Abstract
Vedolizumab, an α4β7-integrin antagonist, is the first gut-selective monoclonal antibody that has been approved for the treatment of moderate-to-severe ulcerative colitis and Crohn’s disease in many countries in the world. However, questions still remain regarding its appropriate use and placement in current treatment algorithms. Therefore, we sought out to evaluate the existing literature on the use of vedolizumab in inflammatory bowel disease. From inception to 21 June 2015 we searched MEDLINE for phase III randomized control trials assessing the utility of vedolizumab in inflammatory bowel disease, of which three were identified. The GEMINI trials demonstrate that vedolizumab is an effective and safe treatment for patients suffering from moderate-to-severe ulcerative colitis (GEMINI I) and Crohn’s disease (GEMINI II and III). However, further studies are needed comparing its efficacy directly with anti-tumor necrosis factor therapies to allow for further delineation of current treatment algorithms as well as ensuring its long-term safety profile.
Introduction
Inflammatory bowel disease (IBD), which encompasses both Crohn’s disease (CD) and ulcerative colitis (UC), is an incurable chronic inflammatory disorder primarily affecting the gastrointestinal tract. It represents a significant problem for populations in industrialized countries with prevalence estimates in North America of up to 319 per 100,000 for CD and 249 per 100,000 for UC [Molodecky et al. 2012]. This is in conjunction with an increase in the incidence of IBD across many nations [Molodecky et al. 2012]. Consequently, IBD represents a substantial economic burden with annual disease-attributable costs estimated at $6.3 billion within the United States [Kappelman et al. 2008]. With pharmaceutical claims accounting for 35% and 27% of costs for CD and UC, respectively [Kappelman et al. 2008], the importance of effective maintenance strategies for patients with IBD is paramount.
Arguably the most significant therapeutic advancement in IBD over the last two decades has been the anti-tumor necrosis factor (anti-TNF) biologic agents including infliximab [Rutgeerts et al. 2005; Targan et al. 1997], adalimumab [Hanauer et al. 2006; Sandborn et al. 2012], golimumab [Sandborn et al. 2014], and certolizumab pegol [Schreiber et al. 2007]. Unfortunately, a notable proportion of patients either do not respond to induction therapy or have a secondary loss of response [Arias et al. 2015; Gisbert and Panes, 2009], which is thought to be due to lack of response to TNF-alpha-driven immune mechanisms, inter-individual pharmacokinetic variation or the formation of anti-drug antibodies [Maser et al. 2006; Seow et al. 2010; Rutgeerts et al. 2004]. Moreover, there are notable concerns regarding the risk of infection after initiating anti-TNF therapy [Ford and Peyrin-Biroulet, 2013]. Therefore, a need exists for new therapeutic agents for those who lose response to anti-TNF therapy, as well as among patients with moderate-to-severe IBD who are anti-TNF naïve but have safety concerns.
Recently, in the United States and Europe, vedolizumab (VDZ), a monoclonal antibody targeting α4β7-integrin [Feagan et al. 2005] has been approved for use in UC and CD. α4β7-integrin is an adhesion molecule expressed on the surface of gut-specific lymphocytes; by binding to mucosal vascular addressin cell adhesion molecule-1 (MAdCAM-1) on intestinal vasculature it plays a critical role in the mediation of leukocyte trafficking to the gut [Berlin et al. 1993; Hesterberg et al. 1996]. VDZ has gained notable attention due to its gut-selective nature, a clear advantage over its predecessor natalizumab, an antibody targeting α4-integrin, whose lack of specificity has been implicated in the development of progressive multifocal leukoencephalopathy (PML) [Langer-Gould et al. 2005; Van Assche et al. 2005]. Therefore, given this breakthrough in the management of IBD, alongside its unclear position in current treatment algorithms, we sought out to systematically review the evidence behind VDZ use in IBD.
Methods
To identify full-text citations, in the English language, of phase III randomized controlled trials evaluating the use of VDZ in IBD, we searched MEDLINE (1948 to 21 June 2015) using the following search strategy: (‘inflammatory bowel diseases [MeSH]’ OR ‘inflammatory bowel disease*’ OR ‘Crohn disease [MeSH]’ OR ‘ulcerative colitis [MeSH]’ OR ‘IBD’ OR ‘Crohn*’) AND (‘vedolizumab’). The authors selected these search terms, based on a recently well-received systematic review in IBD [Shahidi et al. 2012]. The authors subsequently searched the bibliographies of relevant reviews, guidelines and included studies to identify further citations for inclusion. In total, three citations [Feagan et al. 2013; Sandborn et al. 2013; Sands et al. 2014] that met our inclusion criteria were identified from the search protocol.
Results
GEMINI I
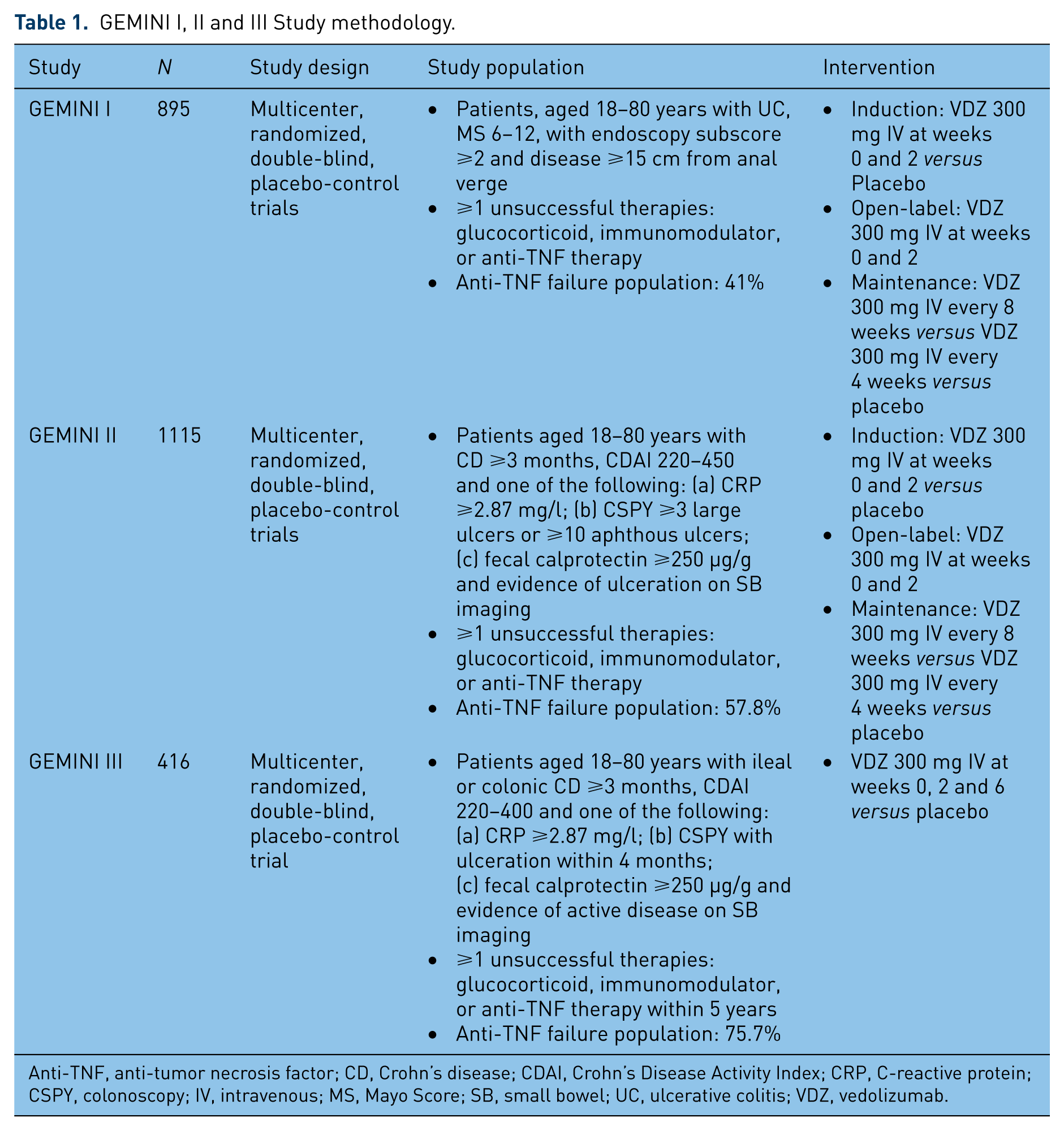
GEMINI I [Feagan et al. 2013] was an adaptive design, multicenter, randomized, double-blind, placebo-control trial assessing the efficacy of VDZ for inducing and maintaining remission among patients with moderate-to-severe UC (Mayo Score 6 to 12 points, with endoscopy subscore ⩾2 points and disease ⩾15 cm from anal verge) and previous failure or intolerance to corticosteroids, immunosuppressants or TNF antagonists (Table 1). For the induction trial, patients were randomized to either VDZ 300 mg at 0 and 2 weeks or placebo, with the primary outcome being clinical response (reduction in Mayo Score ⩾3 points, and decrease ⩾30% from baseline score, with a decrease of ⩾1 points on the rectal bleeding subscore or absolute rectal bleeding score ⩽1 point) at 6 weeks. In total, 374 patients underwent randomization, of which 47.1% receiving VDZ versus 25.5% receiving placebo (p < 0.001) achieved a clinical response at 6 weeks (Table 2). Significant differences in clinical remission (Mayo Score ⩽2 points and no subscore ⩾1 point) and mucosal healing (Mayo endoscopic subscore ⩽1 point) were also found to favor VDZ versus placebo.
GEMINI I, II and III Study methodology.
Anti-TNF, anti-tumor necrosis factor; CD, Crohn’s disease; CDAI, Crohn’s Disease Activity Index; CRP, C-reactive protein; CSPY, colonoscopy; IV, intravenous; MS, Mayo Score; SB, small bowel; UC, ulcerative colitis; VDZ, vedolizumab.
GEMINI I, II and III induction outcomes.

Anti-TNF, anti-tumor necrosis factor; CDAI, Crohn’s Disease Activity Index; VDZ, vedolizumab.
Reduction in Mayo Score ⩾3 points, and decrease ⩾30% from baseline score, with decrease of ⩾1 points on rectal bleeding subscore or absolute rectal bleeding score ⩽1 point.
Mayo Score ⩽2 points and no subscore ⩾1 point.
Mayo endoscopic subscore ⩽1 point.
CDAI score ⩽150 points.
⩾100-point decrease in CDAI score.
Once completed further patients were enrolled into a second cohort to receive open-label VDZ 300 mg at 0 and 2 weeks to facilitate an appropriately powered sample size for the maintenance trial. Patients (n = 373), from either the induction trial or those receiving open-label VDZ, who achieved clinical response at 6 weeks were then randomized to either VDZ 300 mg every 4 weeks, every 8 weeks or placebo as part of the maintenance trial. The primary outcome for the maintenance trial was clinical remission at 52 weeks (Table 3), which was achieved by 41.8% of patients on VDZ every 8 weeks, 44.8% of patients on VDZ every 4 weeks and 15.9% of patients receiving placebo (both p < 0.001 versus placebo). Significant differences in favor of VDZ (either every 4 or 8 weeks versus placebo) were also found for durable clinical response (clinical response at weeks 6 and 52), durable clinical remission (clinical remission at weeks 6 and 52), mucosal healing and glucocorticoid-free clinical remission. No significant differences were identified when comparing every 8 weeks versus every 4 weeks VDZ or among those receiving concomitant glucocorticoid or immunomodulator therapy. No differences in adverse events, including serious infections, were found between study groups. No cases of PML were identified.
GEMINI I and II maintenance outcomes.

CDAI, Crohn’s Disease Activity Index; NS, not significant; Q4W, every 4 weeks; Q8W, every 8 weeks; VDZ, vedolizumab.
Mayo Score ⩽2 points and no subscore ⩾1 point.
Reduction in Mayo Score ⩾3 points, and decrease ⩾30% from baseline score, with decrease of ⩾1 points on rectal bleeding subscore or absolute rectal bleeding score ⩽1 point at weeks 6 and 52.
Clinical remissiona at weeks 6 and 52.
Mayo endoscopic subscore ⩽1 point.
CDAI score ⩽150 points.
⩾100-point decrease in CDAI score.
Clinical remissione at ⩾80% of study visits including final week 52 visit.
GEMINI II
GEMINI II [Sandborn et al. 2013] was two multicenter, randomized, double-blind, placebo-control trials assessing the ability of VDZ to induce and maintain remission in patients with moderate-to-severe CD (CD ⩾3 months, Crohn’s Disease Activity Index [CDAI] 220 to 450 points and one of the following: (a) CRP ⩾2.87 mg/l; (b) colonoscopy (CSPY) ⩾3 large ulcers or ⩾10 aphthous ulcers; (c) fecal calprotectin ⩾250 µg/g and evidence of ulceration on small bowel imaging) and previous therapeutic failure (Table 1). Similar to GEMINI I, patients were either randomized to VDZ or placebo within the induction trial or were given open-label VDZ with patients showing response at 6 weeks (CDAI decrease ⩾70 points) subsequently being randomized to VDZ every 4 weeks, every 8 weeks or placebo for 52 weeks. The two primary outcomes of the induction trial were clinical remission (CDAI score ⩽150 points) and CDAI-100 response (⩾100-point decrease in CDAI score) at 6 weeks (Table 2). The primary endpoint of the maintenance phase trial was clinical remission at 52 weeks (Table 3).
Among the 368 patients within the induction trial, 14.5% receiving VDZ versus 6.8% receiving placebo were in clinical remission at 6 weeks (p = 0.02). No significant difference in CDAI-100 response (VDZ 31.4% versus placebo 25.7%; p = 0.23), and mean change in CRP or CRP normalization (⩽2.87 mg/l) were found between groups. During the maintenance trial (n = 461), clinical remission was achieved in 39% of patients on VDZ every 8 weeks, 36.4% of patients on VDZ every 4 weeks and 21.6% of patients receiving placebo (p < 0.001 and p = 0.004 versus placebo, respectively). Significant differences in CDAI-100 response, glucocorticoid-free clinical remission but not in durable clinical remission (clinical remission at ⩾80% of study visits including final week 52 visit) were found in favor of patients receiving VDZ. Among participants with previous anti-TNF failure, nonsignificant differences between VDZ and placebo were found for clinical remission (10.5% VDZ versus 4.3% placebo; p = 0.11) and clinical response (23.8% VDZ versus 22.9% placebo; p = 0.88) at 6 weeks. During the maintenance trial VDZ every 8 weeks (28.0%) and VDZ every 4 weeks (27.3%) were superior to placebo (12.8%) for clinical remission at 52 weeks (both p < 0.02 versus placebo).
As with GEMINI I, no cases of PML were identified. However, the incidence of serious adverse events, infections, serious infections and cancers were higher in the VDZ group.
GEMINI III
GEMINI III [Sands et al. 2014] was a multicenter, randomized, double-blind, placebo-control trial assessing the efficacy of VDZ among patients with moderate-to-severe CD (ileal or colonic CD ⩾3 months, CDAI 220 to 400 points and one of the following: (a) CRP ⩾2.87 mg/l; (b) CSPY with ulceration within 4 months; (c) fecal calprotectin ⩾250 µg/g and evidence of active disease on small bowel imaging), specifically focusing on previous anti-TNF failure. Patients were randomized to either VDZ 300 mg at 0, 2 and 6 weeks or placebo. The primary outcome was the ability of VDZ to induce remission (CDAI score ⩽150 points) at 6 weeks among patients with previous anti-TNF failure.
In total, 416 patients were randomized, with 76% suffering previous anti-TNF failure (Table 1). Among this large subgroup, 15.2% receiving VDZ versus 12.1% receiving placebo, achieved remission by 6 weeks (p = 0.433) (Table 2). On further analysis, 39.2% receiving VDZ versus 22.3% receiving placebo, achieved a CDAI-100 response (⩾100-point decrease in CDAI score) at 6 weeks (p = 0.001) and 26.6% receiving VDZ versus 12.1% receiving placebo achieved remission at 10 weeks (p = 0.001). When assessing the overall population, 19.1% receiving VDZ and 12.1% receiving placebo achieved remission at 6 weeks (p = 0.048) and 28.7% receiving VDZ and 13% receiving placebo achieved remission at 10 weeks (p < 0.001). There was no difference in adverse events, serious adverse events and serious infections between groups. No cases of PML were identified.
Discussion
While anti-TNF therapy has revolutionized the management of IBD, there still remains limited options for those who suffer primary nonresponse, secondary nonresponse and those with infectious-related limitations, consequently adding to their already notable morbidity. This underlines the importance of identifying a safe and effective treatment option for these disenfranchised populations. With the above in mind, we sought out to evaluate the evidence for VDZ in moderate-to-severe UC and CD. This review, focusing on the GEMINI trials, highlights VDZ’s emergence as an effective tool for inducing and maintaining remission in patients with moderate-to-severe disease activity who have undergone previous unsuccessful attempts with conventional therapies, thus ushering in a new era which now incorporates VDZ into current treatment guidelines [Bressler et al. 2015].
Safety profile
VDZ’s intrinsic appeal lies in its gut-specific mechanism of antagonizing α4β7-integrin, recently highlighted by a randomized trial which showed reduced seroconversion to oral cholera vaccination compared to parenteral hepatitis B vaccination in healthy participants who received VDZ [Wyant et al. 2015]. Nevertheless, concern has lingered regarding its adverse events profile, mostly due to its predecessor natalizumab, and its association with the development of PML; a rare demyelinating disease of the central nervous system caused by the reactivation of JC virus. This is believed to be due to natalizumab’s nonspecific antagonism of both α4β7- and α4β1-integrin, with United States incidence estimates ranging from <1/1000 to 13/1000, dependent on duration of exposure, anti-JC virus antibody positivity, and previous immunosuppressive therapy [FDA, 2015]. Of note, with the availability of anti-JC virus antibody testing, there may still be a role for natalizumab in CD, given its efficacy in maintaining remission [Sandborn et al. 2005]. Among all participants in GEMINI I, II and III, no cases of PML were diagnosed. Moreover, in GEMINI LTS [Colombel et al. 2013], an ongoing open-label extension study, no cases of PML have been identified in more than 2 years of follow up.
Aside from PML, both GEMINI LTS, and a recent meta-analysis [Wang et al. 2014], have demonstrated the favorable risk profile of VDZ, with no significant association compared to placebo for either serious adverse events or serious infections. The most common adverse events, with relatively low frequency are: headache, nasopharyngitis, nausea, arthralgia, upper respiratory infection, and fatigue (all ⩽6%) [Colombel et al. 2013]. Given the above, VDZ has the potential for being an ideal agent and should be considered in those with infectious-related concerns, such as the impending burden of elderly patients suffering from IBD [Katz and Pardi, 2011].
Ulcerative colitis
In the setting of UC, GEMINI I is quite convincing with all primary and secondary outcomes, both in the induction and maintenance trials, confirming the superiority of VDZ versus placebo. This is further supported by a Cochrane meta-analysis for VDZ in UC [Mosli et al. 2015]. In four studies, encompassing 606 patients, VDZ was superior to placebo for achieving clinical response, clinical remission and endoscopic remission. In a recent network meta-analysis of seven randomized, double-blind, placebo-control trials for biologic therapy in UC, there was no significant difference between infliximab and VDZ for inducing remission [Danese et al. 2014]. Therefore, given VDZ’s efficacy and safety profile, it is a favorable therapeutic option in patients with UC who have shown a lack of response to glucocorticoids, immunomodulators and anti-TNF therapy; and while head-to-head comparisons are needed, VDZ may emerge as an equivalent or superior option to anti-TNF therapy in the management of ambulatory patients with UC. These sentiments have been echoed in the 2015 Toronto Consensus guidelines for nonhospitalized UC [Bressler et al. 2015] recommending the use of VDZ among those with moderate-to-severe UC who do not respond to corticosteroids, immunomodulators or anti-TNF therapy.
Crohn’s disease
GEMINI II and III are less compelling for the use of VDZ in CD; most concerning being VDZ’s ability to induce remission at 6 weeks and the lack of mucosal healing data which was not collected in the phase III program. Some of this has been attributed to differences in study design most notably the percentage of anti-TNF failures included in the study. In addition, the inherent mechanism of action may mean that a subpopulation of patients may require a longer duration of treatment in order to obtain remission. GEMINI III showcased VDZ’s superiority to placebo in inducing remission at 10 weeks alongside GEMINI II demonstrating VDZ’s superiority to placebo for achieving clinical remission and glucocorticoid-free remission at 52 weeks, both of which are reassuring. This is further supported by a large network meta-analysis of 39 randomized control trials assessing the utility of biologic therapy in CD [Hazelwood et al. 2015]. VDZ was superior to placebo for inducing and maintaining remission in CD; of note, VDZ did show inferiority to adalimumab for maintaining remission. Taking this altogether and the lack of mucosal healing data, VDZ is an effective therapy for CD, with its most appropriate use being after failed anti-TNF therapy and potentially among those who have failed glucocorticoid or immunomodulator therapy with clinical factors that increase the risk of anti-TNF therapy.
Therapeutic drug monitoring
Quantifying drug trough levels and anti-drug antibodies have garnered notable attention given their correlation with treatment outcomes and their potential for refining current treatment algorithms [Vande Casteele et al. 2015a]. In GEMINI I, II and III mean VDZ trough concentrations at 6 weeks were 27.9, 26.8 and 26.5 µg/ml, respectively. Alongside this, a positive correlation was shown in GEMINI I and II between VDZ levels and clinical efficacy. However, an intriguing finding was that regardless of dosing frequency (either every 4 or 8 weeks), both led to α4β7 saturations ⩾95% in serum lymphocytes. Concerning anti-VDZ antibodies, between 1–4.1% of participants had positive results, where only 0.4–1% were persistently positive (⩾2 consecutive samples) in GEMINI I and II, with no participants in GEMINI III having persistently positive results. In both GEMINI I and II, concomitant immunosuppressive therapy was associated with decreased immunogenicity, which is consistent with results for anti-TNF therapy [Vande Casteele et al. 2015b]. Interestingly, a recent study aggregating study participants from phase I, phase II and phase III VDZ studies showed that only extreme albumin and body weight are potentially meaningful to drug clearance with anti-drug antibodies and concomitant immunotherapy being deemed not clinically relevant [Rosario et al. 2015].
The findings above suggest some level of discordance between serum saturation levels of α4β7 and clinical efficacy as well as that anti-drug antibody levels and concomitant immunotherapy may not have as much relevance as with anti-TNF therapy; however, these results need to be interpreted with caution with further studies evaluating this critical area. Of note, VDZ therapeutic drug monitoring is currently not commercially available.
Future directions
Subsequent to the GEMINI studies, large multicenter cohorts have gone on to showcase VDZ’s efficacy and safety in ‘real-world’ settings for both UC and CD [Amiot et al. 2015a, 2015b; Shelton et al. 2015]. However, aside from the obvious need for head-to-head comparisons of currently available biologic agents, and the evaluation of VDZ to achieve mucosal healing in CD, there are areas of interest that require further evaluation, including the role of VDZ in pregnancy, perianal fistulizing Crohn’s disease, and VDZ’s effects on extra-intestinal manifestations and primary sclerosing cholangitis.
Concerning pregnancy, although no studies have assessed this specifically a total of 24 study participants who received VDZ had unplanned pregnancies: nine live births, three spontaneous abortions, five elective terminations and seven having unknown outcomes as of June 2013 [Takeda, 2013]. One congenital anomaly, agenesis of the corpus callosum with left frontal polymicrogyria, was diagnosed in a male child of a healthy 28-year-old participant who had received one dose of VDZ. Alongside this, animal studies have not shown fetotoxicity in high doses (100 mg/kg) of VDZ [Takeda, 2013]. Noting the above, appropriate pregnancy and breastfeeding-related protocols are needed, with those for anti-TNF therapy being an initial foundation. Concerning extra-intestinal manifestations, no data are available at this time, however, results will be intriguing given the gut-selective nature of VDZ potentially limiting its clinical utility in these settings. However, in the setting of primary sclerosing cholangitis, VDZ may show promise given the hepatic expression of MAdCAM-1 [Grant et al. 2001].
Conclusion
In summary, the GEMINI trials have marked the arrival of VDZ as an effective therapy for patients with moderate-to-severe UC or CD who have failed previous conventional therapy. However, to further delineate treatment algorithms, head-to-head randomized trials comparing VDZ versus anti-TNF therapy, VDZ versus emerging biologic therapies (e.g. ustekinumab) as well as VDZ versus VDZ and concomitant immunomodulator therapy are needed. Moreover, as our experience with VDZ continues to grow, further needed data will become available, allowing us to assess VDZ’s potential superiority among those with malignancy or infectious-related concerns as well as shine a light on its utility in those with extra-intestinal manifestations.
Footnotes
Acknowledgements
We have no financial or intellectual acknowledgements to disclose.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Neal Shahidi: No conflicts of interest.
Brian Bressler: Consultant for Celltrion, Pendopharm. Speaker’s Bureau for Abbvie, Janssen, Takeda, Actavis. Advisory Board for Abbvie, Janssen, Takeda, Shire, Genentech, Ferring, Warner Chilcott. Research Support from Abbvie, Amgen, BMS, Genentech, Janssen, BI, GSK, Pfizer and RedHill Biopharma.
Remo Panaccione: Consultant for Abbvie, Abbott, Amgen, Aptalis, AstraZeneca, Baxter, Eisai, Ferring, Janssen, Merck, Schering-Plough, Shire, Centocor, Elan, GSK, UCB, Pfizer, Bristol-Myers Squibb, Warner Chilcot, Takeda, Cubist, Celgene. Speaker’s Bureau for Abbvie, Abbott, AstraZeneca, Janssen, Schering-Plough, Shire, Centocor, Elan, Prometheus, Warner Chilcott, Takeda. Advisory Board for Abbvie, Abbott, Amgen, Aptalis, AstraZeneca, Baxter, Eisai, Ferring, Genentech, Jansen, Merck, Schering-Plough, Shire, Centocor, Elan, GSK, UCB, Pfizer, Bristol-Myers Squibb, Warner Chilcott, Takeda, Cubist, Celgene, Salix.
Research/Educational Support from Abbvie, Abbott, Ferring, Janssen, Schering-Plough, Centocor, Millenium, Elan, Proctor and Gamble, and Bristol-Myers Squibb.
