Abstract
Background/Aims:
Anti-tumor necrosis factor (anti-TNF) drugs have been the mainstay therapy for moderate to severe inflammatory bowel disease (IBD) over the past 25 years. Nevertheless, these drugs are associated with serious opportunistic infections like tuberculosis (TB). Brazil is ranked among the 30 countries with the highest incidence of TB in the world. This study aimed at identifying risk factors for the development of active TB and describing clinical characteristics and outcomes in IBD patients followed at a tertiary referral center in Brazil.
Methods:
We conducted a retrospective, case–control study between January 2010 and December 2021. Active TB cases in IBD patients were randomly matched 1:3 to controls (IBD patients with no previous history of active TB) according to gender, age, and type of IBD.
Design:
This was a retrospective, case–control study.
Results:
A total of 38 (2.2%) cases of TB were identified from 1760 patients under regular follow-up at our outpatient clinics. Of the 152 patients included in the analysis (cases and controls), 96 (63.2%) were male, and 124 (81.6%) had Crohn’s disease. Median age at TB diagnosis was 39.5 [interquartile range (IQR) 30.8–56.3]. Half of the active TB cases were disseminated (50%). Overall, 36 patients with TB (94.7%) were being treated with immunosuppressive medications. Of those, 31 (86.1%) were under anti-TNF drugs. Diagnosis of TB occurred at a median of 32 months after the first dose of anti-TNF (IQR 7–84). In multivariate analysis, IBD diagnosis older than 17 years and anti-TNF therapy were significantly associated with the development of TB (p < 0.05). After the TB treatment, 20 (52.7%) patients received anti-TNF therapy, and only one developed ‘de novo’ TB 10 years after the first infection.
Conclusions:
TB remains a significant health problem in IBD patients from endemic regions, especially those treated with anti-TNFs. In addition, age at IBD diagnosis (>17 years old) was also a risk factor for active TB. Most cases occur after long-term therapy, suggesting a new infection. The reintroduction of anti-TNFs agents after the anti-TB treatment seems safe. These data highlight the importance of TB screening and monitoring in IBD patients living in endemic areas.
Introduction
Crohn’s disease (CD) and ulcerative colitis (UC) are chronic inflammatory disorders of the gastrointestinal tract, characterized by periods of relapse and remission and risk of progression over time. 1
Despite the evolution of the therapeutic arsenal available for treating inflammatory bowel disease (IBD) in recent years, anti-tumor necrosis factor (anti-TNF) agents have been the mainstay therapy for moderate to severe IBD over the past 25 years. 2 Anti-TNF drugs have shifted the therapeutic paradigm from symptomatic control to deep remission and disease clearance. Moreover, anti-TNF drugs have reduced surgery, hospitalization rates, and disability, improving patients’ health-related quality of life.2,3
Nevertheless, concerns about the safety of anti-TNF therapy remain in clinical practice. Anti-TNFs are associated with an increased risk of malignancies and serious opportunistic infections, especially tuberculosis (TB). 4 TNFα is critical in regulating inflammatory signals in the granuloma formation following primary exposure to Mycobacterium tuberculosis. Its inhibition impairs the host defense mechanisms, increasing the risk of active TB. 5 Indeed, previous studies have demonstrated that anti-TNF-treated patients exhibit an increased risk of TB reactivation compared with the general population.6 –8 Furthermore, in a systematic review, combining anti-TNF agents with either methotrexate or azathioprine resulted in a 13-fold increased risk of TB reactivation compared with anti-TNF monotherapy. 9
Ideally, before any immunosuppression, guidelines have widely recommended screening IBD patients for latent tuberculosis infection (LTBI) with a combination of chest X-ray, tuberculin skin test (TST), and/or interferon-gamma release assay (IGRA), as well as patient clinical data and epidemiological factors. 10 Moreover, recent data have shown that stringent LTBI screening and chemoprophylaxis could significantly decrease the risk of TB reactivation in endemic countries. 11
TB is a global public health problem characterized by high morbidity and mortality worldwide and was the leading cause of death from a single infectious agent until the coronavirus pandemic. 12 Brazil is among the 30 countries with the highest incidence of TB globally, and in 2021, the incidence rate was 32 cases/100,000 inhabitants. 13 In addition, the prevalence of IBD continues to increase worldwide, particularly in newly industrialized countries such as Brazil.14,15 However, the incidence and characteristics of TB among IBD patients remain poorly known in our population.
This study aimed at identifying risk factors for the development of active TB following IBD diagnosis and describing clinical characteristics and outcomes of active TB in IBD patients followed at a tertiary referral center in Brazil.
Methods
Study population
We performed a retrospective, case–control study at the Department of Gastroenterology, Clinics Hospital of the University of São Paulo, School of Medicine, a referral center for treating IBD in Latin America, between January 2010 and December 2021. This timeframe was established due to the incorporation of electronic medical charts in our hospital in 2010.
Definitions of case and control groups
The case group (active TB) comprised patients who developed active TB following IBD diagnosis. Active TB was defined as typical respiratory or extrapulmonary signs and symptoms associated with consistent radiological, bacteriological, and microbiological findings along with bacterial confirmation (smear and/or rapid molecular test and/or culture) according to the Manual of Recommendations for the Control of Tuberculosis in Brazil by the Ministry of Health. 16
The control group consisted of patients with IBD who were followed up at the same outpatient clinics but had no previous history of active TB.
Patients in the case group (IBD and active TB) were randomly matched in a 1:3 ratio with controls according to gender, age, and type of IBD.
For the purpose of this study, medical records of IBD patients were screened for the keywords ‘active TB’ and ‘Tuberculosis.’ In addition, each medical record identified was reviewed thoroughly to ensure the diagnosis of IBD and TB was accurate, and other inclusion and exclusion criteria were met.
Inclusion criteria:
Diagnosis of IBD (CD, UC, or unclassified colitis) according to the criteria established by the European Crohn’s and Colitis Organization (ECCO). 1
Regular follow-up, for at least three consultations or 6 months, in the clinical gastroenterology outpatient clinics at the Clinics Hospital of the Medical School of the University of São Paulo.
Diagnosis of active TB (cases) according to the second updated edition of the Manual of Recommendations for the Control of Tuberculosis in Brazil by the Ministry of Health during outpatient follow-up between January 2010 and December 2021. 16
Exclusion criteria:
Patients under the age of 18.
Patients without a confirmed diagnosis of IBD.
Information on demographic variables (sex, age), smoking, relevant comorbidities, type of IBD (CD or UC), history of TB before IBD diagnosis, clinical aspects of IBD (time of diagnosis, Montreal classification, 17 surgery, and ongoing treatment) and TB characteristics (time between the introduction of immunosuppressive therapy and TB diagnosis, latent TB, clinical features, and treatment) were obtained from electronic medical records. Follow-up variables were outcomes of TB (need for hospitalization, intensive care unit admission, and death), the clinical course of IBD during TB treatment, and the safety of restarting anti-TNF after the TB diagnosis.
LTBI was defined by clinical history, image exams (chest X-ray or computed tomography), and immunological assays, such as TST or IGRA. 18 A positive TST is defined by an induration diameter ⩾5 mm. 16 The national health program in Brazil did not provide IGRA for patients with IBD during the study period. Therefore, our institution had only TST available. According to local standard recommendations, patients were screened for active and ILTB with TST and image exams immediately before introducing anti-TNF therapy.
All study data were collected by reviewing preexisting records, and no information from any patient was revealed. Thus, the study protocol was exempted from the participants’ need for informed consent. This study was conducted with the approval of the Clinical Research Ethics Committee of Clinics Hospital.
Statistical analysis
Quantitative variables were described using mean and standard deviation, medians with IQRs, and qualitative variables using absolute and relative frequencies. The standard features of all patients were described according to the development of TB using absolute and relative frequencies, and the association was verified using Chi-square tests or exact tests (Fisher’s exact test or likelihood ratio test).
Univariate logistic regression analysis assessed the association between patient characteristics and IBD treatment with active TB development. The odds ratios were estimated with 95% confidence intervals using simple logistic regression for all unadjusted analyses. In addition, variables with a descriptive level lower than 0.20 (p < 0.20) were selected in the unadjusted analysis to be included in the joint model using multiple logistic regression.
The software IBM-SPSS, Armonk, NY: IBM Corp. for Windows version 22.0 was used to carry out the analyses, and the software Microsoft Excel 2013 was used to tabulate the data. The tests were performed with a significance level of 5%.
Results
Demographic and clinical characteristics of IBD patients
A total of 38 (2.2%) cases of active TB were identified out of 1760 patients on a regular follow-up at our outpatient clinics between January 2010 and December 2021 (Figure 1). TB cases were randomly matched 1:3 to controls according to gender, age, and type of IBD. Of the 152 patients included in the analysis (cases and controls), 96 (63.2%) were male, and 124 (81.6%) had CD.
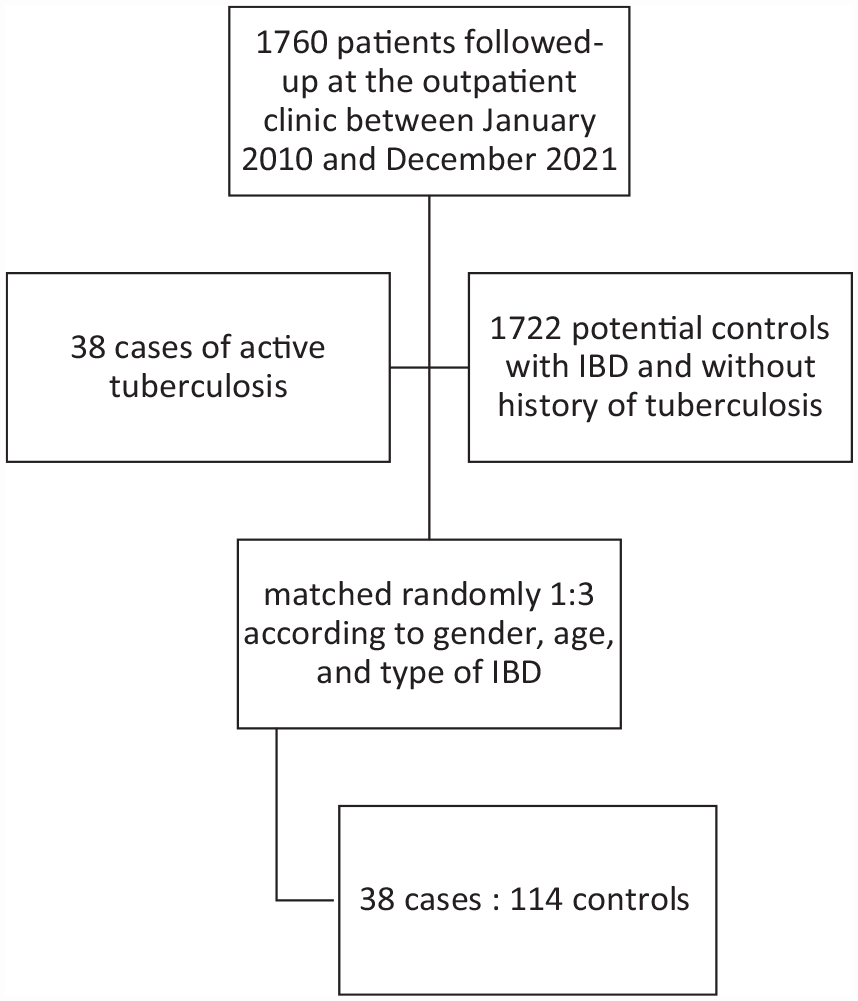
Flow chart of patients with inflammatory bowel disease included in the study.
The demographics and clinical characteristics of the study subjects (cases and controls) are listed in Table 1.
Baseline clinical and demographic characteristics of case and control group from an IBD referral center in Brazil.

Cirrhosis, chronic obstructive pulmonary disease, active oncological disease, diabetes mellitus with target organ damage, chronic cardiovascular disease, chronic renal failure.
CD, Crohn’s disease; IBD, inflammatory bowel disease; LTBI, latent tuberculosis infection; TB, tuberculosis; UC, ulcerative colitis.
Clinical characteristics of TB
Among 38 patients diagnosed with active TB, the median age at TB diagnosis was 39.5 (IQR 30.8–56.3). Half of the cases of active TB were disseminated (50%), 13 patients (34.2%) developed isolated pulmonary disease, and six (15.8%) had other extrapulmonary forms (five cases of pleural TB and one case of meningoencephalitis). The median time period between IBD diagnosis and active TB was 78 months (IQR 13.8–156) (Table 2).
Demographic, clinical, and IBD treatment-related characteristics of active tuberculosis patients from a referral center in Brazil (N = 38).

Immunosuppressant drug or anti-tumor necrosis factor.
IBD: inflammatory bowel disease; ICU: intensive unit care; IQR: interquartile range; TB: tuberculosis; TNF: tumor necrosis factor.Number of patients evaluated in each variable is provided in Bold.
Overall, 36 patients (94.7%) were treated with immunosuppressive medications. Of them, 31 (86.1%) were under anti-TNF drugs, 9 (29%) in monotherapy, and 22 (71%) in combination with either thiopurines or methotrexate. Diagnosis of TB occurred at a median of 32 months after the first dose of anti-TNF (IQR 7–84). In 28 cases (77.8%), TB developed after more than 2 years of treatment with immunosuppressive medications, and in 19 cases (61.3%), after more than 2 years of anti-TNF drugs. Only seven patients (22.6%) developed active TB in the first 6 months of anti-TNF therapy (Table 2).
Among the 38 cases of patients with active TB, 71% did not modify their treatment during the 6 months preceding the development of TB. However, 11 (28.9%) patients required corticosteroids in the 6 months before the development of TB, of which nine (23.7%) maintained the corticosteroid until the diagnosis of active TB (Table 2). No patient underwent de-escalation of anti-TNF drugs or oral immunosuppressants within 6 months.
TST was available to 95 patients, and 13 were diagnosed and treated for LTBI with isoniazid for 6–9 months; three were in the case group and 10 in the control group. Most patients (65.8%) who developed active TB had non-reactive TST before biological treatment; in 10 patients, that information was not available.
All patients with TB received anti-TB treatment following the recommendations of the Brazilian Ministry of Health. Regarding the treatment time, 15/37 (40.5%) patients were treated for 6 months, 18/37 (48.6%) for 9–12 months, and 4/37 (10.8%) for more than 12 months due to severe disseminated disease (Table 2). One patient died in the first month of TB treatment.
Eight patients in the 38 with active TB (21.1%) developed TB treatment-related adverse effects; five were hepatotoxicity, two were skin reactions, and one developed angioedema (Table 2). Nine patients received an alternative scheme for the treatment of TB, two of which were chosen by the infectious diseases team and seven due to an adverse effect of anti-TB therapy.
Outcomes
Anti-TNF and immunosuppressive therapy were stopped in all patients immediately after TB diagnosis. Almost half of the patients 18/38 (47.3%) had active IBD at the time of TB diagnosis, according to inflammatory biomarkers, endoscopic, radiological exams, and/or physician’s global assessment.
Most patients needed hospital admission 26/38 (68.4%), seven patients (18.4%) needed intensive care admission, and a 26-year-old man with ileocolonic CD, using infliximab and methotrexate, died after disseminated TB, including the central nervous system (Table 2). Most patients (81%) were hospitalized for active TB, and the minority (19%) were admitted for IBD flare.
The course of IBD after TB treatment and anti-TNF retreatment
After TB treatment, 20/38 (52.6%) patients received anti-TNF therapy, and five had not previously been exposed to biologics. In two patients, anti-TNF was reintroduced during anti-tuberculosis treatment owing to IBD severity and after multidisciplinary discussions with the infectious diseases team; in one patient after 3 months and another after 5 months of starting TB treatment (Table 2).
The median interval between TB diagnosis and anti-TNF initiation was 8 months, and patients were followed for a median of 31 months (range 2–132). Only one patient developed ‘de novo’ tuberculosis 10 years after the first infection and after 48 months of treatment with infliximab and azathioprine (Table 2).
Risk factors for TB
In the univariate analysis, age at IBD diagnosis was the sole factor significantly associated with the development of TB (p < 0.05) (Table 3). LTBI, disease location or behavior, perianal CD, anti-TNF therapy, smoking, serious comorbidities, previous intestinal surgery, and time of immunosuppression were not risk factors for active TB between cases and controls.
Univariate and multivariate analysis of factors associated with the development of active tuberculosis in IBD patients from a referral center in Brazil.

AZA, azathioprine; CD, Crohn’s disease; IBD, inflammatory bowel disease; IMS: immunossupressor; LTBI, latent tuberculosis infection; MTX: methotrexate; TNF: tumor necrosis factor; UC, ulcerative colitis; &, Not possible to estimate.Significance for Bold p < 0.05.
In the multivariate analysis, both age at IBD diagnosis and anti-TNF therapy were significantly associated with the development of TB (p < 0.05). The chance of active TB in patients diagnosed at age 17 or older was 6– 7 times higher than in patients younger than 17 years old. Patients with anti-TNF therapy had a 3.36 times higher chance of TB than those not treated with anti-TNF (Table 3).
Discussion
This study reports the characteristics, outcomes, and risk factors of active TB in IBD patients followed at a referral center in Brazil. To our knowledge, this study has the highest number of active TB cases being followed up at a single center. Based on the results, 38 (2.2%) patients developed active TB. These rates correspond to more than five-fold the cumulative incidence of TB in Brazil during the study period, justifying the importance of expanding the knowledge of TB in IBD patients.13,16 In contrast, two Spanish series19,20 showed lower rates than the ones we described in our study, probably due to the lower incidence of TB in Spain compared to Brazil. 21 Nevertheless, our results align with a recently published Brazilian study. 22
In our population, age at IBD diagnosis (>17 years old) was identified as a risk factor for active TB. This finding should be attributable to occupational risk because of workplace exposure. Added to this, the everyday lifestyle in this period (stress, hectic schedules, irregular diet, crowded places) can also be an aggravating factor for the higher incidence of the disease. 23 Another possible explanation is a methodological shortcoming, given that the A2 of the Montreal classification (age at diagnosis between 17 and 40) corresponds to the mean age related to the diagnosis of IBD in most Brazilian populations.24–27
In the present study, 36/38 (94.7%) IBD patients who developed active TB were under immunosuppressive medications. Of those, 31/36 (86.1%) were under anti-TNF drugs, 9/31 (29%) in monotherapy, and 22/31 (71%) in combination with either thiopurines or methotrexate.
Studies assessing the risk of active TB in Brazil and Latin America in IBD patients are scarce. Fortes et al., 22 with a different methodology, recently performed a study in north-eastern Brazil and described 8 (2.6%) cases of active TB in 301 IBD patients. Azathioprine, anti-TNF, and a combination of these drugs were associated with a higher risk of active TB. After adjusting for sex, age, type of IBD, and LTBI, anti-TNF combined with azathioprine was associated with a relative risk of 17.8 compared with conventional treatment. 22
We reported a 3.36-fold increased risk of TB in patients with IBD on anti-TNF therapy compared with patients without anti-TNF. However, the risk was lower than in other studies9,22,28 probably due to the high proportion of patients using anti-TNF in monotherapy or combotherapy in the control group (approximately 70%). Furthermore, this data can be justified because the study was conducted in a highly specialized Brazilian IBD referral center, where patients present a predominantly severe disease profile.
A few studies of active TB among IBD patients treated with non-TNF-α inhibitor biologics or small molecules have been reported. For example, in a retrospective South Korean study, a country with an intermediate TB burden, followed 238 IBD patients treated with vedolizumab or ustekinumab for a median of 18.7 months. 29 Active TB did not develop in any patient, suggesting that the risk of TB appears to be low with these drugs. 29 In addition, clinical trials and post-marketing data in patients with IBD receiving vedolizumab or ustekinumab showed a low rate of serious opportunistic infection, including TB.30,31 On the other hand, Janus kinase (JAK) inhibitors may increase the risk of TB infection. A multicenter study with rheumatological patients, including 5671 tofacitinib-treated patients and 12,664 patient-years, described TB as the most common opportunistic infection, with more severe and extrapulmonary disease. 32 Still, the available data are insufficient to assess the real risk of non-TNF-α inhibitor therapy and active TB; further observation is required before making any definite conclusions.
Our study also demonstrated that extrapulmonary TB occurs more frequently than pulmonary. Half of the cases were disseminated, and 34.2% developed isolated pulmonary forms. Given the significant number of disseminated TB cases, we observed high hospitalization rates (68.4%) and need for intensive care admission (18.4%), in addition to a case of death. Similar to our findings, Carpio et al. 7 reported in a Spanish multicenter study 50 cases of active TB in IBD patients treated with anti-TNFs, with 60% developing disseminated or extrapulmonary forms, and 42 (84%) requiring hospitalization, two patients needed intensive care unit admission, and one of them died. TNF blockade can induce an imbalance in the structure of the granuloma and favor the dissemination of the bacillus throughout the body. 5 In the general population, most TB cases are pulmonary; however, extrapulmonary and disseminated forms are more common in immunosuppressed persons.16,23,33 Therefore, IBD patients must be closely monitored since many develop atypical symptoms in the absence of classic respiratory TB symptoms and have the potential to evolve into more severe forms of disease and poor outcomes.
Several studies report a short time between the start of anti-TNF therapy and the development of active TB,19,34 which suggests the reactivation of LTBI. In the present study, TB diagnosis occurred at a median of 32 months after the first dose of anti-TNF (IQR 7–84). In most cases (61.3%), after more than 2 years of anti-TNF drugs, which suggests a new infection. Accordingly, a cohort study from the Brazilian Registry of Biological Therapies in Rheumatic Diseases described five cases of TB, and the mean drug exposure time until the occurrence of TB was 27 months for the anti-TNF group. 35 Other authors described similar data, with figures varying from 14.5 36 to 23 28 months of anti-TNF therapy before the diagnosis of TB, also suggesting a new infection. This explanation is supported by a recent systematic review and meta-analysis showing that the risk of TB in IBD patients on anti-TNF agents depends on the local disease burden. 37 Therefore, patients living in endemic areas may have recurrent exposure to M. tuberculosis and can become infected after a long period of anti-TNF therapy.
Although some studies showed that re-screening for LTBI might be necessary for patients with immune-mediated inflammatory diseases (IMID) on biological therapy,38 –41 data from countries with intermediate to high incidences of TB are still lacking. This is particularly important in Latin America, which is also experiencing a rising prevalence of IBD accompanied by increased use of biologics in the last 20 years. 14 Under special conditions (e.g. live, travel, or work in endemic TB areas), ECCO guidelines consider annual TB screening while continuing immunosuppressive therapy in IBD patients. 10 However, the efficacy of repeating tests for LTBI is still to be established. A prospective study to monitor TB during anti-TNF therapy in patients with IBD and IMID showed that conversion of LTBI is common (17/90−18.9%) during biologic treatment in an area with intermediate TB prevalence. 39 Hence, the risk of developing TB should be considered during any follow-up period, especially in patients exposed to anti-TNF therapy. Moreover, the best strategy for LTBI screening should always be considered individually. An informative note from the Ministry of Health of Brazil recently recommended annual screening for LTBI while the patient uses an immunobiological and/or immunosuppressant. 42
Anti-TNF and immunosuppressive therapy were stopped in all patients immediately after TB diagnosis, but in two patients, anti-TNF was reintroduced during anti-tuberculosis treatment owing to IBD severity. Despite the absence of prospective or controlled data regarding the best time to start immunosuppressive drugs after initiating TB treatment, biologics or small molecules therapy should ideally be reintroduced only after the completion of treatment. 10 Nevertheless, in exceptional cases with more severe disease activity, IBD therapy should be restarted after at least 2 months of anti-tuberculosis treatment. 10 Patients should be evaluated by the infectious diseases specialist and closely monitored clinically.
In our study, 20 patients (52.6%) received anti-TNF therapy after TB treatment. TB re-occurred in just one patient after long-term follow-up, suggesting ‘de novo’ infection rather than reactivation. In a multicenter retrospective, observational study conducted by GETECCU, anti-TNF was restarted after a median of 13 months in 17 patients (34%), and there were no cases of TB reactivation. 7 In a descriptive study on the GETAID cohort, 44 patients with IBD on anti-TNF therapy developed active TB, and biologics were restarted in 27 patients, after a median period of 11.2 months, without any recurrence of the infection. 36 Thus, our data corroborate anti-TNF treatment is safe even after previous TB infection and in endemic areas.
There are some limitations concerning the results of this study. Firstly, the retrospective study design may result in patient loss, missing data, and some selection bias. Secondly, we did not include ethnicity, education, and socioeconomic status in the analysis, co-variates that could substantially impact the results. In addition, some patients did not undergo TST or IGRA due to the unavailability of the test and/or lack of adherence to the test reading. The absence of these data can lead to a misinterpretation of whether some cases of active TB are a new infection or reactivation of LTBI. Another limitation is the absence of reports on patients’ contact with TB. Additionally, a prospective design would provide better data on risk factors for developing active TB. Despite these limitations, this comprehensive analysis describing characteristics, risk factors, and outcomes of active TB in IBD patients sheds light on crucial challenges physicians from developing countries, like Brazil, may confront during IBD care.
In conclusion, TB remains a significant health problem in IBD patients, especially in high-prevalent areas and in patients on anti-TNF therapy. Age at IBD diagnosis (>17 years old) was also identified as a risk factor for active TB. Most cases occur after long-term anti-TNF treatment, suggesting a new infection and highlighting the importance of close monitoring. Reintroduction of anti-TNF drugs after anti-tuberculosis treatment seems safe. A personalized approach to choosing and monitoring therapy in IBD patients is essential and should consider individual characteristics and local epidemiological factors.
