Abstract
Background:
Needle-based confocal laser endomicroscopy (nCLE) can accurately identify the pathological types of pancreatic cystic lesions (PCLs), but there are no reports of real-time intraoperative guidance for lauromacrogol ablation (LA) therapy.
Objectives:
This study aimed to assess the diagnostic accuracy and safety of nCLE in identifying the candidates for LA in the same session as endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA).
Design:
Prospective study.
Methods:
We performed a prospective study of patients with PCL who underwent EUS-FNA and nCLE at the First Medical Center of Chinese PLA General Hospital between February 2024 and June 2025. The diagnostic performance of nCLE in intraoperatively differentiating concomitant ablation-eligible lesions (CAELs) from concomitant ablation-ineligible lesions (CAILs) was evaluated and compared with that of EUS morphology and the cyst fluid string sign.
Results:
A total of 29 patients were enrolled in the study, and the mean size of the cyst was 34.1 ± 12.5 mm. The mean duration of EUS-nCLE was 4.61 min. EUS-nCLE identified CAELs with 94% sensitivity, 77% specificity, and 86% accuracy. The diagnostic accuracy of nCLE was superior to that of EUS morphology and the string sign (86% vs 66%,
Conclusion:
Confocal laser endomicroscopy demonstrates favorable real-time diagnostic performance for PCLs, allowing accurate assessment of the suitability for ablation during puncture and thereby avoiding the need for a repeat procedure.
Plain language aummary
The pancreatic cystic lesions (PCLs) have varying degrees of malignant potential and may cause compressive symptoms. Lauromacrogol ablation has been proven to be safe and efficacious for the treatment of PCLs, but it has strict indications. Evaluating whether a cyst is suitable for ablation relies on cyst fluid analysis and biopsy results obtained from an initial diagnostic puncture. However, this entails a subsequent therapeutic puncture for patients who are eligible for ablation. Confocal laser endomicroscopy provides intraoperative diagnosis of pancreatic cystic pathology and determines suitability for concomitant ablation. Our study demonstrated that confocal laser endomicroscopy identified lesions suitable for concomitant ablation with 86% diagnostic accuracy, higher than that of traditional intraoperative cyst morphology. Furthermore, the incidence of postoperative acute pancreatitis was 6.9%, and all instances were mild in severity. Therefore, due to its favorable intraoperative diagnostic performance, confocal laser endomicroscopy serves as a valuable tool for optimizing the management strategy of patients with pancreatic cystic lesions.
Keywords
Introduction
Pancreatic cystic lesions (PCLs) comprise a heterogeneous group of lesions with varying degrees of malignant potential. 1 Cystic lesions with the potential for malignant transformation include intraductal papillary mucinous neoplasm (IPMN), mucinous cystic neoplasm (MCN), pancreatic neuroendocrine tumors (NET), and solid pseudopapillary neoplasm (SPN). Serous cystic neoplasms (SCN) and pseudocysts (PC) are regarded as benign cystic lesions. The current management strategies for PCLs involve surveillance or surgical resection. 2 Recently, the development of endoscopic ultrasound (EUS)-guided ablation has gained considerable attention as a minimally invasive alternative in treating PCLs. Lauromacrogol, as a sclerosant, was reported for the ablation of PCLs in 2015 by Linghu et al. 3 EUS-guided lauromacrogol ablation (EUS-LA) was proven to be safe and efficacious for the treatment of PCLs. 4 However, EUS-LA has strict indications, mainly focusing on selected SCN and non-malignant MCN. It is of utmost importance to accurately identify the pathological type of PCLs before EUS-LA. However, the current assessment of PCLs relies mainly on cyst fluid analysis and/or EUS-guided through-the-needle biopsy (EUS-TTNB), which means that patients need to be re-punctured if they meet the indications for ablation.
EUS-guided needle-based confocal laser endomicroscopy (EUS-nCLE) offers real-time microscopic imaging of the inner wall of pancreatic cysts during an EUS-guided fine-needle aspiration (EUS-FNA) procedure, providing virtual biopsies with high resolution (1–3.5 mm). 5 Imaging patterns from EUS-nCLE have been associated with specific PCLs, which have excellent diagnostic performance. 6 The nCLE can identify the specific pathological type of PCLs during the puncture and evaluate on-site whether the patient meets the LA indications. It helps to make clinical decisions and reduce the number of punctures. This study aimed to assess the efficacy and safety of using nCLE features to guide the decision for concomitant LA during the same session as EUS-FNA.
Methods
Study design
Patients with PCLs who underwent EUS-FNA were prospectively and consecutively screened at the First Medical Center of Chinese PLA General Hospital between February 2024 and June 2025. A total of 33 patients underwent EUS-nCLE. Diagnostic nCLE images were successfully obtained in 29 of these patients. The study flow diagram is shown in Figure 1. The inclusion criteria were as follows: (1) age ⩾18 years; (2) provision of informed consent to EUS-nCLE; (3) PCLs with an appropriate pathway for puncture with a 19-G needle; and (4) cyst size ⩾15 mm. The exclusion criteria were as follows: (1) recently identified episode of pancreatitis; (2) known allergy to fluorescein contrast agent; (3) an inability to safely tolerate intravenous anesthesia; (4) microcystic lesions or those with predominantly solid components; and (5) patients with conditions indicative of a high surgical risk such as pregnancy, coagulopathy, or severe cardiovascular disease. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 7

The study flow diagram.
Procedures
EUS examinations were carried out by an endosonographer using a linear-array echoendoscope (Prosound F75 (Aloka, Tokyo, Japan) and GF-UCT260 (Olympus, Tokyo, Japan)) to observe the morphology and size of the cyst and whether it was connected to the pancreatic duct. Then EUS-FNA and nCLE procedures were carried out as previously reported.6,8 A 19-G needle (Cook, Limerick, Ireland) preloaded with nCLE mini-probe (CelTouch, Wuxi, China) was inserted into the cyst and securely positioned under EUS guidance through a trans-gastric or trans-duodenal approach. Fluorescein (2.5 mL; 10% fluorescein sodium) was intravenously injected, and the video of the inner structure of the cyst was simultaneously recorded within 10 min. After image acquisition, the miniprobe was retrieved from the needle, and the images were analyzed by two trained gastroenterologists (>70 nCLE examinations for gastrointestinal and PCLs), who were blinded to the EUS images. At the same time, the maximum possible volume of cyst fluid was aspirated, followed by instillation of an appropriate amount of normal saline into the cyst, and EUS-TTNB was then performed. After biopsy, the saline within the cyst was aspirated, leaving a small rim of fluid around the needle tip. The cystic fluid was sent for cytological and biochemical analyses after recording the cystic fluid characteristics, such as its color, viscosity, clarity, and volume. The biopsy specimens were sent for histological analysis.
During the procedure, the endosonographer assessed the cyst type based on a comprehensive evaluation of both EUS features and nCLE diagnosis. If the cyst was considered suitable for ablation during the puncture, the EUS-LA procedure would be carried out. 4 Lauromacrogol was injected into cysts until the cystic wall was completely soaked in solution, followed by lavage (repetitive aspiration and reinjection of lauromacrogol) for 3–5 min to increase its concentration in the cyst. Directly following lavage, half to two-thirds of the lauromacrogol was retrieved, leaving one-third to half of the lauromacrogol in the cyst. Then, the needle was carefully retracted.
Patients were closely monitored after the procedure and were assessed for any adverse events (AEs) such as abdominal pain, fever, nausea, bleeding, pancreatitis, or an increase in serum amylase or lipase levels. Post-procedural management differed according to whether ablation was performed. Patients who did not undergo ablation fasted for 1 day and received intravenous proton pump inhibitor (PPI) and antibiotic for 1 day, followed by oral antibiotic for 3 days and oral PPI for 3–7 days. Patients undergoing ablation fasted for 2–3 days. An intravenous PPI and antibiotic were administered for 2–3 days, followed by oral PPI therapy for 3–7 days. Octreotide was intravenously administered for at least 1 day until the serum amylase levels returned to normal.4,9
Indications and contraindications of EUS-LA
The patients who were enrolled to undergo EUS-LA were required to meet the following criteria: (1) SCNs in selected cases, such as those with increasing size during radiological imaging surveillance, those causing symptoms, or patients with a strong desire to undergo the procedure; (2) non-malignant MCN. The exclusion criteria were as follows: (1) SCN with pre-procedural refusal of same-session ablation; (2) cysts communicated with pancreatic duct on EUS or suspected IPMN on nCLE; (3) PC or pancreatic necrosis; (4) highest risk features of malignancy or suspicious malignancy 2 ; (5) multilocular PCLs (>6 locules within the cyst); and (6) cystic degeneration of neuroendocrine tumor or SPN. SCN and non-malignant MCN were pooled in the overall group of concomitant ablation-eligible lesions (CAELs). IPMN, pseudocyst, and malignant MCN were classified as concomitant ablation-ineligible lesions (CAILs).
PCLs diagnosis
EUS-based diagnosis
Intraoperative comprehensive evaluation based on the cyst morphology and the characteristics of cystic fluid (e.g., the string sign).2,10–14 The presence of malignant signs in cysts is evaluated according to the 2023 Kyoto guidelines. 2
nCLE diagnosis
The nCLE diagnosis was established based on the previous studies (Figure 2).6,8,15–19 The findings highly specific for SCN are a “superficial vascular network” or “fern pattern,” which is a parallel or interconnected network of blood vessels. For IPMN, characteristic findings include finger-like papillae consisting of an overlying epithelium and an underlying vascular core. The increased papillary epithelial width and darkness on nCLE suggest malignant potential of IPMN. For MCN, characteristic findings include single or multiple layers of epithelium (epithelial bands) without a papillary configuration. The dark aggregates of compact cells surrounded by various quantities of irregular small vessels or gray tissue were related to the malignancy of MCN. The “branched” or “rope-ladder” vascular patterns can also be observed in IPMN and MCN. The features of a pseudocyst on nCLE are fields of bright or black particles and a lack of discernible epithelial and vascular pattern.
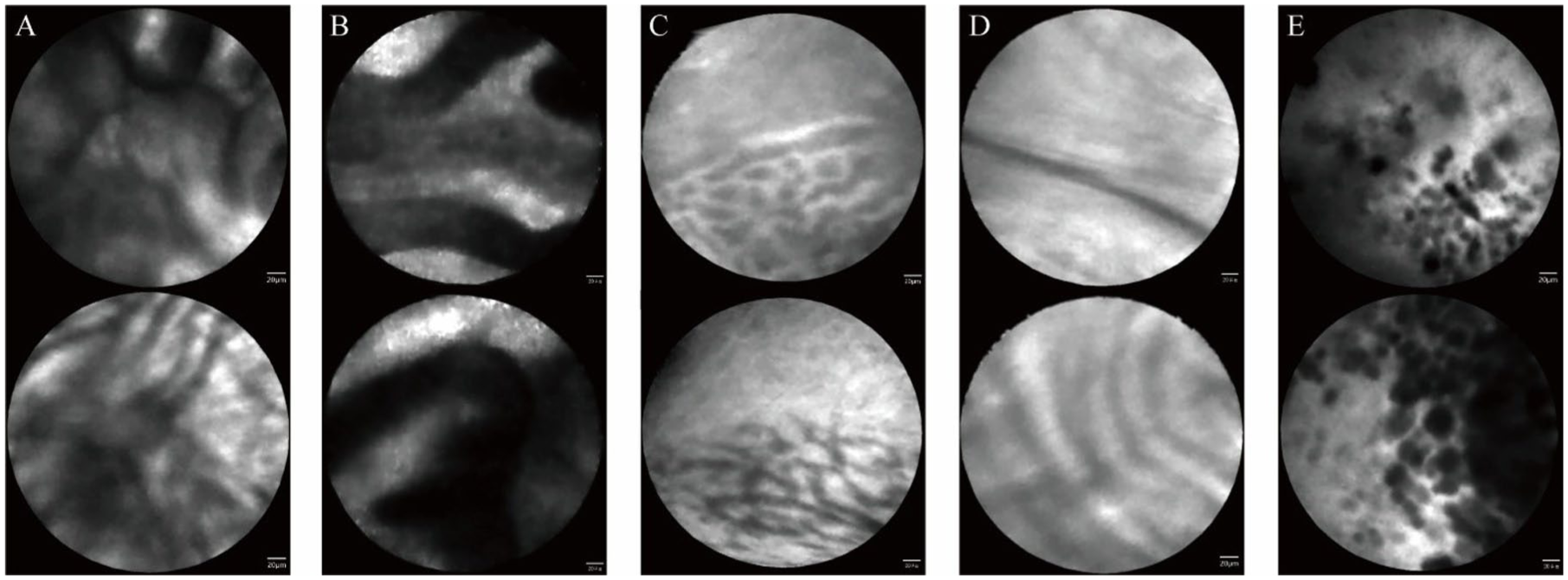
Representative nCLE images of pancreatic cystic lesions. (a) IPMN displays finger-like papillae, consisting of an overlying epithelium and an underlying vascular core. (b) Malignant IPMN is characterized by increased papillary epithelial width and darkness. (c) SCN exhibit a “superficial vascular network” or “fern pattern,” representing a parallel or interconnected network of blood vessels. (d) MCN typically shows single or multiple layers of epithelial bands, without a papillary configuration. (e) Pseudocysts present a field of bright or dark particles.
Reference standard diagnosis
The final diagnosis was established using histopathology from surgical specimens (when available) or EUS-TTNB samples, and, in the absence of diagnostic histology, was determined by integrating cyst fluid cytology and cyst fluid biomarkers (including carcinoembryonic antigen (CEA), glucose, amylase, and lipase). Biomarker thresholds were used to support classification of mucinous versus non-mucinous cysts: an elevated CEA level (>192 ng/mL) and/or a low glucose level (<50 mg/dL) were considered suggestive of a mucinous lesion.11,20
Follow-up evaluation
The treatment response to EUS-LA was assessed by comparing cyst volume at the final follow-up with the original volume prior to the procedure. Cyst volume was estimated on MRI by measuring the maximal cyst diameters in three orthogonal dimensions—the length (a) and width (b) on axial images, and the height (c) on coronal images—and applying the ellipsoid formula:
Statistical analysis
Categorical variables were tabulated as counts and percentages. Continuous variables were presented as means with standard deviations or median with interquartile range (IQR). For normally distributed continuous data, statistical significance was assessed by the Student’s
In the diagnostic performance analysis, the accuracy, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated for each index test (EUS-based diagnosis and nCLE diagnosis) against the reference standard, with 95% confidence intervals (CIs). Sensitivity was defined as the proportion of reference-standard CAELs correctly identified as CAELs, and specificity as the proportion of reference-standard CAILs correctly identified as CAILs. Overall accuracy was calculated as the proportion of correctly classified lesions among all lesions. The McNemar test was used to compare classification performance between EUS-based diagnosis and nCLE diagnosis. When the number of discordant pairs was small, an exact McNemar test was applied.
Results
Cohort characteristics
A total of 29 patients were enrolled in the study between February 2024 and June 2025 at a single center. As shown in Table 1, the mean age of all subjects was 51.0 ± 15.9 years, and the mean size of the cyst was 34.1 ± 12.5 mm. Eight patients reported a history of abdominal pain and/or bloating, while jaundice was observed in two patients. The mean duration of EUS-nCLE was 4.61 min. After integrated intra-procedural assessment, 15 of the 29 patients were deemed to have CAELs. LA was performed in 13 of these patients. The remaining two patients had stated during pre-procedural counseling that they would decline ablation if the lesion was suspected to be an SCN; therefore, they did not undergo ablation and were followed up conservatively.
Cohort characteristics.
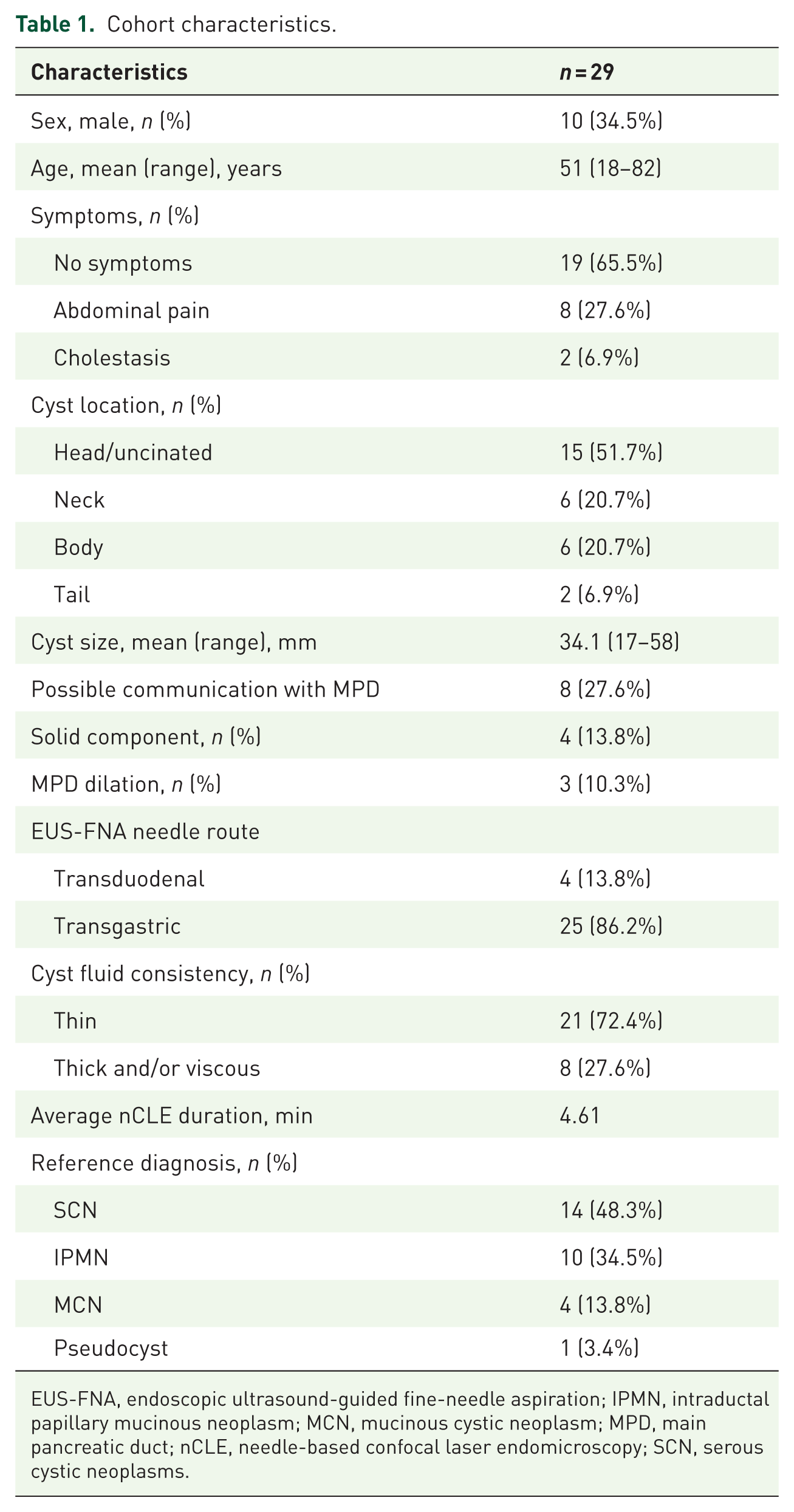
EUS-FNA, endoscopic ultrasound-guided fine-needle aspiration; IPMN, intraductal papillary mucinous neoplasm; MCN, mucinous cystic neoplasm; MPD, main pancreatic duct; nCLE, needle-based confocal laser endomicroscopy; SCN, serous cystic neoplasms.
Diagnostic accuracy for differentiating CAELs versus CAILs
The diagnostic performance of the tested modalities is summarized in Table 2. The sensitivity, specificity, and overall accuracy of EUS-based diagnosis for classifying lesions as CAELs versus CAILs were 0.50, 0.85, and 0.66, respectively. nCLE diagnosis yielded a sensitivity of 0.94, a specificity of 0.77, and an overall accuracy of 0.86. Notably, nCLE diagnosis showed significantly higher overall classification accuracy than EUS-based diagnosis (
Diagnostic parameters for the diagnosis of CAELs and CAILs using EUS-based diagnosis and nCLE diagnosis.

CAELs, concomitant ablation-eligible lesions; CAILs, concomitant ablation-ineligible lesions; CI, confidence interval; EUS, endoscopic ultrasound; nCLE, needle-based confocal laser endomicroscopy; NPV, negative predictive value; PPV, positive predictive value.
Adverse events
The overall incidence of AEs was 13.8% (4 of 29). Two patients developed hyperamylasemia. And procedure-related pancreatitis occurred in two patients (6.9%), all of which were classified as mild according to the revised Atlanta classification. 22 The details of AEs stratified by ablation status are as follows. Among the 13 patients who underwent LA, 2 developed postoperative hyperamylasemia without abdominal pain. Of the 16 patients who did not undergo ablation, 2 developed postoperative mild acute pancreatitis. All patients who experienced AEs recovered with conservative symptomatic treatment. No AEs were attributed to intra-cystic hemorrhage, infection, or intravenous fluorescein administration.
Treatment responses
Of the 13 patients who underwent ablation therapy, 8 were successfully followed up, with a mean follow-up period of 10 months (Table 3). Treatment responses of eight patients were CR in three patients (37.5%), partial response in two patients (25.0%), and persistent in three patients (37.5%). The median cyst volume decreased from 7537 mm3 (IQR, 6150–16,003.25) before ablation to 1113 mm3 (IQR, 87.75–9805.75) after ablation (
Treatment outcomes after EUS-guided lauromacrogol ablation.

EUS, endoscopic ultrasound; IQR, interquartile range.

The MRI images of the cyst before and after ablation. (a, b) MRI images of Patient A. (a) Pre-procedural imaging showing a 25 mm × 24 mm cystic lesion in the pancreatic body. (b) Follow-up imaging at 8 months after ablation, demonstrating complete radiological resolution of the cyst. (c, d) MRI images of Patient B. (c) Pre-procedural imaging showing a 28 mm × 18 mm cystic lesion in the pancreatic head. (d) Follow-up imaging at 9 months after ablation, showing the cyst decreased to 12 mm × 13 mm, achieving partial resolution.
Discussion
PCLs demonstrate heterogeneous malignant potential, and the appropriate therapeutic strategy depends on an accurate diagnosis. Although cross-sectional imaging and EUS can provide morphological information for the diagnosis of PCLs, their diagnostic accuracy remains unsatisfactory. 23 Glucose and CEA have shown promising diagnostic value in EUS-FNA cyst fluid analysis, and the advent of EUS-TTNB has further enhanced the diagnostic accuracy for PCLs.24,25 However, these methods can only provide relevant results postoperatively and cannot achieve real-time diagnosis of cystic lesions. By contrast, nCLE enables real-time visualization of the cyst wall at 1000-fold magnification during EUS-FNA, with reported diagnostic accuracies ranging from 71% to 94%. In a large multicenter prospective study, EUS-nCLE demonstrated high diagnostic performance for identifying specific PCL subtypes, with overall accuracy consistently exceeding 90%.10,26 EUS-guided ablation offers the potential for early intervention in selected lesions, but its application is constrained by strict indications. In practice, diagnosis sometimes needs to be confirmed at the initial puncture, followed by a second puncture for ablation, which increases both patient trauma and healthcare costs. The real-time diagnostic capacity of confocal laser endomicroscopy effectively compensated for this limitation.
Treatment options are based on the risk stratification of the PCLs, and generally include surveillance, ablation therapy, and surgical resection. EUS-guided ablation is a promising approach for PCLs, with a significantly lower incidence of Branch duct IPMN (BD-IPMN) progression over 10 years compared to surveillance alone.27,28 In addition, frequent surveillance for low-risk PCLs may act as a burden for the patients and society. It has been demonstrated that the intensity of image-based surveillance can be safely and significantly reduced after successful EUS-guided ablation. 29 Moreover, in a long-term analysis, EUS-guided ablation demonstrated superiority over surgery in terms of safety profile and preservation of pancreatic function. 21 Ablation therapy has reported long-term efficacy and safety, and ablation agents, including ethanol, paclitaxel, and polidocanol, are available options. 30 A recent meta-analysis reported an overall CR rate of 44% after EUS-guided ablation. 31 The first human pilot study to illustrate the safety and feasibility of EUS-guided ethanol ablation was conducted in 2005. 32 Ethanol monotherapy achieved a pooled CR rate of 32%. Paclitaxel has been incorporated into EUS-guided chemoablation because of its antineoplastic activity and hydrophobicity, which may facilitate retention within the cyst cavity and prolong local drug exposure. In a pooled analysis, combined paclitaxel and ethanol ablation achieved CR in 63.6% of PCLs. 33 EUS-guided radiofrequency ablation appears to be a feasible, minimally invasive option for selected pancreatic lesions, including microcystic SCNs, BD-IPMN with worrisome features, and small NETs, with meaningful lesion reduction and an acceptable adverse-event profile.34–37 EUS-LA safely treats PCLs with a CR rate of 51.4%. LA mainly focusing on selected SCN and non-malignant MCN. The communication between the cyst and the pancreatic duct of IPMN may result in a poor treatment response and a higher risk of pancreatitis, and there have been no reports on the safety and efficacy of LA for IPMN. Therefore, all types of IPMNs were excluded for EUS-LA in this study. We are currently undertaking studies to evaluate the safety and efficacy of EUS-LA in patients with BD-IPMN.
During the puncture procedure, the cyst type is assessed based on the intraoperative factors, including cyst morphology, communication with the pancreatic duct, and the results of the string sign. However, the diagnostic accuracy of these intraoperative indicators is relatively low. And they are insufficient for accurately identifying the pathological type of the cyst, being able to correctly recognize only 66% of CAELs. Definitive assessment of whether a lesion is suitable for ablation requires postoperative results of cyst fluid analysis and cyst biopsy pathology. nCLE offers real-time imaging that allows for detailed visualization of cystic lesions, providing additional diagnostic information by assessing cystic epithelium, vascularity patterns, and potential malignancy features. This enhanced imaging capability facilitates more accurate lesion characterization, helping to identify lesions that are truly suitable for concomitant ablation. Our study demonstrated that the intraoperative diagnostic accuracy of nCLE was higher than that of cyst morphology-based diagnosis (86% vs 66%,
In clinical practice, SCN and BD-IPMNs are sometimes difficult to distinguish based solely on morphological features, as both can appear as multilocular, lobulated lesions.11,38 Furthermore, when the pancreatic duct is not markedly dilated, accurate assessment of communication between the PCLs and the pancreatic duct becomes challenging. With a diagnostic accuracy exceeding 90% for distinguishing mucinous from non-mucinous lesions, nCLE offers a powerful tool to overcome this limitation.6,39,40 However, confocal laser endomicroscopy still encounters several diagnostic challenges. The multiple parallel epithelial bands observed in MCNs may sometimes be difficult to distinguish from the papillary structures of IPMNs, 15 which can result in diagnostic ambiguity and misjudgment regarding the eligibility of cysts for ablation therapy. Incorporating EUS findings provides key information on cyst morphology and, importantly, the relationship with the pancreatic duct, which can aid in distinguishing MCN from IPMN. Thus, EUS and nCLE offer complementary strengths, and integrating both modalities may improve cyst-type classification. In our study, to preserve independent interpretation of confocal images, EUS morphological features were not disclosed to the nCLE reviewers. Nonetheless, in routine practice, combining nCLE with EUS findings is likely to support more accurate lesion classification. 41 In research on ablation techniques using ethanol and paclitaxel, contraindications for ablation mainly include malignant mucinous lesions and pseudocysts.42,43 Confocal laser endomicroscopy effectively detects malignant cystic lesions, with malignant IPMNs exhibiting increased papillary epithelial width and darkness 19 and malignant MCNs presenting as dark aggregates of neoplastic cells. 18 Accordingly, nCLE could play an expanding role as an intra-procedural tool to assist in identifying cystic lesions suitable for concomitant ablation.
In terms of AEs, EUS-FNA, cyst biopsy, nCLE examination, and LA may all predispose patients to postoperative pancreatitis. Our study also observed a higher incidence (13.8%) of postoperative AEs. Two patients (6.9%) developed post-procedural acute mild pancreatitis. One of them was ultimately diagnosed with pancreatic pseudocyst and reported upper abdominal pain 3 months before the procedure. The other patient had a cyst located at the pancreatic uncinate process. For the nCLE examination, a 19-G needle was introduced transgastrically, traversing a relatively large segment of the pancreatic parenchyma, which may have contributed to the development of postoperative pancreatitis in this patient. To mitigate this risk of procedure-related pancreatitis, we recommend minimizing the duration of nCLE examination and terminating the procedure once typical diagnostic features are observed. In addition, postoperative management may be considered to include acid suppression, enzyme inhibition, and anti-infective therapy.
Whether SCN with essentially no malignant potential should be treated with ablation remains controversial. In our study, LA for SCN was considered in carefully selected circumstances after shared decision-making. During pre-procedural counseling, patients were explicitly informed that observation is an appropriate option for SCN and the potential ablation-related AEs. For some patients, the presence of the cyst can impose a substantial psychological burden, and a surveillance strategy may lead to cumulative and potentially unnecessary economic costs. For others, the presence of symptoms motivated the choice to undergo ablation. Procedure-related pancreatitis is a recognized AE of pancreatic cyst ablation. Our previous data showed that the incidence of AEs related to LA was 3.6%. In our cohort of ablated SCN cases, no clinical acute pancreatitis occurred, although the sample size remains limited. While indications for pancreatic cyst ablation vary across centers and ablation agents, nCLE may provide reliable real-time cyst characterization to support appropriate same-session decision-making. Further studies are warranted to assess the robustness of nCLE-guided intra-procedural classification across different ablation indications and ablation agents.
Our study has certain limitations. First, it was a single-center study with a relatively small sample size, which may limit the power to detect modest differences between modalities. Therefore, larger, multicenter studies are warranted in the future to further evaluate the intraoperative diagnostic efficacy of nCLE. In addition, confocal laser endomicroscopy has inherent limitations when applied in EUS-FNA, as the nCLE probe can only be introduced through the 19-G needle. When cysts are located in the pancreatic head or the uncinate process, and a 19-G needle is not feasible, nCLE examination cannot be performed, restricting its clinical applicability. Finally, post-ablation follow-up was available for only eight patients, and the limited sample size and relatively short follow-up duration may have introduced bias in the assessment of ablation outcomes.
Conclusion
Our study demonstrated that confocal laser endomicroscopy provides favorable real-time diagnostic performance for PCLs. By enhancing diagnostic confidence at the time of the EUS-guided puncture, nCLE may help reduce the need for repeat procedures and thus represents a valuable adjunctive tool to optimize patient management.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261422841 – Supplemental material for Real-time guidance of lauromacrogol ablation for pancreatic cystic lesions using confocal laser endomicroscopy
Supplemental material, sj-docx-1-tag-10.1177_17562848261422841 for Real-time guidance of lauromacrogol ablation for pancreatic cystic lesions using confocal laser endomicroscopy by Xinwei Hao, Xiuxue Feng, Fang Liu, Xiaotong Niu, Longsong Li, Bo Zhang, Yi Yao, Nan Ru, Enqiang Linghu, Yawei Bi and Ningli Chai in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We sincerely thank the endoscopy team of the First Medical Center of Chinese PLA General Hospital for their professional expertise and continuous support throughout the data collection and clinical implementation stages of this research.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
