Abstract
Background:
The growing prevalence of eosinophilic esophagitis (EoE) represents a considerable burden to patients and health care systems. Optimizing cost-effective management and identifying mechanisms for disease onset and progression are required. However, the paucity of large patient cohorts and heterogeneity of practice hinder the defining of optimal management of EoE.
Methods:
EoE CONNECT is an ongoing, prospective registry study initiated in 2016 and currently managed by EUREOS, the European Consortium for Eosinophilic Diseases of the Gastrointestinal Tract. Patients are managed and treated by their responsible specialists independently. Data recorded using a web-based system include demographic and clinical variables; patient allergies; environmental, intrapartum, and early life exposures; and family background. Symptoms are structurally assessed at every visit; endoscopic features and histological findings are recorded for each examination. Prospective treatment data are registered sequentially, with new sequences created each time a different treatment (active principle, formulation, or dose) is administered to a patient. EoE CONNECT database is actively monitored to ensure the highest data accuracy and the highest scientific and ethical standards.
Results:
EoE CONNECT is currently being conducted at 39 centers in Europe and enrolls patients of all ages with EoE. In its aim to increase knowledge, to date EoE CONNECT has provided evidence on the effectiveness of first- and second-line therapies for EoE in clinical practice, the ability of proton pump inhibitors to induce disease remission, and factors associated with improved response. Drug effects to reverse fibrous remodeling and endoscopic features of fibrosis in EoE have also been assessed.
Conclusion:
This prospective registry study will provide important information on the epidemiological and clinical aspects of EoE and evidence as to the real-world and long-term effectiveness and safety of therapy. These data will potentially be a vital benchmark for planning future EoE health care services in Europe.
Keywords
Introduction
Eosinophilic esophagitis (EoE) is a chronic immune-mediated inflammatory disorder defined symptomatically by esophageal dysfunction and histologically by eosinophil predominant inflammation of the esophagus. 1 A Th2 immune reaction to food and/or aeroallergens is involved in the pathophysiology of EoE, 2 and most patients also present with a personal and/or family history of several atopic manifestations, 3 which are recognized as a factor in the appearance of the disease. 4
EoE is diagnosed in children and adults up to their 50s5–7 after demonstrating an eosinophil-rich inflammation in esophageal biopsies, taken during an upper gastrointestinal (GI) endoscopic exam, and carried out to study the origin of symptoms of esophageal dysfunction. In younger children and infants, this mainly consists of reflux-like symptoms, vomiting, abdominal pain, food refusal, and failure to thrive. 8 Older children and adults with EoE most commonly report solid food dysphagia, food impaction, and non-swallowing-associated chest pain. 9 Left untreated, symptoms and esophageal inflammation tend to persist over time, 7 and patients can develop esophageal rings, focal strictures, or a long narrowing in the esophageal caliber (Straumann, Spichtin and Grize, 2003), risk being directly related to patient age and diagnostic delay (Dellon, Kim and Sperry, 2014).12,13 Therefore, the natural history of EoE has been proposed as consisting of a chronic inflammation that may progress into fibrous remodeling of the esophageal wall, with collagen deposition, lamina propria fibrosis, and esophageal rigid strictures, as the disease evolves from childhood into adulthood. Esophageal remodeling may result in several disease-inherent and procedure-related complications. 14 A small number of patients, however, do not seem to follow this trend and currently cannot be identified before the esophageal caliber is reduced. Although not associated with mortality or risk of malignancy, the chronic and progressive nature of EoE and associated symptoms negatively impact patient quality of life15,16 and indicate a need for treatment.
The first description of EoE in 1977 considered it as a particular form of eosinophilic gastroenteritis with esophageal involvement. 17 During the 1980s, a handful of case reports and short case series of young patients with dysphagia and radiological demonstration of ringed and narrow caliber esophagi highlighted its association with atopy.18–22 The first consecutive series of patients with EoE were published in the 1990s23–25 and stated the basis for defining EoE as an independent clinicopathological disorder. However, the disease went largely unrecognized until the new millennium, when the interest in EoE increased with the release of the first clinical guidelines, 26 a sharp growth in the number of reported patients, and exponential efforts in research defining some clinical and epidemiological aspects of the disease. Three decades on from its first descriptions, many aspects of EoE still remain elusive, with most pathophysiological and epidemiological features being poorly understood. New therapeutic agents are currently being tested in numerous clinical trials, but their real utility is not yet known. Furthermore, we lack information on the effect of novel treatment options in special populations, which are often excluded from clinical trials, such as children, pregnant women, or the elderly, as well as patients with other associated systemic or digestive conditions. Finally, the long-term effects of the different therapies and their ability to modify the natural history of EoE have not yet been defined. Therefore, there is a need for a prospective registry that will provide information on the real-world effectiveness and safety of EoE therapies, both well-established and newer agents, the epidemiology and risk factors, and the long-term consequences and disease-associated costs for EoE.
Rationale for the registry
Almost four decades after its first descriptions, EoE remains full of pathophysiological, epidemiological, clinical, and therapeutic uncertainties. EoE is currently recognized as the most common cause of chronic or recurrent esophageal symptoms, after gastroesophageal reflux disease (GERD), and it is the leading cause of dysphagia in children and young adults in Europe and North America, with a prevalence that exceeds 100 cases per 100,000 inhabitants, according to several studies.5,27 Lately, EoE has also emerged as a disorder with increasing frequency in other regions, such as North Africa,28,29 Central and South America,30,31 and Asia. 32 As a result, EoE now represents a chronic common health problem, which implies a significant impairment in the quality of life of patients suffering from it. 15 Figure 1 summarizes the prevalence estimates for EoE according to the results of a recent systematic review with meta-analysis. 33
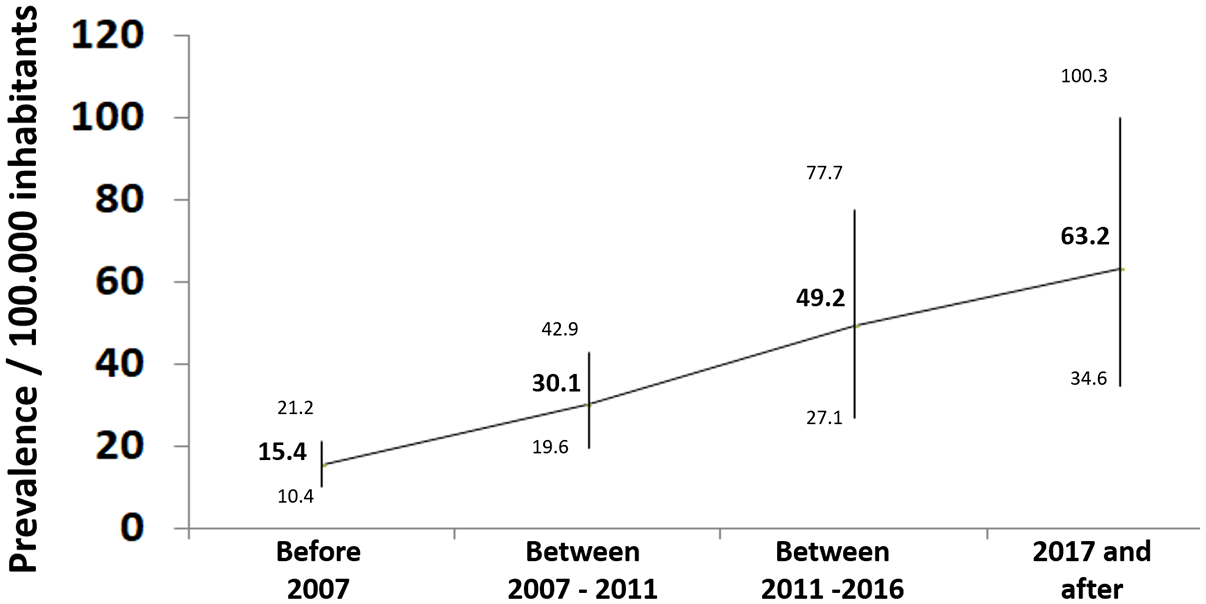
Prevalence rates for EoE with 95% confidence intervals (CIs; in patients per 100,000 inhabitants), resulting from meta-analyses of individual studies grouped according to the diagnostic criteria for EoE used in each study.
In addition, EoE represents a significant burden on the health care systems, due to the diagnostic delay, the need for endoscopy with biopsies to diagnose the disease and monitor the response to treatments, and the costs of new drugs. The mean annual cost per patient attributable to EoE in the United States has been estimated to be US$2300 per year 34 and increases considerably in pediatric patients up to $4001 per year, far exceeding the cost of care of Crohn’s disease (US$985) and celiac disease (US$856). 35
Understanding the continuous increase in the number of patients who have developed the disease in recent decades clearly requires the definition and identification of risk factors independently associated with the development of EoE, which recent studies have just begun to do. 36 Among the factors, geography, including population density and weather, has been proposed, with a negative correlation when population density and risk of EoE were compared between rural and urban areas. 37 Cold climate was also found to increase the odds of EoE compared with tropical and arid zones, 38 as well as indoor and housing contaminants, including gas and forced air heating systems, and brick exteriors. 39 Family association in cases has also been shown throughout the literature, with 7−8% of EoE family members being affected by the disease, 40 with a clustering of EoE in families that can largely be attributed to the common family environment. 41 Despite numerous genetically susceptible variants having been shown to contribute to EoE, 42 the environment seems to play the dominant role. This is supported by the fact that members of the same family living together are more likely to be exposed to similar environmental factors 41 and the finding that there is also an increased risk of EoE in patients’ spouses, who share no common genetic background. 43
The risk of developing EoE has been linked to specific environmental factors described over the last decade, including preterm labor, cesarean delivery, non-exclusive breastfeeding, neonatal intensive care unit (ICU) admission, and use of antibiotics and anti-secretive drugs during childhood,44–48 with some interactions between variants within the CAPN14 and LOC283710/KLF13 genes having been observed. These risk factors are, however, almost identical to those already reported in children predisposed to bronchial asthma. 49 As some but not all genetic studies of EoE have controlled results for another allergic disease, it is not completely clear to what extent these risk factors are specific to EoE or an atopic nature. Either way, the early life factors examined so far are all related to dysbiosis in gut colonization during infancy and involve the esophageal microbiome in regulating the functioning of the esophageal immune system. Differences in esophageal microbiota in terms of abundance and composition have been described in patients with EoE and non-EoE controls,50–53 and evidence of activation of toll-like receptor-dependent signaling pathways in EoE supports the involvement of the innate immune system in the pathogenesis of EoE. 54 Identifying the risk factors and defining the potential interactions among gene polymorphisms, esophageal microbiota, and EoE-related immunological pathways, even before the onset of EoE in predisposed individuals, are essential in the implementation of potentially preventive strategies and therapeutic measurements to address the continuously increasing number of patients who are developing the disease.
Since the initial descriptions of the disease in the early 1990s, EoE has been recognized as a particular form of food allergy, 25 with dietary management having variable effectiveness according to the type of intervention. 55 At the same time, a wide range of options have been developed to treat patients with EoE, from endoscopic dilations aimed at resolving esophageal strictures by enlarging the caliber of the esophagus, 56 to different drugs, mainly including topic steroids,57,58 inhibitors of acid secretion, 59 anti-allergic agents, 60 and even monoclonal antibodies. 61 In recent years, high-quality designed clinical research, including randomized controlled trials (RCTs) and prospective studies, has evaluated the efficacy of the different treatment options available to achieve and maintain disease remission in patients with EoE. This has been summarized in several systematic reviews and meta-analyses56,62–64 by providing evidence on the complex management of this disorder in order to help doctors in their decision making. However, this management remains subject to a wide variety of practices,65–68 mainly due to the difficulties of matching the individual circumstances of each patient with available resources in the clinical settings they attend, the absence in many settings of specific drugs approved for use in EoE, the absence of solid data on the origin of the disorder, the factors that determine their prognosis, and personal and social costs involved in the clinical course of the disease.
EoE currently represents a health and social challenge, therefore, with a considerable proportion of young patients and with a complex management that requires the collaboration of multiple specialists (gastroenterologists, allergists, pathologists, nutritionists, pediatricians, and others) in order to properly manage the disease. However, at the same time, EoE also represents an extraordinary opportunity for research in the field of immunoallergic-based diseases, which continue to expand in the countries of our environment.
In 2013, a group of European researchers and clinicians gathered in EUREOS, the European Consortium for Eosinophilic Diseases of the GI Tract (www.eureos.online), and began designing an ambitious project, the foundation for future clinical and epidemiological research on EoE in Europe. Synergies from their respective National Scientific Societies integrated into the United European Gastroenterology (UEG) allowing for the design of a common research project and its submission to the UEG Link Award program. The program facilitates links between European National Societies in the field of Gastroenterology, supporting them in promoting transnational activities of scientific and professional cooperation. The ‘Harmonizing diagnosis and therapy of Eosinophilic esophagitis (EoE) across Europe’ (HaEoE-EU) project was granted in the 2014 LinkAward call, beginning in November 2014 with an implementation period up to December 2016. After its completion, the whole project was transferred to EUREOS, as the main international collaborative scientific organization focused on EoE, to its further development and implementation.
Study objectives
The overall objective of the EoE CONNECT project is to establish an infrastructure and operating procedures necessary for the continued development of multicenter collaborative studies (including clinical, environmental, and epidemiological) regarding the different factors involved in the etiology, pathophysiology, therapy, and prognosis of EoE.
The specific goals of EoE CONNECT include the following: (1) to promote studies about the influence of environmental factors in the onset and course of EoE, (2) to promote studies to phenotypically characterize patients with EoE, (3) to promote studies about the efficacy and safety of drugs used in EoE patients, (4) to promote epidemiological studies related to EoE, (5) to analyze the use of health resources in the care and assistance of EoE patients, and (6) to disseminate scientific and technical knowledge through documentation and researcher meetings.
Study design and population
EoE CONNECT is a multicenter, international, observational, prospectively maintained registry of pediatric and adult patients diagnosed with EoE and maintained in participating study sites around Europe.
Recruitment onto EoE CONNECT began in December 2015. Registration of the patient, and obtaining patient permission, is undertaken by the EoE specialists at each participating center. Study investigators are responsible for data collection. Inclusion of both retrospectively and prospectively diagnosed patients is permitted, differentiated by the date of granted informed consent and patient registration. Prospective inclusion of incident diagnosed EoE is preferred, and at present, prospectively recruited incident cases represent ~85% of registered patients in EoE CONNECT.
To be included on the registry, patients are required to have a confirmed diagnosis of EoE based on the following criteria, according to the evidence-based guidelines: 1 (1) symptoms of esophageal dysfunction, (2) a peak eosinophil count ⩾15 per high-power field (HPF; 400× magnification) at any esophageal level, and (3) exclusion of other systemic and local causes of esophageal eosinophilia. Patients with EoE and concomitant GERD can be included if the diagnosis of EoE and GERD has been established based on accepted diagnostic criteria. Patients are excluded where they refuse to participate or sign the informed consent, or have additional causes for esophageal eosinophilia, such as eosinophilic gastroenteritis with esophageal involvement, Crohn’s disease, vasculitis, or esophageal infection. 1 Details are imputed onto EoE CONNECT and updated by investigators at every study site during face-to-face or remote clinical appointments.
Variables and definitions
The variables included in the EoE CONNECT are grouped into several forms: demographic data, potential risk factors, clinical characteristics of the disease and length of symptoms at diagnosis, endoscopic and histological features at the point of diagnosis as well as in every subsequent examination, visits to clinic, and additional examinations (pH monitoring, barium swallow, and esophageal manometry). Treatment data are registered sequentially, and new sequences are created each time a different treatment (drug or diet modality, active principle, formulation, or dose) is administered to a patient. Clinical and histological responses to therapies are also registered. Allergology issues collected on EoE CONNECT consist of concomitant atopic diseases, serum-specific immunoglobulin E values and relevant analytical values, results from skin prick testing and atopic patch tests, and food allergy background. Major therapies for concomitant atopies are also collected.
Demographic data include sex, date and place of birth, time of diagnosis, and address and length of time living there at the point of diagnosis. Environmental factors, which include smoking habits, exposure to tobacco during pregnancy or childhood, any family background of EoE, birth delivery details − including gestation time, weight at birth, and cesarean section − feeding during first year of life divided by quarters, use of antibiotics in childhood, sharing a bedroom, tonsillectomy, residence up to being 20 years old (rural, urban, or mixed), contact with animals and pets in childhood, food consumption habits, and profession, are collected in order to explore potential risk factors.
Details of type and duration of symptoms before diagnosis are selected from a scroll down menu with predefined options; free text is also allowed. Endoscopic features are registered according to the EREFS classification system: 69 Total EREFS (0–9) is calculated by summing the severity scores of the five individual major components (Edema 0–1, Rings 0–3, Exudates 0–2, Furrows 0–1, and Strictures 0–1), and the minor finding of crepe paper esophagus (mucosal fragility or laceration upon passage of endoscope, 0–1), with higher scores indicating more severe endoscopic findings: rings and strictures are classified as fibrotic features, while edema, furrows, and exudates are defined as inflammatory ones.
Results from esophageal biopsies include peak eosinophil density at any esophageal third (in eosinophils per HPF) and the HPF size used by local pathology microscopes. Details on findings in gastric and duodenal biopsies, if available, can also be provided at each endoscopic examination.
Regarding treatment options, EoE CONNECT collects information on the use and effectiveness of proton pump inhibitors (PPIs), topical and systemic steroids (by including all principles in each family), and other empirical or investigational drugs; doses and length of therapy are also registered. Dietary interventions include exclusive feeding with elemental formulas, allergy testing–directed food elimination, all variants of empirical food elimination diets, and other options, endoscopic dilation, and other therapies.
Symptoms in EoE CONNECT are measured in adults and adolescents by the Dysphagia Symptoms Score (DSS), a non-validated measuring instrument developed by Alex Straumann and colleagues in 2010. 70 Briefly, DSS assesses frequency of dysphagia, ranging from none (0) to several times per day (5); the intensity of dysphagia, ranging from unhindered swallowing (0) to long-lasting complete obstruction requiring endoscopic intervention (5); and the duration of dysphagia, ranging from no attacks (0) to lasting up to endoscopic removal of the impacted food (5). Total scores range from 0 to 15. Subjective symptom intensity reported by either children or parents is considered for younger children. As a second point of clinical evaluation, EoE CONNECT includes an assessment by physicians of symptoms from the initiation of a therapy for EoE, to capture the short-term effectiveness of an intervention.
Active disease in EoE CONNECT is defined as a peak eosinophilic infiltrate by >15 cells per HPF at any esophageal level together with ⩾5 points in the DSS. A DSS ⩾8 points is considered as severe dysphagia.
Response to therapy is evaluated independently according to clinical, endoscopic, and histological criteria (except for endoscopic dilation, which does not include histological assessment). Symptomatic improvement is independently assessed by changes in DSS reported by patients and by clinicians’ perceptions. A decrease of more than 50% in baseline DSS after therapy is considered clinical remission in older children and adults, as previously defined;71–73 a symptomatic improvement ⩽50% from baseline is considered as a clinical response. For younger children, any subjective improvement in symptoms reported by either children or parents is considered as clinical remission. In addition, clinicians semi-quantitatively score changes in symptoms from the initiation of any therapy as complete clinical remission, partial remission, or no response.
Histological remission is defined as an eosinophil peak count below the diagnostic threshold of 15 cells per HPF at all esophageal levels after therapy. In addition, deep histological remission is considered when the eosinophil peak count is <5 eos/HPF at all esophageal levels after therapy. Clinico-histological remission is defined as the simultaneous combination of symptomatic remission and all degrees of histological remission (peak eosinophil count <15 eosinophils per HPF) in the same patient after therapy.
Lack of efficacy is defined either as maintenance or worsening of the patient’s symptoms, combined with persistence of histological activity of the disease at the end of any therapy, or a situation that leads the physician to escalate the dose of drug, the level of food restriction, or change to an alternative drug or diet modality in a patient.
The occurrence of complications (esophageal strictures, perforation, need for hospital admission for EoE, changes in disease phenotype) is also recorded.
Finally, the registry includes a tool that allows investigators at each site to search for their own patients based on a range of criteria, from dates of inclusion or date of visits, to type of treatment used, responses to them, or cases with incomplete information.
Recruitment, data collection, and follow-up
At present, EoE CONNECT is being conducted at 39 centers in Spain, Italy, Denmark, and France. Study data are collected and managed using an electronic data capture tool funded by EUREOS, which is a non-profit medical and scientific society. EUREOS provided this service free of charge, with the aim of promoting investigator-driven research. Demographic and clinical data, as well as risk factors and concomitant diseases, are collected from each patient at the time of inclusion in the study. Thereafter, all other visits conducted throughout their medical care are also prospectively registered, as well as changes in symptoms and results from endoscopic or additional examinations. As EoE is based on individual clinical practice and local protocols, no predefined visit schedule is provided.
Monitoring and data verification
Data entered on the registry are verified by the investigators at the coordinating center (Hospital General de Tomelloso). The database is monitored and individual data are manually revised to evaluate whether the study selection criteria were met, to evaluate whether the information on diagnosis and therapy was correctly registered, and, ultimately, to ensure the correct inclusion of therapies to guarantee the highest scientific and ethical standards. Data completion is assessed at the point of every database utilization, based on a pivotal group of variables relevant for each database analysis; depending on the nature of analysis, variables could include ‘baseline characteristics’, ‘type of treatment’, and ‘effectiveness of results’. Duplicates are manually removed. Data discrepancies are resolved by questioning the investigators and through group e-mailing. In addition, after data extraction and prior to any statistical analysis, the database is reviewed for inconsistencies and subsequently subjected to data cleaning.
Ethics and data protection
The study is being conducted in accordance with the 1964 Declaration of Helsinki and its subsequent revisions. The study protocol, informed consent forms, assessment for patients under 12 years old, and agreement for participation have all been approved by the Ethics Research Committee of Hospital Universitario de La Princesa, Madrid, Spain, which acts as the central Ethics Committee for EoE CONNECT. Updates in the study protocol or its essential documents are all approved by the central ethics committee (the last revision on 6 July 2021, approval code 2649). Mere assent is not a valid form of agreement to participate in the registry. Translation of all participating patients’ documents into the native language of each participating center is provided. In accordance with the Spanish regulations for observational studies and best practice principles, ethics committees at each individual participating study site must also provide approval to participate in the registry before the site is activated and patients can be recruited. All patients or legal guardians must provide informed written consent to participate in EoE CONNECT.
The EoE CONNECT application is not freely accessible; access is established through credentials (username and password) that have been authorized by those responsible for the registry. No one except the user is aware of the password, which is encrypted on the database server. As a control measure, the application is equipped with an auditing system that monitors all access to the application (who and when a user has logged into the application). EoE CONNECT has a dedicated server where all the information collected in the application is stored. Direct access to the server is restricted to personnel who administer and maintain the correct functioning of the server. The web server is provided with a Thawte SSL (Secure Socket Layer) certificate that provides a secure and encrypted 256-bit connection to any user who connects to the application. In addition, access to the application server by administrators is subject to three layers of security: (1) Security by virtual private network (VPN): access to the server is not made directly through the Internet – it is necessary to have a VPN client specifically configured and installed in order to have access; (2) Windows Server Security, provided by the Windows Server operating system, requires a new username and password; and (3) Microsoft SQL Server Security: the database server where the information is housed is only accessed by using additional new credentials (a new username and password).
The information collected and stored by the EoE CONNECT application is located in servers in Spanish national territory and therefore subject to Spanish and European legislation on data protection. Therefore, the company that manages the servers where the information is hosted, the company that manages, maintains, and administers the application, and those in charge of data processing all comply fully with the obligations stipulated within the EU regulations which correspond to the General Data Protection Regulation (GDPR).
Scientific exploitation and information dissemination of the EoE CONNECT registry
Although each researcher is free to use their local EoE CONNECT data for research without requiring any additional approval, the exploitation of central data is regulated by a specific standard operating procedure. All participating researchers who provide clinical information on a minimum of 30 patients can request EUREOS to use the information contained in the registry. Only those EUREOS members whose centers contribute to EoE CONNECT can request a study using registered data: independent researchers, other scientific societies, or public or private companies are excluded. Once the study is approved by the EoE CONNECT Scientific Committee, the information on the selected variables is delivered to the researcher and anonymized for subsequent use, analysis, and publication. There is a clear authorship policy, so that for each study all EoE CONNECT project researchers who have collaborated on patient data from their own center have the right to authorship, provided they meet the other ethical requirements. Both articles and authorship must be approved by the Scientific Committee, the EoE CONNECT coordinator, and the main researchers of the centers involved.
In order to disseminate the information generated from the registry, the EoE CONNECT coordinator presents an annual update of the registry and its scientific findings. This coincides with the annual meetings of EUREOS.
Results
The EoE CONNECT project has already begun to meet the objective of generating scientific knowledge about EoE. The first collaborative article based on EoE CONNECT data was published in 2020 and evaluated the effectiveness of first- and second-line therapies for EoE in clinical practice. 71 Determinants for treatment choice were evaluated by multivariate analyses, which found the stricturing phenotype of EoE to be associated with selection of topical steroids over PPIs or diets as the first-line therapy; a lack of fibrotic features at initial endoscopy was associated with selection of elimination diets over topical steroids as a second-line therapy. A subsequent paper assessed the effectiveness of PPI therapy for EoE in real-world practice and found this drug-induced histological remission in 48.8% and a decreased symptom score in 71.0% of patients, irrespective of the drug used or patients’ age. More EoE patients with an inflammatory rather than stricturing phenotype accomplished clinico-histological remission after PPI therapy (odds ratio, OR = 3.7, 95% CI = 1.4–9.5). In addition, prolonging treatment length from 8 to 12 weeks increased chances of clinico-histological remission (OR = 2.7, 95% CI = 1.3–5.3), with treatments over 12 weeks providing no additional benefit. Among patients who responded to PPI, dosage reduction was able to maintain clinico-histological remission in 69.9% of patients (Laserna-Mendieta, Casabona and Guagnozzi).
The ability of PPI to stop and reverse fibrous remodeling and endoscopic features of fibrosis in EoE has been recently assessed through the EoE CONNECT registry: 75 among responder adult patients, a short course of PPI treatment reduced significantly EREFS scores in patients treated with PPI (in a similar way to swallowed topic corticosteroids), as well as rings and strictures. Achieving deep histological remission (<5 eosinophils/HPF) provided further reduction in the total EREFS score. Endoscopic improvement was confirmed with EndoFLIP in a subcohort of patients, who improved esophageal distensibility after treatment with PPI.
In addition, EoE CONNECT has provided seven reports to the United European Gastroenterology Week meetings for the period 2018–2021, the results of which will be the subject of forthcoming publications. Figure 2 summarizes the main information currently recorded in EoE CONNECT.

Graphical summary of the information currently collected in EoE CONNECT, including country of origin of patients, major recruiting centers, annual inclusion of patients, ratio of children and adults, and distribution by sex of registered participants. EoE phenotype, age at diagnosis, and follow-up time are also summarized.
Discussion
The EoE CONNECT project constitutes the largest prospective clinical registry of patients with EoE in Europe and represents a paradigmatic example of collaborative effort in clinical research framed within a scientific society. Similarly, it values the disinterested participation of patients and researchers, with high-quality usage procedures and scientific production, giving EUREOS visibility and international prestige as a consolidated group.
In order to plan health care for patients with EoE, it is important to define its current epidemiology, to evaluate the characteristics of patients at diagnosis, and to analyze their disease course and need for appropriate treatments. This information is being provided by EoE CONNECT at present. The potential of the registry to recruit large series of patients with a structured assessment of potential risk factors for EoE will also deepen the knowledge of the causes of the disease and its dramatic expansion in recent decades, and pave the way toward the development of early diagnosis, or even preventive strategies capable of minimizing the impact of the disease.
The incidence of EoE continues to increase from affecting approximately 5 to 10 new cases per 100,000 inhabitants annually5,7,76 to close to or greater than 20.27,77 As EoE predominantly affects young people and does not shorten life expectancy, the growth in the prevalence has been considerable in recent years 33 and will likely continue for the foreseeable future. This striking increase cannot be explained by the expanding use of endoscopic examinations78,79 but should be framed within the recent general expansion of immune-mediated and allergic diseases, and explained by the hygiene hypothesis. 80
In the case of EoE, the lifelong use of resources per patient can be high. Studies carried out in the United States suggest that the economic burden of EoE is noticeable and may increase over time, either because of drug-associated costs or the need for invasive endoscopy–based procedures with biopsies to diagnose and monitor the disease.34,81 The expense of EoE is not restricted to health care systems and insurance providers, but patients themselves and their families frequently have to face increased costs imposed by dietary therapy. 82 These aspects are especially relevant for a disease of such a chronic nature as EoE, where the care for sufferers is provided, in most European countries, by public health care services. The dependence on ever expensive techniques and drugs to treat an increasingly affected population is likely to cause a dramatic increase in public resources and costs in coming years, adding to the growing burden of other chronic immune-mediated conditions.83–85 As this rate of increase will become unsustainable for most developed countries, evaluation of diagnostic and therapeutic interventions in terms of their efficiency and cost–benefit ratio is a necessity. EoE CONNECT is a suitable tool to help identify strategies for early diagnosis and effective treatment, both initially and to maintain remission; to optimize management and prevent complications, especially those that appear in the long term such as strictures and perforation, and are not usually identified in protocolled clinical trials; 86 and to reduce the overall burden on both patients and health care providers.
In addition, in a predictable scenario where expensive biological therapies currently under development for EoE will be incorporated into clinical practice in the next few years,61,87 an independent evaluation of the real-world effectiveness of therapy to inform best practices is essential. Data from our registry, which includes both specialist EoE units and general gastroenterology departments, will provide valuable insight into this issue.
A key strength of the EoE CONNECT registry is the inclusion of a large, multicenter series of patients with EoE, who are recruited by physicians engaged with EoE at multiple sites in different countries. By including a range of hospitals with and without EoE specialized units, the EoE CONNECT study will assess diagnostic and treatment practices across a range of settings and will allow comparison of patient outcomes between settings and regions. The results provided by EoE CONNECT may reflect actual clinical practice rather than that reported in protocolized studies and provide more representative data for special populations usually not included in clinical trials. Patients are recruited based on strict diagnostic criteria, and clinical and biological disease activity is assessed with instruments designed for EoE. Follow-up information is captured at each clinic visit, ensuring data completeness. The prospective nature of the registry and the active monitoring of data ensure reliability of the registered information.
Some limitations of EoE CONNECT should also be acknowledged: to begin with, the registry is not population based at all study sites, and at present no patients with other esophageal conditions or controls with healthy esophagus are being recruited, which prevents the development of translational projects. This point, however, as well as the implementation of a biobank to collect blood and tissue samples from patients at different stages along the disease’s course, will be implemented in the near future. Instruments to measure EoE symptoms have been developed and validated, for children 88 and adults,89–91 but at present no validated version is available in the majority of European languages, including Spanish and Italian; in addition, the questionnaires are protected by copyright and their use is not for free. Therefore, symptoms reported by patients at baseline and changes induced by treatment are assessed in EoE CONNECT with the DSS, which is a non-validated measuring instrument. 70 The DSS has been, however, repeatedly used in previous studies to document changes in dysphagia in patients with EoE, in research performed in the United States and Europe, a reduction of 50% or more, with regard to the baseline DSS, having been defined as the criterion for clinical remission.72,73,92–94 Although not yet validated, the DSS has shown sufficient responsiveness to changes in symptoms in the medium and long term, but of limited utility for short-term changes. 71 Due to this, the EoE CONNECT registry incorporates a second point of evaluation provided by the clinician after the assessment of symptoms from the initiation of a therapy for EoE, in order to capture the short-term effectiveness of an intervention.
To conclude, the EoE CONNECT registry study will provide important information about the epidemiological and clinical aspects of EoE in patients of all ages, as well as about the optimal management of the disease and its natural history. These data will be a vital benchmark for planning future EoE health care services in Europe.
Footnotes
Collaborators
Sergio Casabona-Francés: Department of Gastroenterology, Hospital Universitario La Princesa, Madrid, Spain
Teresa Pérez-Fernández: Department of Gastroenterology, Hospital Universitario La Princesa, Madrid, Spain
Roland Llerena Castro: Department of Gastroenterology, Hospital Universitario Vall d’Hebron, Barcelona, Spain
Matteo Ghisa: Department of Surgery, Oncology and Gastroenterology, Università di Padova, Padova, Italy
Daria Manie: Department of Surgery, Oncology and Gastroenterology, Università di Padova, Padova, Italy
Gaia Pellegatta: Personalized Medicine, Asthma and Allergy Clinic, IRCCS Humanitas Research Hospital, Rozzano, Italy
Adolfo Suárez: Department of Gastroenterology, Hospital Universitario Central de Asturias, Oviedo, Spain
Javier Alcedo: Department of Gastroenterology, Hospital Universitario Miguel Servet, Zaragoza, Spain
Paula Gil Simón: Department of Gastroenterology, Hospital Universitario Río Hortega, Valladolid, Spain
María Teresa Palomeque: Department of Allergy, Complejo Universitario Hospitalario de Albacete, Albacete, Spain
Teresa Asensio: Department of Allergy, Complejo Universitario Hospitalario de Albacete, Albacete, Spain
Alicia Granja-Navacerrada: Department of Gastroenterology, Hospital Universitario de Fuenlabrada, Fuenlabrada, Spain
Lonore Hurtado de Mendoza Guena: Department of Gastroenterology, University Hospital Fundación Jiménez Díaz, Madrid, Spain
Alba Rodríguez Sánchez: Department of Gastroenterology, Hospital Universitario Santa Lucía, Cartagena, Spain
Lluïsa Masiques Mas: Pediatrics Department, Granollers General Hospital, Granollers, Spain
Raffaella Dainese: Department of Allergy, Center Hospitalier d’Antibes Juan-les-Pins, France
Sara Feo-Ortega: Pediatric Gastroenterology Unit, Hospital General de Tomelloso, Tomelloso, Spain
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alfredo J. Lucendo has served as a speaker, and/or has received research and/or education funding and/or consulting fees from Adare/Ellodi, Dr. Falk Pharma, Regeneron, and EsoCap. Cecilio Santander has received training and consultant fees from Laborie/MMS. The rest of the authors have no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The establishment and design of the EoE CONNECT registry was developed with a grant from the United European Gastroenterology through the National Societies Link Award program. The maintenance of the database is financed by EUREOS (European Society of Eosinophilic Oesophagitis). Funding agencies had no role in the study design, in the writing of this manuscript, or the decision to submit for publication.
