Abstract
Background:
Japanese guidelines recommend triple therapy with vonoprazan or a proton pump inhibitor (PPI) in combination with antibiotics to treat Helicobacter pylori (H. pylori) infection. While studies have shown improved eradication rates and reduced costs with vonoprazan versus PPIs, there is little data describing healthcare resource use (HCRU) and treatment patterns.
Objectives:
To compare patients treated with a vonoprazan-based or PPI-based regimen for H. pylori infection in Japan in terms of their characteristics, HCRU, healthcare costs, clinical outcomes, and treatment patterns.
Design:
Retrospective matched cohort.
Methods:
We used data from the Japan Medical Data Center claims database (July 2014–January 2020) to identify adult patients with H. pylori infection and a first observed use of vonoprazan or a PPI in 2015 or later (index date). Patients prescribed a vonoprazan-based or a PPI-based regimen were matched 1:1 using propensity score matching. HCRU, healthcare costs, diagnostic tests, a proxy for H. pylori eradication (i.e. no triple therapy with amoxicillin in combination with metronidazole or clarithromycin >30 days after the index date), and second-line treatment were described during the 12-month follow-up period.
Results:
Among 25,389 matched pairs, vonoprazan-treated patients had fewer all-cause and H. pylori-related inpatient stays and outpatient visits than PPI-treated patients, resulting in lower all-cause healthcare costs [185,378 Japanese yen (JPY) versus 230,876 JPY, p < 0.001]. Over 80% of patients received a post-treatment test for H. pylori. Fewer vonoprazan-treated than PPI-treated patients subsequently received an additional triple regimen for H. pylori infection (7.1% versus 20.0%, p < 0.001) or a prescription for vonoprazan or a PPI as monotherapy (12.4% versus 26.4%, p < 0.001) between 31 days and 12 months after the index date.
Conclusion:
Patients with H. pylori infection who were treated with vonoprazan-based therapy had lower rates of subsequent H. pylori treatment, lower overall and H. pylori-related HCRU, and lower healthcare costs than patients treated with PPI-based therapy.
Keywords
Introduction
Helicobacter pylori (H. pylori) is a gram-negative bacterium that is classified by the World Health Organization as a Group 1 carcinogen. 1 H. pylori causes chronic inflammation of the gastric mucosa, which, if left untreated, can persist and cause more serious conditions including peptic ulcer disease and gastric adenocarcinoma.1,2 H. pylori is estimated to be responsible for around 89% of non-cardia gastric cancers worldwide and more than 95% in Japan.3,4
The estimated prevalence of H. pylori infection in Japan is less than 30%, 5 and it is typically spread from family members to infants. 6 While prevalence rates have been decreasing over the past few decades, H. pylori infection is highly correlated with the incidence of gastric cancer. Therefore, continued efforts to reduce the rates of infection are of utmost importance. 7 In an attempt to facilitate the prevention of gastric cancer, Japanese health insurance coverage was approved for H. pylori eradication therapy in patients with gastric or duodenal ulcers in 2000. 2 This coverage was subsequently expanded in 2013 to include all patients with H. pylori infection. 2
In Japan, current treatment options for H. pylori infection include either a proton pump inhibitor (PPI) or vonoprazan, given in combination with two antibiotics.6,8 Vonoprazan is a potassium-competitive acid blocker that was approved in Japan in December 2014, with use in Japan starting in 2015. Vonoprazan was approved in 2022 for use in the United States and has shown efficacy over PPI-based therapy in other South Eastern Asian countries.9–11 The PPIs that are available in Japan are omeprazole, lansoprazole, rabeprazole and esomeprazole. 6 Rabeprazole and esomeprazole have been associated with slightly higher eradication rates in clinical trials (77–94%) than earlier PPIs such as omeprazole (75–88%). 12 In contrast to PPIs, vonoprazan is acid-stable and does not require acid for activation, thus producing greater and longer lasting suppression of gastric acid secretion than PPIs. 13 Accordingly, vonoprazan-based therapy has been shown to be superior to PPI-based therapy in both first- and second-line treatment of H. pylori infection, with eradication rates of over 90%.13–15 Notably, vonoprazan-based therapy has demonstrated superior efficacy against clarithromycin-resistant H. pylori strains.13,16
The 2016 Japanese guidelines for the treatment of H. pylori infection recommended a 7-day triple combination of vonoprazan or a PPI with amoxicillin and clarithromycin as first-line treatment. 6 For patients infected with known clarithromycin-resistant strains of H. pylori, metronidazole is recommended in place of clarithromycin. For patients who are truly penicillin-allergic, sitafloxacin or clarithromycin with metronidazole may be used in place of amoxicillin.6,17,18 However, national health insurance only covers vonoprazan or a PPI in combination with amoxicillin and clarithromycin for first-line treatment and replacement of clarithromycin with metronidazole for second-line treatment, although additional regimens may be covered under employer-based plans. 19 Therefore, real-world treatment patterns in clinical practice may differ from guideline recommendations.
Real-world studies have been conducted in Japan to evaluate eradication rates and costs associated with vonoprazan and/or PPIs.2,8,20,21 In one claims-based study, patients treated first line with vonoprazan had significantly higher eradication rates than those treated with a PPI (93.6% versus 79.7%; p < 0.001), resulting in lower total treatment costs [12,952 versus 13,146 Japanese yen (JPY)]. 8 However, there is little data describing and comparing healthcare resource use (HCRU) and treatment patterns between patients treated with a vonoprazan-based or a PPI-based regimen. In addition, the comparative economic impacts of these two approaches have not been well characterized in a routine clinical setting. Therefore, this study was conducted to compare patients treated for H. pylori infection in Japan with a vonoprazan-based or a PPI-based regimen in terms of their characteristics, HCRU, healthcare costs, clinical outcomes and treatment patterns.
Methods
Data source
We used data from the Japan Medical Data Center (JMDC) claims database from July 2014 to January 2020. The JMDC includes employees of companies and their dependents, representing over 7.3 million beneficiaries. Information on treatments, procedures, confirmed and suspected diagnoses, HCRU and costs are available. Most prescription medications (including vonoprazan and PPIs) are covered by the national health insurance system in Japan. Inpatient data were also pulled from the Diagnosis Procedure Combination (DPC) system. In this dataset, diagnoses, HCRU and costs were available on a monthly level, while treatment information and diagnostic tests were available on a daily level. The data were anonymized and not linkable to personal data for privacy protection. Therefore, submission to ethical committees was not required in accordance with the Ethical Guideline of Epidemiological Research in Japan.
Study design
For the analysis of vonoprazan and PPI uptake, we used an unmatched sample of eligible patients to descriptively summarize the number of patients receiving treatment with vonoprazan-based or PPI-based therapy each year from 2015 to 2019. The year 2020 was not included in this descriptive sample since data were only available for January of that year.
For the remaining analyses, we conducted a retrospective, propensity score–matched cohort study among patients with H. pylori infection who initiated a vonoprazan-based or a PPI-based regimen during or after 2015. The index date was the date of the first observed use of vonoprazan or a PPI in 2015 or later, with the encompassing month being defined as the index month. The 6-month period prior to the index date was defined as the baseline period. Given the data availability, variables available on a monthly level were assessed in the six calendar months before the index month (excluding the index month). The 12-month period after and including the index date was defined as the follow-up period. Given the data availability, variables available on a monthly level were assessed in the 12 calendar months including and after the index month.
Study population
Patients were included if they met the following criteria: (1) had a first observed use of vonoprazan or a PPI in 2015 or later, (2) had at least one confirmed diagnosis of H. pylori infection during the baseline period or index month, (3) were aged ⩾18 years on the index date, (4) had continuous insurance enrollment during the baseline and follow-up periods, and (5) had no evidence of gastric malignancy or erosive esophagitis during the baseline period or index month.
Patients with claims for both vonoprazan and a PPI within 2 weeks of the index date were excluded. If only the month but not the exact date of the first vonoprazan or PPI claim was available, the patient was still excluded if both treatments were used in the same month.
Study outcomes
Study outcomes measured during the follow-up period included HCRU, costs and clinical outcomes (i.e., diagnostic tests, second-line treatment). HCRU and healthcare costs (in 2020 JPY) comprised all-cause and H. pylori-related inpatient/DPC and outpatient visits and gastroenterologist visits. All-cause costs included pharmacy costs. H. pylori-related HCRU and healthcare costs were identified as claims with a confirmed or suspected H. pylori diagnosis code.
Since post-treatment test results for confirmation of eradication were not available, we used a proxy to represent H. pylori eradication. This was defined as the patient receiving no subsequent prescriptions for triple therapy including amoxicillin with either metronidazole or clarithromycin >30 days after the index date. Second-line treatment was defined as the first vonoprazan or PPI prescription claim filled between 31 days after the index date and the end of follow-up, including the antibiotics filled within 14 days of that claim. In addition, time from the index date to initiation of second-line treatment was reported.
Statistical analysis
Patients receiving a vonoprazan-based regimen were matched 1:1 to patients receiving a PPI-based regimen using propensity score matching. We created a propensity score model with greedy-match algorithm using a logistic regression model comprising age at index date, sex, index year, care setting, smoking status, Charlson comorbidity index, H. pylori-related symptoms and clinical characteristics, any H2-receptor antagonist use, any antibiotic use, any gastroenterologist visit, and any H. pylori-related inpatient/DPC or outpatient visits during baseline. The success of the propensity score model in balancing characteristics between the patients receiving a vonoprazan-based regimen and a PPI-based regimen was assessed by examining the standardized mean difference, which, if less than 0.02, would indicate that the characteristics were well balanced. Demographics and characteristics of patients treated with either a vonoprazan-based or a PPI-based regimen were described using medians, means and standard deviations, and were compared using Wilcoxon rank-sum tests for continuous variables. The Wilcoxon rank-sum test was used rather than a t-test since the former is less sensitive to outliers. Patients treated with either a vonoprazan-based or a PPI-based regimen were described using counts and proportions and compared using chi-squared tests for categorical variables. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 22
Results
Uptake of vonoprazan in unmatched sample since approval
Since the introduction of vonoprazan in 2015, the number of patients in our sample who were prescribed it steadily increased from 6594 in 2015 to 28,956 in 2019. Correspondingly, the number of patients prescribed a PPI decreased from 11,238 in 2015 to 2629 in 2019 (Figure 1).

Vonoprazan versus PPI uptake for the treatment of H. pylori in Japan in the unmatched sample.1
Characteristics of matched vonoprazan-treated and PPI-treated patients
We identified 1,454,4497 individuals who used vonoprazan or a PPI from 2010 to 2020. Among 130,732 patients with H. pylori infection who met the sample selection criteria before propensity score matching, 100,701 (77.0%) were treated with a vonoprazan-based regimen at the index date and 30,031 (23.0%) with a PPI-based regimen. There were 25,389 matched pairs after propensity score matching.
Matched patients who received a vonoprazan-based or a PPI-based regimen were similar as indicated by a standardized mean difference <0.2 (Table 1). The mean age in both cohorts was approximately 51 years and about 59% were male. The most common H. pylori-related indication was gastritis (38.1% of vonoprazan-treated patients and 39.5% of PPI-treated patients). While all patients had a diagnosis code for H. pylori infection during the baseline period, 81.5% of vonoprazan-treated patients and 77.4% of PPI-treated patients also received a diagnostic test for H. pylori during baseline. Few patients received treatment with an antibiotic (12.4% and 13.0%) or had an H. pylori-related outpatient visit (25.3% and 26.5%) during the baseline period.
Baseline a patient characteristics for matched vonoprazan-treated and PPI-treated patients in Japan.
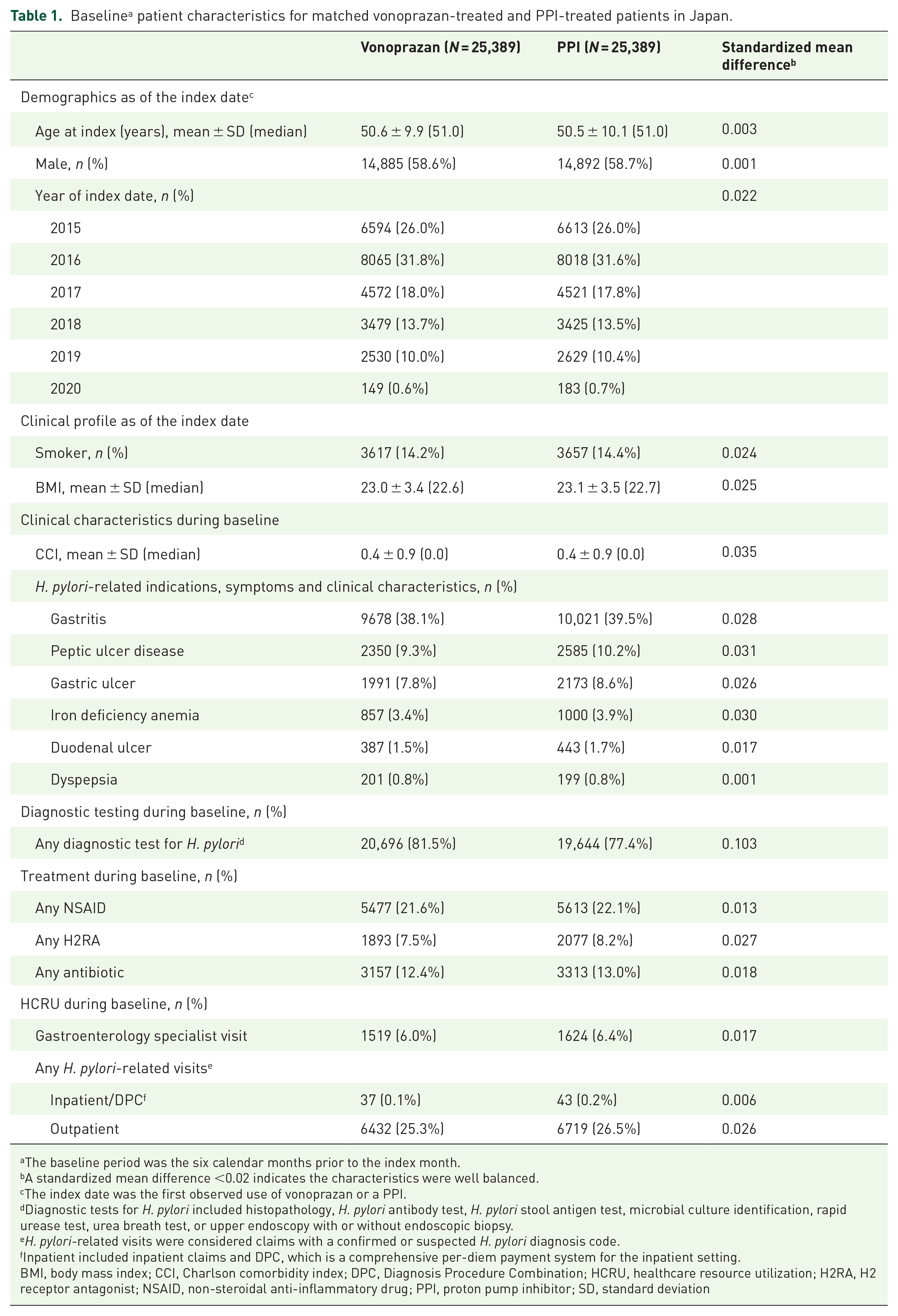
The baseline period was the six calendar months prior to the index month.
A standardized mean difference <0.02 indicates the characteristics were well balanced.
The index date was the first observed use of vonoprazan or a PPI.
Diagnostic tests for H. pylori included histopathology, H. pylori antibody test, H. pylori stool antigen test, microbial culture identification, rapid urease test, urea breath test, or upper endoscopy with or without endoscopic biopsy.
H. pylori-related visits were considered claims with a confirmed or suspected H. pylori diagnosis code.
Inpatient included inpatient claims and DPC, which is a comprehensive per-diem payment system for the inpatient setting.
BMI, body mass index; CCI, Charlson comorbidity index; DPC, Diagnosis Procedure Combination; HCRU, healthcare resource utilization; H2RA, H2 receptor antagonist; NSAID, non-steroidal anti-inflammatory drug; PPI, proton pump inhibitor; SD, standard deviation
Most matched patients were prescribed a guideline-recommended first-line regimen (i.e., either vonoprazan or a PPI with amoxicillin and clarithromycin). Significantly more vonoprazan-treated patients than PPI-treated patients were managed according to the guideline (94.7% versus 85.1%, respectively, p < 0.001). Of note, 1.8% of vonoprazan-treated patients and 10.9% of PPI-treated patients did not have evidence of antibiotic use in the 2-week window around vonoprazan or PPI initiation (i.e., they were prescribed either drug as monotherapy, p < 0.001).
HCRU of matched vonoprazan-treated and PPI-treated patients
During the 12-month follow-up period, vonoprazan-treated patients had slightly fewer all-cause inpatient stays (mean: 0.07 versus 0.10, p < 0.001) and fewer all-cause outpatient visits (mean: 12.2 versus 13.0, p < 0.001; Table 2) than PPI-treated patients. Similarly, vonoprazan-treated patients had slightly fewer H. pylori-related inpatient stays (mean: 0.007 versus 0.011, p < 0.001) and had fewer H. pylori-related outpatient visits (mean: 3.8 versus 4.4, p < 0.001; Table 2) than PPI-treated patients. Visits to gastroenterologists were slightly more common among vonoprazan-treated than PPI-treated patients (mean: 0.49 versus 0.46, p < 0.001).
HCRU and H. pylori diagnostic testing during follow-up of matched vonoprazan-treated and PPI-treated patients. a

The follow-up period was the 12 calendar months after and including the index month.
Inpatient included inpatient claims and DPC, which is a comprehensive per-diem payment system for the inpatient setting.
H. pylori-related visits were considered claims with a confirmed or suspected H. pylori diagnosis code.
Diagnostic tests for H. pylori included histopathological sample, H. pylori antibody test, H. pylori stool antigen test, microbial culture identification, rapid urease test, urea breath test, 24-h intragastric/intraesophageal pH measurement, or upper endoscopy with or without endoscopic biopsy.
DPC, Diagnosis Procedure Combination; HCRU, healthcare resource utilization; PPI, proton pump inhibitor; SD, standard deviation
Healthcare costs of matched vonoprazan-treated and PPI-treated patients
Mean total all-cause healthcare costs were statistically significantly lower for patients treated with vonoprazan than for those treated with a PPI (185,378 ± 456,470 (median: 94,262) JPY versus 230,876 ± 689,312 (median: 101,988) JPY, p < 0.001; Figure 2). Mean costs of visits to gastroenterologists were also lower for patients treated with a vonoprazan-based regimen than for those treated with a PPI-based regimen [8194 ± 62,049 JPY versus 10,199 ± 107,213 JPY (both medians: 0), p < 0.001]. Similar comparative trends were observed for H. pylori-related costs (Figure 2).

Clinical outcomes of matched vonoprazan-treated and PPI-treated patients
Most patients received a diagnostic test for H. pylori during the follow-up period (86.3% of vonoprazan-treated patients and 81.8% of PPI-treated patients; Table 2), within a mean of 79 days from the index date to the first post-index test in both cohorts.
Fewer vonoprazan-treated than PPI-treated patients subsequently received a prescription for triple therapy between 31 days and 12 months after the index date (7.1% versus 20.0%, respectively, p < 0.001; Figure 3(a)). In addition, fewer vonoprazan-treated than PPI-treated patients received a prescription for vonoprazan or a PPI as monotherapy between 31 days and12 months after the index date (12.4% versus 26.4%, p < 0.001; Figure 3(b)). The mean time from the index date to second-line treatment initiation was 121.5 days for vonoprazan-treated patients and 103.7 days for PPI-treated patients.
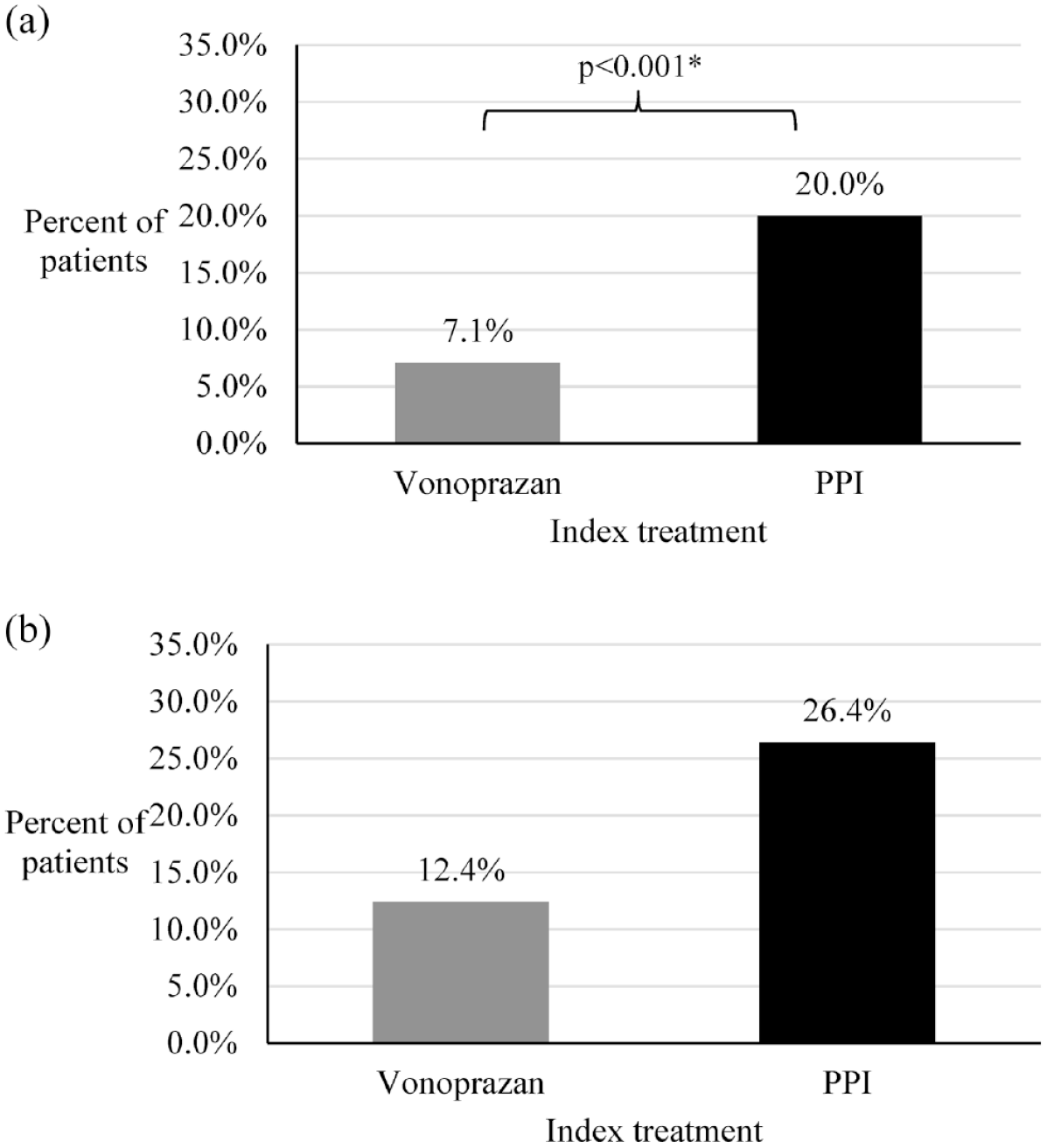
Subsequent prescriptions following first-line treatment. (a) Subsequent triple therapy1 31 days to 12 months after index date.2 (b) Subsequent vonoprazan or PPI prescription3 31 days to 12 months after index date. 2
Discussion
This retrospective matched cohort study used a large sample from closed Japanese insurance claims data to compare vonoprazan-based and PPI-based H. pylori eradication therapy in routine clinical practice. Patients diagnosed with H. pylori infection and treated with a vonoprazan-based regimen had lower rates of subsequent H. pylori treatment, lower overall and H. pylori-related HCRU, and lower healthcare costs than patients treated with a PPI-based regimen. These findings contribute to the growing literature demonstrating the clinical and economic benefits of vonoprazan-based therapy for H. pylori eradication.
The prevalence of H. pylori infection in Japan has decreased in the last few decades, largely due to improved socioeconomic standards and sanitary conditions.7,23 Furthermore, this decline has been accelerated by the expansion of Japanese health insurance to cover H. pylori-positive gastritis in 2013. 23 The success rate of eradication has also increased since 2015, which coincided with the launch of vonoprazan. 2 Indeed, a positive correlation was observed between success rate and the proportion of patients treated with vonoprazan (R2 = 0.9764), suggesting that increasing use of vonoprazan is related to higher efficacy in the treatment of H. pylori infection. The rapid uptake of vonoprazan 2 since its approval is also corroborated by the current study findings. Despite the decreasing prevalence of H. pylori infection in Japan, nearly 30% of the population was infected in 2016, 5 signaling a need for continued national eradication efforts.
Since direct reporting of the results of post-treatment tests of H. pylori status was not available in the JMDC database, we examined the use of subsequent triple therapy for H. pylori infection as a proxy for eradication. A separate, claims-based study by Tokunaga et al used a similar proxy for H. pylori eradication, defined as patients who completed any diagnostic test after first-line therapy and who had not been prescribed any second-line therapy. 8 Based on their findings, vonoprazan-based regimens used first-line were associated with significantly higher eradication rates than PPI-based regimens (93.6% versus 79.7%; p < 0.001). These rates are very similar to our results based on subsequent prescription of a triple regimen (i.e., 7.1% versus 20.0%, corresponding to estimated eradication rate of 92.9% and 80.0% among vonoprazan-treated and PPI-treated patients, respectively).
The benefit of vonoprazan-based over PPI-based eradication therapy has been consistently demonstrated. In a network meta-analysis of randomized controlled trials (RCTs), comparative effectiveness ranking showed that vonoprazan triple therapy was the most efficacious of all the eight first-line treatments that were considered, with 3.8-times higher odds of eradication compared to PPI-based triple therapy [odds ratio (OR; credible interval (CrI)) = 3.80 (1.62, 8.94)]. 14 A separate meta-analysis of RCTs found that vonoprazan-based triple therapy was associated with higher relative efficacy than PPI-based triple therapy [OR (95% CrI) = 2.73 (2.11, 3.54)] and bismuth subcitrate quadruple therapy [OR (95% CrI) = 1.60 (1.07, 2.38)] for the eradication of H. pylori. 24 In addition, vonoprazan-based triple therapy had a 72.1% probability of being the most efficacious compared to the other dual, triple and quadruple regimens that were considered. These trends have been upheld in real-world clinical practice; one retrospective, hospital-based study demonstrated that first-line PPI-based therapy had significantly lower odds of eradication than vonoprazan-based therapy [OR (95% confidence interval (CI)) = 0.28 (0.23, 0.33)]. 21 Of note, the benefit associated with vonoprazan was also extended to second-line treatment [OR (95% CI) = 0.71 (0.56, 0.89)]. Our results add to the growing body of evidence demonstrating improved H. pylori eradication rates with vonoprazan-based treatment compared to PPI-based treatment.
This study has also demonstrated significant reduction in HCRU with associated cost savings among patients treated with vonoprazan-based rather than PPI-based therapy. These findings are consistent with those of the claims-based study by Tokunaga et al, which found that costs per patient for first- and second-line treatments were approximately 200 JPY lower with vonoprazan-based than PPI-based therapy (12,952 versus 13,146 JPY, respectively) because of higher eradication rates with the first-line use of vonoprazan. 8 Since Tokunaga et al evaluated different cost components (i.e., first- and second-line medication, diagnostic test, and subsequent visit), 8 their results are not directly comparable to ours, although the trend is similar. Of note, we showed reductions in both all-cause and H. pylori-related HCRU and costs, suggesting that the economic benefit of vonoprazan may extend beyond the costs of treatment.
Despite the higher cost of vonoprazan over PPIs, its use may lower total healthcare costs due to more reliable eradication of H. pylori infection. 8 A cost-effectiveness analysis by Kajihara et al demonstrated that vonoprazan-based triple therapy was more cost-effective than rabeprazole-based triple therapy, with an incremental cost-effectiveness ratio of 147 JPY per percent difference in eradication rate. 20 However, Seko et al found no difference between vonoprazan- and lansoprazole-based eradication therapy. 25 Further research is therefore warranted to characterize more fully the comparative economic impacts of vonoprazan-based and PPI-based eradication therapy.
Limitations
Our findings are subject to some limitations. Since the JMDC database is restricted to beneficiaries employed by companies and their dependents, our results may not be generalizable to the broader Japanese population. Although we used propensity score matching to account for differences in key baseline characteristics between the two cohorts, results may have been subject to residual confounding due to unmeasured characteristics. Administrative claims databases may be subject to data omissions or coding inaccuracies. For example, not all patients had a diagnostic test for H. pylori infection during the baseline period despite H. pylori positivity being a requirement for reimbursement in Japan. Also, some variables of interest, such as reasons for treatment selection, were not available in the claims data. Lastly, records of pharmacy claims do not specify whether the prescription was written solely to treat H. pylori infection and do not guarantee that patients took the medication as prescribed.
Since some information, such as HCRU and costs, was only available at the month level, we could not assess the exact timing of these events (i.e., before or after index). Furthermore, the index month was not included in the baseline estimates but was included in the follow-up period estimate; therefore, some of the pre-index HCRU and costs may have been captured in the follow-up period instead. Lastly, results of post-treatment tests for H. pylori infection were not available in claims data. Therefore, confirmed eradication rates could not be determined using these data. However, the high rate of diagnostic testing after first-line therapy combined with the lower subsequent rate of prescriptions among those who received vonoprazan suggests that more in that group than in the PPI-treated group achieved successful eradication.
Conclusion
Vonoprazan-based therapy had lower total healthcare costs and HCRU than PPI-based therapy in the 12 months after treatment initiation. Based on prescription data for subsequent treatments, H. pylori eradication rates may have been higher with vonoprazan-based regimens, consistent with the results of comparative clinical trials from Japan and elsewhere in Asia. Future studies should explore the health consequences and direct and indirect costs of patients with H. pylori infection that was not eradicated by initial therapy.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231168714 – Supplemental material for Real-world outcomes associated with vonoprazan-based versus proton pump inhibitor-based therapy for Helicobacter pylori infection in Japan
Supplemental material, sj-docx-1-tag-10.1177_17562848231168714 for Real-world outcomes associated with vonoprazan-based versus proton pump inhibitor-based therapy for Helicobacter pylori infection in Japan by Colin W. Howden, Erin E. Cook, Elyse Swallow, Karen Yang, Helen Guo, Corey Pelletier, Rinu Jacob and Kentaro Sugano in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
