Abstract
Background:
Treatment with antitumor necrosis factor alpha (anti-TNF-α) is safe and effective as first-line therapy; however, its efficacy is limited due to primary nonresponse (PNR) and secondary loss of response (LOR), resulting in treatment discontinuation in approximately 40%–50% of cases. Vedolizumab (VDZ) and ustekinumab (UST) therapies could be good alternatives in patient with anti-TNF failure; however, no head-to-head randomized comparison of these drugs as second- or third-line treatments has been made.
Objectives:
This study aimed to assess the treatment persistence and comparative effectiveness of UST and VDZ in patients with refractory Crohn’s disease (CD).
Design:
In this nationwide retrospective study, patients with CD on UST or VDZ maintenance therapy were enrolled. Clinical data at baseline, after induction, and at week 52 were obtained.
Methods:
Clinical and biochemical activities as well as corticosteroid-free remission (SFR) rates were assessed, while concomitant medications, comorbidities, hospitalizations, and surgeries were recorded during the follow-up to detect any predictors.
Results:
A total of 161 UST- and 65 VDZ-treated patients completed the follow-up. No significant difference in clinical or biochemical remission rates was observed after induction between the two treatment groups; however, clinical remission rate at week 52 was higher in UST group. UST showed superior drug persistence than VDZ (86.5%, 57.9%, p < 0.0001). The drug type was predictive of clinical SFR at week 52 [p = 0.011, odds ratio (OR) = 2.39 with UST]. Drug failure rates were higher for VDZ than those for UST (PNR rates: 21.54% and 4.97%, respectively, p < 0.001, OR = 8.267, p = 0.001). LOR and escalations were more common during UST treatment (61.5% versus 36.9%, p < 0.001; 64.2% versus 23.1%, p < 0.001). Hospital and surgical admission rates did not differ significantly. Only one adverse event occurred with VDZ at week 20, which led to drug cessation.
Conclusions:
VDZ and UST were safe and effective for treating patients with CD in whom anti-TNF therapy failed. UST showed superior drug persistence than VDZ, but dose escalation was more frequent. Biologicals used in lower treatment lines resulted in better drug persistence.
Introduction
Inflammatory bowel disease (IBD), Crohn’s disease (CD), and ulcerative colitis (UC) are chronic inflammatory conditions of the gastrointestinal tract. 1 Increased understanding of the immunological pathways underlying these diseases has led to new therapeutic options, but the exact pathological mechanisms remain unknown. The use of biological therapies for IBD has revolutionized the treatment of moderate to severe disease.2,3 The first approved molecular targeted antibody, which was against tumor necrosis factor alpha (TNF-α), was infliximab (IFX) introduced in 1998 by the US FDA. Later, other drugs targeting TNF showed proven efficacy and safety in patients with moderate to severe IBD. 4 However, approximately 30% of patients fail to achieve an initial response to anti-TNF therapy because of primary nonresponse (PNR).5,6 Furthermore, 25%–45% of patients need to stop using anti-TNF agents because of loss of response (LOR) in the first year. 7 Besides primary and secondary LOR, infusion-related adverse events also limit the long-term use of anti-TNF agents.8,9 Vedolizumab (VDZ) and ustekinumab (UST) are newer therapeutic options in patients with anti-TNF failure. Although these drugs provide alternative therapeutics with different mechanisms of action, evidence indicates that the probability of treatment success declines with each line of treatment. 10 The increase in available therapeutic options has broken down the long-standing barrier that limited randomized head-to-head comparisons of the efficacy and safety of different biological therapies. Recently, two head-to-head trials comparing the efficacies of adalimumab (ADA) versus VDZ and of ADA versus UST in patients with moderate to severe UC and CD were published.11,12 However, these trials did not answer the question of whether or not integrin inhibition or IL12/23 blockade is the better therapeutic option after anti-TNF failure. Reviewing the results of two baseline studies, GEMINI versus CERTIFI, conducted nearly 10 years earlier does not help in estimating odds because both concluded that patients with moderate to severe CD who were resistant to TNF antagonists had an increased rate of response to induction with VDZ/UST relative to that with placebo. Patients with an initial response to both agents had significantly increased rates of response and remission as maintenance therapy.13–15
Although physicians in many countries use a hierarchical prescribing order requiring prescribing one or two anti-TNF agents prior to using newer biological agents, real-life effectiveness usually override study efficacies. In Hungary, at the time of the current study, both VDZ and UST could be prescribed as second- or third-line treatments after one or two anti-TNF failures or as first-line treatments if individual permission was obtained by the National Health Insurance Fund of Hungary. Therefore, switching to a newer biological agent with a different molecular target may be delayed by months. Moreover, we are not able to predict treatment success, so the choice of the drugs in the second-, third-, or even fourth-line mainly depends on the local practice or central regulations of the country. Although clinical experience with both biologicals continues to grow, predicting the effectiveness of therapies in terms of baseline disease severity and comorbidities is currently difficult, although defining the ideal patient profile is expected to significantly improve efficacy and safety. Consequently, more real-life data are needed to reveal easily determinable and routinely available clinical or laboratory parameters to predict the success of therapy and to help select the best treatment choice for each patient.
The primary study aim was to compare the effectiveness and treatment persistence of VDZ and UST in patients with CD who had mostly failed anti-TNF treatment in a real-life setting. The secondary aim was to identify the predictive factors and comorbidities associated with treatment success and persistence in patients who used both agents.
Materials and methods
Study design and data source
This was a retrospective multicentre cohort study that included five tertiary referral IBD centers in Hungary. The patients’ data were collected consecutively from the medical records systems and stored in a uniform database. Data were obtained at induction of both agents, at 16–20 weeks after induction and at week 52. Patients ⩾18 years old with an established diagnosis of CD who were receiving VDZ or UST therapy because of any contraindication of first-line anti-TNF agents or after failure of one or two anti-TNF agents were enrolled in each participating center. Patients were treated with UST or VDZ according to the current regulations of the National Health Insurance Fund of Hungary according to a summary of the products’ characteristics. The patients received intravenous VDZ treatment with an induction regimen of 300 mg at weeks 0, 2, and 6 and received maintenance therapy with the same dose after induction at every 8 weeks. An escalated regimen because of LOR was applied every 4 weeks. UST therapy was initiated with a weight-based intravenous infusion at baseline according to the following specifications (260 mg for <55 kg, 390 mg for 55–85 kg, and 520 mg for >85 kg). The first subcutaneous 90-mg induction dose was administered at week 8 followed by a subsequent maintenance dose of 90 mg administered subcutaneously at every 4–12 weeks depending on disease activity. Enrolled patients provided written informed consent for both treatments.
Second-, third-, and fourth-line therapies were interpreted as the application of a different drug after the first, second, and third switch from the first-line biological agent, respectively.
Baseline parameters and outcomes
Clinical and demographic baseline data, including sex, age at diagnosis, disease duration, location (using the Montreal classification), 15 disease behavior (using the Montreal classification 15 ), perianal phenotype, prior resections and extraintestinal manifestations (EIMs), were collected. Data regarding the period of the prior anti-TNF treatment were also obtained. If patients received both UST and VDZ treatments, data were obtained only until the first treatment. Patients with incomplete follow-up and no anti-TNF treatment were excluded.
At baseline, data, including UST and VDZ treatment lines, clinical activity based on the Clinical Disease Activity Index (CDAI), 16 biochemical activity based on C-reactive protein (CRP) level, concomitant corticosteroid or thiopurine therapy and EIMs, were recorded. Clinical and biochemical activity, concomitant medications, EIMs, and perianal symptoms were obtained at 16–20 weeks after induction and at the end of follow-up at week 52. If the drug was terminated after induction, a PNR was classified between weeks 16 and 20 because of the lack of efficacy. A LOR was defined as the occurrence of disease activity that led to treatment termination during the maintenance phase after effective induction or an escalated regimen was applied during treatment. EIMs and perianal complaints were evaluated based on both patients’ and physicians’ assessments using improved and not improved binary categories. The primary outcome was defined as a corticosteroid-free composite remission (SFR) after the induction at weeks 16–20 and at week 52. Remission was defined by clinical (CDAI < 150) and biochemical (CRP < 10 mg/L)17,18 activities. The primary outcome was used to identify the possible predictors of 1-year efficacy of both agents. The secondary outcome was defined as drug persistence at week 52.
Comorbidities were recorded and the Charlson comorbidity index (CCI), including age, myocardial infarction, chronic heart failure, peripheral vascular diseases, cerebrovascular accidents and/or transient ischemic attack, dementia, chronic obstructive pulmonary disease (COPD), connective tissue disease, peptic ulcer disease, liver diseases, diabetes mellitus, hemiplegia, chronic kidney injury, solid tumor, leukemia, lymphoma, and acquired immune deficiency syndrome, was calculated at baseline. The CCI is a weighted index that is being validated for stratifying risk of comorbid conditions. 19 The CCI could range from a minimum of zero to a maximum of 33 points and was used to assess the effect of comorbidities on drug survival in combination with biological agents.
Hospitalization rates, surgical procedures, immunomodulator, and/or corticosteroid necessity during the follow-up due to disease activity were also obtained. The escalated regimens defined above were also recorded in case both agents were used.
Statistical analysis
Statistical tests were performed using R statistical software version 4.1.1 (R Foundation, Vienna, Austria) and IBM SPSS software (IBM SPSS Statistics for Windows, Version 26.0, IBM Corp., Armonk, NY, USA). Descriptive statistics were presented as the mean ± standard deviation of the mean (SD) or median + interquartile range (IQR) for continuous variables and as the count + percentage for categorical variables. Normality was tested by visual interpretations. Continuous variables were tested via the Welch test or Mann–Whitney U-test for independent samples to compare differences between groups (after assumptions checked in cases of each test), whereas categorical variables were analyzed using the chi-squared test and Fisher’s exact test to compare proportions of groups. Kaplan–Meyer analysis was performed to describe the persistence of a therapeutic effect of both agents. After identification of the possible predictive factors of primary outcomes, PNR and LOR multivariate logistic regression models were constructed (overall model fit was assessed by the Nagelkerke R 2 and goodness of fit was determined by performing the Hosmer–Lemeshow test). A multivariable Cox proportional hazards model was created to assess the effect of the CCI and type of any drug-on-drug survival. Continuity of the hazard ratio (HR) over time was tested. Values of p < 0.05 were considered to be indicative of statistical significance.
Results
Baseline characteristics
In all, 200 UST- and 96 VDZ-treated patients were included in the study. Finally, after excluding data for sequential treatments with UST and VDZ, incomplete follow-up data, and data of patients without any prior anti-TNF treatment, the data of 226 patients were considered eligible for analysis (Figure 1).

Flowchart of exclusion depending on prior UST or VDZ treatment, follow-up period or no prior anti-TNF exposure.
In the UST and VDZ cohorts, 161 and 65 patients were eligible for analysis, respectively (median age: 36 and 35 years, male/female ratio: 57/104 and 21/44, median treatment duration: 13.3 and 12.0 years, respectively). There were no differences in the baseline demographic data between the UST and VDZ cohorts. Among the baseline clinical characteristics of the patients, perianal disease phenotype was significantly more common in the UST cohort than in the VDZ cohort (44.7% and 24.6%, p < 0.001), respectively. Differences in treatment level, disease localization and behavior, disease activity parameters, prior bowel resections, and concomitant medications were not observed between the two groups at baseline. The most relevant extraintestinal symptom was arthritis in each group; however, neither the prevalence of EIMs nor any of the other baseline parameters differed significantly between the treatment groups. Prior IFX treatment was terminated in 61 UST- and 38 VDZ-treated patients due to LOR (37.9% versus 58.5%, p = 0.004), and in case of 19.3% versus 12.3% due to PNR (p = 0.21). Prior IFX exposure resulted in cessation in 23.6% and 7.7% (p = 0.005) in the UST and VDZ treatment groups due to infusion-associated reactions. In 9.9% of UST- and 13.9% of VDZ-treated patients’ (p = 0.40) prior IFX treatment was terminated due to side effects. IFX treatment was stopped due to pregnancy in 3.1% and 4.6% (p = 0.58) of patients in the UST and VDZ cohorts. In patients treated with IFX previously, 6.2% in the UST and 3.1% in the VDZ groups (p = 0.34), the cessation of therapy was due to other patient associated causes. Prior ADA was ceased due to LOR in 48.0% and 61.5% (p = 0.075) in the UST and VDZ groups, respectively. In 18.6% and 12.3% (p = 0.25) of patients in UST and VDZ cohorts with prior ADA treatment was terminated due to PNR. In addition, ADA treatment was stopped due to treatment-associated reactions in 13.0% and 10.8% (p = 0.64) in the UST and VDZ, respectively, groups. Previous ADA therapy was terminated due to side effects in 7.5% and 4.6% (p = 0.44) in the UST and VDZ cohorts. Pregnancy resulted in cessation of ADA therapy in 3.7% and 6.2% (p = 0.42) of patients in UST and VDZ groups. While other patient-associated termination occurred in 9.3% and 4.6% (p = 0.24) in the UST and VDZ groups. The baseline demographic and clinical characteristics of the cohorts are summarized in Table 1.
Baseline clinical and demographic characteristics of the patients.
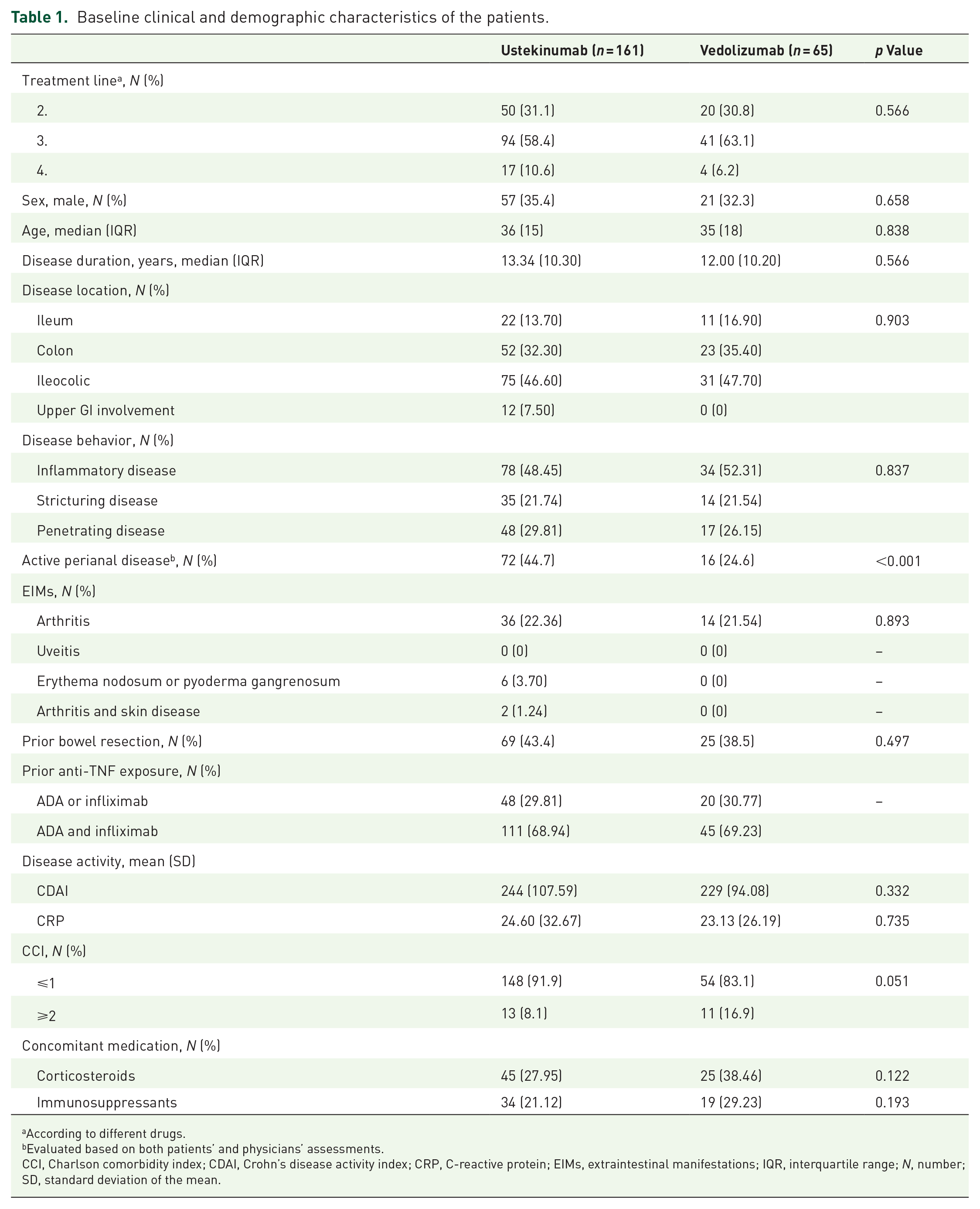
According to different drugs.
Evaluated based on both patients’ and physicians’ assessments.
CCI, Charlson comorbidity index; CDAI, Crohn’s disease activity index; CRP, C-reactive protein; EIMs, extraintestinal manifestations; IQR, interquartile range; N, number; SD, standard deviation of the mean.
Comorbidity prevalence and CCI at baseline
Data of 226 patients were available for assessing CCI at baseline. In all, 19 patients (8.4%) had at least one comorbidity of which 11 (57.9%) were on UST and 8 (42.1%) were on VDZ therapy (p = 0.179).
In the cohort of patients with at least one comorbidity, four patients were >60 years old (21.05%), 5 were between 40 and 60 years old (26.32%), and 10 were <40 years old (52.63%). Of the four patients >60 years old, three were on VDZ treatment. Males were numerically more common in the comorbidity group than in the noncomorbidity group; however, statistical significance was not measured (52.6% versus 32.9%, p = 0.083). There were no significant differences between groups according to the age, median disease duration, and follow-up time. The disease location was statistically similar in both groups (p = 0.982). The inflammatory phenotype was more common in patients with comorbidities than in those without comorbidities (73.7% versus 47.3%), whereas penetrating disease was less frequent in patients with comorbidities (5.3% versus 30.9%) (p = 0.04). There was no difference in the baseline disease activities between the comorbidity cohorts (CDAI p = 0.80, CRP p = 0.837). The baseline characteristics of patients with and without comorbidities are presented in Table 2.
Comorbidity characteristics at baseline.

At baseline.
CDAI, Crohn’s disease activity index; CRP, C-reactive protein; IQR, interquartile range; N, number; SD, standard deviation of mean.
Comorbidities did not differ significantly between UST and VDZ groups (6.79% versus 12.3%, p = 0.179). There was a significant difference in the CCI between the VDZ and UST cohorts. More patients had a ⩾1 higher score in the VDZ cohort than in the UST cohort (p = 0.046). CCI diseases were numerically higher in the VDZ group than those in the UST group (16.9 versus 7.4, p = 0.059). The data referring comorbidities are presented in Tables 2 and 3. The CCI score was not associated with drug persistence in our model (CCI HR = 0.977, p = 0.874).
Comorbidity prevalence at baseline.

CCI item scores.
AIDS, acquired immune deficiency syndrome; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; N, number; TIA, transient ischemic attack.
SFR and therapy persistence
The clinical SFR rate was higher in the UST group than that in the VDZ group at week 52 (59.46 versus 39.68, p = 0.008); however, neither biochemical SFR rates at week 52 (40.13 versus 34.92, p = 0.48) nor SFR rates after the induction period differed significantly between the two groups (clinical: UST 56.94% versus VDZ 46.30%, p = 0.18; biochemical UST 45.26% versus VDZ 41.81%, p = 0.66). Coupling data are presented in Tables 4 and 5, and Figures 2 and 3 summarize these results. UST-treated patients were more likely to remain on therapy than VDZ-treated patients [UST: 86.5%, 95% confidence interval (CI) = 81.2%–92.0%; VDZ: 57.9%, 95% CI = 46.9%–71.3%; p < 0.0001] (Figure 4).
Nonparametric tests on corticosteroid-free remission at week 52 using CDAI.

Number of patients to assess data.
CCI, Charlson comorbidity index; CDAI, Crohn’s disease activity index; CRP, C-reactive protein; N, number of; SD, standard deviation of the mean; SFR, corticosteroid-free remission; UST, ustekinumab; VDZ, vedolizumab.
Nonparametric tests on corticosteroid-free remission at week 52 using CRP.

Number of patients to assess data.
CCI, Charlson comorbidity index; CDAI, Crohn’s disease activity index; CRP, C-reactive protein; N, number of; SD, standard deviation of the mean; SFR, corticosteroid-free remission; UST, ustekinumab; VDZ, vedolizumab.
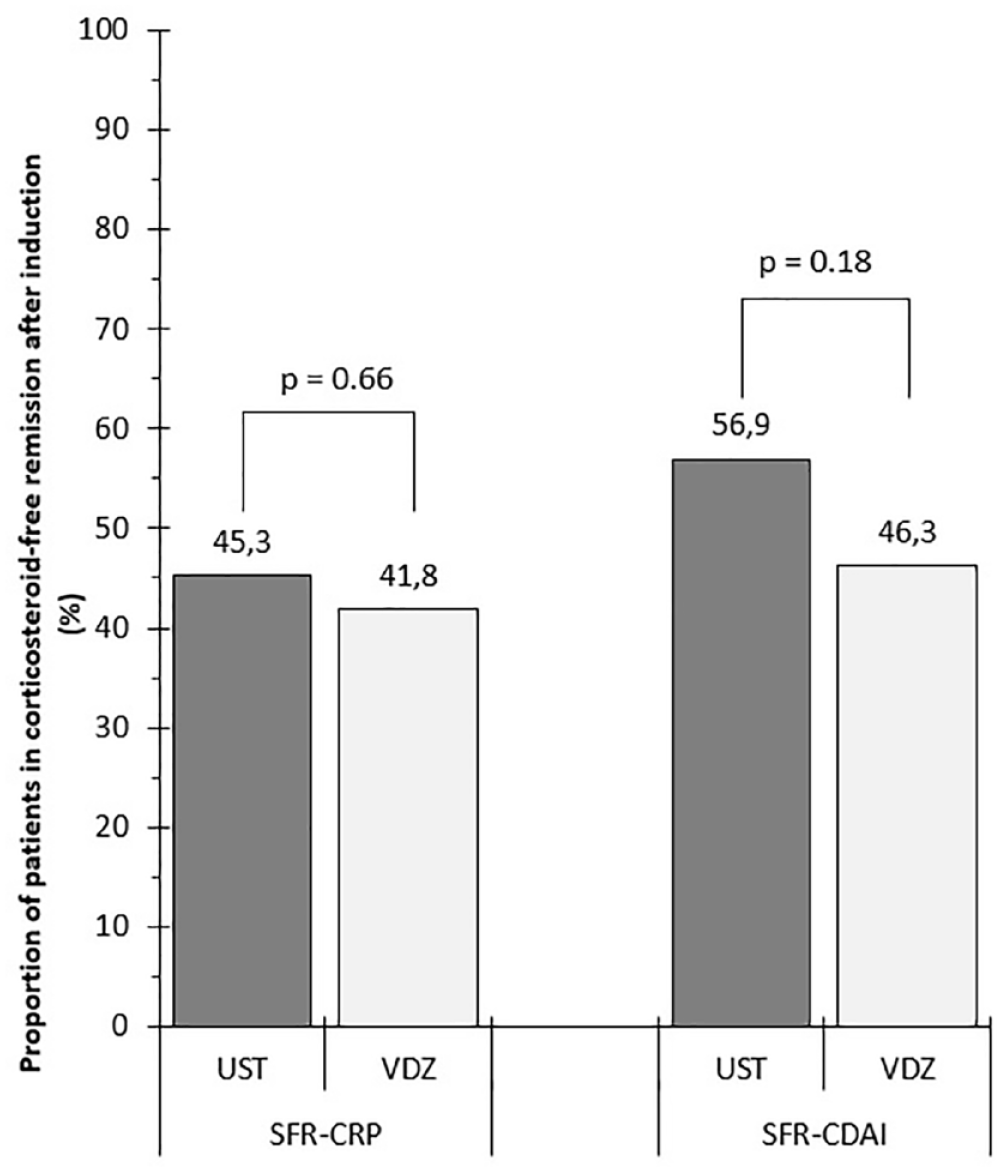
Proportion of patients achieving SFR after the induction period (16–20 weeks) based on clinical (CDAI < 150) and biochemical (CRP < 10 mg/L) parameters.
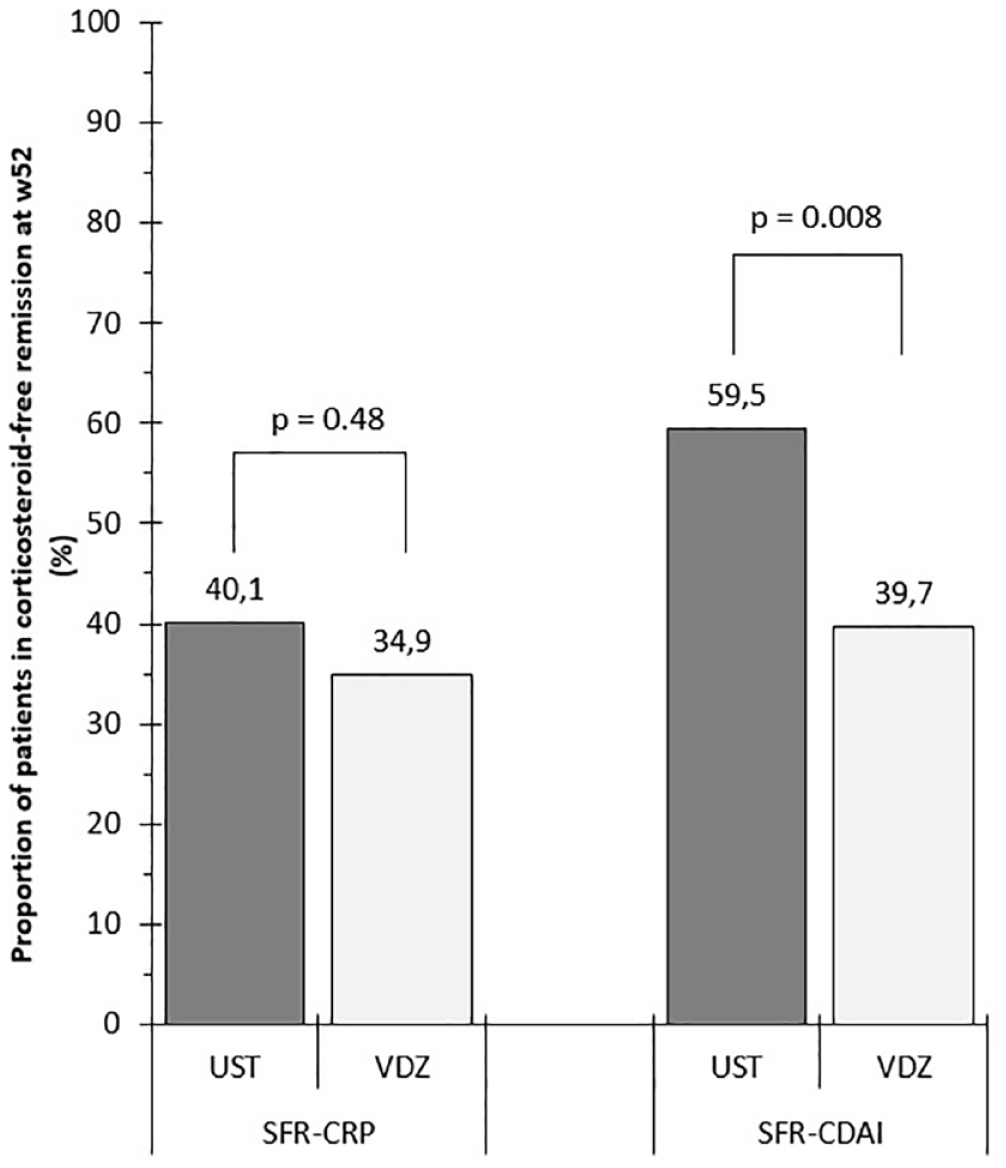
Proportion of patients achieving SFR at week 52 based on clinical (CDAI < 150) and biochemical (CRP < 10 mg/L) parameters.

Kaplan–Meyer survival analysis showed the superiority in drug persistence of UST- versus VDZ-treated group (p = 0.001).
Predictors of SFR
In addition to the lines of treatment, the possible predictors of 52-week SFR were age, baseline CDAI and CRP levels, and CCI score. The possible effect of the drug type on achieving clinical remission was suggested (p = 0.008); however, differences in biochemical outcomes were not detected (p = 0.477). Differences between the groups who did and did not achieve SFR according to sex, comorbidity, combined immunomodulatory therapy, escalated regimen, and disease duration were not significant. Further details containing the other possible predictors that were excluded are summarized in Tables 5 and 6.
Multivariate logistic regression model for prediction of clinical (CDAI) SFR at week 52.
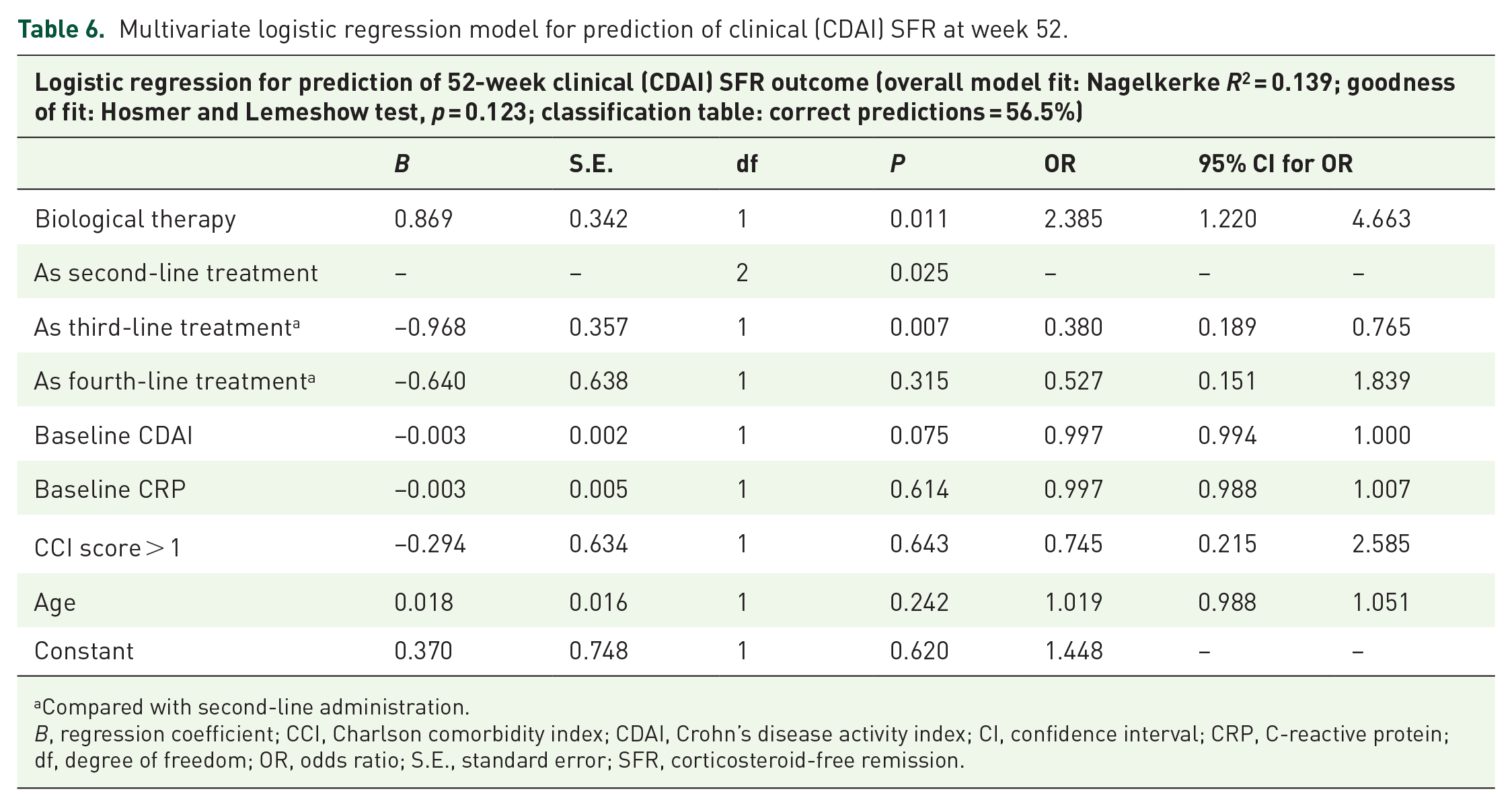
Compared with second-line administration.
B, regression coefficient; CCI, Charlson comorbidity index; CDAI, Crohn’s disease activity index; CI, confidence interval; CRP, C-reactive protein; df, degree of freedom; OR, odds ratio; S.E., standard error; SFR, corticosteroid-free remission.
According to the significant parameters mentioned above, the logistic regression models showed that drug type was a predictor of achieving clinical SFR (UST OR = 2.385, p = 0.011), while it was not confirmable on biochemical SFR (OR = 1.337, p = 0.403). By line of therapy, the probability of achieving SFR based on CDAI decreased with lines greater than third-line treatment (third line versus second line, p = 0.007, OR = 0.38, fourth line versus second line, p = 0.315, OR = 0.527). Baseline CDAI had a possible weak effect on the primary clinical outcome (OR = 0.997, p = 0.075). In our models, older age was a predictor of achieving SFR based on CRP (OR = 1.034, p = 0.037). The models’ reliabilities were weak (Nagelkerke R 2 : CDAI = 0.139 and CRP = 0.120). Other model details are presented in Tables 6 and 7.
Multivariate logistic regression model for prediction of biochemical (CRP) SFR at week 52.

Compared to second-line administration.
B, regression coefficient; CCI, Charlson comorbidity index; CDAI, Crohn’s disease activity index; CI, confidence interval; CRP, C-reactive protein; df, degree of freedom; OR, odds ratio; S.E., standard error; SFR, corticosteroid-free remission.
Therapy discontinuation, LOR, and drug safety
Treatment discontinuation due to any reason was more common in the VDZ cohort compared with UST group (VDZ 41.54% and UST 13.04%, p < 0.001) with a median duration of 28 (IQR: 28) and 20 (IQR: 32) weeks, respectively (p = 0.535). The PNR rate was more common in the VDZ cohort than in the UST cohort (21.54% and 4.97%; p < 0.001). The type of drug and baseline CRP were predictors of PNR (UST: OR 8.267, p = 0.001; baseline CRP: OR 1.012, p = 0.062). Coupling data and model details are presented in Supplemental Tables 1 and 2.
LOR was more common in the UST group compared with the VDZ cohort (61.5% and 36.9%, p < 0.001). Only biological agent was identified as a possible predictor of LOR (coupling data are available in Supplemental Tables 3 and 4). Therapy escalation was more commonly required by the UST-treated patients than by the VDZ-treated patients [n = 97 (64.2%) versus 15 (23.1%); p < 0.001]. Escalation regimens of 12–8 weeks and 8–4 weeks were administered in 36 and 67 UST-treated patients (34.95% and 65.05%, respectively).
There was only one infusion-associated reaction resulting in treatment termination in the VDZ cohort at week 20. In the UST cohort, none of the patients presented drug-associated adverse events. Serious infections were not recorded in any of the treatment groups. In the VDZ cohort, one patient stopped treatment at week 16 because of pregnancy with an inactive disease.
Extraintestinal manifestations
The prevalence of EIMs did not differ at baseline between the UST and VDZ cohorts (27.32% and 21.53%, respectively; p = 0.367). During the induction period, existing complaints were improved in 13 patients in the UST cohort and in 3 patients in the VDZ cohort (29.55% versus 21.43%, respectively; p = 0.554). During the 52-week follow-up, the symptoms of 14 UST- and 4 VDZ-treated patients improved from baseline (31.82% versus 28.57%, respectively; p = 0.819).
Perianal disease
Active perianal disease was more common in the UST cohort than in the VDZ cohort at baseline (44.72% versus 24.62%, p = 0.005). After the induction period, perianal symptoms were improved in 30 patients in the UST cohort and in six patients in the VDZ cohort (41.67% versus 40.00%, respectively; p = 0.905). During the 52-week follow-up, perianal symptoms were improved in 39 patients in the UST cohort and in five patients in the VDZ groups (54.17% and 33.33%, respectively; p = 0.142).
Hospitalizations
Hospital admission was required in 10 UST- and 8 VDZ-treated patients because of relapse (6.2% and 12.3%, respectively, p = 0.126) at a mean of 27.6 and 13.5 weeks after inclusion (p = 0.022). Rates of hospitalization because of surgery did not differ between the treatment groups. Eighteen (11.2%) UST- and 13 (20.0%) VDZ-treated patients were admitted to surgery (p = 0.081) after a mean of 32.6 weeks from initiation of both drugs (p = 0.998). Resections were performed in all surgically hospitalized UST-treated patients; whereas seven patients required bowel resection (p = 0.633) and five patients needed perianal surgery in the VDZ group. None of the examined possible predictors differed significantly between the hospitalized and non-hospitalized groups and between the surgically treated and non-surgically treated groups. Coupling data are presented in Supplemental Tables 5 and 6.
Discussion
This real-life multicentre nationwide study analyzed data of 161 UST- and 65 VDZ-treated patients and demonstrated higher clinical SFR rates during the UST compared with VDZ treatments at week 52; however, biochemical SFR rates and SFR rates after the induction did not differ between the two groups. Remaining on therapy was more likely among the UST-treated patients than among the VDZ-treated patients. This finding may have been influenced by the fact that there was a significant difference in the frequency of dose optimization between the cohorts. The frequency of PNR was higher in the VDZ cohort than that in the UST cohort, whereas the frequency of LOR was higher in the UST cohort than that in the VDZ cohort. Our results also showed that using biologicals in a sequential way has negative impact on achieving clinical SFR.
Trials that have investigated the possible pharmacokinetic backgrounds of VDZ response loss have been contradictory. Ungar et al., in their multicentric-observational trial, did not find a consistent pharmacokinetic–pharmacodynamic correlation in the possible mechanism of LOR to VDZ, in contrast to anti-TNF drugs. 20 Consistent with that data, a Danish dual-center cohort study verified that the long-term clinical remission rates of VDZ in IBD groups with escalated and non-escalated regimens were similar. 21 Data from the ENTERPRISE study showed a VDZ treatment outcome with an additional 10th week of infusion, similar to that of the original regimen in perianal CD. 22 On the other hand, a systematic review and meta-analysis proved that escalated regimens could restore response to more than half of VDZ-treated patients. 23 The excess frequency of UST-escalated regimens could be explained by the fact that at the time of the introduction of UST, the standard subcutaneous dose was administered every 12th week in the maintenance phase, which later changed to a standard 8-week maintenance regimen per the label specification change.
Biemans et al. reported the significant superiority of clinical and biochemical SFR at week 52 for UST versus VDZ therapy from a Dutch propensity-score-matched trial. They did not prove the superiority of UST either after induction or at week 12 and week 24. 24 A German single-center study showed that 78.9% of patients with CD and 68.1% of patients with UC continued VDZ treatment after week 52. 25 A meta-analysis that compared the efficacies of UST and VDZ in CD also collected data about clinical SFR and treatment persistence and found significant superiority of UST in both clinical SFR at week 52 and in drug persistence. 26 A recent multicentre-retrospective study from Italy displayed the equality of UST and VDZ in terms of achieving objective remission among patients with anti-TNF refractory CD at week 52; however, clinical remission rates differed. 27 Our study found 1-year persistence rates of 86.5% for UST and 57.9% for VDZ treatment, which are higher than the rates found by Ylisaukko-Oja et al. of 46.2%–37.1% for ADA and 47.1% for UST during 12 months of treatment in a real-life setting. 28
In this study, regression models were created to identify the significant predictors of achieving SFR at week 52. The regressions showed that baseline UST treatment and lower treatment line were significant predictors of outcomes, although the Nagelkerke R 2 of the models indicated only weak correlations. Mühl et al. proved that in patients with UC, a lower partial-Mayo score was associated with better SFR rates after 1 year of VDZ treatment, whereas in patients with CD, a lower HBI was associated only with short-term SFR rates. 25 The PANIC study reported worsening persistence rates with increasing treatment line, which was seen in our cohort as well. 29
Drug survival or long-term efficacy and safety are influenced by age and presence of any comorbidity. A previous study found that the prevalence of comorbidities could predict the long-term treatment outcome in IBD via medication interactions, reduced adherence and poorer response to treatment. 30 Asscher et al. verified that the presence of any comorbidity had higher predictive value than age alone in their VDZ- and UST-treated IBD cohort. 31 They did not find a strong association between CCI score and efficacy/safety parameters. In our study, the comorbidity frequency did not differ significantly between cohorts; however, Asscher et al. have reported higher comorbidity rates during VDZ treatment. According to our study, the most relevant diseases were chronic heart failure, diabetes mellitus, and solid tumors. In this study, the CCI scores at baseline were higher in the VDZ cohort than those in the UST cohort, but the CCI score did not have a clear effect on drug survival in our model. Furthermore, age and comorbidity prevalence were not associated with SFR outcomes. The increased safety suggested by the different mechanism of action of VDZ in the presence of serious comorbidities explains the choice in our real-life setting.
UST and VDZ have been shown to have reassuring safety profiles. A meta-analysis of UST in patients with IBD found 16.7% incidence rate of adverse events, 8% infection rate, and 3.9% serious infection rate. 32 A safety analysis of VDZ treatment from 46 studies has been reported. 33 The overall adverse event rate ranged from 1% to 67% and the infection and serious infection rates ranged from 5% to 24% and 4% to 10%, respectively. That meta-analysis also reported that infusion-related reactions were uncommon. In our cohort, there was only one infusion-associated reaction, which resulted in treatment termination in the VDZ group. In the UST group, none of the patients presented drug-associated adverse events. Infections were not recorded in any of the treatment groups.
Hospitalization rates due to surgery or disease flare did not differ between the treatment cohorts in our study. In real-world studies, hospitalization rate reduction has been observed for VDZ treatment and the pooled estimate from 13 studies was 5.2% for UST treatment. 32 In our study, the hospitalization rates were 6.2% for UST and 10.4% for VDZ, without any statistically significant difference.
The improvement in EIMs differed between UST and VDZ because of their various mechanisms of action. In our study, UST did not show superior improvement of EIMs versus VDZ. A systematic review proved the efficacy of UST for the treatment of EIMs, especially for dermatological and rheumatological manifestations. 34 Only a few comparative clinical trials have been reported.
Perianal disease phenotype was more common in UST-treated patients, but there was no significant difference in symptomatic improvement between the treatment groups. Post-hoc analyses of clinical trials have provided only weak evidence that any investigated drugs could induce and maintain perianal fistula remission, so additional data are needed.35–37
Several other clinical trials have proven the efficacy and safety of new-generation biological agents in patients with IBD. In these trials, data were obtained from a selected population and financial restrictions, physicians’ individual decisions, and prior biological treatments, including biosimilars, were not examined.13,38 There have been few head-to-head data comparisons of second- and third-line applications of both UST and VDZ in real-world study settings.24,39–40
Our retrospective comparative study was conducted in five tertiary IBD centers in Hungary. Data from 161 UST- and 65 VDZ-treated patients were analyzed with the aim of assessing drug persistence and identifying the possible predictors of non-first-line administration of UST and VDZ in patients with CD in addition to assessing their safety profiles. On the basis of the statistically significant difference between the groups that achieved SFR and did not achieve SFR, we identified age as a positive predictor of achieving SFR, but the difference over 4–5 years between the groups was not clinically relevant. Both UST and VDZ were found to be safe and effective in patients with refractory CD, but both drugs were more effective in lower treatment lines. Patients with higher age and/or with more comorbidities could be treated by VDZ because of its mechanism of action, but EIMs limited the usability of VDZ.
There were several limitations in our study that should be considered. In Hungary, except in some special indications (e.g. prior malignancy or coexisting dermatological disease), anti-TNF agents are used as first-line therapy in IBD, so in our study, the patients were receiving second- or third-line treatments with UST or VDZ. However, this treatment approach is also used in many other European countries. In this multicentre study, data from a relatively high number of patients in Hungary were analyzed, but the patient sample was not representative of all Hungarian patients with IBD because of the demographic discrepancies and multi-centric design. In addition, the high number of statistical tests performed could have increased the risk of type I errors. In addition, the retrospective nature of this study did not allow extrapolation of the data. However, a study strength was that our data represent real-life situations regarding the difficulty of choosing the most effective biological agent with different mechanisms of action after TNF failure in CD.
Conclusion
Our study results showed difference in clinical SFR but not in biochemical SFR, hospitalization, and surgery rates between patients treated with UST and those treated with VDZ. However, UST was associated with superior drug persistence versus VDZ at 1 year, which was not influenced by comorbidities. PNR was more common in VDZ-treated patients. UST and VDZ were found to be safe alternatives to anti-TNF refractory or intolerant patients, although the use of these drugs in lower-lines of therapy is recommended.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221144349 – Supplemental material for Ustekinumab is associated with superior treatment persistence but not with higher remission rates versus vedolizumab in patients with refractory Crohn’s disease: results from a multicentre cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848221144349 for Ustekinumab is associated with superior treatment persistence but not with higher remission rates versus vedolizumab in patients with refractory Crohn’s disease: results from a multicentre cohort study by Péter Bacsur, Mária Matuz, Tamás Resál, Pál Miheller, Tamás Szamosi, Eszter Schäfer, Patrícia Sarlós, Ákos Iliás, Kata Szántó, Mariann Rutka, Anita Bálint, Ágnes Milassin, Anna Fábián, Renáta Bor, Zoltán Szepes, Tamás Molnár and Klaudia Farkas in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The manuscript was reviewed by Enago and funded by Klebelsberg Library, University of Szeged. The authors are grateful to Csilla Tóth-Káli, Gabriella Pócsik, Brigitta Csorba, and Krisztina Ördög of the Department of Medicine, University of Szeged for the study support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
