Abstract
Background:
Inflammatory bowel diseases (IBD) are chronic, relapsing-remitting inflammatory conditions with a substantial negative impact on health-related quality of life and work productivity. Treatment of IBD has been revolutionized by the advent of biologic therapies, initially with anti-TNF agents and more recently with multiple alternatives targets, and yet more under development.
Objectives:
Approximatively one third of patients do not respond to biologic therapy and more importantly a significant proportion experiences partial response or loss of response during treatment. The latter are common clinical situations and paradoxically are not addressed in the commercial drug labels and available guidelines. There is therefore a clinical need for physicians to understand when and how eventually to optimize the biologic therapy.
Design:
This consensus using a Delphi methodology was promoted and supported by the Emirates Society of Gastroenterology and Hepatology to close this gap.
Data Sources and Methods:
Following an extensive systematic review of over 60,000 studies, 81 studies with dose escalation and five addressing drug monitoring were selected and in addition five systematic reviews and three guidelines.
Results and Conclusion:
after three rounds of voting 18 statements were selected with agreement ranging from of 80% to 100%
Keywords
Introduction
Inflammatory bowel diseases (IBDs) consisting of Crohn’s disease (CD) and ulcerative colitis (UC), are chronic, relapsing, and progressive, disabling conditions which affect the gastrointestinal (GI) tract. 1 During the 20th century, IBD was considered mainly a disease of ‘westernized’ countries of North America, Europe, and Oceania. Since the turn of the 21st century, IBD is recognized as a global disease with rapidly increasing incidence in the newly industrialized countries of Asia, South America, and Africa, where societies have become more westernized. 2
The goal of the treatment is the induction and maintenance of remission to avoid complication and disability. 3 The achieving of these targets has been made realistic with biologic therapies, since the advent of anti-tumour necrosis factor (TNF) agents and more recently with several other mechanistic targets and more under development. However, the health care cost of IBD, which is already three times that of general population, is increasing progressively with the wider use of biologic therapy, although with substantial beneficial outcomes, such as reduced need for hospitalization and surgery, which in turn mitigates against the pharmaceutical cost in the long term. 4
However, approximatively 30% of patients do not respond to anti-TNF therapy and nearly 50% patients experience loss of response during treatment. 5 The latter are common clinical scenarios and paradoxically are not addressed in the commercial drug labels and available guidelines. There is therefore an urgent need for physicians to understand when and how to optimize biologic therapy, and this has been the focus of this systematic review and Delphi consensus.
Methods
The study was performed following the preferred reporting items for systematic reviews (PRISMA).
Literature search
Studies that investigated (a) patients: adults OR paediatric with established CD or UC; (b) intervention: infliximab (IFX), adalimumab (ADA), golimumab (GOL), vedolizumab (VDZ), ustekinumab (UST), tofacitinib (TOF); (c) outcome: patients developed loss of response (LOR) and/or required dose intensification; (d) use of therapeutic drug monitoring (TDM) were considered. We identified relevant literature (only published articles) by performing a systematic search until 31st December 2020 of three databases: PubMed, Cochrane Library, and Embase. Keywords used were (all fields): anti-TNF OR TNF-alpha OR TNF-α OR infliximab OR adalimumab OR Golimumab OR vedolizumab OR ustekinumab OR tofacitinib OR therapeutic drug monitoring (TDM) AND inflammatory bowel disease OR Crohn OR ulcerative colitis. For PubMed, all relevant MeSH terms were used. The final queries were validated by manual review and matching results. The reference list of eligible studies and reviews articles were hand-searched to identify further relevant publications (Figure 1).

Flowchart of the selection of studies in the systematic review.
Study selection
Two authors (VA and NR) independently checked the retrieved articles to eliminate duplicates and reviews. In duplicate reports, the most comprehensive article was chosen. The variables recorded were year of publication, country, sample size, diagnosis, therapeutic regimen, duration of follow-up, percentage of patients receiving dose intensification and TDM, and success rate when available.
Methodology of the consensus
The consensus was initiated and supported by the Emirates Society of Gastroenterology and Hepatology by inviting the members of the committee among the public and private sectors across different Emirates of United Arab Emirates that already produced the UAE guidelines on IBD. 1 Following the extensive research of the relevant scientific literature, a proposed list of recommendations was compiled by two authors (VA and NR) and distributed online to the entire panel with all relevant literature for the first assessment of the agreement. The final agreement was finalized by the panel in a face-to-face web meeting. A Likert-type scale (1, strongly disagree; 2, disagree; 3, neutral; 4, agree; and 5, strongly agree) was used to measure the agreement. In cases of disagreement, uncertainty, or agreement less than 75% of participants, the panellists were required to submit comments and propose changes. In case of debate or conflict, re-voting online was repeated. The updated recommendations were then re-evaluated by the entire panel in the second round of face-to-face meeting. An agreement of 75% or more represented a strong recommendation; 50–74.9% represented a recommendation, and less than 50% represented a suggestion. Percentage of the final agreement is given between brackets in Table 1.
List of recommendations and percentage of agreement.

IBD, inflammatory bowel disease; TDM, therapeutic drug monitoring.
Results – dose intensification
Five systematic reviews were identified,6–10 and 81 case series selected and listed on Table 211–89
ADA, Adalimumab; CD, Crohn’s disease; GOL, Golimumab; IBD, Inflammatory bowel disease; IFX, infliximab; LOR, loss of response; TOF, Tofacitinib; UC, ulcerative colitis; UST, Ustekinumab; VDZ, Vedolizumab.
Dose escalation has been performed either increasing the dose or shortening the administration interval.
In a meta-analysis of 23 studies 6 loss of response to ADA was seen in 21% at the end of one year in the pooled data for those patients who had either initial response to, or primary non-response to induction therapy. Among those who had dose intensification for loss of response, for whom data were available, 71% achieved a symptomatic response and 40% achieved symptomatic remission as evaluated with CDAI. Subgroup analysis revealed that nearly 20% of patients with an initial response lost response annually, and among those for whom data were available, about 25% underwent dose intensification by the end of the year. Overall, around one fifth of adult patients require dose intensification and experience a loss of response after initiation of ADA therapy.
A review of 16 studies 7 calculated the annual incidence of loss of response to IFX to be 13%. In the studies included in that review, response to dose intensification was noted in 54–90%, with 31% achieving symptomatic remission in one study.
Qiu et al. 8 have reported 86 eligible studies using anti-TNF therapy in CD; the rate of LOR ranged from 8% to 71% with a median of 33% (95% CI 29–38) at 1-year follow-up, with an annual risk of LOR of 20.9% per patient-year. The pooled rate of dose intensification calculated with the random effect was 34% (95% CI 28–41) at a median follow-up of 1 year, with no clear difference for IFX and ADA (38% and 36%, respectively). The annual risk for dose intensification was 14.9% and 26.3% per patient year for IFX and ADA, respectively.
In the same year, Einarson et al. 9 published a critical review of studies using dose intensification in CD in 12 European countries. Fifty-eight studies including nine abstracts were evaluated for a total of 7850 patients. Overall, 29.9% ± 3.5% of patients required dose escalation; 25.2% ± 2.4% with IFX and 32.8% ± 6.2% with ADA (P = 0.35). Interestingly, rates of dose-escalation increased according to order of treatment: 19% for first line, 37% second line and 41% for third. Of note, short-term response rates to escalation were 63% for ADA and 45% for IFX (P = 0.08).
In the most recent systematic review by Gemayel et al., 10 dose escalation was investigated in patients with UC. Thirty-five studies reporting dose escalation were evaluated. Dose-escalation of IFX ranged between 5% and 50% at median 0.67 years of all patients included at induction. Similarly, dose escalation for ADA on anti-TNF naïve patients ranged from 5% to 45.9% at a median of 6.5 months. Dose escalation under VDZ after failure of anti-TNF occurred in 20% of patients and in 47% of responders at the induction at 1 year of follow-up. Factors associated with an increased likelihood of dose escalation included: initiating IFX in acute severe colitis (hazard ratio (HR) = 2.75, P = 0.01), having UC compared to CD (HR = 2.73, P = 0.007) and using immunomodulator therapy before a treatment with IFX (HR = 3.9, P = 0.008). The overall response rate after dose escalation was 62.4% ± 6% and 45.2% ± 11.4% for ADA and IFX, respectively. There was significant heterogeneity among studies and the effectiveness of dose escalation was available only for a short-term follow-up. Only one study with a more prolonged follow-up reported that the efficacy was lost at the rate of 43% patient-year. 38
Interestingly, adverse events following dose escalation appear to be less extensively monitored; in the review by Gemayel et al. 10 reporting on five studies following ant-TNF dose escalation, the highest rate of AE was attributed to acute or delayed infusion reactions.
Recently Ehrenberg et al. 70 reported the rate of dose escalation of biologic therapy in over 7,000 IBD patients tracked in Healthcare Analytics database in the period 2015–2017 in the United States. Dose escalation occurred in 39% of patients on IFX, 28% of those on ADA, 23% on VDZ, 22% on UST, and 14% on GOL. The magnitude of dose escalation was greatest for UST (131%) and lowest for GOL (45%).
A possible explanation of the efficacy of dose-intensification is to revert the immunogenicity induced by anti-TNF agents by producing anti-drug antibodies. Recently Battat et al. 90 have reported a large retrospective evaluation of over 100,000 IBD patients evaluated at the Prometheus Biosciences Laboratories in San Diego, Ca, USA. Anti-drug antibodies were detected in 23.6% and 19.6% of patients treated with IFX and ADA, respectively. In patients with antibodies (n = 453), IFX dose-escalation yielded a significantly higher proportion achieving the primary outcome (47.5% vs 30.9%, P < 0.001), higher drug concentration (P < 0.001) and reduction of antibodies (P = 0.002) compared to no escalation (n = 204). In contrast no patients receiving dose escalation with ADA (n = 87) achieved the primary outcome defined as achieving a serum level of ⩾5 µg/mL and of ⩾7.5 µg/mL for IFX and ADA, respectively and undetectable antibodies. However, the sample size for ADA was probably too small to draw firm conclusions.
In our systematic review, we have identified 81 published studies, mainly retrospective (n = 63) with a mean follow-up ranging from 6 months to 3.5 years but more frequently of 1 year. The majority of studies reported the use of IFX and ADA with few exceptions; six studies with UST, four VDZ, two GOL and two with TOF. The reported percentage of dose escalation varied widely from 5% to 70% with nine studies reporting all patients receiving dose escalation because of secondary LOR. The success rate of dose escalation is reported only in 20 studies with percentage ranging from 15% to 100%.
No randomized controlled trial (RCT) data are available for dose intensification, and in most studies, data from primary non-responders (in contrast to partial response) and patients with loss of response are generally pooled together. Loss of response to anti-TNF therapy occurs at a rate of about 10–20% annually, and between 50% and 90% will regain symptomatic response following treatment intensification. Data for dose intensification in patients with a partial initial response (or have achieved symptomatic but not complete remission) are scarce, although the ongoing cluster randomization trial, REACT-2, aims to address this. 91 Data from cohorts of patients treated with VDZ or UST are scarce but appear to follow a similar trend82,85,92
Results – therapeutic drug monitoring
TDM is the cornerstone in optimizing biologic therapy so as to enable the maximum benefit that can be obtained from a drug before considering dose escalation or initiating a switch. TDM has assumed even greater significance, given the loss of response that occurs with anti-TNF medications over a period of time. 5
TDM typically involves measuring the trough level of the drug along with the presence of anti-drug antibodies (ADAbs). Ideally, TDM assays should be drug tolerant, where quantitative trough and ADAbs levels are both reported. Drug-sensitive assays, although cheaper, are sub-optimal as they cannot measure ADA in the presence of a drug. 93
Typically, trough levels should be drawn 24 hours before the next infusion to get a true trough level, but this may not always be possible.
Although trough antibody levels are comparable across assays, quantitative ADA titres are not comparable across assays, and hence it is ideal if the same laboratory is used to perform serial TDMs on the same patient to enable comparisons.
TDMs can be used reactively or proactively. Reactive TDM is typically used in patients with evidence of active inflammation who are not responding to treatment (have never responded, i.e., primary non-responders, or who have lost response after initially responding, i.e., secondary loss of response), in order to guide decision-making. 92
A randomized controlled trial was performed that compared reactive TDM versus empiric dose escalation in CD disease patients. 94 No difference was observed in primary end point of achieving remission; however, the therapeutic trough level of IFX in the study was ⩾0.5 mcg/mL, which is much lower than the accepted trough infliximab level of ⩾5 mcg/mL, with the potential for patients inappropriately deemed as having mechanistic failures. However, cost savings were noted in the reactive TDM group. 94
Proactive TDM may be performed irrespective of the clinical status but is generally performed in a patient with clinical response, to optimize therapy and prevent future flares.95,96 Two trials have evaluated the concept of proactive TDM in comparison with clinically guided dosing during maintenance therapy. The TAXIT trial 95 used a ‘treat to trough strategy’ where all IBD patients were first optimized to a target trough concentration between 3 and 7 mcg/mL, following which they were randomly assigned to receive IFX based on their clinical features versus continued therapy based on their trough concentrations. The study failed to meet its primary end point of clinical and biochemical remission after 1 year of treatment; however, several secondary end points were met such as fewer flares and less-acute infusion reactions in the groups that had proactive trough level measurements. It has been suggested that meaningful differences in the proactive TDM group may have shown up after 1 year, and that the trial was stopped prematurely.
Another similar trial 96 (TAILORIX), looked at comparing incremental dose increases of IFX based on clinical symptoms, biomarkers, and IFX trough concentrations, in comparison to empiric dose escalation based on clinical symptoms alone, in bio-naïve CD patients post induction at week 14. This study also failed to meet its primary end point, and no differences were observed on corticosteroid free remission both the treatment groups.
Proactive TDM has also been studied in the induction phase. Certain aggressive phenotypes such as perianal/penetrating CD, acute severe UC may require higher than the normal anti-TNF trough concentrations, which are best evaluated post-induction, to guide future maintenance therapy, and may be even more important than proactive TDM done in the maintenance phase. 97 Early IFX trough concentration optimisation has been associated with increased short-term mucosal healing rates and lower rates of ADAbs for IFX with effects seen as early as week 2 in patients with UC. 98
Data such as these have prompted the Sydney consensus 99 and BRIDGe consensus-Rand panel 95 to advocate measuring trough levels for anti-TNFs in responders post-induction therapy. The AGA guidelines, in contrast, do not unequivocally support proactive TDM. 100
Proactive TDM should probably also be performed while optimizing anti-TNF monotherapy. 95 In patients on concomitant anti-TNF and immunomodulatory therapy, in whom there is consideration of discontinuing the immunomodulatory due to risk of opportunistic infections and hepato-splenic T-cell lymphoma, anti-TNF trough concentrations should be checked both before and after stopping the immunomodulator. This phenomenon has been well documented with IFX, wherein a fall in IFX trough concentration occurs after stopping the immunomodulator, necessitating dose escalation commensurate with this fall, in order for the patient to continue maintain clinical remission. 101
There have been three consensus statements on TDM in IBD published in recent years.97,99,100 They all agree that TDM should be performed reactively, in patients with evidence of active ongoing inflammation associated with a primary or secondary loss of response. The premise of reactive TDM is that disease activity has to be confirmed following which the TDM can be measured. The algorithm below (Table 3) represents how to interpret reactive TDM and the decisions involved after incorporating these results.
Suggested algorithm to manage anti-TNF therapy in IBD.
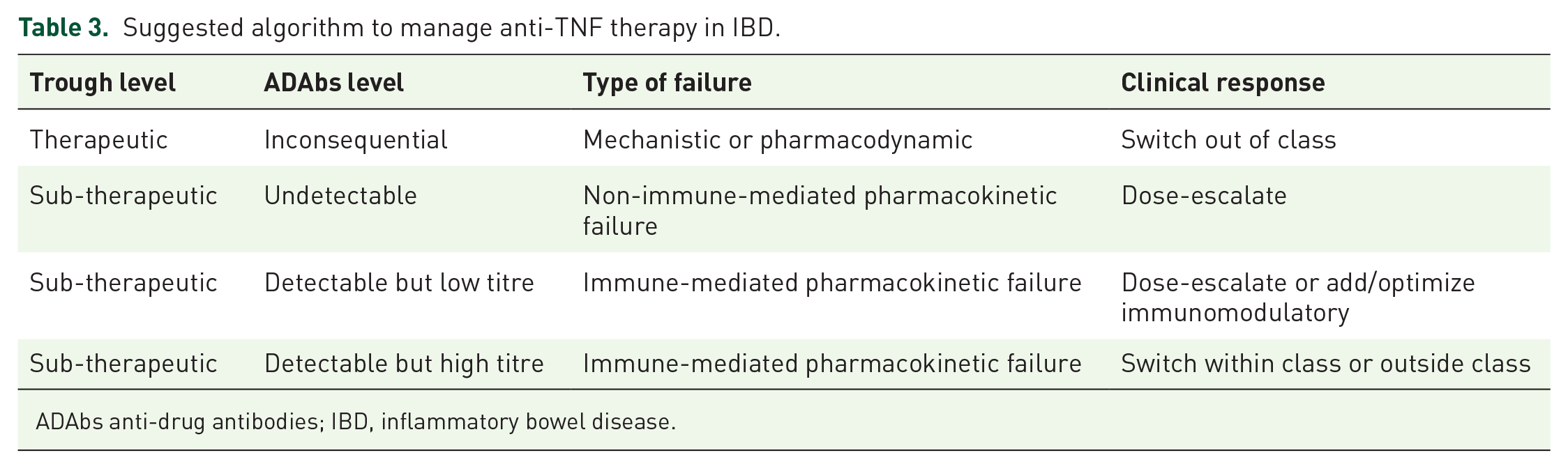
ADAbs anti-drug antibodies; IBD, inflammatory bowel disease.
Guidelines on proactive TDM monitoring are less consistent. Current AGA guidelines make no recommendation on proactive TDM in IBD. 100 The Sydney consensus 99 and BRIDGe consensus-Rand panel 97 both recommend proactive TDM to be done at the end of induction therapy to guide further management in the maintenance phase. The Sydney consensus 99 recommends TDM testing during the maintenance phase periodically, if the results are likely to change management in patients on anti-TNF therapy. The BRIDGe consensus-Rand panel 97 recommends proactive TDM at least once during the maintenance phase in patients on anti-TNF therapy.
Despite endorsements of proactive TDM by the Sydney consensus 99 and BRIDGe consensus-Rand panel, 97 there are certain drawbacks to proactive TDM which must be highlighted. First, target trough concentrations for patients in remission have been poorly defined with sub-optimal discriminatory thresholds leading to inappropriate dose changes. Second, there is no consensus with regard to what to do in situations where incidental findings of ADAbs are found. Finally, and perhaps most importantly, the feasibility of doing such frequent drug levels and resource utilization comes into question.
Target trough concentrations for different anti-TNFs have been suggested. However, the target trough concentration is a dynamic number and may differ depending on the inflammatory burden, timing of assessment (induction or maintenance) or the target goal attempting to be achieved (e.g clinical remission vs deep remission vs histological remission). However, the underlying theme is the same, to consider higher trough concentrations for more aggressive disease phenotypes (perianal or penetrating CD, acute severe UC), and when more targeted outcomes are being considered (endoscopic and histological remission).
For example, the AGA recommends target trough concentrations for IFX of at least 5 mcg/mL, adalimumab of at least 7.5 mcg/mL and certolizumab of at least 20 mcg/mL. 100 The Sydney consensus recommends an IFX trough concentration of between 3 and 8 mcg/mL for luminal disease and >10 mcg/mL for perianal disease. For adalimumab, they recommend a trough level of between 5 and 12 mcg/mL for luminal disease. 99 The BRIDGe consensus-Rand panel recommends IFX trough level of at least 3 mcg/mL for maintenance and at least 7 mcg/mL for mucosal healing. For ADA, they recommend a trough concentration of at least 5 mcg/mL for maintenance and at least 7 mcg/mL for mucosal healing and not considering switching in active disease unless the trough concentration is at least 10 mcg/mL. For golimumab, they recommend a trough concentration of at least 1 mcg/mL during maintenance and a maintenance trough concentration for certolizumab of at least 15 mcg/mL. 97
Data on quantitative ADAbs titers are more limited and have been studied mainly for IFX. A high ADAbs titre for IFX differs depending on the type of assay used. For the ANSER (Promethus) assay ⩾10 U/mL is considered a high titre and for the Inform Tx/Lisa tracker (Miraca) assay a level of ⩾200 ng/mL is considered high. 97 A high quantitative ADAbs titre, which ever assay is used, should warrant switching within class to another anti-TNF assuming the trough concentrations are sub-therapeutic or switching out of class altogether. A low ADAbs titre can be overcome by dose escalation or adding/optimizing an immunomodulatory when the trough levels are sub-therapeutic (See Table 3).
There is no consensus from any of the groups regarding TDM either reactively or proactively in patients using vedolizumab or ustekinumab or small molecules. Accumulating evidence 93 suggests that drug levels for new biologics such as vedolizumab and ustekinumab may be clinically meaningful, but more data are needed before optimal positioning of TDM for newer biologics can be recommended routinely. 99
The pharmacokinetics of small molecules is different from that of large monoclonal antibodies. Thus far, no immunogenicity has been described for small molecules including tofacitinib. There is a linear association between tofacitinib dose and trough levels, hence TDM is not applicable for tofacitinib. 102
Conclusion
The main focus of this topic and systematic review has been to evaluate how to optimize the therapy of IBD with biologics and small molecule with the two available options of dose intensification and therapeutic drug monitoring (TDM). In the absence of controlled study on dose intensification and given the somewhat contrasting data of the studies applying TDM methodology, we tried to reach a consensus among experts with a Delphi consensus, on the basis of the available literature with the aim to provide the best patient’s care and to support the management even with regard to the interface with third payer.
Up to a third of patients with IBD have a primary non-response or sub-optimal response to biologics and small molecules. In addition, up to 50% of patients with an initial response, may experience loss of response, and the trend is not changed after introduction of agents with different mechanism of action. Reasons for loss of response include low serum drug level, high titre of anti-drug antibodies, obesity, faecal loss, and/or malnutrition although these are not mutually exclusive.
Despite the low quality of evidence, optimisation of biological therapy through dose intensification, may recapture response. This situation often may be an argument against the approval from the third payer. The consensus group, after reviewing the literature, has agreed that dose intensification, defined as either an increased dose or a shortening of the dosing interval (in the case of anti-TNF), should be attempted with the aim of achieving complete remission in patients with an initial sub-optimal response or subsequent loss of response. Moreover, this strategy should be used before swap to a different class, given attrition with response to biological therapy in previously biologic-exposed patients. After dose intensification and achieving and stabilizing the needed target, the dosage can be de-escalated to standard schedule, following a case-by-case evaluation. Of note, dose intensification with biologic therapy has not adversely modified the safety and very frequently re-capture response.
More specifically when using anti-TNF therapy, decision of dose escalation or switching within or outside the class can be made in a timely and structured manner using TDM (suggested algorithm-Table 3). In this regard, the choice of the appropriate assay is key, with a view to obtaining information regarding presence and titre of anti-drug antibodies (‘reactive’ TDM) and is endorsed by consensus guidelines as appropriate. More recently, data are accumulating to support the ‘proactive’ use of TDM at the end of the induction phase, during the maintenance phase, or to optimize serum levels with more aggressive disease such as with a high inflammatory burden, acute severe UC, or fistulizing CD disease.
More data are needed to elucidate the efficacy and usefulness of TDM for the newer biologics, such as vedolizumab and ustekinumab. In contrast, there is no role for TDM while using tofacitinib due to lack of immunogenicity and its different pharmacokinetic profile as compared to biologics.
Footnotes
Author contributions
VA and RN conducted the literature review, drafted the first recommendations, and supportive text. JKL critically reviewed and corrected the manuscript. All authors have discussed and voted the recommendations, revised, and approved the manuscript.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

