Abstract
Objective
Pancreatic cancer is a deadly disease that may initially present as acute pancreatitis. This study aimed to examine the survival and outcomes of patients with pancreatic cancer presenting with acute pancreatitis.
Methods
A retrospective review was conducted of alcohol-abstinent adult patients diagnosed with pancreatic cancer between February 2002 and February 2024 at a single tertiary care center. Patients with and without acute pancreatitis were compared with respect to clinical characteristics, tumor features, management strategies, and outcomes.
Results
A total of 284 patients were diagnosed with pancreatic cancer during the study period, of whom 84 met the inclusion criteria. Among these, 24 presented with acute pancreatitis. Sixty control patients without acute pancreatitis were matched for comparison. There were no significant differences between the groups in terms of demographics or comorbidities. Tumor stage, location, and histological subtype were also comparable. Acute pancreatitis was not associated with improved survival (p = 0.252). Patients in the acute pancreatitis group experienced significantly longer hospital stays (p = 0.044) and had a higher rate of sudden cardiac arrest (p = 0.032).
Conclusion
The presence of acute pancreatitis in patients with pancreatic cancer is not associated with earlier diagnosis or improved survival outcomes. Although acute pancreatitis may lead to earlier clinical evaluation, it does not confer a prognostic advantage. Further studies are needed to clarify the clinical significance of acute pancreatitis in pancreatic cancer.
Introduction
Pancreatic cancer (PC) is considered a deadly and aggressive disease, ranking 12th worldwide among the most common cancers. It is the 11th most common cancer in men and women. 1 The asymptomatic nature of early-stage PC profoundly affects its prognosis. Approximately 8% of PC cases are expected to survive for 5 years following diagnosis, and approximately 15% of cases are considered treatable when detected early. 2
PC accounts for approximately 1% of acute pancreatitis (AP) cases, suggesting that AP may serve as an early sign of the disease. 2 This may be explained by tumor occluding the outflow of the pancreatic duct, resulting in the infiltration of digestive enzymes into the pancreatic interstitium, which leads to autodigestion and inflammation.
Only 20% of patients with PC are found to have resectable disease at the time of diagnosis. 3 These findings emphasize the importance of early detection in improving survival rates; however, effective early diagnostic tools remain lacking. The onset of symptoms, including weight loss, jaundice, and abdominal pain, may indicate that the disease has progressed to an advanced stage.
Despite this theoretical association between AP and PC, the prognostic significance of AP in PC remains unclear. This study aimed to evaluate whether presentation with AP is associated with earlier tumor detection or improved outcomes in patients with PC at a tertiary center in Jordan.
Methods
Study design and data collection
This retrospective study included patients diagnosed with PC at King Abdullah University Hospital (KAUH). Data were collected from the medical records of patients managed between February 2002 and February 2024. The study was approved by the ethics committee at Jordan University of Science and Technology (Reference Number: 20230025). All patient information was deidentified, and no direct or indirect identifiers were included in the dataset. This study was conducted in accordance with the Declaration of Helsinki 1975, as revised in 2024, and was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies. 4
We conducted a survival analysis to evaluate the difference in overall survival at 12-month intervals, up to 96 months after diagnosis, comparing patients with and without AP. Data collected included information on patient demographics (age, sex, and body mass index (BMI)), medical history (comorbidities and risk factors), oncological characteristics (tumor stage, location, histological type, and paraneoplastic symptoms), presenting symptoms, and treatment approaches (surgical and nonsurgical interventions). Secondary outcomes included length of hospital stay, admissions to intensive care units (ICUs), use of mechanical ventilation, dialysis requirements, and survival rates.
Inclusion criteria
Adult patients diagnosed with PC (limited to pancreatic ductal adenocarcinoma, including all variants, and pancreatic neuroendocrine tumors confirmed by histopathology) at King Abdullah University Center between February 2002 and 2024 were included. Patients with PC were further categorized into two groups: those with at least one episode of AP (AP group) and those with no episodes (non-AP group). Given the limited number of cases, we increased the number of controls to enhance the statistical power and reliability of the study. Each case of AP was ratio matched with 2–3 control participants (mean ratio, 2.5:1) based on age, sex, and comorbidities. Patients with chronic pancreatitis or those who had undergone endoscopic retrograde cholangiopancreatography (ERCP) within 3 days prior to the onset of AP were excluded. None of our patients had a history of alcohol intake. Other causes of AP, including medications and infections, were also excluded. The severity of AP was mild in all patients.
Definition
The diagnosis of PC was established on the basis of histopathological examination and/or imaging evidence, including computed tomography (CT), ultrasonography (US), magnetic resonance cholangiopancreatography (MRCP), ERCP, and positron-emission tomography (PET). Histological confirmation in patients who did not undergo surgical resection of primary tumor was obtained using endoscopic ultrasound (EUS) guided biopsy, ERCP brush cytology, CT guided percutaneous biopsy, or laparoscopic biopsy.
The diagnosis of AP was defined by the presence of epigastric pain, serum lipase levels equal to three times or more above the permitted limit, and/or radiographic evidence of acute inflammatory processes.
Statistical analysis
Statistical analyses were conducted to compare the two groups using R version 4.3.3 (2024-02-29 ucrt). Chi-square tests and Fisher’s exact tests were employed for categorical variables, whereas t tests and Wilcoxon rank-sum tests were used for continuous variables. The Kaplan–Meier (KM) approach was used to generate survival curves, and the differences in survival curves were assessed via the log-rank test. A p value of less than 0.05 was considered statistically significant.
Results
A total of 284 patients were diagnosed with PC during the study period. Of these, 84 patients were included in the analysis. Twenty-four patients had at least one episode of AP. For each patient with PC and AP (n = 24), up to 2.5 (60/24) patients with PC and without AP (n = 60) were matched and used as a control group for comparison.
Clinical and demographic data
Examination of clinical and demographic data revealed no age disparity at presentation between the AP group (mean age, 59.2 ± 13.1 years) and the non-AP group (mean age, 63.4 ± 12.7 years) (p = 0.175). The sex distribution was comparable, with males comprising 54.2% of patients with AP and 58.3% of patients without AP (p = 0.727). Table 1 shows the clinical and demographic characteristics of both groups. Moreover, no significant differences were observed in the prevalence of comorbidities.
Clinical and demographic characteristics of all patients (84) with pancreatic cancer (PC).

DM: diabetes mellitus; IHD: ischemic heart disease; BMI: body mass index.
Oncologic features
Although there was a trend toward higher cancer stages at diagnosis in the AP group, analysis of the tumor stage distribution did not reveal a statistically significant difference (p = 0.058). Similarly, the location of the tumor (head of the pancreas vs. the body and tail) and the histological subtype (adenocarcinoma or nonadenocarcinoma) were comparable across both groups. Table 2 summarizes the main oncologic features of all patients across different categories.
Comparison of oncological features between patients with pancreatic cancer diagnosed with and without acute pancreatitis.

PC: pancreatic cancer, AP: acute pancreatitis.
Presenting symptoms
Abdominal pain was the most common presenting symptom among all patients and varied significantly between the two groups. It was reported in 95.8% of the AP group compared with 65% of the non-AP group (p = 0.004). Other symptoms, including jaundice, loss of appetite, nausea, and vomiting were comparable between the two groups.
Clinical outcomes
Patient survival was the primary endpoint of this study. AP was not associated with improved survival in patients with PC. Overall, 37.5% of patients with AP survived for more than one year, compared with 25% of patients without AP (p = 0.252). There was no significant difference between the survival curves of the two groups (log-rank test, p = 0.5). Figure 1 shows the KM curves comparing the survival differences between the two groups. In the AP group, there was no difference in survival between patients diagnosed with PC at least 3 months after an AP episode and those diagnosed more than 3 months later (p = 0.9) (Figure 2).

Kaplan–Meier (KM) curve showing the survival difference between the AP and non-AP groups.

Kaplan–Meier (KM) curve showing the survival difference between patients diagnosed with acute pancreatitis ≤3 months before cancer diagnosis and those diagnosed with acute pancreatitis >3 months before cancer diagnosis (Log-rank test, p = 0.9).
Overall, the number of admission days was significantly greater in the AP group than in the non-AP group (chi-square, p = 0.044). Other measures, including ICU admissions, mechanical ventilation, and dialysis requirements, were similar between the groups. Unexpectedly, patients with AP were more likely to die of sudden cardiac arrest (70.8% vs. 45%; p = 0.032). Table 3 summarizes the clinical outcomes of patients with PC who were diagnosed with and without pancreatitis.
Clinical outcomes compared between patients with pancreatic cancer diagnosed with and without acute pancreatitis.
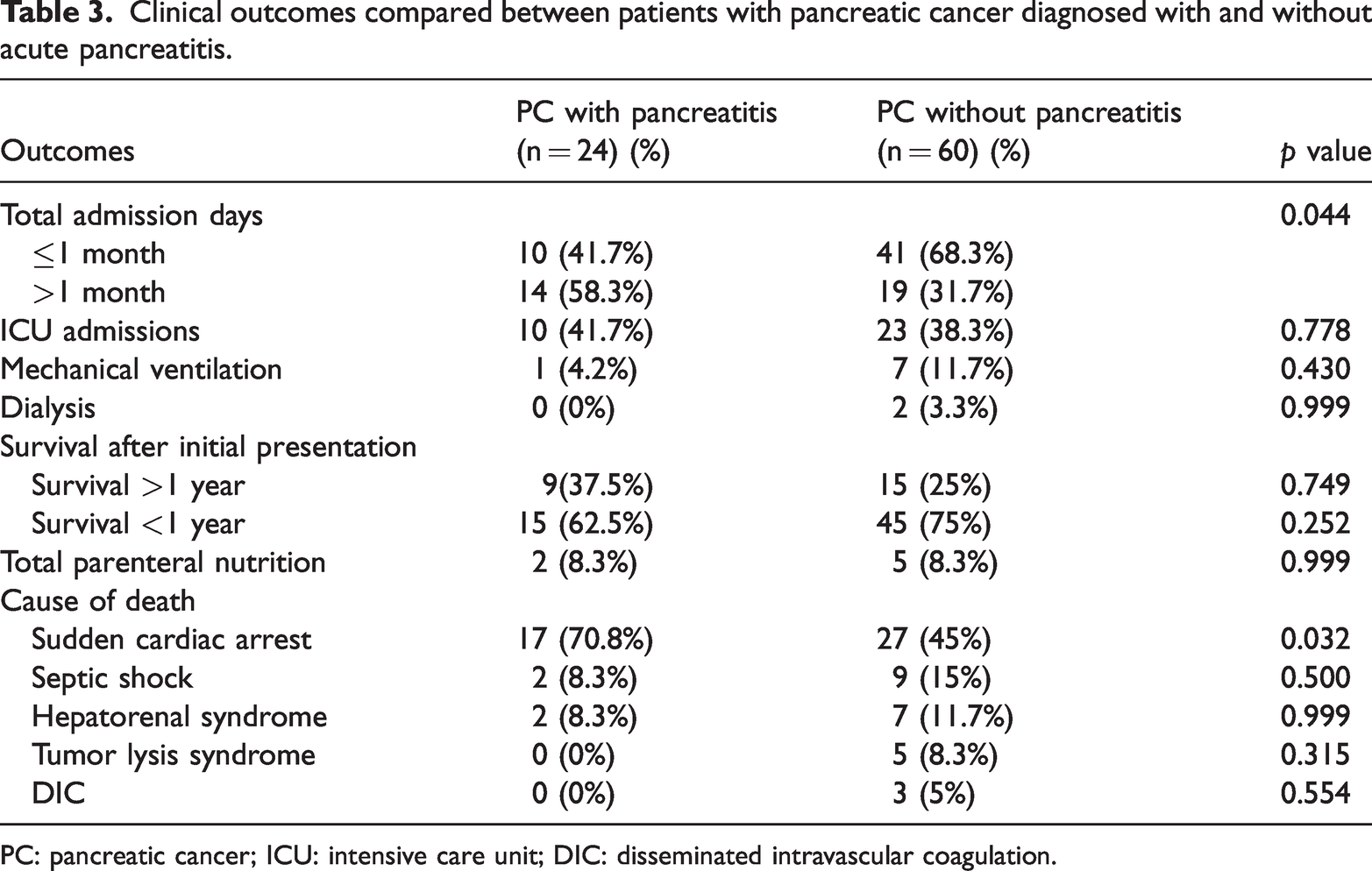
PC: pancreatic cancer; ICU: intensive care unit; DIC: disseminated intravascular coagulation.
Discussion
This study investigated whether AP serves as an early clinical marker of PC and whether it is associated with improved outcomes. Although AP may facilitate earlier clinical presentation, our findings suggest that it does not lead to earlier-stage diagnosis or enhanced survival.
Our results also revealed that patients with AP were more likely to present with abdominal symptoms, experienced longer hospital stays, and had significantly higher rates of sudden cardiac arrest as a cause of death.
Theoretically, AP may serve as an early indicator of underlying PC. This could be explained by the hypothesis that the tumor, particularly in the head of the pancreas, occludes the pancreatic duct lumen, resulting in the infiltration of digestive enzymes into the pancreatic interstitial space rather than only in the duct lumen. This infiltration leads to autodigestion and inflammation.5–7 The resulting abdominal symptoms may prompt patients to seek medical attention and undergo imaging work-up, potentially facilitating earlier detection of PC.
Our study found no association between early tumor stage and AP (Fisher’s, p = 0.058; Wilcoxon rank = 0.360), consistent with the findings of Minato et al., 8 who also reported no association between AP and PC stage. In their study, no significant differences were observed between patients with and without AP in terms of tumor location, clinical stage, resection rate, or survival duration between the two groups. In contrast, other studies have demonstrated a substantial correlation, with the AP cohort being diagnosed at an earlier stage than the non-AP cohort. In an analysis of 50,074 patients with PC from the United States and Denmark, Kirkegård et al. 1 reported that patients with AP had a lower prevalence of metastatic tumors at diagnosis and higher resection frequencies than those without AP. Patients with PC who had AP diagnosed up to 90 days prior to cancer detection had lower tumor stage and better survival. Following surgery, Danish and US patients with AP demonstrated higher 5-year survival rates than those without AP. Similarly, Mujica et al. 7 reported that the 1-year survival rate of patients with PC who initially presented with AP was >25%, whereas the 1-year overall survival rate of patients with PC was 20%. 7
Unsurprisingly, our findings differ from those of previous studies. Adenocarcinoma, the most common histologic subtype, is an exocrine tumor that arises from the ductal epithelium. Approximately 60%–70% of cases involve the head of the pancreas. In patients with cancer, pancreatitis typically results from duct obstruction. Larger tumors increase the likelihood of duct occlusion through the mass itself (infiltration vs. compression), increased locoregional lymph nodes, or the surrounding desmoplastic reaction, which is a significant pathologic feature of PC. This explains the increased probability of more advanced malignancy in patients who present with pancreatitis. However, if cancer cells multiply and extend into the ductal lumen, patients may develop pancreatitis at an early tumor stage. To date, screening for PC in young patients with idiopathic pancreatitis has not been recommended. The PC–AP association will likely require further analysis for better understanding before any recommendations can be made.
The presenting symptoms differed between the two groups, with the AP group showing a significant association with abdominal pain (chi-square, p = 0.004) and a near-significant association with nausea and vomiting (chi-square, p = 0.062). These findings are likely attributable to AP, given its correlation with severe pain. Gapp et al. 9 reported that AP results from premature activation of zymogen and trypsinogen, leading to local pancreatic destruction and the initiation of the inflammatory cascade. Liddle and Nathan 10 suggested that neurogenic pain in AP may be associated with trypsin secretion by the pancreas, potentially modulated by proteinase-activated receptor 2 (PAR2). The enteric nervous system is essential for regulating pancreatic secretion through enteropancreatic reflexes. PAR2 is present on pancreatic neurons and can activate them, resulting in the release of inflammatory neuropeptides. These findings indicate that PAR2 may contribute to pancreatic inflammation. Rosenbaum et al. 11 reported that activation of transient receptor potential (TRP) channels on primary sensory neurons promotes the release of neuropeptides, such as substance P and calcitonin gene-related peptide, in the spinal cord, resulting in pain and localized pancreatic inflammation. Nausea and vomiting may be explained by the hypothesis that a pancreatic tumor exerts pressure on the stomach or the proximal duodenum, thereby resulting in various gastrointestinal symptoms. Additionally, digestive symptoms may arise due to obstruction in the pancreatic duct, which impedes the flow of pancreatic enzymes, or due to alterations in the production of these enzymes.
Hospitalization duration was significantly greater in the AP group than in the non-AP group (chi-square, p = 0.044). Several factors may account for this discrepancy. Patients in the AP group were more likely to be diagnosed at an earlier tumor stage, although the difference was not statistically significant; this may influence the management plan and allow for more aggressive chemotherapy or radiotherapy interventions. Another contributing factor may be the recurrence of AP episodes. Additionally, the higher incidence of complications and paraneoplastic features may require more frequent hospitalization.
Patients in the AP group demonstrated a significant association with cardiac arrest as a cause of death (chi-square, p = 0.032). No associations were observed between tumor lysis syndrome (Fisher’s, p = 0.315) or disseminated intravascular coagulation (DIC; Fisher’s, p = 0.554) as causes of death. Conversely, the non-AP group showed all three associations, although none were statistically significant. The exact underlying mechanism responsible for this phenomenon remains unclear. One potential explanation may involve the hypercoagulable state in PCs and/or electrolyte imbalances. Huang et al. 12 reported that angiotensin II, a component of the renin–angiotensin–aldosterone system (RAAS) in pancreatic tumor cells, upregulates insulin-like growth factor II receptor (IGF-IIR) expression by activating downstream kinases. This activation leads to the degradation of sirtuin 1 (SIRT1) via proteasomes, thereby preventing SIRT1 from deacetylating heat shock transcription factor 1 (HSF1). Increased acetylation of HSF1 compromises its ability to bind to the IGF-IIR promoter region, which results in ventricular hypertrophy and apoptosis, thereby affecting cardiac contractility. Takano et al. 13 reported that insulin-like growth factor 1 (IGF-1) is a peptide with prohypertrophic and anti-apoptotic properties. Khorana et al. 14 suggested that PC may induce various thromboembolic disorders through activation of the coagulation cascade, promoting tumor progression and angiogenesis, which directly facilitate the formation of distal thrombi and ultimately leads to pulmonary embolism. Sancho-Chust et al. 15 demonstrated that pulmonary tumor emboli (PTEs) are rare in patients with cancer, corroborating previous findings. PTE involves tumor cells that initiate coagulation, leading to the formation of microthrombi within the pulmonary arteries. This mechanism alters the pulmonary vasculature, resulting in pulmonary hypertension, which is the principal cause of cor pulmonale and right-sided heart failure.
Study limitations
This study has several limitations. First, it was a retrospective study. Although 284 patients were initially identified in the archives, only 84 had complete data available for evaluation.
Second, the sample size of patients with PC and AP was relatively small to assess risk factors with statistical significance, and this may have affected the generalizability of the results.
Finally, we were unable to control for changes in medical practices, diagnostic standards, and treatment options that changed over time, which may have influenced patient prognoses and mortality rates, as the study spanned more than 20 years (2002–2024).
Conclusion
This retrospective study demonstrates that the presence of AP in patients with PC is not associated with earlier tumor stage at diagnosis or improved survival outcomes. Although pancreatitis may prompt earlier clinical evaluation, it did not translate into a prognostic advantage in our cohort. Notably, patients with AP experienced longer hospitalizations and a higher incidence of sudden cardiac arrest. These findings suggest that although AP is suggestive of underlying malignancy, it should not be assumed to confer a survival benefit. Further multicenter studies with larger sample sizes are warranted to clarify the prognostic significance of pancreatitis in patients with PC.
Footnotes
Acknowledgments
The authors wish to acknowledge that AI-based tools were used solely to assist in language editing and refinement of the manuscript, without influencing the scientific content or interpretation of the results.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by all authors. The first draft of the manuscript was written by AM and MR. All authors commented on previous versions of the manuscript. AM supervised the study. All authors read and approved the final manuscript.
Availability of data and materials
The dataset supporting the conclusions of this article is available from the corresponding author upon reasonable request, following the removal of patient-identifying information.
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request. To protect patient confidentiality, the dataset cannot be shared publicly.
Declaration of conflicting interests
The authors declare no competing interests.
Funding
The authors received no financial or material support.
