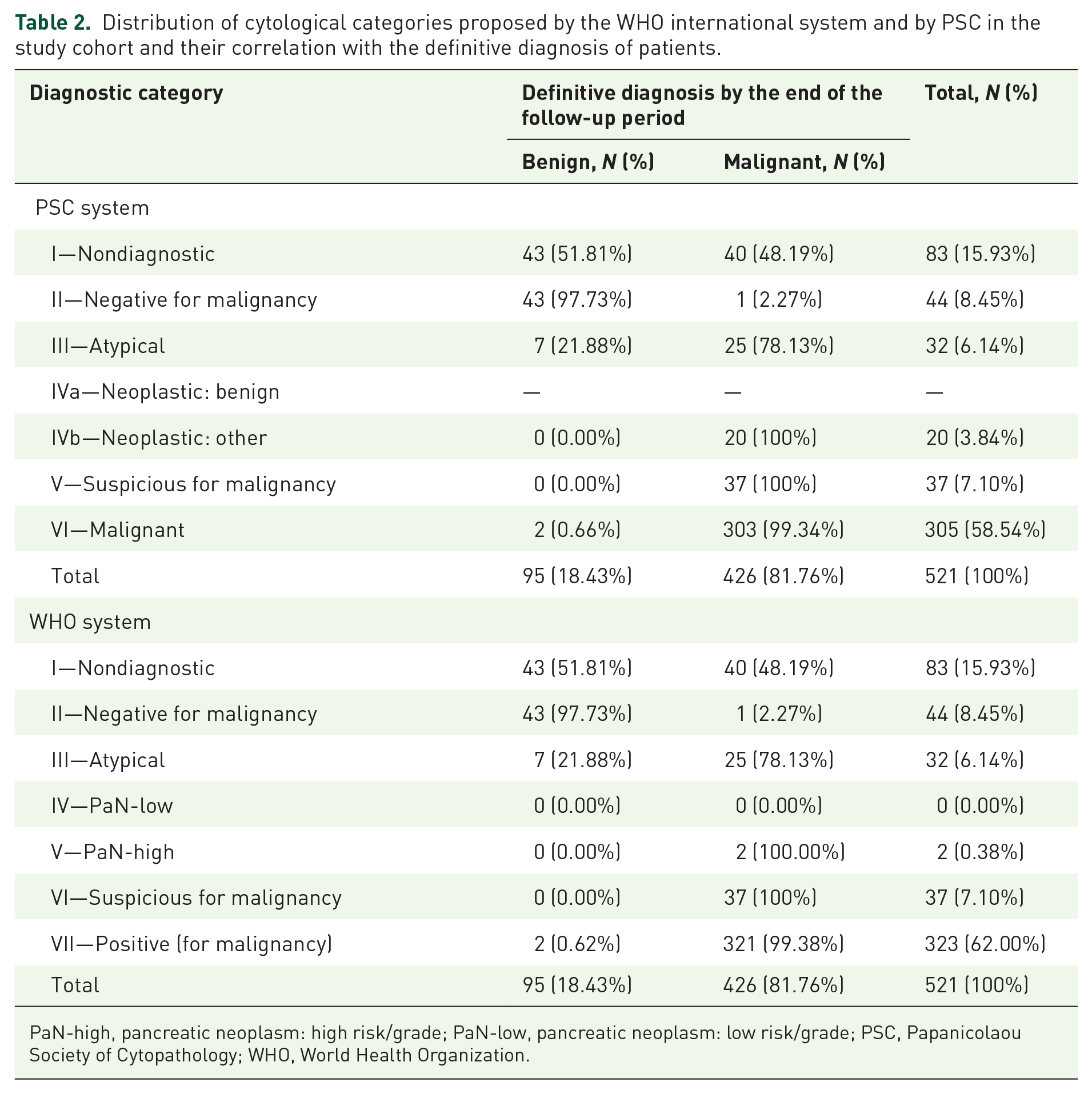
PaN-high, pancreatic neoplasm: high risk/grade; PaN-low, pancreatic neoplasm: low risk/grade; PSC, Papanicolaou Society of Cytopathology; WHO, World Health Organization.
Absolute and relative ROM of cytological categories proposed by the WHO international system and by PSC for reporting pancreaticobiliary cytopathology.
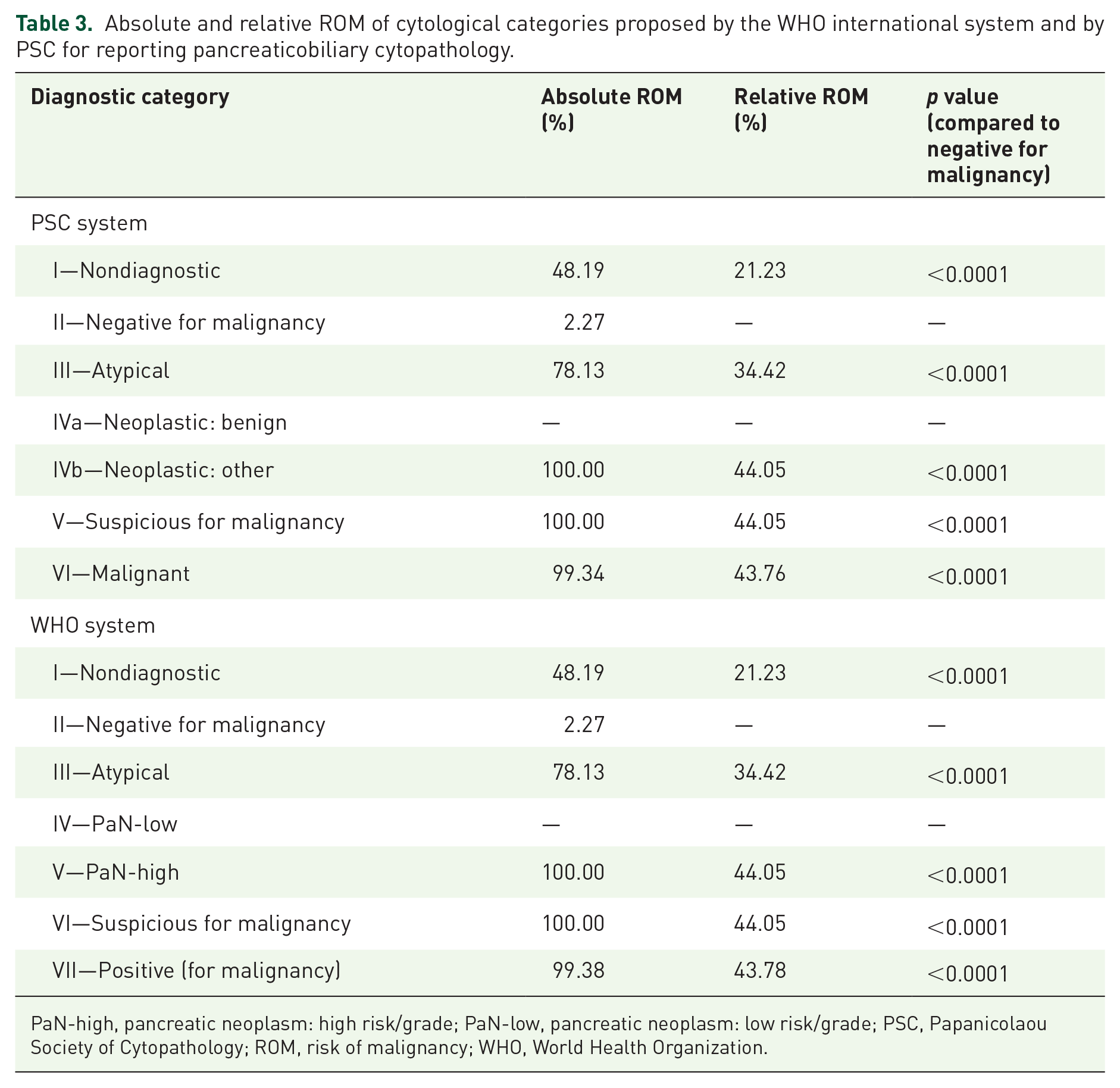
PaN-high, pancreatic neoplasm: high risk/grade; PaN-low, pancreatic neoplasm: low risk/grade; PSC, Papanicolaou Society of Cytopathology; ROM, risk of malignancy; WHO, World Health Organization.
Comparison of the predictive value of cytological categories proposed by the WHO international system and by PSC based on the sensitivity, specificity, PPV, and NPV.

NPV, negative predictive value; PPV, positive predictive value; PSC, Papanicolaou Society of Cytopathology; WHO, World Health Organization.
Within the “negative for malignancy” (PSC II and WHO II) category, ductal adenocarcinoma was demonstrated during the follow-up of patients in one case, giving an absolute ROM rate of 2.27%. The clinical and endosonographic picture of this case suggested autoimmune pancreatitis, and the repeated transabdominal ultrasound-guided biopsy confirmed the final diagnosis within 1 month.
The absolute and relative ROM of the “atypical” (PSC III and WHO III) category was 78.13% and 34.42%, respectively, which is significantly higher compared to the “negative for malignancy” (PSC II and WHO II) category (p < 0.0001). The reason for indeterminate diagnosis was the markedly low cellularity of the smears and FFPE tissues in all cases, which was aggravated by a disturbing degree of blood contamination in 13 cases, pronounced inflammatory cell infiltration in 8 cases, and significant contamination with upper gastrointestinal epithelial cells in 1 case. In the “atypical” (PSC III and WHO III) category, the final diagnosis was based on histopathological examination in 62.50% of cases (repeated EUS-FNA n = 5, transabdominal US-guided biopsy n = 8, surgical sample n = 4, autopsy n = 3). The clinical course of the disease during follow-up was used to determine the final diagnosis in 12 cases. In five of these patients, the endosonographic image was suggestive of benign disease, and no lesion was detected by follow-up EUS examination requiring repeat sampling. The re-biopsy of seven patients with rapidly progressive underlying disease and deteriorating general condition was waived due to lack of clinical relevance because they were no longer suitable for oncological treatment or refused it. The definitive diagnosis at the end of follow-up was malignant in 25 cases (ductal adenocarcinoma n = 22, primary bile duct carcinoma n = 1, metastatic small-cell lung cancer n = 1, and hematolymphoid tumor n = 1) and benign in 7 cases (chronic pancreatitis n = 3, focal lesion disappeared during follow-up n = 2, acute necrotizing pancreatitis n = 1, and low-grade IPMN n = 1).
Despite the heterogeneous nature of the “neoplastic: other” (PSC IVb) category of the PSC system, the absolute and relative ROM for solid lesions were 100% and 44.05%. This category included 14 well-differentiated NETs, 2 high-grade IPMNs, 3 SPNs, and 1 ductal adenocarcinoma. It is important to note that one ductal adenocarcinoma was misdiagnosed by cytology as NET and therefore included in this category; however, this did not affect the ROM. The two high-grade IPMN cases of the “PaN-high” (WHO V) category were considered malignant due to the high risk of malignant transformation with 100% absolute and 44.05% relative ROM. In these cases, the malignant nature of these tumors is supported only by clinical and radiological progressive disease course, histological verification of invasion was impeded by patients’ refusal of curative surgery.
All the cases in the “suspicious for malignancy” (PSC V and WHO VI) category also had a definitive diagnosis of malignancy (ductal adenocarcinoma n = 35, signet ring cell carcinoma n = 1, and metastatic lung adenocarcinoma n = 1). In the “malignant” (PSC VI and WHO VII) category, two ductal adenocarcinoma diagnoses were overestimated, being chronic pancreatitis with acute inflammation (false positive). The absolute and relative ROMs were 99.34% and 43.76% for “malignant” (PSC VI) and 99.38% and 43.78% for “positive for malignancy” (WHO VII) categories, respectively, due to the different case numbers.
Predictive value of cytological categories
Excluding “non-diagnostic” (PSC I and WHO I) and inconclusive “atypical” (PSC III and WHO III) categories, the sensitivity, specificity, NPV, PPV, and validity of the cytopathological evaluation using the PSC and WHO systems were identical (99.72%, 95.56%, 97.73%, 99.45%, and 99.26%, respectively). No substantial improvement in the sensitivity of pathological assessment could be achieved by including the “atypical” category in the analysis. By considering the “atypical” category as malignant, a substantial reduction in specificity was seen in both the PSC and the WHO systems (from 95.56% to 82.69%). When the “atypical” category was considered benign, a comparable reduction in sensitivity (from 99.72% to 93.26%), NPV (from 97.73% to 65.79%), and validity (from 99.26% to 93.61%) was observed (Table 4).
Discussion
In the last decade, EUS-FNA has become a first-line modality for the diagnosis of solid pancreatic lesions 6 ; however, the pathological evaluation of the obtained cytological specimens and the clinical interpretation of the findings are often challenging. The purpose of standardized reporting categories of the previously used PSC system was to summarize the morphologic criteria and provide risk stratification in each reporting category, as well as to support the communication among the members of the interdisciplinary team involved in the management of pancreatic cancer.9,10 The WHO classification system, published in 2022, aimed to address the interpretation problems encountered in the application of the PSC system, specifically regarding the PSC IVb category. Currently, limited data are available on the risk stratification of the diagnostic categories of this new classification system and its advantages over the PSC system in clinical practice have not yet been confirmed. Our retrospective single-institution cohort study affirmed the absence of differences between the PSC and WHO classification systems in the diagnosis of solid pancreatic lesions when analyzing the ROM values of diagnostic categories and the predictive values of conclusive cytology findings (sensitivity, specificity, NPV, PPV, and the validity). However, the WHO classification system, as illustrated in Figure 1, offers a clearer and more easily interpretable situation for interdisciplinary teams through the reclassification of the PSC IVb category.
Several prospective and retrospective studies have assessed the ROM values of the standardized categories of the PSC system, showing significant variation in most categories. Exceptions to this trend are PSC V (suspicious for malignancy) and PSC VI (malignant) categories, where consistently high ROM has been reported. In the systematic review including eight studies conducted by Nikas et al. before shifting into the new WHO system, the absolute ROM values for PSC I, II, III, IVb, V, and VI categories were 8%–50%, 0%–40%, 28%–100%, 0%–34%, 82%–100%, and 97%–100%, respectively. 19 The common categories between the PSC and WHO reporting systems, which have not undergone substantial changes in relation to solid pancreatic lesions, are “non-diagnostic” (PSC I and WHO I), “negative for malignancy” (PSC II and WHO II), “atypical” (PSC III and WHO III), and “suspicious for malignancy” (PSC V and WHO VI), so there should be no difference in the value of ROM between the reporting systems. The difference in the risk stratification of the two reporting systems could only be the result of the reclassification of the PSC IVa and PSC IVb categories. This reclassification resulted in the establishment of PaN-low (WHO IV) and PaN-high (WHO V) categories. In addition, malignant lesions were transferred to the “positive for malignancy” (WHO VII) category. In our study cohort, only 3.84% of cases needed to be reclassified due to the transition to the WHO system, which did not result in significant changes in the relative and absolute ROM values of the categories, which were as follows: PSC IVb 100% and 44.05%; PSC VI 99.34% and 43.76%; PaN-high (WHO V) 100% and 44.05%; and WHO VII 99.38% and 43.78%, respectively. A retrospective study carried out by Lui et al. evaluated the absolute ROM of 2562 EUS-FNA samples in seven standardized categories of the WHO system and found that it was 50%, 29%, 70%, 15%, 100%, 99%, and 100% for solid pancreas lesions. 20 Simultaneously, it was emphasized that the absolute ROM value is significantly influenced by whether the lesion exhibits cystic or solid morphology. In their study cohort, the absolute ROM for cystic lesions exhibited notable distinctions from those of solid lesions, with values of 7%, 0%, 19%, 13%, 38%, 78%, and 100% in WHO I-VII categories. Remarkably, in this study, the ROM value for the “negative for malignancy” (WHO II) category is very high and exceeds the value observed in the majority of other studies.15,19,21 The observed differences may primarily stem from a notable level of interobserver variability among cytologists in the assessment of EUS-FNA samples. 22 In our cohort, the absolute ROM in this category was only 2.27%. The explanation for this is that pathologists categorized cytological findings as “negative for malignancy” (WHO II) in very selective cases, specifically when the cytologic features were characteristic for certain non-neoplastic lesions (chronic pancreatitis, autoimmune pancreatitis, ectopic spleen, etc.) and/or when the complete absence of cellular atypia correlated with EUS findings indicating a lack of malignant involvement.
The clinical significance of the “atypical” (PSC III and WHO III) category is notable as it does not permit a definitive differential diagnosis between benign and malignant conditions. This often results in delays in treatment, heightening the burden of patients, and increasing medical costs due to repeated interventions. Interpreting this category poses challenges in establishing the predictive value of cytological categories. If categorized as positive for malignancy, specificity decreases (82.69%) and the number of false positives (n = 9) rises. Conversely, if classified as negative for malignancy, the NPV decreases (65.79%) and the number of false negatives (n = 26) increases. The classification system demonstrates its highest validity (99.26%) when the atypical category is excluded from the analysis; consequently, our results confirmed the inconclusive nature of this category and that it does not contribute to the diagnostic process. Our findings align with studies assessing the predictive value of both the PSC system and the WHO system.12,14,15,23 Reducing the proportion of cytological findings in the “atypical” (PSC III and WHO III) category may be the ideal solution for these problems; therefore, institutions (both cytopathologists and endoscopists) should monitor and keep the rate of this low. In terms of sampling, the proportion of inconclusive cytological findings including the “non-diagnostic” (PSC I and WHO I) and “atypical” (PSC III and WHO III) categories is influenced by the characteristics of the lesion (uncinate process location, less than 2 cm in size, the presence of necrotic areas, increased vascularization, benign etiology) and the technical aspects of EUS-FNA (smaller needle diameter, suction technique, the use of contrast-enhanced EUS or EUS elastography, the technique of smear preparation, obtaining FFPE, ROSE, macroscopic on-site evaluation).24–28 Previous studies have shown a significant variation among cytopathologists in the use of indeterminate diagnostic categories.22,29,30 The enhancement of cytological diagnostic criteria, standardization of specimen quality evaluation, and training for cytopathologists have the potential to enhance agreement among cytopathologists, which would lead to increased repeatability of cytological diagnosis and reduction of inconclusive and false negative cases. 31 Unfortunately, currently, there is no guideline that defines atypical cytology rate as a quality indicator or determines its minimum standard value. 32
The most important limitation of our study is its single-center retrospective design. In addition, it is important to note that the final diagnosis relied on clinical follow-up data in more than 60% of cases, so the histological diagnosis was confirmed by follow-up histologic reports in only 39.35% of cases. A significant strength of our study is the relatively large number of included cases and the creation of a clinically uniform cohort by including only solid pancreas lesions. Solid and cystic pancreatic lesions exhibit substantial differences in their incidence, malignant potential, clinical behavior, diagnostic and therapeutic protocols, as well as prognosis. Furthermore, previous studies have also highlighted that the standardized categories of the PSC and WHO systems demonstrate significant variations in the ROM value for solid and cystic lesions, respectively.
Conclusion
Our study revealed that the WHO system was identical to the PSC system in terms of ROM and predictive values of categories for diagnosing solid pancreatic lesions. However, the reclassification of malignant lesions (such as SPN and pancreatic NET) from the “neoplastic: other” (PSC IVb) category to the “positive for malignancy” (WHO VII) category not only harmonizes the systems but also enhances interdisciplinary communication, reducing the likelihood of misinterpreting pathological findings. In our cohort, the absolute ROM of the “negative for malignancy” category (PSC II and WHO II) was only 2.27%, a rate relatively low compared to findings in most other similar studies. This low rate is attributed to its usage only when a specific diagnosis of a non-neoplastic or benign neoplastic lesion can be made, or when cytology reveals solely normal pancreatic cells without any evident mass lesion on ultrasound, as defined by the WHO system. Therefore, we recommend a judicious application of the “negative for malignancy” category due to the potential for false negatives, which may be caused by sampling error. Our study also confirmed that specimens categorized as “atypical” (PSC III and WHO III) are associated with malignancy in almost 80% of cases but may lead to delay in diagnosis due to their inconclusive nature. Therefore, the proportion in this category should be reduced, which could be facilitated by specific training of pathologists or by the evaluation of questionable cases by multiple pathologists.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241271958 – Supplemental material for Comparison of risk of malignancy and predictive value of diagnostic categories defined by Papanicolaou Society of Cytopathology system and WHO reporting system for pancreaticobiliary cytopathology in solid pancreatic lesions
Supplemental material, sj-docx-1-tag-10.1177_17562848241271958 for Comparison of risk of malignancy and predictive value of diagnostic categories defined by Papanicolaou Society of Cytopathology system and WHO reporting system for pancreaticobiliary cytopathology in solid pancreatic lesions by Béla Vasas, Anna Fábián, Zsófia Bősze, Sándor Hamar, László Kaizer, Tibor Tóth, Péter Bacsur, Tamás Resál, Anita Bálint, Klaudia Farkas, Tamás Molnár, Zoltán Szepes and Renáta Bor in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
