Abstract
Background:
The risk of serious infection and active tuberculosis in patients with inflammatory bowel disease (IBD) has not been concurrently evaluated based on the use of anti-tumor necrosis factor (TNF)-α agents versus non-anti-TNF biologics (vedolizumab/ustekinumab) in the Korean population.
Objectives:
We compared the risk of serious infection and active tuberculosis in Korean patients with IBD treated with non-anti-TNF biologics (vedolizumab/ustekinumab) or anti-TNF-α agents.
Design:
This study was a population-based cohort analysis of nationwide administrative claims data.
Methods:
Health Insurance Review and Assessment Service claims data (representing 97% of the South Korean population) from between January 2007 and February 2021 were reviewed, and adults with IBD who initiated vedolizumab/ustekinumab or anti-TNF-α treatment (n = 6123) between 2017 and 2020 were enrolled. Intergroup differences in the risk of serious infection requiring hospitalization/emergency department visits or active tuberculosis during the follow-up period were analyzed.
Results:
In the patients treated with anti-TNF-α agents or vedolizumab/ustekinumab during a mean follow-up of 1.55 ± 1.05 and 0.84 ± 0.69 years, the incidence rates of serious infection were 9.43/100 and 6.87/100 person-years, respectively. Multivariable analysis showed no significant intergroup difference in the risk of serious infection with vedolizumab/ustekinumab or anti-TNF-α treatment; the adjusted relative risk of vedolizumab/ustekinumab compared with anti-TNF-α agents was 0.81 (95% confidence interval 0.46–1.44, p = 0.478). Among patients treated with anti-TNF-α agents and vedolizumab/ustekinumab, the incidence rates of active tuberculosis were 0.87 and 0.37 per 100 person-years, respectively. The relative risk of vedolizumab/ustekinumab compared with anti-TNF-α agents was 0.31 (95% confidence interval 0.07–1.26, p = 0.101). In a subset analysis comparing vedolizumab and ustekinumab with anti-TNF-α agents, similar results were observed.
Conclusion:
In Korean patients with IBD, non-anti-TNF biologics (vedolizumab/ustekinumab) tended to be associated with a lower risk of serious infection or active tuberculosis than anti-TNF-α agents.
Introduction
Biologics are the mainstay therapy for managing patients with inflammatory bowel disease (IBD) refractory to conventional medical treatment, including steroids or other immunomodulators.1–3 Among biologics, monoclonal antibodies against tumor necrosis factor alpha (anti-TNF-α) agents constitute the cornerstone of IBD treatment owing to their proven efficacy. 4 Recently, two novel non-anti-TNF biologics with different mechanisms of action have been developed for treating IBD: (i) vedolizumab, a recombinant humanized IgG1 monoclonal antibody to the homing receptor α4β7 integrin complex, prevents the migration of lymphocytes to the intestinal tissue; and (ii) ustekinumab, a humanized IgG1 monoclonal antibody targeting the p40 subunit of interleukin-12/23, which alleviates intestinal inflammation by inhibiting interleukin-12/23 binding to T cells and natural killer cells.1,4,5 These two drugs have a clinical efficacy similar to that of anti-TNF-α agents in the treatment of IBD.6,7
The increased risk of serious infection, reported in clinical trials and real-world studies,8–12 especially within the first 90 days of treatment initiation, 13 constitutes the biggest drawback of the use of anti-TNF-α agents. Furthermore, as TNF plays an essential role in the host defense against Mycobacterium tuberculosis infection, 14 anti-TNF-α treatment inadvertently increases the risk of tuberculosis, 15 which is the most serious infectious complication associated with anti-TNF-α treatment in tuberculosis-endemic countries. In contrast, vedolizumab and ustekinumab are relatively safe in terms of the risk of serious infection16,17 or active tuberculosis development. 18 However, few studies have compared the risk of infectious complications between anti-TNF-α and vedolizumab/ustekinumab treatment.19,20 In particular, the risk of serious infection and active tuberculosis development in patients with IBD has not been concurrently evaluated based on the use of anti-TNF-α agents versus vedolizumab/ustekinumab in the Korean population.
Therefore, this study aimed to compare the risks of serious infection and active tuberculosis in South Korean patients with IBD treated with non-anti-TNF biologics (vedolizumab/ustekinumab) or anti-TNF-α agents.
Methods
Data source
This population-based retrospective cohort study involved the analysis of data from South Korea’s Health Insurance Review and Assessment Service (HIRA) database, which comprises 97% of the entire South Korean population of approximately 50.0 million who are obligated to enroll in the National Health Insurance program. The accuracy of claims for National Health Insurance was assessed by HIRA, a government-affiliated agency. After reviewing the incurred medical costs and reports from healthcare providers with regard to medical services, the HIRA provides reports of health insurance claims data that include diverse information, including diagnoses according to the International Classification of Diseases, 10th Revision (ICD-10), inpatient/outpatient status, demographics, procedures, drugs, prescription dates and periods, and the tests performed 21 Furthermore, the database provides registration details for rare incurable diseases (RIDs) for several diseases, including Crohn’s disease (CD) and ulcerative colitis (UC). The RID system is highly reliable, as it requires the attending physician to provide additional certification through specified diagnostic criteria, such as histologic examinations, before such registration. 22 The data provided by the HIRA were obtained using anonymized identifiers in accordance with the Act on the Protection of Personal Information, which is maintained by public agencies.
Patients and ethics
We obtained the HIRA claims data of adult patients (age ⩾19 years) with (i) ICD-10 codes for UC (K51.0–51.9) or CD (K50.0–50.9) and (ii) who received biologics treatment with anti-TNF-α agents (infliximab, adalimumab, and golimumab) or non-anti-TNF biologics (vedolizumab/ustekinumab), between January 2007 and February 2021. We first excluded patients (i) who did not have RID registration codes for UC (V131) or CD (V130) and (ii) whose ICD-10 codes for UC or CD were not in the respective principal or subsidiary diagnostic fields. To eliminate the influence of prior use of anti-TNF-α agents, we excluded patients with a history of anti-TNF-α treatment between July and December 2016. Next, we selected patients whose biologic treatment was initiated after 2017, as vedolizumab and ustekinumab were approved for use in South Korea in 2017. 23 Furthermore, to ensure at least a 90-day observation period, we excluded patients for whom biologics were started after December 2020. Of the remaining patients, those with medical comorbidities that could affect the development of serious infection or tuberculosis, such as organ transplantation, were excluded. Finally, we identified 6123 patients with IBD who initiated anti-TNF-α or vedolizumab/ustekinumab treatment between January 2017 and November 2020 (Figure 1).
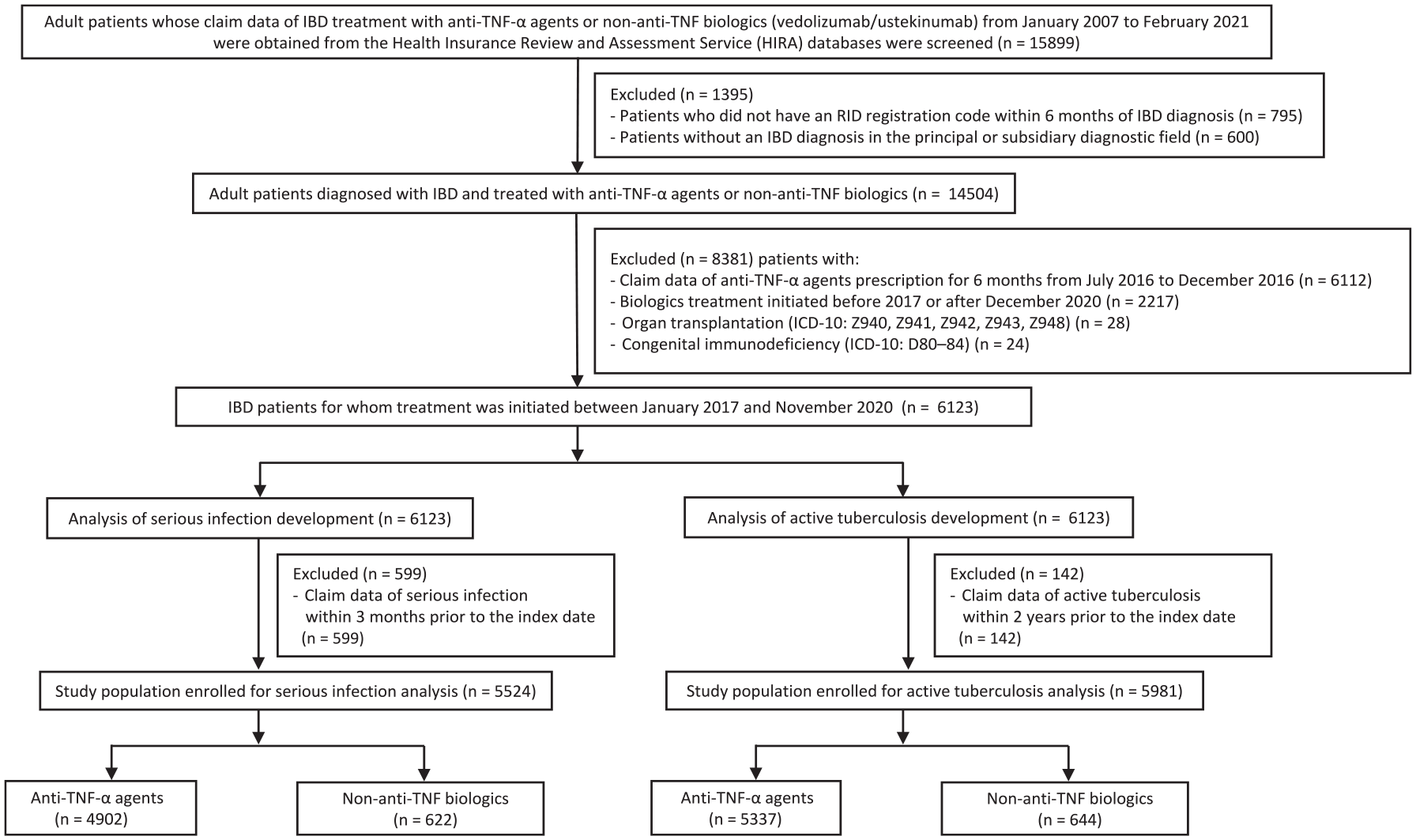
Patient selection flowchart.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 24
Analysis of risk for serious infection
The primary outcome of interest was the development of serious infections, defined as a diagnosis of infection requiring hospitalization or emergency department visit. The detailed diagnosis of serious infections was determined in accordance with a previous study based on ICD-10 codes pertaining to all diagnostic fields of hospitalization or emergency department visits, which were subcategorized according to the infection site: pulmonary, gastrointestinal, skin, urinary tract, ear/nose/throat, musculoskeletal, and other infections 20 Supplemental Table 1 presents the details of the diagnostic codes for serious infections.
The exposures of interest were anti-TNF-α agents and vedolizumab/ustekinumab. As a previous study demonstrated that the risk of serious infection related to treatment with anti-TNF-α agents increased within the first 90 days of treatment and subsequently declined 13 we excluded patients with a history of serious infection within 90 days preceding the initiation of biologics (Figure 1). Figure 2 presents detailed definitions of serious infection outcomes attributed to biologics treatment. After the initiation of biologics-based treatment, patients were considered continuously exposed from the index date, which was defined as the date of the first biologics prescription. If the development of serious infection was noted, it was considered to be associated with biologics use until 90 days after treatment discontinuation [Figure 2(a)]. We considered each event a separate occurrence if the time gap between outcome events [Figure 2(b)] or the interruption period between biologics treatments was longer than 90 days [Figure 2(c)]. Moreover, if biologics were switched (anti-TNF-α agents to vedolizumab/ustekinumab or vice versa), serious infections within 90 days of prescription modifications were attributed to the previous drug treatment [Figure 2(d)].

Study-specific definition of serious infection development attributed to drug therapy and follow-up duration in patients treated with one type of biologics during the study period. (a) Development of the outcome was considered to be associated with biologics use if the outcome occurred during biologics treatment (black star) or within 90 days after discontinuation of biologics treatment (empty star). Outcome occurrence more than 90 days after the withdrawal of biologics was not included in the outcome analysis (asterisk), as the follow-up period was terminated 90 days after the withdrawal of biologics. (b) In the case of multiple-outcome development, if the interval between the outcomes was >90 days, each event was counted as a separate outcome event (upper line). In contrast, if the interval between the multiple outcomes was ⩽90 days, only the first event was counted as a single outcome (lower line). (c) For cases (i) wherein biologics treatment was discontinued for >90 days and then resumed, and (ii) if the outcome was noted in both treatment periods, these events were separately counted as multiple outcome occurrences. (d) Study-specific definition of serious infection analysis in patients who switched to another biologic agent during the study period. Outcome occurrence was attributed to the use of initial biologics if it occurred during the period when the initial biologics were used (black star) or within 90 days after discontinuation of the initial biologics treatment (empty star). The follow-up period was defined as up to 90 days after the discontinuation of the initial biologics treatment. Therefore, events that occurred in the period after switching to another biologic were not included in the main analysis, irrespective of the time of development (asterisk).
In the analysis of serious infection risk, the follow-up duration was determined as lasting until (i) 90 days after the discontinuation of biologics, (ii) 90 days after switching the biologic treatment (anti-TNF-α agents to vedolizumab/ustekinumab or vice versa), or (iii) the end of the follow-up period, whichever came first.
Analysis of active tuberculosis development
Another primary outcome was the development of active tuberculosis. A tuberculosis diagnosis was defined as a patient with a tuberculosis-based claim (ICD-10 codes A15–19) who had received a prescription for any antituberculosis drug (isoniazid, rifamycin, ethambutol, pyrazinamide, prothionamide, cycloserine, para-aminosalicylic acid, levofloxacin, moxifloxacin, bedaquiline, linezolid, or delamanid) at least once within 90 days from the time of diagnosis25,26 As patients with active tuberculosis are usually treated in the outpatient department, the tuberculosis definition did not require hospitalization or emergency department visits.
A history of tuberculosis is a significant risk factor for subsequent tuberculosis development27,28 and the likelihood of relapse is significantly higher within the first 2 years after the completion of tuberculosis treatment. 29 Therefore, for the analysis of active tuberculosis development, we excluded patients who were diagnosed with active tuberculosis within 2 years preceding the first date of their biologics prescriptions (Figure 1). Figure 3 presents the definition of active tuberculosis onset with biologics treatment. Briefly, tuberculosis was considered to be related to biologics treatment if the tuberculosis developed during treatment with biologics or within 90 days of the discontinuation of biologics treatment [Figure 3(a)]30,31 Additionally, only the first event was considered a study outcome, whereas subsequent events associated with initial or switched biologics were not included in our analysis [Figure 3(b)].

Study-specific definition for active tuberculosis development attributed to drug therapy and follow-up duration. (a) Development of active tuberculosis was determined to be related to biologics use if the outcome occurred during the biologics treatment (black star) or within 90 days after withdrawal of biologics (empty star). If active tuberculosis occurred ⩾90 days after the withdrawal of biologics, the occurrence was not included in the outcome analysis (asterisk). (b) In the case of multiple-outcome development, only the first event of active tuberculosis was counted as the single outcome associated with the prescribed biologics (black star), whereas the subsequent development of active tuberculosis was not included, irrespective of whether it occurred during the use of initial or switched biologics (asterisk).
For the active tuberculosis analysis, the follow-up duration was defined as until (i) the study outcome occurred, (ii) 90 days after the discontinuation or switching of biologics, or (iii) the end of the follow-up period, whichever came first.
Covariates
A number of covariates were extracted from the HIRA data, including age, sex, use of steroids or immunomodulatory drugs, comorbidities, and the Charlson Comorbidity Index (CCI), which was measured using the ICD-10 code diagnoses for several major comorbidities, as previously defined. 32 The presence of comorbidities and CCI measurements were assessed based on a time point within 1 year prior to the index date of IBD diagnosis.
Statistical analysis
Categorical variables were compared using McNemar’s test (two categories) and the Test of Marginal Homogeneity (three categories). Continuous variables were analyzed using the Wilcoxon signed-rank test. We counted the number of episodes of serious infection and active tuberculosis outcomes and calculated the incidence rate per 100 person-years of exposure period for non-anti-TNF biologics versus anti-TNF-α agents. Infection rates were calculated as follows:
The relative risk (RR) of vedolizumab/ustekinumab use compared with the use of anti-TNF-α agents was analyzed, and the 95% confidence interval (CI) was determined by Poisson regression based on the crude rates. Multivariable analysis using Cox proportional hazards modeling was performed after adjusting for covariates, such as age and sex. Statistical analyses were performed using SAS EG version 7.1 (SAS Institute, Cary, NC, USA) and R version 3.5.1 (R Foundation for Statistical Computing, Vienna, Austria), and p-values <0.05 were considered statistically significant.
Results
Patients in the serious infection analysis
For the serious infection analysis, we selected 5524 patients with IBD after excluding 599 with a history of serious infection (Figure 1).
Table 1 shows the baseline characteristics of the patients. They were predominantly male (69.3%), with a median age of 27 (interquartile range, 21–41) years. CD and UC were diagnosed in 3339 (60.4%) and 2185 (39.6%) patients, respectively. Anti-TNF-α treatment was initiated in 4902 (88.7%) patients; infliximab was most frequently prescribed (57.9%), followed by adalimumab (35.6%) and golimumab (6.5%). The remaining 622 patients (11.3%) received vedolizumab (n = 284) or ustekinumab (n = 338). Statistically significant differences in the baseline characteristics were noted between the two groups in terms of the type of IBD, the proportion of patients treated with steroids or thiopurine, and CCI (Table 1).
Baseline characteristics of patients with IBD and serious infection stratified by biologics-based treatment.
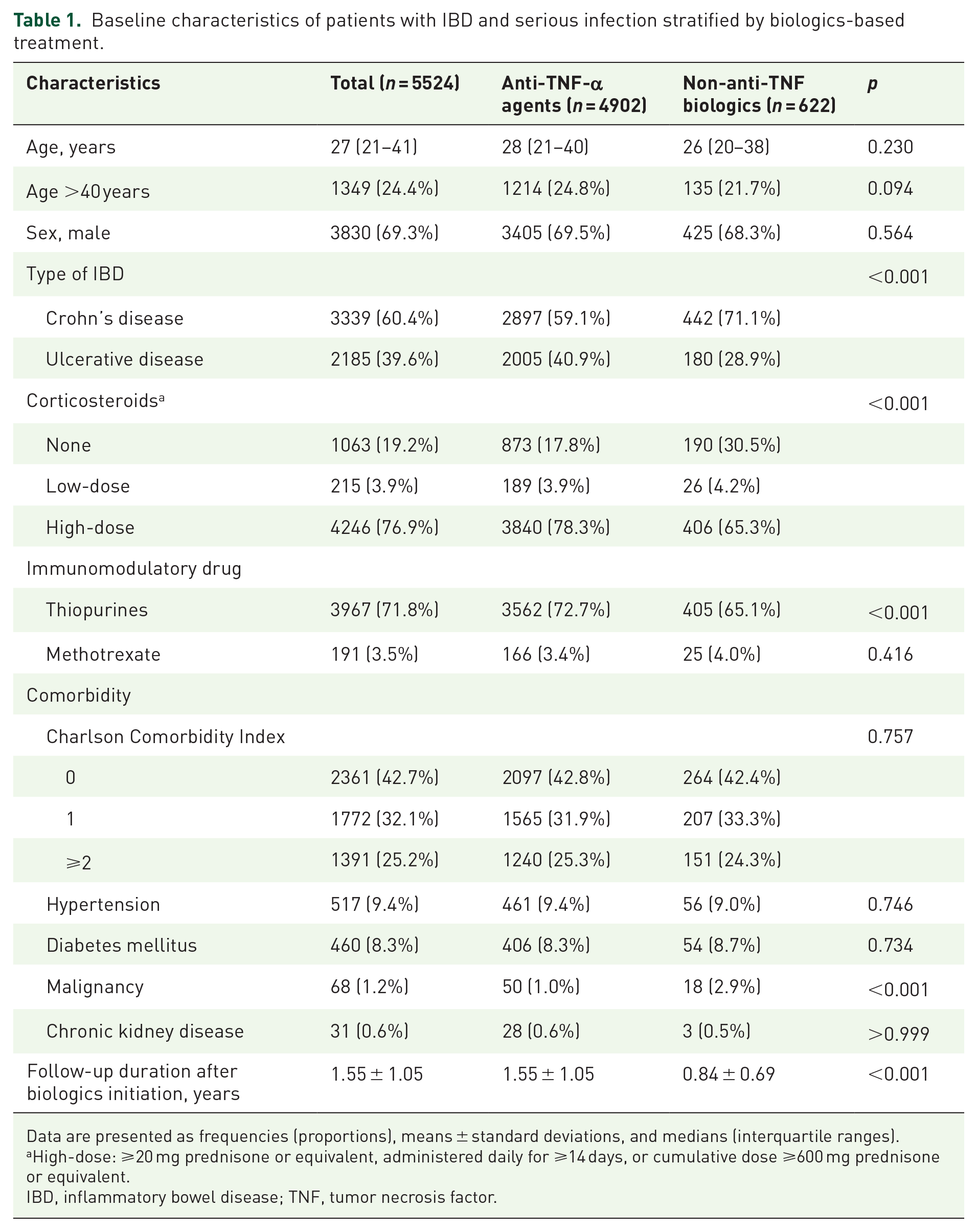
Data are presented as frequencies (proportions), means ± standard deviations, and medians (interquartile ranges).
High-dose: ⩾20 mg prednisone or equivalent, administered daily for ⩾14 days, or cumulative dose ⩾600 mg prednisone or equivalent.
IBD, inflammatory bowel disease; TNF, tumor necrosis factor.
Serious infection development according to the biologics used
Of the 4902 patients who received anti-TNF-α agents, 718 developed serious infection during a mean follow-up period of 1.55 ± 1.05 years, with an incidence rate of 9.43 per 100 person-years. Of the patients who were prescribed non-anti-TNF biologics, 36 developed serious infections during a mean follow-up period of 0.84 ± 0.69 years, with an incidence rate of 6.87 per 100 person-years in the non-anti-TNF biologics group. Univariate analysis showed that treatment with vedolizumab/ustekinumab was not associated with a higher risk of serious infection relative to anti-TNF-α agent use (RR, 0.73; 95% CI 0.40–1.34, p = 0.308). Several covariates, including age, sex, comorbidities, CCI, and steroid use, were associated with serious infections in univariate analyses (Supplemental Table 2). After adjustments for these confounding factors, no significant intergroup difference in serious infection development was noted; the adjusted RR of serious infection for non-anti-TNF biologics for serious infection versus anti-TNF-α agents was 0.81 (95% CI 0.46–1.44, p = 0.478, Table 2).
Risk of serious infection of non-anti-TNF biologics compared to anti-TNF-α agent treatment in patients with inflammatory bowel disease.
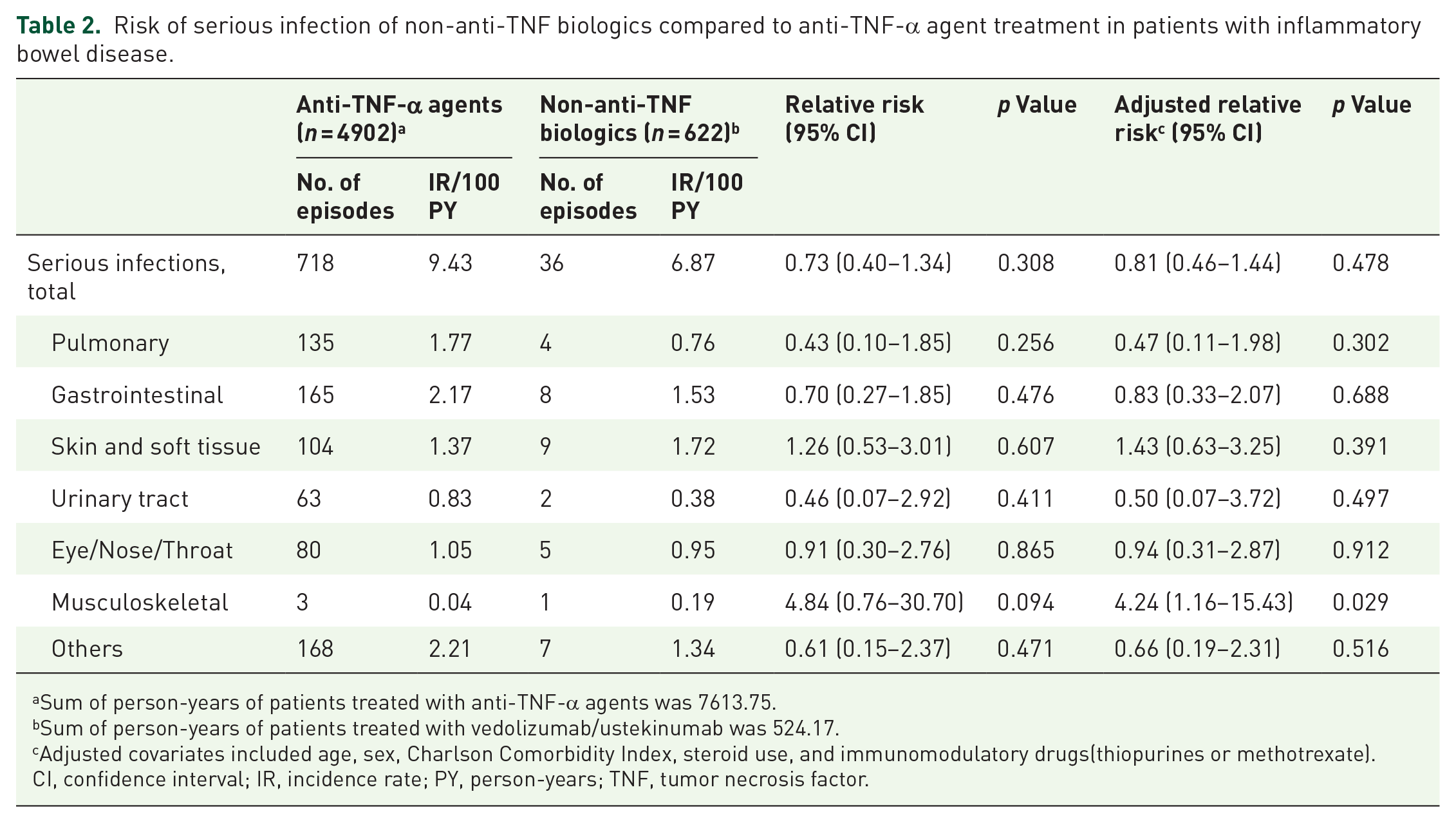
Sum of person-years of patients treated with anti-TNF-α agents was 7613.75.
Sum of person-years of patients treated with vedolizumab/ustekinumab was 524.17.
Adjusted covariates included age, sex, Charlson Comorbidity Index, steroid use, and immunomodulatory drugs(thiopurines or methotrexate).
CI, confidence interval; IR, incidence rate; PY, person-years; TNF, tumor necrosis factor.
Supplemental Table 3 presents a detailed classification of serious infections according to infection site subgroups. We found similar results in a subset analysis that compared the risk of serious infections for vedolizumab and ustekinumab separately with anti-TNF-α agents (Supplemental Tables 4 and 5).
Patients included in the active tuberculosis analysis
For the analysis of active tuberculosis development, 5981 patients with IBD were selected after excluding 142 patients with a history of active tuberculosis (Figure 1).
Table 3 presents the baseline characteristics of patients with active tuberculosis. In this group, the median age was 28 years (interquartile range, 21–41 years), and male patients predominated (68.9%). CD was diagnosed in 58.9% of patients. Anti-TNF-α agents were selected as biologic therapy in 5337 patients (89.2%) [the most frequently prescribed drug was infliximab (59.3%), followed by adalimumab (34.6%), and golimumab (6.1%)], and the remaining 644 (10.8%) patients received vedolizumab (n = 294) or ustekinumab (n = 350). Several characteristics, such as the type of IBD, use of thiopurine, and CCI, showed significant intergroup differences.
Baseline characteristics of patients with IBD and active tuberculosis stratified by biologics treatment.
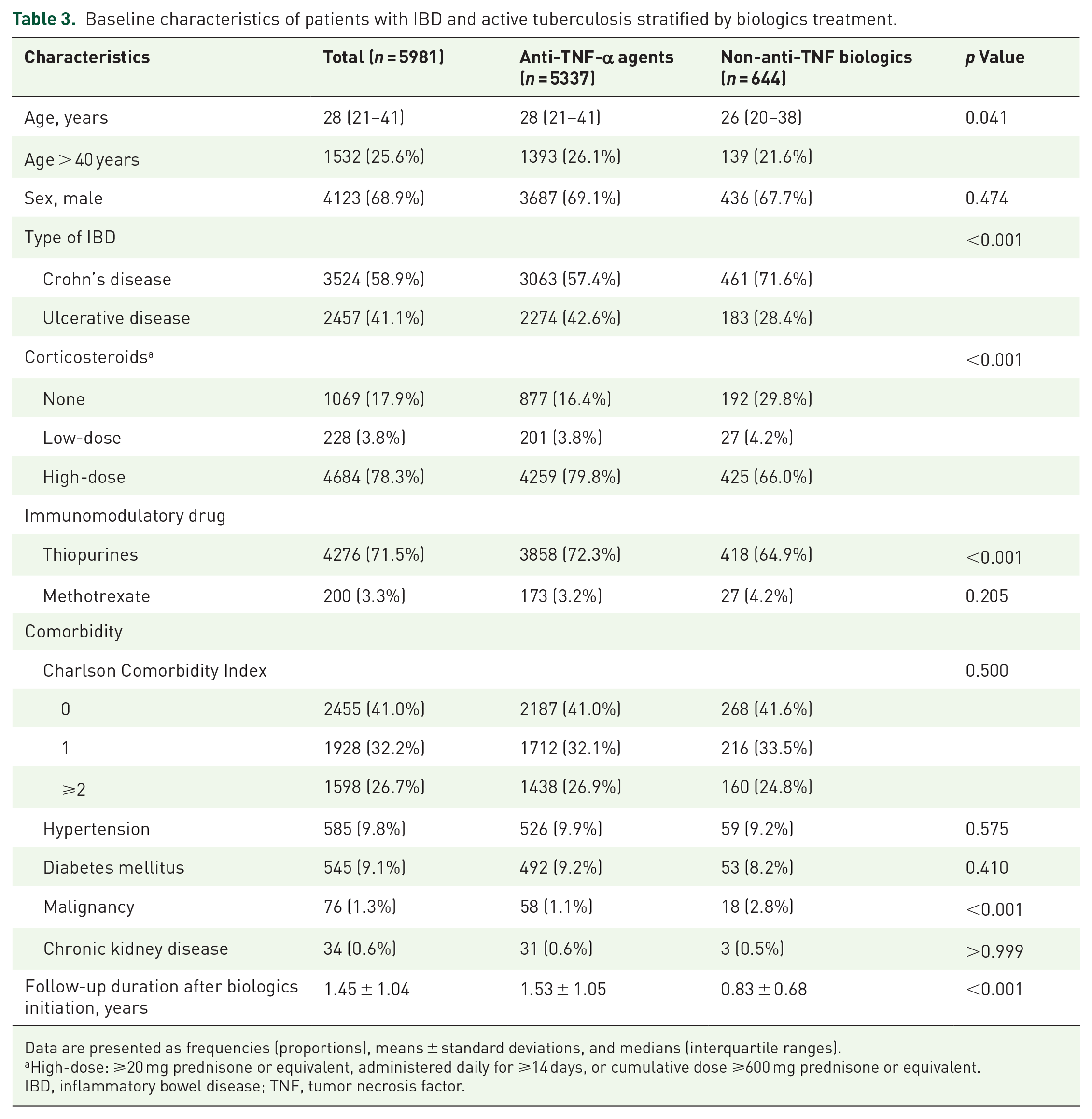
Data are presented as frequencies (proportions), means ± standard deviations, and medians (interquartile ranges).
High-dose: ⩾20 mg prednisone or equivalent, administered daily for ⩾14 days, or cumulative dose ⩾600 mg prednisone or equivalent.
IBD, inflammatory bowel disease; TNF, tumor necrosis factor.
Analysis of active tuberculosis development
During a mean 1.53 ± 1.05 years of follow-up, 71 of 5337 patients treated with anti-TNF-α agents were diagnosed with active tuberculosis (Table 4). Moreover, the development of active tuberculosis was noted in 2 of 644 patients who received non-anti-TNF biologics during a mean follow-up of 0.83 ± 0.68 years. The incidence rates of active tuberculosis were 0.87 per 100 person-years and 0.37 per 100 person-years, respectively. The univariate analysis revealed that the risk of active tuberculosis did not differ according to the biologics used; the RR of non-anti-TNF biologics compared with anti-TNF-α agents was 0.31 (95% CI 0.07–1.26, p = 0.101). Supplemental Table 6 shows the baseline characteristics of the two patients who developed active tuberculosis during vedolizumab/ustekinumab treatment. Supplemental Tables 7 and 8 present the results of the subset analysis investigating the risk of active tuberculosis associated with vedolizumab/ustekinumab separately from the risk of active tuberculosis associated with anti-TNF-α agents.
Risk of tuberculosis with vedolizumab/ustekinumab relative to risk with anti-TNF-α agent treatment in patients with inflammatory bowel disease.

Sum of person-years of patients treated with anti-TNF-α agents was 8155.87.
Sum of person-years of patients treated with vedolizumab/ustekinumab was 535.22.
CI, confidence interval; IR, incidence rate; PY, person-years; TNF, tumor necrosis factor.
Discussion
We conducted a population-based cohort analysis of nationwide administrative claims data of patients with IBD in South Korea and evaluated whether the risk of serious infection and active tuberculosis differed with non-anti-TNF biologics (vedolizumab/ustekinumab) versus anti-TNF-α agent use. To our knowledge, this was the first study to investigate this issue in Korean IBD patients. The key findings were as follows: (i) the risk of serious infections in patients with IBD treated with non-anti-TNF biologics showed a lower tendency than in those who received anti-TNF-α agents, and (ii) the risk of tuberculosis tended to be lower in those treated with non-anti-TNF biologics.
Several previous studies have reported the rates of serious infection associated with anti-TNF-α agents and non-anti-TNF biologics. Singh et al. found that vedolizumab was associated with a lower risk of serious infections than anti-TNF-α agents in patients with UC, whereas no difference in risk was noted in patients with CD in an analysis of data from a US administrative claims database 20 Recently, a study that assessed data from a US Medicare administrative claims cohort of older patients found that treatment with vedolizumab was associated with a lower risk of infections that necessitated hospitalization than treatment with anti-TNF-α agents. 33 Another study that analyzed data from a national commercial health insurance plan in the United States from 2008 through 2019 showed that the use of ustekinumab led to a significantly lower risk of infection. 19 We also found that there was a lower tendency toward the development of serious infection in patients who received non-anti-TNF biologics than in those who received anti-TNF-α agents.
Notably, among serious infections, the risk of musculoskeletal infection development differed significantly between the non-anti-TNF biologics and anti-TNF-α agent groups (Table 2), which was attributed to the higher rate of infective myositis in those treated with non-anti-TNF biologics (Supplemental Table 3). In the study cohort, one patient treated with vedolizumab had infective myositis, and the detailed clinical characteristics and medication history are presented in Supplemental Table 9. Although there have been some previous case reports on the development of myositis after the use of vedolizumab34,35 the insights drawn from the present study alone are insufficient to determine whether vedolizumab treatment increases the risk of infective myositis. Further research is required to clarify this issue.
As TNF plays a pivotal role in the formation and maintenance of granuloma integrity, 14 there is an increased risk of active tuberculosis with anti-TNF-α agent use that varies by geographical variation in tuberculosis burden 36 South Korea has a intermediate incidence of tuberculosis (36/100,000 per year in 2021), 37 so patients with IBD in South Korea who are treated with anti-TNF-α agents face a higher risk of tuberculosis than those in Western countries.38,39 In contrast, as the mechanism of action of ustekinumab is indirectly related to the inhibition of TNF and the drug target of vedolizumab is not associated with TNF, 40 the risk of tuberculosis associated with these drugs appears to be theoretically lower than that associated with the anti-TNF-α agents. Previous studies have reported that the risk of active tuberculosis is considerably lower in patients with IBD who receive vedolizumab or ustekinumab18,41,42 and the rate of active tuberculosis also tended to be lower in patients treated with vedolizumab or ustekinumab in our study cohort. We should also consider that immunosuppressive agents such as steroids or azathioprine can substantially increase the risk of active tuberculosis 43 so the cause of active tuberculosis in our patients might be related to immunosuppressive agents other than non-anti-TNF biologics.
The present study had several limitations. First, we had a small number of patients treated with vedolizumab/ustekinumab compared with those who received anti-TNF-α agents, and the follow-up duration after biologics treatment was relatively short, particularly for those who received non-anti-TNF biologics. These limitations were mainly because we only included patients whose biologics treatment was initiated after 2017. This time point was selected because it was only in 2017 that vedolizumab and ustekinumab were approved for the treatment of IBD in South Korea. 23 Additionally, although we excluded patients with IBD who had a history of anti-TNF-α treatment between July and December 2016, it is unclear whether the prior use of anti-TNF-α agents before June 2016 influenced our study outcome. Third, we could not assess unobserved confounders, particularly those related to treatment selection by the attending physician, although we included steroids or other immunomodulator use as surrogate markers of disease activity. Another important confounder concerning biologics selection was that physicians generally tend to choose non-anti-TNF biologics for patients at a higher risk of infection. Nevertheless, the risk of infection tended to be lower in association with non-anti-TNF biologics. Fourth, information regarding the positivity of latent tuberculosis infection screening tests was not available from the nationwide claims data we analyzed. However, according to the regulations of the National Health Insurance system in South Korea, patients with IBD planning to undergo biologic treatment (including both anti-TNF-α agents and non-anti-TNF biologics) were mandated to undergo screening for latent tuberculosis infection as a prerequisite during the study period. Moreover, given that the present study was an administrative claims database–derived study, the subjective or objective disease activity of IBD was not evaluated. Finally, as detailed microbiological data were unavailable in the claims databases, we used ICD-10 codes for serious infections or active tuberculosis. Importantly, definitions encompassing hospitalization-inducing serious infections and active tuberculosis have been validated in previous studies.20,44
In conclusion, we found that treatment with non-anti-TNF biologics was associated with a tendency toward a lower risk of serious infections necessitating hospitalization than treatment with anti-TNF-α agents among Korean patients with IBD. Additionally, there tended to be a lower risk of active tuberculosis development in association with non-anti-TNF biologics than with anti-TNF-α agents.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241265013 – Supplemental material for Comparative risk of serious infections and tuberculosis in Korean patients with inflammatory bowel disease treated with non-anti-TNF biologics or anti-TNF-α agents: a nationwide population-based cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848241265013 for Comparative risk of serious infections and tuberculosis in Korean patients with inflammatory bowel disease treated with non-anti-TNF biologics or anti-TNF-α agents: a nationwide population-based cohort study by Min Jee Kim, Ye-Jee Kim, Daehyun Jeong, Seonok Kim, Seokchan Hong, Sang Hyoung Park and Kyung-Wook Jo in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

