Abstract
Background:
Anti-tumor necrosis factor (TNF) agents are now accepted as the first-line medical treatment for stricturing Crohn’s disease (CD). However, data are lacking about the effectiveness of advanced therapies after anti-TNF failure.
Objectives:
To compare the effectiveness of ustekinumab and a second-line anti-TNF agent after failing to respond to a first-line anti-TNF for a symptomatic stricturing CD.
Design:
Multicenter retrospective study.
Methods:
We included consecutive adult patients with CD treated with ustekinumab or anti-TNF for symptomatic stricture (confirmed on imaging or endoscopy) after prior failure of one anti-TNF for the current stricture. Short-term endpoints were symptomatic remission (composite endpoint) at 6 months, defined as no abdominal pain, no vomiting, no food restriction, no sub-occlusive episode, no steroid, no surgery, and no drug discontinuation or symptomatic response (same definition except for tolerating mild abdominal pain). Long-term endpoints were time to drug discontinuation for treatment failure and to bowel damage progression. The comparisons were performed after using propensity score analysis.
Results:
Seventy patients were included (34 on ustekinumab, 36 on anti-TNF). After propensity score adjustment, symptomatic remission at 6 months was achieved in 73.9% of patients receiving ustekinumab compared to 42.7% in the anti-TNF group (p = 0.24), while symptomatic response was observed in 84.0% and 49.5%, respectively (p = 0.13). Predictors of remission in the ustekinumab group were prior bowel resection (p = 0.001) and stricture length <12 cm (p = 0.042). The risk of treatment discontinuation (hazard ratio (HR) = 2.86 (1.33–6.15); p = 0.008) and bowel damage progression (HR = 3.90 (1.64–9.24); p = 0.003) were higher in the anti-TNF group.
Conclusion:
Ustekinumab appears more effective than a second-line anti-TNF in patients with symptomatic stricturing CD after failing to respond to a first-line anti-TNF.
Plain language summary
In the USTEKNOSIS study, we showed that ustekinumab appears more effective than a second anti-TNF in patients with symptomatic stricturing Crohn’s disease after failing to a first anti-TNF.
Introduction
Crohn’s disease (CD) is a chronic inflammatory bowel disease (IBD) characterized by transmural lesions that can evolve to progressive bowel damage, including strictures or penetrating complications such as fistulas and abscesses, leading ultimately to bowel resection.1,2 Stricture is more common in the small bowel than in the colon.3,4 While stricture is observed between 5% and 10% of CD at the time of diagnosis, up to 40% of the patients can experience a stricturing phenotype over time. 3 Therapeutic management of stricturing CD is also challenging and has widely changed during these last decades. Surgical resection was the reference standard for treating symptomatic CD strictures. Despite the emergence of biological therapies, the use of anti-tumor necrosis factor (TNF) agents was slowed by the hypothesis that this kind of medication could increase the risk of intestinal obstruction suggested by preliminary data.5–7 Fortunately, the TREAT registry demonstrated that taking into account the confounding factors, the patients who received infliximab were not more likely to develop a stenosis. 8 The CREOLE study dramatically modified the practice showing that two-thirds of patients achieved therapeutic success at 6 months using adalimumab therapy and thus could benefit from a medical approach with half of patients avoiding bowel resection at 4 years post-initiation. 9 More recently, the STRIDENT trial confirms that CD strictures are responsive to anti-TNF therapy leading to improved symptoms and stricture morphology in most of the patients. 10 As patients with symptomatic stricturing CD are systematically excluded from clinical trials, data on efficacy of other therapeutic options such as ustekinumab, vedolizumab, JAK inhibitors, and anti-interleukin 23 (IL23) agents are lacking in this situation. The potential relevance of the second line of advanced therapy to treat stricturing CD is attractive, but it is questionable, as the potential efficacy of advanced therapy after failing to respond to at least one anti-TNF agent has not been investigated so far. In addition, whether continuing with anti-TNF agents owing to a higher level of evidence regarding this mechanism of action is a better option than swapping to another class remains unexplored.
Ustekinumab is a biological therapy (IgG1 antibody), targeting the p40 subunit shared by IL-12 and IL-23. It is an attractive option thanks to demonstrated efficacy against placebo, 11 similar data compared to anti-TNF therapy in luminal CD, 12 favorable safety profile, 13 and high level of acceptability by the patients (SC injections with large interval between two injections). 14 However, the efficacy of ustekinumab has never been investigated so far, except for data from a Chinese study observing higher rate of steroid-free remission according to CDAI at week 52 in patients treated with ustekinumab compared to those receiving infliximab (69.8% vs 51.8%). 15
In the USTEKNOSIS study, we aimed to compare the effectiveness of ustekinumab versus a second-line anti-TNF agent in patients with symptomatic stricturing CD who have previously failed a first-line anti-TNF therapy for stricturing CD.
Methods
Ethical considerations
The study was approved by the local Ethics Committee (IRB00013412, “CHU de Clermont Ferrand IRB #1, numéro IRB 2022-CF032”), compliance with the French policy of individual data protection. All authors had access to the study data and reviewed and approved the final manuscript.
Study design and inclusion criteria
We conducted a retrospective multicenter study in two expert IBD centers in France (CHU Clermont-Ferrand and CHU Amiens) according to STROBE guidelines (Supplemental Appendix 1). From comprehensive pharmacy databases of patients receiving biological therapies, we included all consecutive CD patients treated with ustekinumab or a second-line anti-TNF (adalimumab or infliximab) for symptomatic stricturing CD between January 2012 and June 2022, and who had been exposed to at least one anti-TNF for the current stricture. Symptomatic stricturing CD was defined as the presence of clinical symptoms such as abdominal pain, nausea, vomiting, or hospitalization due to (sub-) occlusive symptoms and confirmed using cross-sectional imaging (intestinal ultrasound, CT scan, or magnetic resonance enterography) 16 or endoscopy. 17 Patients who underwent surgery for the stricture after failure of the last anti-TNF, but before inclusion, were not eligible.
Data collection
Study data were collected and managed using REDCap electronic data capture tools hosted at the Centre Hospitalier Universitaire de Clermont. 18 Data were collected retrospectively by reviewing computerized patients’ records.
The following data were collected at baseline: gender, age, smoking status, age, CD duration, CD location, behavior, and prior history of bowel resection. The type of anti-TNF failure was classified as primary failure, secondary loss of response, or intolerance. Disease activity at the time of inclusion was assessed based on clinical data (presence and severity of obstructive pain categorized as none, mild, moderate, or severe), obstructive symptoms, nausea, vomiting, dietary restrictions, hospitalization, and biological parameters (level of C-reactive protein (CRP) or fecal calprotectin). Morphological characteristics were also retrieved from imaging analyses. During the follow-up (ending at drug discontinuation, bowel resection, or the date of the last follow-up for patients without events), data from routine medical visits were collected up to the end of the study, including patient-reported outcomes (abdominal pain and stool frequency), symptoms of bowel obstruction, nausea, vomiting, dietary restrictions, CD-related hospitalizations, steroid initiation, serum CRP levels, fecal calprotectin levels, bowel resections, and imaging data (when available) to assess bowel damage progression.
Investigated drugs
Two groups were considered in this study based on the drug delivered at baseline: either ustekinumab or a second-line anti-TNF (infliximab or adalimumab). In our centers, therapeutic escalation is standardized and based on objective markers of inflammation but not on proactive therapeutic drug monitoring, especially for prior anti-TNF therapy. Patients treated with ustekinumab received one intravenous (IV) infusion (260 mg if <55 kg, 390 mg if 56–85 kg, 520 mg if >85 kg) followed by a subsequent subcutaneous injection of 90 mg every 8 weeks from week 8. Therapeutic escalation to 90 mg every 4 weeks was allowed and based on the physician’s judgment. Infliximab was administered with IV infusions of 5 mg/kg at week 0, week 2, and week 6 and then every 8 weeks. Dose escalation to 10 mg/kg as well as interval shortening was left to the physician’s discretion. Adalimumab was given according to its label using subcutaneous injections: 160 mg at week 0, 80 mg at week 2, and 40 mg every other week. Therapeutic escalation to 80 mg every other week, 40 mg every week, or 80 mg every week was authorized and based on the physician’s judgment. The use of concomitant immunosuppressive therapy was based on the physician’s choice.
Endpoints
Regarding the effectiveness at 6 months, symptomatic remission was defined as the absence of all of the following symptoms or criteria: abdominal pain (subscore from CDAI = 0), vomiting, food restrictions, sub-occlusive episodes, steroid use, bowel resection, endoscopic dilation, or treatment discontinuation. Symptomatic response was defined as no or mild abdominal pain (graded ⩽ 1 according CDAI definition), and absence of vomiting, food restrictions, sub-occlusive episodes, steroid use, bowel resection, endoscopic dilation, or treatment discontinuation.
Regarding long-term effectiveness, we assessed drug discontinuation for failure-free survival and bowel damage progression-free survival. Bowel damage progression was considered in the case of bowel resection, new stricture or fistula, or worsening of preexisting stricture (increase in prestenotic dilation on imaging or occurrence of major obstructive symptoms) or fistula (new abscess or new fistula tract) as previously defined in several studies.19–21
Sample size calculation
The sample size was estimated to provide satisfactory power to detect long-term effectiveness, especially progression of bowel damage, with a hazard ratio (HR) greater than 2. For a two-sided type I error of 5% and power greater than 80%, 29 patients in each arm were required to confirm this assumption. Taking into account the primary analysis conducted with propensity score weighting, we estimated that at least 32 patients were needed in each group.
Statistical analysis
Patients’ characteristics were expressed as mean and standard deviation or median and interquartile range for continuous data. The assumption of normality was assessed by using the Shapiro–Wilk test. As indication bias is frequent in retrospective studies (the choice of treatment is generally influenced by patients’ characteristics), we used a propensity score to balance for these differences and recreate, as much as possible, the gold standard of a randomized trial. Inverse probability of treatment weighting (IPTW) was used by assigning each participant an inverse weighting of the probability of receiving ustekinumab and anti-TNF, estimated by the propensity score, to make ustekinumab and anti-TNF groups comparable. The validity of the matching was tested by analyzing the standardized differences. Considering the characteristics of the subjects at baseline and clinical relevance, the propensity score model included the following variables: gender, age, CD duration, CD location, prior bowel resection, concomitant immunosuppressant or steroids, number of strictures, length of stricture, CD severity, number of prior biologics, type of anti-TNF failure, and adalimumab versus infliximab as first-line anti-TNF agent. The validity of the weighting was tested by analyzing the standardized differences (|d|), with |d| >0.2 considered to be an imbalance.
Then, the comparisons between groups for continuous variables were performed using Student’s t test or Mann–Whitney test when the assumptions of t test were not met. The homoscedasticity was studied using the Fisher–Snedecor test. For categorical data, Chi-squared and Fisher’s exact tests were applied.
Among the variables collected at baseline, predictive factors of endpoints were analyzed as aforementioned in patients treated with ustekinumab and those who received anti-TNF separately. Time-to-event censored data (time to bowel damage or drug discontinuation) were estimated using the Kaplan–Meier method. Log-rank test was used to determine prognostic factors associated with the censored outcomes. The proportional-hazard hypothesis was studied using Schoenfeld’s test and plotting residuals. Statistical analyses were performed using Stata 15 software (StataCorp, College Station, TX, USA). All tests were two-sided, with a Type I error set at 0.05.
Results
Baseline characteristics of the population
A total of 70 patients with CD were included, encompassing 34 in the ustekinumab group and 36 in the anti-TNF group (29 patients receiving infliximab and 7 patients treated with adalimumab). Patients’ characteristics at baseline are detailed in Table 1. No significant difference has been found between the two groups except for a higher rate of prior infliximab exposure (52.9% vs 22.2%, p = 0.007) and prior exposure to at least two biologics (41.2% vs 8.3%; p = 0.001, respectively) for patients treated with ustekinumab (Table 1). In addition, a higher proportion of patients had concomitant immunosuppressive therapy among the second-line anti-TNF group (63.9% vs 8.9%, p < 0.001). Of note, we did not find any significant difference between the two groups regarding prior infliximab use for treating stricturing CD (32.3% vs 19.4%, p = 0.21) as well as primary failure to anti-TNF therapy for stricturing CD (20.5% vs 16.7%, p = 0.19; Table 1). Stricture characteristics, including number, length, pre-stenotic dilation, and location, were similar between the two groups, even though a higher rate of concomitant fistula was noted in the anti-TNF group (26.5% vs 3.7%, p = 0.033; Table 1).
Patients’ baseline characteristics.
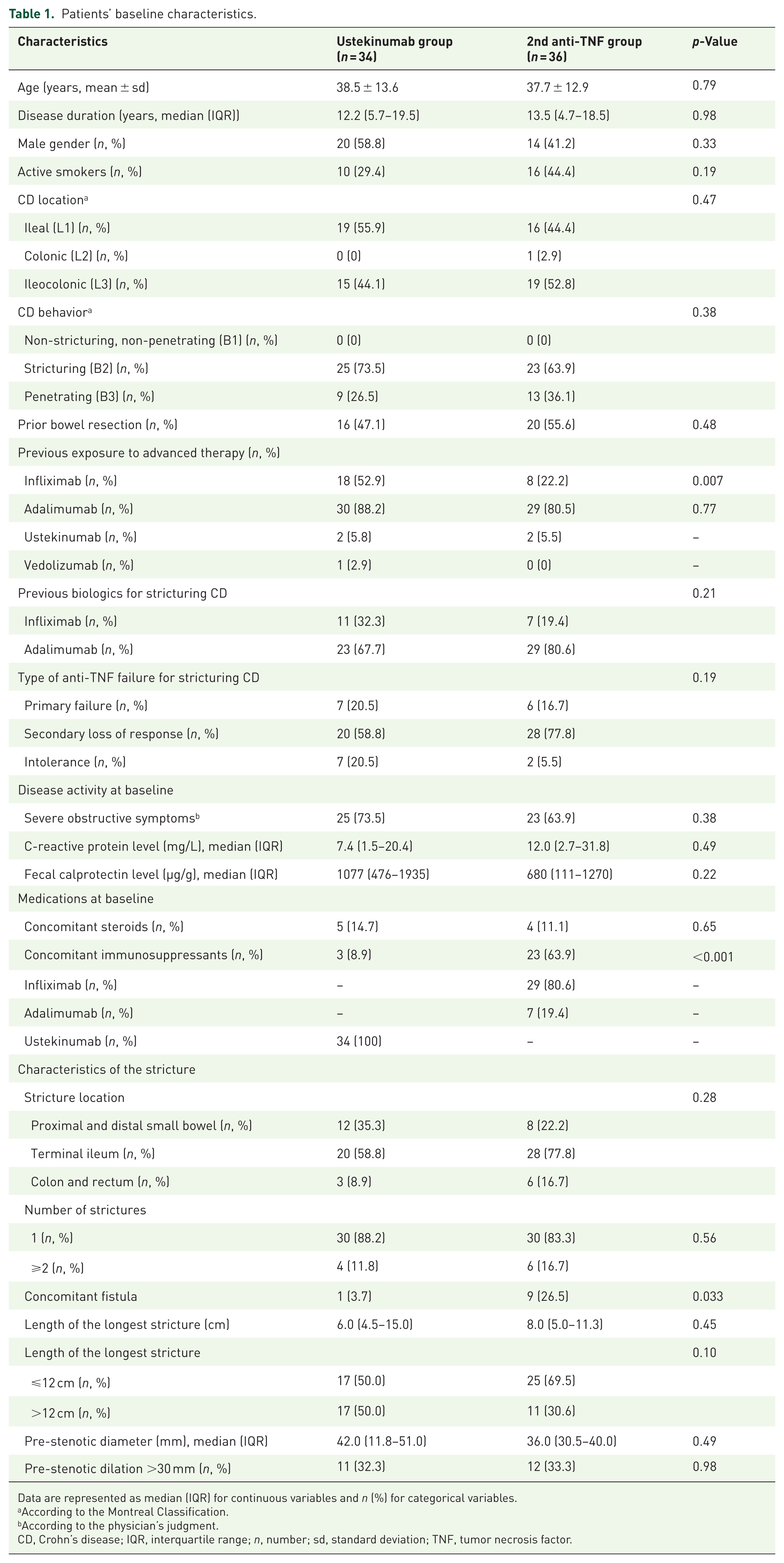
Data are represented as median (IQR) for continuous variables and n (%) for categorical variables.
According to the Montreal Classification.
According to the physician’s judgment.
CD, Crohn’s disease; IQR, interquartile range; n, number; sd, standard deviation; TNF, tumor necrosis factor.
Effectiveness at 6 months
After adjustment for propensity scores, symptomatic remission at 6 months was achieved in 73.9% in the ustekinumab group compared to 42.7% in the anti-TNF group without reaching statistical significance (p = 0.24; Figure 1). In the same way, symptomatic response was observed in 84.0% and 49.5% among patients treated with ustekinumab and a second-line anti-TNF, respectively (p = 0.13; Figure 1).

Symptomatic response or remissiona at 6 months in patients with Crohn’s disease treated with a second-line anti-TNF (red), and ustekinumab (yellow) for symptomatic stricture after failure of at least one anti-TNF agent.
In univariate analyses, prior bowel resection (p = 0.001) and a stricture length <12 cm (p = 0.042) were associated with a higher likelihood of being in symptomatic remission 6 months after starting ustekinumab therapy for symptomatic structuring CD. It has been confirmed in multivariable analysis showing that short structure (>12 cm had a higher likelihood) and prior bowel resection were predictive of a higher likelihood of achieving symptomatic remission at 6 months in patients treated with ustekinumab for stricturing CD. On the other hand, no predictor of effectiveness has been identified in the second-line anti-TNF group.
Long-term effectiveness
After adjustment for potential confounders using propensity score analysis, the patients treated with a second-line anti-TNF agent had a significantly higher risk of treatment discontinuation due to therapeutic failure compared to those treated with ustekinumab (HR = 2.86 (1.33–6.15); p = 0.008; Figure 2). In addition, the risk of bowel damage progression was significantly increased in the anti-TNF group (HR = 3.90 (1.64–9.24); p = 0.003; Figure 3) but was not only driven by the risk of bowel resection (HR = 2.60 (0.70–9.61); p = 0.15).

Kaplan–Meier curves comparing drug discontinuation-free survival between a second-line anti-TNF agent, and ustekinumab in patients with symptomatic stricturing Crohn’s disease previously exposed to at least one anti-TNF (results are adjusted after propensity score analyses and expressed as adjusted hazard ratio).

Kaplan–Meier curves comparing bowel damage progression-free survival between a second-line anti-TNF agent, and ustekinumab in patients with symptomatic stricturing Crohn’s disease previously exposed to at least one anti-TNF (results are adjusted after propensity score analyses and expressed as adjusted hazard ratio).
Discussion
In the real-world evidence USTEKNOSIS study, we reported that ustekinumab showed a clear trend for higher effectiveness than a second-line anti-TNF agent to achieve symptomatic remission or response in patients with stricturing CD who have previously failed a first-line anti-TNF therapy. Our long-term findings also highlight, for the first time, that ustekinumab may be a more effective option than a second-line anti-TNF agent in preventing bowel damage progression and reducing the risk of treatment discontinuation.
To address the question of the best option after anti-TNF failure in patients with stricturing CD, as well as to compare treatment efficacy in general, the best option is a head-to-head randomized controlled trial (RCT). Unfortunately, no RCT has been conducted so far in this situation. An alternative will be network meta-analyses based on clinical trials against placebo, but none have been published so far owing to the lack of placebo-controlled trials in stricturing CD. The last option is a real-world evidence comparative study, such as the USTEKNOSIS study. As the choice of treatment by the physician is generally influenced by deliberate or unaware parameters, addressing the potential issue of indication bias in real-world data is a key point leading to an increased use of propensity score analyses. Although one may think that this type of analysis could require a very high number of patients, it is not mandatory for the IPTW method, as highlighted by a comprehensive review demonstrating that even in the case of small study samples or low prevalence of treatment, IPTW can yield correct estimations of treatment effect as long as the confounders are included within the model. 22 In the USTEKNOSIS study, we paid particular attention to take into account the most relevant confounders related to the patients, the disease, the characteristics of the stricture, as well as prior history with medications. For example, prior infliximab exposure or exposure to at least two biologics was more frequent in the ustekinumab group. This imbalance has been taken into account in the propensity score analysis. However, this disequilibrium could favor second-line anti-TNF rather than ustekinumab. As our data showed better long-term outcomes with ustekinumab, it reinforces our findings.
Short-term results showed a high rate of symptomatic response or remission with ustekinumab (84.0% and 73.9%, respectively). These encouraging results are in line with those reported by a Chinese retrospective study comparing ustekinumab and infliximab in CD, including a subgroup of patients with stenosis. 15 In this substudy, the rate of steroid-free remission was 64.0% in patients treated with ustekinumab. 15 These data suggest that ustekinumab could be an interesting option in patients with stricturing CD. To our best knowledge, this Chinese study is the only other real-world evidence study, except for ours, comparing ustekinumab and anti-TNF in patients with stricturing CD. 15 They reported a higher rate of steroid-free remission according to CDAI in patients treated with ustekinumab compared to those receiving infliximab at week 52 (69.8% vs 51.8%). 15 The data from the USTEKNOSIS study also suggest that ustekinumab could be a better option than a second-line anti-TNF to achieve short-term response or remission. However, this Chinese study can be distinguished from ours regarding several important points. 15 They selected CD patients based on CDAI, which is known as not suitable for stricturing CD. In addition, they pooled bio-naïve and bio-experienced patients without indicating whether the biological therapy was given for stricturing CD. Finally, they did not provide data on long-term follow-up. 15
Until the end of the last century, IBD physicians restricted medical therapy to inflammatory stenosis and considered that fibrosing stricture was associated with therapeutic failure requiring surgery or endoscopic dilation. This differentiation is now considered not exactly reflecting the reality, as it has been shown that inflammation and fibrosis are two overlapping and continuous entities.23–25 In the USTEKNOSIS study, we identified the length of the stricture (<12 cm) and prior history of bowel resection as predictive factors of ustekinumab effectiveness. Previous data on anti-TNF therapy, including the CREOLE study, also identified some morphological characteristics of the stricture as associated with the likelihood of medical treatment success, such as short length (>12 cm), but also direct (decreased stricture luminal diameter) or indirect (pre-stenosis dilation) sign reflecting stricture narrowing.9,23,26 Of note, these morphological parameters of the stricture are known to impact the efficacy of medical therapy independently from the inflammation/fibrosis dichotomy.23,26
Our study is the first to show that ustekinumab could significantly outperform a second-line anti-TNF agent in terms of long-term treatment persistence and prevention of bowel damage progression in patients with stricturing CD. Specifically, patients in the anti-TNF group had a significantly higher risk of treatment discontinuation (HR = 2.86, p = 0.008) and bowel damage progression (HR = 3.90, p = 0.003) compared to the ustekinumab group. Although there is no existing data dedicated to the comparison between cycling anti-TNF agents or swapping to ustekinumab therapy in stricturing CD, our data align with growing evidence suggesting the same trend in patients with luminal non-complicated CD, such as the recent data from the PANIC3 study, demonstrating better drug persistence for ustekinumab over second-line anti-TNF agents in bio-exposed CD patients. 27
Several limitations of this study warrant consideration. First, its retrospective design is inherently subject to recall bias and confounding factors, despite efforts to mitigate these through propensity score adjustment. In addition, even though taking into account potential confounders is more important than the sample size by itself small as aforementioned, 22 our sample size may have limited the power to detect differences in some outcomes, particularly short-term clinical remission. Besides, pharmacokinetic or pharmacodynamic data on anti-TNF would have been of great value, and the absence of systematic combination therapy with an immunosuppressant in the second-line anti-TNF group has to be underlined. Evaluating transmural response assessed by MRI before and after the treatment would have been an interesting additional data, but were not systematically collected. However, USTEKNOSIS was the only study focusing on the literature gap of potential efficacy of medical treatment as second line of biological therapy in stricturing CD, including long-term outcomes, and thus could provide meaningful information to guide the hierarchization of the treatment in this situation.
Conclusion
Ustekinumab seems to be more effective than a second-line anti-TNF agent to treat symptomatic stricturing CD after failure of a first-line anti-TNF. However, these data need to be confirmed by independent prospective data with a larger sample size.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251389476 – Supplemental material for Real-world comparison of effectiveness between ustekinumab and a second-line anti-TNF agent in patients with symptomatic stricturing Crohn’s disease failing to respond to a first-line anti-TNF agent: the USTEKNOSIS study
Supplemental material, sj-docx-1-tag-10.1177_17562848251389476 for Real-world comparison of effectiveness between ustekinumab and a second-line anti-TNF agent in patients with symptomatic stricturing Crohn’s disease failing to respond to a first-line anti-TNF agent: the USTEKNOSIS study by Guillaume Le Cosquer, Bruno Pereira, Arthur Jammet, Mathurin Fumery and Anthony Buisson in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
