Abstract
An estimated 2.5–3 million individuals (0.4%) in Europe are affected by inflammatory bowel disease (IBD). Whilst incidence rates for IBD are stabilising across Europe, the prevalence is rising and subsequently resulting in a significant cost to the healthcare system of an estimated 4.6–5.6 billion euros per year. Hospitalisation and surgical resection rates are generally on a downward trend, which is contrary to the rising cost of novel medication. This signifies a large part of healthcare cost and burden. Despite publicly funded healthcare systems in most European countries, there is still wide variation in how patients receive and/or pay for biologic medication. This review will provide an overview and discuss the different healthcare systems within Western Europe and the barriers that affect overall management of a changing IBD landscape, including differences to hospitalisation and surgical rates, access to medication and clinical trial participation and recruitment. This review will also discuss the importance of standardising IBD management to attain high-quality care for all patients with IBD.
Introduction
Europe is a large continent encompassing 44 countries with an estimated population of 748 million, which is equivalent to 9.8% of the global population. 1 Approximately 2.5–3 million individuals (0.4%) in Europe are affected by inflammatory bowel disease (IBD), a complex, chronic inflammatory disorder of the gastrointestinal tract. IBD can affect individuals of all ages, gender, ethnicities and socio-economic parameters. The chronic nature of this disease demands continuous care, often necessitating repeat invasive investigations, hospital admissions and surgeries, and initiation and maintenance of expensive therapies such as biological medications. Unsurprisingly, IBD can have a significant burden on most healthcare systems 2 with a direct healthcare cost of 4.6–5.6 billion euros per year. 3 In a 5-year follow-up study, Burisch et al. 2 found the mean cost per patient-year for IBD patients was €2609. Whilst hospitalisations and diagnostic procedures accounted for greater than 50% of costs during the first year of diagnosis, subsequent expenditure on biologics accounted for 74% of costs in Crohn’s disease (CD) and 48% in ulcerative colitis (UC). The mean annual cost per patient-year for biologic use was €866 (€1782 for CD and €286 for UC). The medical costs for IBD management is comparable to those of other severe chronic diseases such as diabetes mellitus or cancer. 4 Despite the formation of the European Union (EU), which encompasses 27 countries, there is still considerable variation between countries’ healthcare and socio-economic situations. This review will discuss the different healthcare systems within Western Europe and the barriers that affect overall management of a changing IBD landscape, including differences to hospitalisation and surgical rates, access to medication and clinical trial participation and recruitment.
Healthcare systems within Europe
There are three predominant healthcare systems within Western Europe (Table 1). Finland, Greece, Ireland, Italy, Sweden, Spain, Denmark, Portugal and the United Kingdom operate through public finance funded by general taxation. This is also known as the Beveridge model. Apart from Denmark, these countries also have the option of private voluntary insurance. Healthcare in Denmark, Finland and United Kingdom is covered by the National Health Service, which is almost entirely financed, planned and run by public authorities, resulting in equal and almost free access to healthcare services. In comparison, Italy has a National Health Service but is based on compulsory health insurance with mixed public and private service provision. This has resulted in substantial healthcare inequalities between the north and south of the country. 5 The Greek healthcare system also has unequal access to health services with gaps in service provision. Their healthcare system is based on compulsory social insurance, incorporating considerable private sector involvement for financing and service provision.
Healthcare services available across Europe.
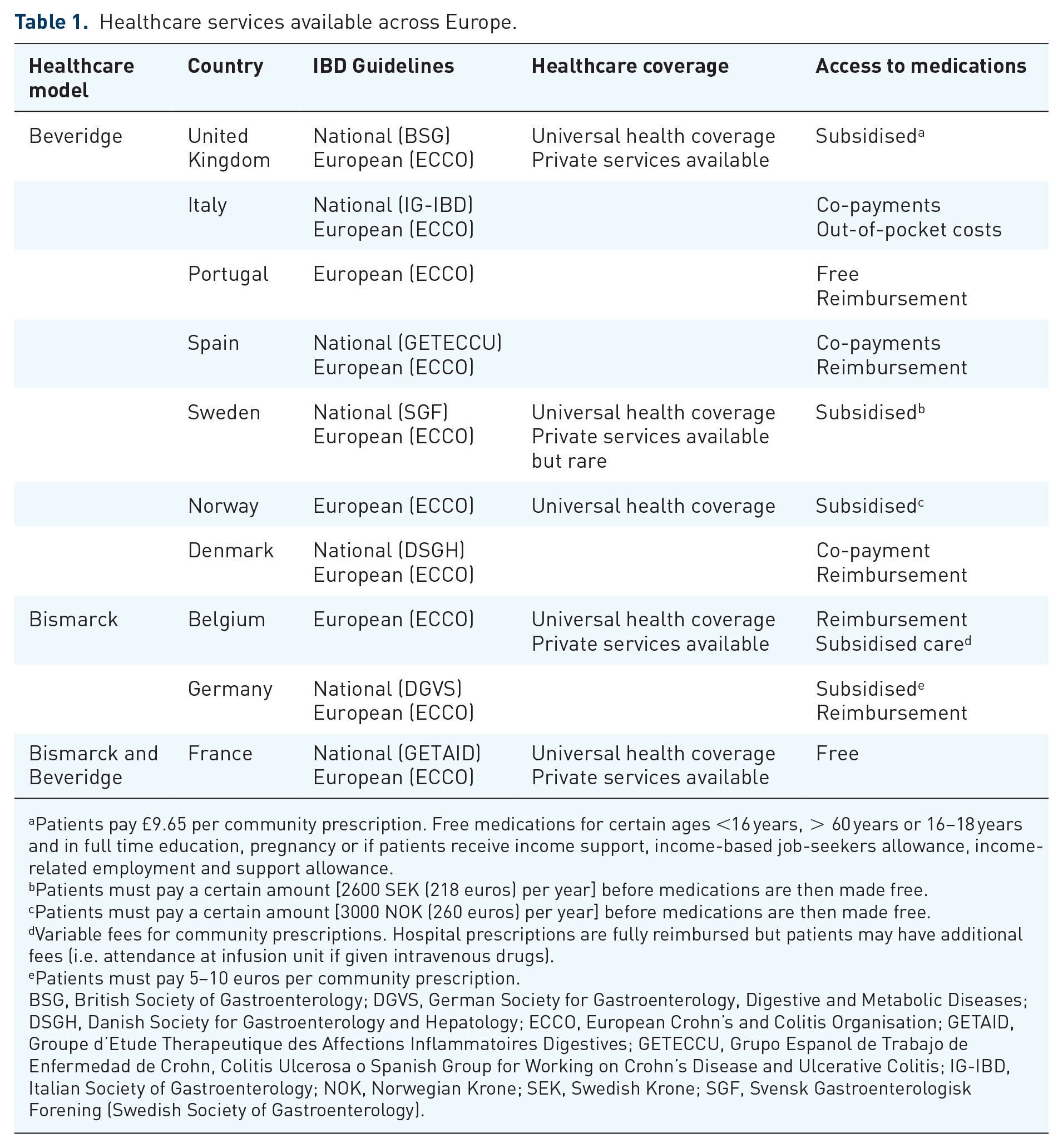
Patients pay £9.65 per community prescription. Free medications for certain ages <16 years, > 60 years or 16–18 years and in full time education, pregnancy or if patients receive income support, income-based job-seekers allowance, income-related employment and support allowance.
Patients must pay a certain amount [2600 SEK (218 euros) per year] before medications are then made free.
Patients must pay a certain amount [3000 NOK (260 euros) per year] before medications are then made free.
Variable fees for community prescriptions. Hospital prescriptions are fully reimbursed but patients may have additional fees (i.e. attendance at infusion unit if given intravenous drugs).
Patients must pay 5–10 euros per community prescription.
BSG, British Society of Gastroenterology; DGVS, German Society for Gastroenterology, Digestive and Metabolic Diseases; DSGH, Danish Society for Gastroenterology and Hepatology; ECCO, European Crohn’s and Colitis Organisation; GETAID, Groupe d’Etude Therapeutique des Affections Inflammatoires Digestives; GETECCU, Grupo Espanol de Trabajo de Enfermedad de Crohn, Colitis Ulcerosa o Spanish Group for Working on Crohn’s Disease and Ulcerative Colitis; IG-IBD, Italian Society of Gastroenterology; NOK, Norwegian Krone; SEK, Swedish Krone; SGF, Svensk Gastroenterologisk Forening (Swedish Society of Gastroenterology).
Austria, Germany, Belgium, France and Luxembourg operate under the Bismarck model, which is also publicly funded but similar to Greece, is based on compulsory social insurance. In Germany, however, individuals can take out private insurance if they are self-employed and their income exceeds a certain amount. Whilst the German healthcare system is amongst the most expensive in Western Europe,6,7 France has a complex mix between the private and public sector in terms of service provision and healthcare financing. In Belgium, patients can buy additional private insurance, which often covers more than what is incorporated by the public system.
The third type of healthcare system is private finance based on voluntary insurance. The Netherlands is the only country, which has mixed compulsory social insurance covering 62% of the population and private voluntary insurance covering 31% of the population. A national insurance scheme covers for catastrophic risks, chronic illness, disability and psychiatric care.
Under the EU law, EU citizens have the right to access healthcare in any EU member state and to be reimbursed for care abroad by their home country. 8 The European Health Insurance Card ensures that necessary healthcare is provided under the same conditions and at the same cost as people insured in that country.
Incidence and prevalence of IBD
With an acceleration in incidence rates in newly industrialised countries becoming more Westernised, IBD is now recognised as a global disease. 2 Western Europe is still known to have one of the highest incidence rates for IBD with an estimated incidence of 1.85–10.5/100,000 person-years for CD and 1.9–17.2/100,000 person-years for UC. 9 Whilst the number of new cases is now stabilising, 9 prevalence rates are rising secondary to the general rise in life expectancy and improvement in disease management.
Overall, the prevalence rates vary among Western Europe from 28.2 to 322/100,000 person-years in CD and 43.1 to 412/100,000 person-years in UC patients. 9 Incidence rates are highest in the Scandinavian countries and the United Kingdom. The Nordic countries, including Norway, Sweden and Denmark, have some of the highest incidence and prevalence rates of IBD in the world, with recent predictions estimating the prevalence of IBD in Norway to reach 1% of the general population by 2030.10,11 Although Southern European countries have often been considered as low prevalent areas for IBD, recent studies have proven this is no longer the case, as can be seen from recent studies from Spain, Italy and Portugal.12–17
Western Europe is vast and disease rates vary not only between countries, but within each country itself. One of the largest UK studies to determine epidemiology rates in IBD explored health records from more than 38.3 million people between 2000 and 2020 and found a population incidence rate of 36 new cases per 100,000 person-years, with greater rates in Scotland and Northern Ireland compared to England and Wales. 18 Similarly, whilst the overall incidence of IBD in Italy is estimated to be approximately 6.3/100,000 person-years, the Northern regions have documented higher rates compared to the Southern regions, 16 although this has been disputed by other studies.19,20 Spain has an overall incidence of 16.2/100,000 person-years but had significant variation between the North (Asturias and Navarra), Central, North-East (Catalunya) and Southern (Andalucia) regions.21–24
Robust epidemiology studies can vary between countries, particularly if there is not a national collective dataset or registry already in place, such as in Belgium and Portugal. Detection rates, methodology and diagnostic criteria differ between studies, making it challenging to compare incidence and prevalence rates across countries within Europe. Whilst prospective population-based studies are preferable, they are time-consuming and expensive. Consequently, institutional or regional studies are conducted instead. The latest epidemiological data for Belgium dates to the 1990s, documenting incidence rates of 4.1/100,000 for CD and 3.7/100,000 for UC patients.25,26 In Portugal, Santiago et al. 14 reported a high IBD incidence rate of 48.6/100,000 person-years. This study’s methodology, however, could not guarantee that all cases investigated had a confirmed diagnoses of IBD and thus may have included false-positive results. Moreover, this contrasts with the systematic review conducted by Ng et al. 9 who reported a case number between 3.77 and 7.71/100,000 person-years. It is thus important to bear in mind that whilst observed differences might be attributed to real differences in environmental factors, lifestyle and genetic susceptibility, the differences may also simply be due to differences in methodology between studies. 3
Surgical and hospitalisation rates
For patients with UC, the most common surgical procedures performed are total proctocolectomy with ileostomy and ileal-anal anastomosis; and for patients with CD, ileo-caecal resections and segmental colectomies. 27 In Europe, the 1-year rate for major surgery in CD patients ranged from 6% to 14%, which increased to 12% to 27% at 5-year following diagnosis.11,28–34 The UC surgery rates are lower, with colectomy rates ranging from 1% to 5% at 1-year and 3% to 8% at 5-year from diagnosis.28,29,31,34,35
In general, the number of surgical procedures performed for IBD in Europe has either stabilised or steadily decreased in recent years, likely due to the availability of new medical treatments, particularly the introduction of biological therapies. This has been noted in United Kingdom, Italy, Sweden, Norway and Denmark.27,36–41 However, caution should be taken when attempting direct comparison of surgical rates across time and between countries due to the lack of concomitant data on disease severity and inter-country differences in treatment practice and availability of novel medication. 42 Furthermore, many countries have not reported surgical trend data over time, such as in Spain and Belgium.
In the United Kingdom, the cumulative incidence of surgery for CD has decreased over three time periods (1986–1991, 1992–1997 and 1998–2003) by 59%, 37% and 25%, respectively. 30 Sweden has similar reduction in surgery rates within 5 years of diagnosis, from 54.8% in 1990–1995 to 17.3% in 2009–2014 27 although the rate of surgical interventions varied by disease subtype and surgical procedure. Interestingly, Portugal is the only country to document an increase in IBD-related surgical procedures, with a shift towards elective surgery.43,44 However, they found that IBD re-operation rates had actually decreased when corrected for disease prevalence. The cause for this remains unclear due to the lack of published data although it is postulated that the main contributing factors are the rising disease prevalence, delayed diagnoses of CD leading to more complex and complicated disease and the European guidelines recommending elective surgery when possible and early surgical interventions for the more complicated cases.14,45,46
A global systematic review from 42 countries with temporal trend analysis from 35 countries has demonstrated stable hospitalisation rates for both UC and CD in countries with rising prevalence rates. 47 This comprises of countries in the Western world, including most countries in North America, Western Europe and Oceania. This reduced hospitalisation rate is likely a consequence of innovations in therapeutics and the adoption of clinical management algorithms, which has allowed for the transition of moderate to severe IBD management from the hospital to community/outpatient settings. The liberated use of IBD specialist nurses and telemedicine has further eased this shift in management. In fact, numerous studies across Europe have demonstrated reduced hospitalisation rates and improved patient satisfaction since the introduction of the IBD specialist nurses and telephone helpline,48–51 and this became even more pronounced during the COVID-19 pandemic.52,53
Recent European studies have shown the cumulative rate of hospitalisation in CD patients range from 23% to 49% at 1 year and 44% to 54% at 5 years after diagnosis.11,28,29,54–56 However, lower hospitalisation rates are reported for UC patients with the 1-year hospitalisation rate ranging from 9% to 33% and the 5-year hospitalisation rate ranging from 18% to 54%.36,57,58 A pan-European study demonstrated that hospitalisation rates declined significantly following the second year with a cumulative 10-year risk of 52.7%. 56 There was considerable variation between countries with the highest hospitalisation rates in Denmark, Ireland and Portugal with lower rates seen in Norway, Greece and Italy. A meta-analysis published in 2010 observed hospitalisation rates from nine European countries and found the highest rates in Denmark and Scotland with the lowest in Spain, Switzerland and the Netherlands. 59 There is variation between studies, however, with some studies demonstrating worsening hospitalisation trends within Spain despite the introduction of biological therapies and the reduction in surgical procedures.43,60
Similar to surgical rates, hospitalisation rates should be interpreted with caution. The data are highly variable between countries and studies are limited by its subjective outcome measure with numerous confounding factors and frequent omissions on the indication for hospitalisation in databases. This includes disease phenotype and severity, diagnostic work-up, healthcare/reimbursement policies, local practice and different physician-thresholds for hospitalisations between centres.
Drug access
The availability of biological and biosimilar drugs has revolutionised the treatment for IBD and represents one of the most striking advances in medical science in healthcare in the last two decades. 61 Biologics have been shown to achieve disease remission, slow disease progression, decrease the need for surgery and subsequently increase work participation and quality of life. 62
In Europe, specific organisations have been created to approve and regulate the use of medications. In the United Kingdom, this is managed through the National Institute of Health and Care Excellence (NICE), the Scottish Medicines Consortium and the All-Wales Medicine Strategy Group. Specifically, NICE is a national drug approval and implementation body, which provides rigorous, independent assessment of complex evidence for new health and drug technologies by combining information on clinical efficacy and cost-effectiveness criteria. Subsequent guidelines and recommendations are published and updated regularly to encourage best practice and improve patient outcomes. Working under the UK Department of Health, the Medicines and Healthcare Products Regulatory Agency are a national oversight body that regulate the safe and appropriate use of medicinal products and medical devices in the United Kingdom. Its activities include overseeing UK notified bodies, regulating clinical trials, monitoring compliance for medicines and medical devices and offering technical and regulatory advice for these products.
For countries that are part of the EU, all medicines produced using biotechnology must be approved through a centralized procedure known as the European Medicines Agency (EMA). 63 The approval of a pharmaceutical product authorized by the EMA is binding to all member states of the EU. Infliximab was the first biological drug to receive authorisation for adult patients with moderately-to-severe CD in 1999 and 2006 for UC by the EMA. This was followed by adalimumab in 2007, vedolizumab in 2014 and ustekinumab in 2016.64,65 In 2018, the EMA licensed the use of the first small molecule named tofacitinib, 66 a janus kinase inhibitor, and since then a plethora of small molecule therapies have entered the European market.
Despite the uniformity in drug registration and international professional guidelines written by the British Society of Gastroenterology and the European Crohn’s and Colitis Organisation (ECCO), treatment practices are still highly variable. 64 One of the key factors for this variability in drug access is the high cost of biologics, with the annual cost of drug therapy being over 10,000 euros per patient. 64 Historically, healthcare costs were primarily driven by hospitalisation and surgery. The Dutch COIN study demonstrated that these costs have now shifted towards the increased use of biological therapy, specifically for patients with CD and those on anti-tumour necrosis factor drugs. 67 This was further validated by a French study that disclosed medication to be the major driver of healthcare costs (84%) with hospitalisation and surgery accounting for 11% and 2%, respectively. 68 This study also demonstrated that the only variable associated with a greater cost of IBD apart from biologic initiation was a history of a switch from one biologic agent to another (p = 0.0018). 68
In the first several years following the introduction of biological drugs, patients from high income countries were the fortunate few to have access to drug therapy. 62 Moreover, depending on the drug, there was large variation in treatment cost per patient with up to threefold variation between countries. 64 This is particularly the case where biological therapies are not subject to reimbursement or are reimbursed with serious restrictions to a given group of patients with specific disease characteristics. 65 Discernible reimbursement coverage is likely to be a strong reason behind the disparate access rates for biological treatment. Whilst most countries apply eligibility criteria based on the ECCO guidelines as a standard for reimbursement, there can be marked variation in requirement between countries such as disease severity and chronicity, and duration of reimbursed maintenance therapy. In contrast, IBD patients in the United Kingdom are not charged for their medication although strict regulations and criteria must be met prior to medication being recommended for use. A pan-European survey was conducted by Ding et al. 69 which highlighted the variability in access to tests such as faecal calprotectin and medication such as thiopurines and anti-tumour necrosis factor agents. Cost was the frequently cited barrier for access with a significant West-East and North-South divide.
Eligibility criteria to initiate biological treatment is highly variable with countries such as France where a high Crohn’s disease activity index of greater than 300 is required before CD patients are entitled to treatment. Additional requirements such as failure of a non-biological drug (steroids and/or immunosuppressants) is seen in most European countries. Anti-tumour necrosis factor therapy is generally the first-line biologic in most centres secondary to its cost compared to other biologics or small molecules. In Belgium, patients must cycle through three months of corticosteroids and/or immunomodulators prior to reimbursement of any advanced therapy. Whilst access to drugs is fully covered by public funds following reimbursement criteria in Spain, only selected centres are allowed to dispense these medications, and only after patients had previously failed at least one non-biologic drug. Portugal has no major limitations on the use of IBD drugs but there is a prolonged time gap between EMA’s approval and Portugal’s Medical Products Agency (INFARMED) authorisation to introduce the drug to the national market. This time gap is mainly due to reimbursement negotiations. In addition, there is further variability between countries on who can prescribe biologics. Countries, such as France and Germany, allow gastroenterologists as well as other specialists (immunologists, internists, surgeons and general practitioners) to prescribe and indicate biologic use whereas countries such as United Kingdom restrict biologics prescription for IBD patients to gastroenterologists only. 64 In Norway, although most of the advanced therapies approved by the EMA for IBD are available, the recommendations for its use are centrally regulated by a public organisation (Legemiddelinnkjøpssamarbeid) in order to minimize costs and establish national recommendation for their use by Norwegian gastroenterologists. Unfortunately, there is a lack of evidence to demonstrate whether there is an optimal approach to healthcare utilisation for the use of biologic agents in IBD. Given the fact that there are diverse and specific differences to the use of these drugs in different healthcare systems, it is conceivable that future research could explore the outcomes from these cohorts to determine if there is a more effective therapeutic approach to the use of biologics.
Sweden, Germany, Denmark and Italy have the least restrictive eligibility criteria, with Sweden’s national guidance focused on the importance of a personalised approach taking into account patient’s wishes, disease phenotype and activity, extra-intestinal manifestations, safety and costs. 70 Germany can also take a personalised approach once drugs have been officially licensed by the EMA. When prescribing, gastroenterologists must consider limitations provided by the label but have the option to apply for cost coverage for shortened application intervals in special treatment situations by the respective issuance company. Denmark has no restrictions on prescribing IBD medication and base their decision-making on the ECCO guidelines. Italy’s national health system provides universal access to healthcare services, including medications for IBD; however, patients may still face co-payments or other out-of-pocket costs. The Italian government has implemented measures such as regional drug formularies and risk-sharing agreements between pharmaceutical companies and healthcare systems in an effort to counteract rising healthcare costs whilst balancing patient needs. 71 Consequently, it is unsurprising that the number of patients on biologics per 100,000 population is highly variable, ranging from the highest in Sweden (53.5), France (34), Spain (32.3) and Germany (32.9). 64
The availability of the first biosimilars for infliximab and adalimumab for use in Europe in 2013 and 2017, respectively helped to improve access whilst reducing the financial burden of associated biologics.62,64,72 Studies have shown that biosimilars have comparable safety and efficacy rates to those of the original biological product but are a substantially cheaper option by up to 70%.62,65 In Germany, pre-specified rates for the use of biosimilar over originators were enacted to ensure most patients are treated with biosimilars. However, there are other factors to consider when issuing and supplying biosimilars to patients, including regulatory issues, biosimilar acceptability amongst clinicians, price and reimbursement policies as well as supply and demand incentives. 73
There are substantial inequalities to accessing biological drugs amongst the European countries. In countries where biological therapy is less affordable, reimbursement conditions including eligibility criteria and administrative requirements are more restrictive. Whilst access is strongly determined by the economic development of the country, there are still large differences among countries with a similar economic development. This suggests that numerous factors such as patient perception, health sector governance, platforms for care delivery, workforce numbers and skills, tools and resources have a role in drug access and must be explored further to improve IBD management.
The health technology assessment (HTA) is a scientific evidence-based process that allows competent authorities within Europe to determine the relative effectiveness of new or existing health technologies. 74 Whilst not a part of national policy making, HTA was created to inform clinical decision-making and improve patient access to health technologies and can help allocate budgetary resources in the field of health, such as for establishing pricing or reimbursement levels of health technologies. 74 To create HTA guidelines, a large amount of data is needed including disease burden evidence from clinical studies, patient reported outcome measures, results from health economics analyses, modelling and budget impact analyses. However, it has proven to be difficult to obtain local data on epidemiology, costs, cost of illness and cost-effectiveness of the specific treatments seeking public funding. A major limitation for IBD patients in Europe is a lack of a consensus registry that provides up-to-date epidemiology and covers the entire patient population on biologic treatment. All five Nordic countries have nationwide prescription databases covering all dispensed drugs61,62 and in 2005, Sweden established a national registry for IBD 75 followed by Denmark in 2013 who established a special registry to document the use of biologics in IBD. 63 Today, the Swedish registry has data on greater than 60,000 IBD patients and many important studies have been based on these registries.64–67,76–81 Establishing registries and integrating registries between countries would allow for big data mining and analyses. Moreover, it would ensure appropriate patient follow-up and provide valid and reliable data on access rates, potentially resulting in favourable financial and reimbursement decision-making.
Clinical trial access
Clinical trials have a pivotal role in supporting advances in IBD management. Whilst the number of IBD clinical trials being initiated each year has more than doubled between 1999 and 2019, patient recruitment has not increased proportionally.82,83 In fact, the greatest challenge in designing and completing IBD studies is patient recruitment. 84 Specific patient concerns for recruitment have resulted in a reduced incentive for trial participation; this includes placebo assignment, study design and attributes and the increased availability of licensed treatments, competitive or overlapping trials and limited number of eligible patients meeting strict inclusion and exclusion criteria.85–87 Another potential challenge is the lack of access to investigational treatment following the completion of a clinical trial. Although many phase III trials include long-term extension phases to allow patients to remain on therapy until regulatory approval is obtained, this may be an issue for the early phase trials particularly if a clinical development program is discontinued. 87 Consequently, the failure to recruit patients quickly and/or in sufficient numbers is a leading cause of clinical trial discontinuation with concerns with the ability to deliver future trials in IBD.86,88 Rubin et al. 88 demonstrated that patients identified healthcare providers as the most helpful resource for researching clinical trials. The study further stated that physicians rarely initiate conversations about clinical trials and patients typically do not ask. Specifically, participants from France and Germany reported that healthcare provider conversations were the most used resource for researching clinical trials, whereas Spanish participants were more likely to use pharmaceutical manufacturer websites and social media. 88
The Nordic countries, particularly Norway and Denmark, actively participate in numerous clinical studies related with IBD. Although the Nordic countries are well-known for their expertise in basic/translational and population studies in IBD (such as the ongoing prospective Inflammatory Bowel Disease in South-Eastern Norway (IBSEN) III study), the inclusion in clinical trials has been challenging due to their relatively small population. Sweden, on the other hand, was previously a very active nation for clinical trials but has become less so in more recent years. The country has seen a steady decline in Clinical Trials applications to the Medical Products Agency in Sweden by pharmaceutical companies, from 361 applications in 2007 to 193 in 2021. 89 Many obstacles have been identified, and among them are lack of policies, poor financing of clinical trials, lack of infrastructure, poor integration of research and innovation into the healthcare and lack of staff in combination with poor patient information regarding the opportunity to be part of clinical trials. 90
While the beginning of this century demonstrated a major shift in enrolment to Eastern and Central Europe with the smaller countries such as Poland, Hungary and Czech Republic taking on considerably more IBD studies, it has become increasingly difficult to recruit study sites due to the complexities of recent clinical trials.86,88 Trial endpoints have become more objective, requiring multiple endoscopies with video recordings sent for central reading. In addition, trials are asking for more information including serum and stool collections for biomarkers with pharmacokinetic measurements, leading to longer and more frequent study visits with higher costs. 86 A German and Italian patient survey identified that two-thirds of patients were willing to participate in clinical trials with favourable factors being higher education, poor quality of life, financial compensation and informative materials. Negative factors included the possibility of receiving a placebo, frequent colonoscopies and the time and effort expenditure.91,92 These complexities along with a greater reliance on technical equipment and procedures have made clinical trials increasingly difficult and time-consuming for less experienced investigators and facilities.85,86
Over the past 10 years, phase I and II trials remain concentrated in Western European countries with Eastern European countries having one-third fewer trials in comparison. Phase III trials are more evenly distributed, which is in line with previously reported trends demonstrating a rise in pharmaceutical clinical trial activity across Europe. 93 There are currently 91 ongoing registered clinical trials in IBD within the EU, of which 45 are sponsored from pharmaceutical companies, 17 from the Nordic countries, 7 from Spain, 7 from Italy, 3 from Germany and 2 from France. 94 In stark contrast, there are currently 78 registered IBD studies within the United Kingdom alone. England has a very strong research framework due to the unique infrastructure of the National Institute for Health and Care Research (NIHR) network. 95 The NIHR supports research by significantly investing in and providing research expertise, specialist facilities, a research delivery workforce and support service, which aids and delivers research that is funded by NIHR and other organisations, including medical research charities and the life sciences industry. Per annum, the NIHR invests greater than £606 millions of infrastructure funding for services, facilities and people to support research and its delivery, which is awarded through a national peer-reviewed funding process. Furthermore, the NIHR helps to support the coordinated delivery of commercial and non-commercial research trials throughout England by providing annual funding of £300 million to the 15 local clinical research networks across the country.
The advantage of living in a European country is the proximity of neighbouring countries with easy access for multinational collaboration particularly for those countries part of the EU. Multinational collaboration is important for clinical research as it improves research quality, maximises access to patients and leads to quicker results. 96 It also allows the sharing of medical and scientific expertise, tools, procedures and costs, increases the applicability of research findings, reduces duplication and enhances methodological standards.97–99 The results from multinational trials can facilitate implementation of healthcare policies, optimise resource use and improve patient care across borders.97,98 However, only 3% of academic trials compared to 30% of industry trials are multinational. 100 Whilst funding may be the greatest barrier for this discrepancy, numerous regulatory and legal barriers can be challenging to conduct pan-European clinical trials.96,101 One of the greatest barriers for patients to access clinical trials across borders is the exclusion of patients’ right to receive medical care and claim reimbursement for clinical trial participation in another European country as per the scope of the Directive on the application of patients’ rights to cross-border healthcare. 102
Lalova et al. 103 conducted a pan-European survey that demonstrated that patients were interested in participating in clinical trials abroad with the strongest reasons to access a new treatment that is either not marketed or unavailable in a similar trial in their own country of residence. This study also found that the primary reason for hesitation to enter cross-border trials were logistical and financial burdens, including travel distance and expenses, accommodation, translation services, loss of income and the costs of baseline therapy when the experimental treatment was an add-on. Countries with the least open clinical trials were Central and Eastern European countries such as Bulgaria, Romania and Greece whilst the two countries with greater access were Germany and United Kingdom. Whilst enrolling foreign patients would enhance recruitment numbers, there are several confounding factors to consider. This includes language barriers, reimbursement of travel expenses and the risk of being accused of undue incentive, travel aspects that may impact patients’ underlying condition and the extensive administrative burden of international relocation. 104 Language is a key barrier to trial recruitment, particularly the challenge of understanding and accepting the informed consent for trial participation. There is also little clarity on follow-up care at the end of the clinical trial, specifically lack of scientific expertise and/or specialised equipment required for follow-up treatment. Another obstacle is that the home country’s health insurance system may refuse to fund follow-up care services for patients who had access to experimental treatment abroad. 103
It is well known that encouraging a research-positive culture in health and care organisations not only improves patient access to clinical research and new treatment options, but has also demonstrated (1) better overall patient outcomes, (2) a happier workforce and (3) provides benefits for the healthcare systems. 105 Improved patient outcomes include reduction in mortality rates106,107 and improved cancer survival outcomes.108,109 There is also evidence to state that a research-friendly environment helps recruit and retain medical workforce, 110 provides meaningful work and reduces risk of burnout. 111 Further studies have demonstrated research improves clinical practice, reduces the cost of healthcare and also drives policy change. 112 This would be an interesting area for future research to investigate and compare different healthcare systems for whether active research centres/countries correlate with better quality of care and overall patient outcomes.
Standardising IBD treatment
One of the most valued targets but monumental tasks in IBD treatment is providing high-level quality of care. 113 Numerous studies have shown huge differences in clinical care in IBD, depending on the type of healthcare system, location of treatment (academic centre versus private practice versus general gastroenterologists) and presence of an integrated model of care (collaboration between specialist IBD clinicians, IBD nurses, registered dietitians, clinical psychologists).114,115 These studies demonstrated that specialist care with a multidisciplinary approach was associated with greater rates of remission, lower IBD-related hospitalisations, lower levels of corticosteroid dependence and higher rates of immunomodulatory and biological use. ECCO conducted a web-based survey study of 4670 patients from 25 different European countries, of which only 52% reported adequate access to care. 116 This could potentially be improved by standardising care and advocating for all IBD units to promote and provide high-level quality of care.
The Spanish Working Group on CD and UC (GETECCU) is a medical society that promotes excellence in healthcare, teaching and research. They set out to establish reliable and consistent standards of IBD in Spain. In collaboration with other stakeholders such as patients’ associations and nurses, the group developed a set of quality indicators based on Delphi methodology. GETECCU selected 53 quality indicators and subjected them to a normalisation process. IBD units would then be assessed by an external organisation and granted official certification if all quality indicators were met to a safe and high standard, with re-certification required every 2–3 years to ensure the high-quality care is maintained. 117 In the United Kingdom, the national IBD audit programme was established to support the development of national standards for IBD care and to establish quality IBD care as a key component of local healthcare delivery.118,119 Whilst both programmes aim to improve quality care standards of IBD, the former (Spain) evaluates each unit onsite by an external association/company while the latter (United Kingdom) submits local data from their unit, which is then compared against a national benchmark.
Another way of standardising care is by creating and adhering to guidelines. The aim of guidelines is to remove variation in care and improve patient outcomes by providing appropriate practice measures for healthcare providers.120–122 They are designed to ensure people receive safe, consistent, high-quality, personalised care that is independent of age, socio-economic status and geographic location. Most countries within Europe have their own national guidelines for managing IBD, such as in the United Kingdom (British Society of Gastroenterology), Sweden (Swedish Society of Gastroenterology), Denmark (Danish Society for Gastroenterology and Hepatology), Germany (German Society for Gastroenterology, Digestive and Metabolic Diseases) and Italy (Italian Society of Gastroenterology). However, many other countries (Norway, Portugal, Belgium, Spain) do not and instead adhere to the guidelines published by the ECCO. Despite these guidelines, barriers still exist due to differences in local policies with challenges as described above with medication issuance and available services.
Conclusion
Recent studies have demonstrated a steady decline in IBD incidence rates. However, prevalence rates are on the rise and as a result, so too is the burden of disease. Whilst hospitalisation and surgical rates are on a downward trend, the rising cost of novel medication is climbing, which signifies a large part of healthcare cost and burden. Despite publicly funded healthcare systems in most European countries, there is still wide variation in how patients receive and/or pay for biologic medication. Moreover, a decline in clinical trial participation will only hinder further development and progression in IBD management. Improving IBD management by standardising care across Europe can be initiated by; implementing national data registries for all patients with IBD, instigating certification programmes for IBD units or auditing IBD units for high levels of clinical care, increasing participation of specialised IBD nurses and other multidisciplinary professionals and improving international research collaborations to promote new developments and innovations in the field of IBD. Whilst it may be unrealistic to standardise care for IBD patients between countries throughout Europe, aiming for cohesive care within a nation should not only be achievable, but championed.
