Abstract
Granulomatous cheilitis, characterized by persistent inflammation of the lips and a granulomatous histology, is sometimes associated with Crohn’s disease and is a therapeutic challenge. Reported evidence indicates treatment with an anti-TNF agent (mainly infliximab) is the most recommended therapeutic option after failure of conventional treatments. The clinical case reported the effectiveness of ustekinumab, a monoclonal antibody against interleukins 12/23, to induce the remission of severe and recurrent granulomatous cheilitis in a patient with Crohn’s disease.
Keywords
Introduction
Up to one-third of patients with Crohn’s disease (CD) may present extraintestinal articular manifestations, and less frequently dermatological and ocular manifestations. 1 Granulomatous cheilitis, characterized by persistent inflammation of the lips and a granulomatous histology, is sometimes associated with CD and is a therapeutic challenge.2–4 When conventional treatments fail, treatment with a tumour necrosis factor antagonist (anti-TNF) has been successful in isolated cases. 4 Ustekinumab is a human IgG1 monoclonal antibody targeting the p40 subunit of interleukins 12/23, with proven efficacy in the treatment of moderate to severe CD 5 and active psoriatic arthritis. 6 However, data on the outcomes of ustekinumab for the treatment of CD-associated granulomatous cheilitis are very limited.
Case report
We present the clinical case of a 70-year-old male ex-smoker with a history of diabetes and high blood pressure. Aged 56 years, an exploratory laparotomy was performed in 2005 due to severe abdominal pain, diarrhoea and weight loss. A phlegmon including terminal ileum and cecum was resected. The gross examination of the resected specimen showed a discontinuous pattern of inflammation with several small aphthous ulcers and two large serpiginous ulcers. Focal chronic inflammation, focal crypt irregularity and granulomas in the histologic examination allowed diagnosis of ileal CD. 7 After surgery, and due to mild abdominal symptoms and recurrent pauciarticular arthritis in the ankles, he was treated with salazopyrin, methotrexate and short steroid cycles. Infliximab was started in 2010 due to corticosteroid dependence.
He remained asymptomatic in the abdominal area for 7 years, but in 2014 he needed infliximab escalation (5 mg/kg every 6 weeks) due to recurrent arthropathy. In 2016, methotrexate was withdrawn due to leukopenia and anaemia. That year the patient presented diffuse lip swelling with speech and swallowing difficulties. The patient had reported a similar episode in 2009, providing an anatomical pathology report from a biopsy taken from the inside lining of the lower lip with a diagnosis of granulomatous cheilitis (non-necrotizing epithelioid granulomas, oedema, lymphangiectasia, and perivascular lymphocytic infiltration). Treatment with oral corticosteroids was given, achieving remission. With infliximab trough levels of 6 μg/mL and negative antibodies, infliximab was optimized (7.5 mg/kg every 6 weeks). Despite optimization, at 1 month cheilitis relapsed and peripheral arthropathy persisted, and he required repeated cycles of prednisone at low doses during 2017. An endoscopy did not reveal lesions in anastomosis or neo-terminal ileum (Rutgeerts score i0).
Given the persistence of recurrent granulomatous cheilitis and arthropathy in a patient with ileal CD, despite infliximab dose escalation and supra-therapeutic drug concentrations (most recent trough levels >14 μg/mL), we decided to switch to a biological with a different mechanism of action, and the patient received an induction dose with ustekinumab 360 mg IV. The patient evolved favourably, and during a follow up of 24 months received maintenance treatment with ustekinumab 90 mg SC every 12 weeks, not presenting gastrointestinal symptoms, recurrence of granulomatous cheilitis or inflammatory arthropathy.
To report the present case, all the data were extracted from the ENEIDA database, in accordance with the approval of the Ethics Committee of our centre (C.P. ENEIDA - C.I.12/148-E). The patient gave his written informed consent for the ENEIDA project and an additional verbal informed consent for the publication of this clinical case. The verbal consent obtained was recorded in the patient’s medical chart.
Discussion
Here we describe a clinical case of CD limited to the ileum with associated type I pauciarticular arthritis that required treatment with salazopyrin, methotrexate and subsequently infliximab. The patient developed severe recurrent granulomatous cheilitis, which was successfully treated by switching from infliximab to ustekinumab.
Granulomatous cheilitis, originally described by Miescher in 1945, is a rare entity of unknown aetiology, characterized clinically by persistent inflammation of the lips and histologically by granulomatous infiltrate.2,3 The term orofacial granulomatosis (OFG) is used to encompass not only isolated cheilitis but a variety of other chronic granulomatous inflammatory manifestations of the oral cavity and maxillofacial region, including deep buccal ulcerations, prominent aphthous stomatitis, oral cobblestoning or angular cheilitis. 8 OFG can occur as an isolated phenomenon or as a manifestation of CD, called oral CD (OCD). Oral disease may predate intestinal CD involvement, especially in young patients. 9 Currently, OCD and OFG are separated on clinical grounds, where OFG tends to present with more labial pathology like isolated cheilitis and less buccal ulceration or cobblestoning. 8
Several drugs have been used for the management of granulomatous cheilitis, with very variable results, and there is no consensus regarding optimal treatment.3,9 Oral or locally injected corticosteroids (triamcinolone) are the initial therapeutic option.3,10 Other drugs such as clofazimine, dapsone, thalidomide, colchicine, hydroxychloroquine, topical tacrolimus, and systemic antibiotics have been used. 11 However, the clinical course of granulomatous cheilitis is unpredictable, with spontaneous remissions and frequent recurrences. Plastic surgery (cheiloplasty) is necessary in cases where drug therapy fails to reduce labial deformities completely.10–12
We searched PubMed in March 2020 for full-text case reports or case series using the following search strategy: (‘cheilitis’ OR ‘granulomatous cheilitis’ OR ‘cheilitis granulomatosa’ OR ‘orofacial granulomatosis’) AND (‘anti-tumour necrosis factor’ OR ‘infliximab’ OR ‘adalimumab’ OR ‘vedolizumab’ OR ‘ustekinumab’). Cases with orofacial granulomatosis were monitored and only included if the granulomatous cheilitis was the predominant manifestation. Cases with predominance of buccal ulcerations, Melkersson–Rosenthal syndrome or pyostomatitis vegetans were excluded. In Table 1 we summarize the characteristics of patients with granulomatous cheilitis, with or without coexistent CD, treated with biologics.4,9,10,13–28 There was a clear predominance of cheilitis in children and young adults (mean age 26 years, SD 15), and 55% of the patients were female. Dupuy et al. have reported a greater male predominance and a young age of onset in CD patients who present with oral manifestations. 29 Harty et al. have noted a higher incidence of concomitant perianal CD in those patients also presenting with OFG. 30 Gale et al. considered that CD in conjunction with OFG represents a distinctive subphenotype of CD that is characterized by extensive inflammation, perianal disease, and pronounced granuloma formation in the intestine. 31 Six of the 13 CD patients (46%) treated with biologics for cheilitis granulomatosa had concomitant perianal disease (Table 1).
Full-text case reports or case series of patients with granulomatous cheilitis, with or without CD, treated with biologics.
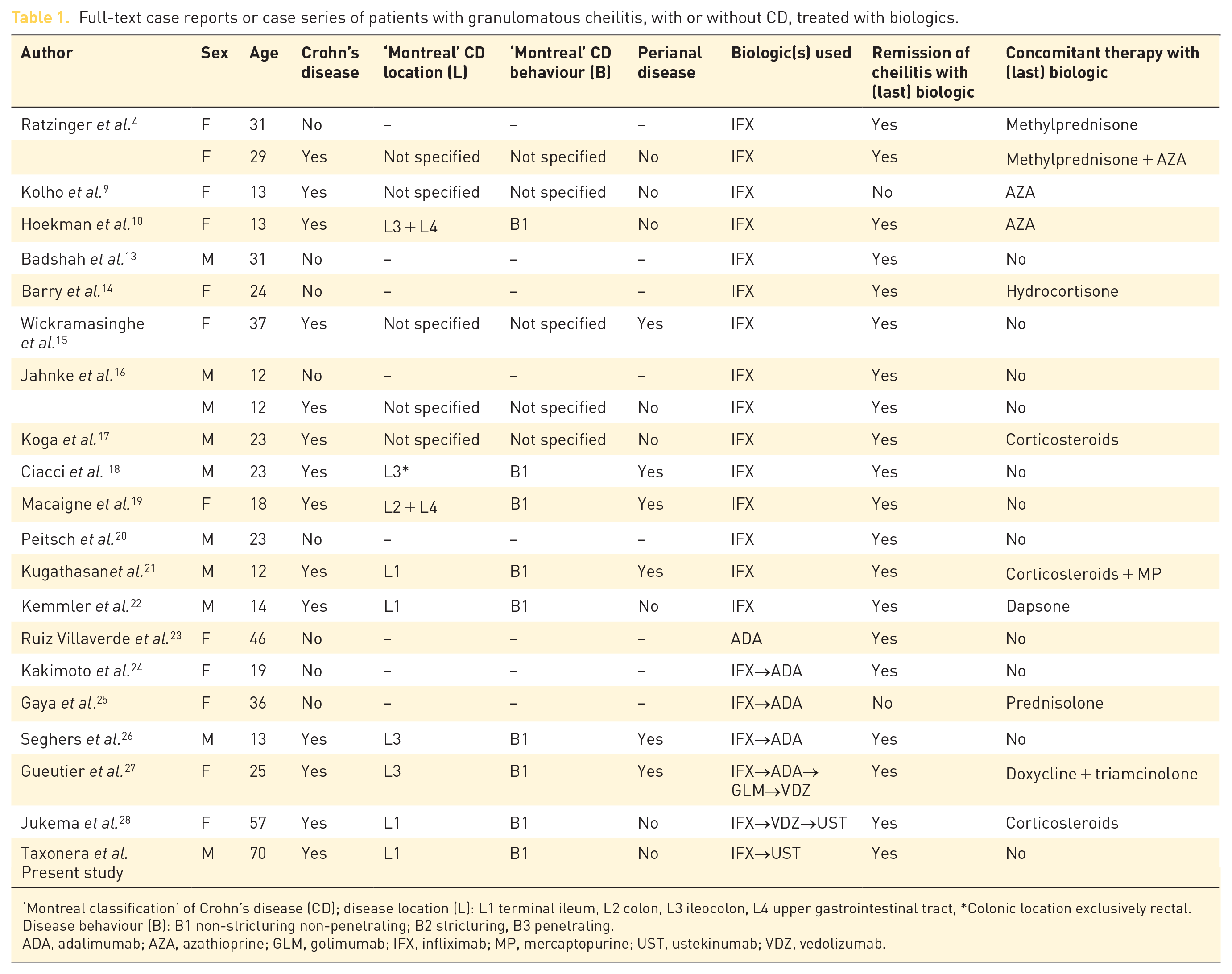
‘Montreal classification’ of Crohn’s disease (CD); disease location (L): L1 terminal ileum, L2 colon, L3 ileocolon, L4 upper gastrointestinal tract, *Colonic location exclusively rectal. Disease behaviour (B): B1 non-stricturing non-penetrating; B2 stricturing, B3 penetrating.
ADA, adalimumab; AZA, azathioprine; GLM, golimumab; IFX, infliximab; MP, mercaptopurine; UST, ustekinumab; VDZ, vedolizumab.
A total of 22 patients (14 with CD) received biologics: 15 patients infliximab, one patient adalimumab, three patients infliximab followed by adalimumab, one patient infliximab, adalimumab, golimumab and finally vedolizumab, one patient infliximab, vedolizumab, and finally ustekinumab, and in the present case, infliximab followed by ustekinumab. Cheilitis went into remission in 18 patients (86%) treated with an anti-TNF agent, regardless of the absence or presence of gastrointestinal disease. Therefore, increased TNF production is considered to play a key role in the damage of the oro-facial mucosa. Ratzinger et al. reported the therapeutic regimens in a series of 14 patients (four with CD) with cheilitis granulomatosa, two of them (one with CD) received infliximab with a satisfactory outcome. 4 The authors considered that infliximab, an effective drug in CD, could be a promising treatment option for severe cases of granulomatous cheilitis. Kolho et al. describe eight paediatric patients presenting with OFG, including a CD case with cheilitis granulomatosa that unfortunately did not markedly improve with infliximab. 9 There were 12 additional published clinical cases (eight with CD) reporting successful treatment of cheilitis granulomatosa with infliximab, especially in childhood.10,13–22 Kakimoto et al. reported a patient with cheilitis without CD in which infliximab induced a good initial response but triggered an acute infusion reaction; remission was achieved after switching to adalimumab. 24 Gaya et al. reported a case of isolated cheilitis successfully treated with infliximab. Cheilitis recurred some years later and partially responded to adalimumab, although the patient developed a perioral cellulitis that led to drug discontinuation. 25 Seghers et al. described a patient with cheilitis granulomatosa, which appeared as a paradoxical manifestation in a CD patient while receiving infliximab and finally resolved with adalimumab. 26 Conversely to what happened in our case, this patient had undetectable infliximab trough levels and positive anti-drug antibodies at the time of switching to adalimumab. Gueutier et al. reported the efficacy of vedolizumab in a single case of granulomatous cheilitis refractory to three anti-TNF agents. 27 Recently, a letter reported a case of granulomatous cheilitis successfully treated with ustekinumab after failing infliximab and vedolizumab.
In conclusion, this review of reported evidence indicates treatment with an anti-TNF agent (mainly infliximab) is the most recommended therapeutic option for granulomatous cheilitis (with or without CD) after failure of conventional treatments. However, in our patient, efficacy of TNF blockade with infliximab was completely lost despite very high doses and supra-therapeutic drug levels. In these cases, it can be assumed that the damage is produced by other mechanisms of action mediated by cytokines other than TNF. IL-12 and IL-23 are linked to the production of IFN-γ, a pivotal mediator of inflammation in peripheral tissues, including orofacial mucosa, by promoting multiple proinflammatory cytokines. 32 Accordingly, in order to treat granulomatous cheilitis, the alternative use of other biological agents such as ustekinumab, a monoclonal antibody against interleukins 12/23 with proven efficacy in CD and psoriasis, is attractive. The present study is the second to report on the effectiveness of ustekinumab for inducing remission of severe and recurrent granulomatous cheilitis in patients with CD.
Footnotes
Acknowledgements
The authors thank Dr. G. Morley for reviewing the English-language manuscript.
Author contributions
CT, CA and MC treated the study patient and drafted the manuscript. DO contributed with the literature review and designed the table. ER contributed with critical revision of the manuscript. All authors read and approved the final manuscript.
Conflict of interest statement
CT has served as a speaker or has received research or education funding from MSD, Abbvie, Hospira, Pfizer, Takeda, Janssen, Ferring, Faes Farma, Shire Pharmaceuticals, Dr. Falk Pharma, Gebro Pharma, and Tillots. This activity is not related to the present work. The remaining authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
