Abstract
Background:
The skin–gut axis, characterized by bidirectional communication between the skin and gut, plays a crucial role in the pathogenesis of psoriasis and inflammatory bowel diseases (IBD).
Objectives:
We aimed to explore the association between psoriasis and IBD and identify predictors associated with IBD development among patients with psoriasis.
Design:
Retrospective cohort study.
Methods:
A retrospective study which utilized an electronic database from the Meuhedet Health Maintenance Organization (MHMO) in Israel. Psoriasis was categorized as severe if any systemic agent or phototherapy was administered. Univariate and multivariate logistic regressions were used to identify specific predictors for IBD, with adjustments made for potential confounders. The study received approval from the Ethical Committee of the MHMO.
Results:
In total, 61,003 adult patients who were diagnosed with psoriasis between 2000 and 2022 were included. Among them, 1495/61,003 patients (2.4%) were diagnosed with IBD, as compared to 3834/244,012 patients (1.6%) in the non-psoriasis group [adjusted odds ratio (OR): 1.47; 95% confidence interval (CI): 1.37–1.56; p < 0.001]. Increased age (OR: 1.01; 95% CI: 1.01–1.02; p < 0.001), male gender (OR: 1.22; 95% CI: 1.03–1.45; p = 0.024), and Jewish ethnicity (OR: 2.5; 95% CI: 1.2–4.1; p < 0.001) were identified as significant risk factors for IBD. Spondyloarthropathies, including psoriatic arthritis (OR: 2.27; 95% CI: 1.86–2.77; p < 0.001) and ankylosing spondylitis (OR: 2.82; 95% CI: 1.5–5.32; p < 0.05), were associated with a higher prevalence of IBD. Furthermore, severe psoriasis was significantly associated with a higher likelihood of IBD, compared to mild psoriasis (OR: 16.03; 95% CI: 11.02–23.34; p < 0.001).
Conclusion:
A significant association between psoriasis and IBD was demonstrated, including its subtypes: Crohn’s disease and ulcerative colitis. Moreover, such association may depend on psoriasis severity as determined by the treatment used. This association warrants further investigation and implies a potential need for closer monitoring of patients with severe psoriasis.
Plain language summary
1- Gut and skin barrier play an integral role in psoriasis and inflammatory bowel disease (IBD) development. 2- Shared genetic and environmental factors could explain the association between both diseases. 3- We report increased association between psoriasis and IBD, a relationship that is more pronounced in patients with severe psoriasis. 4- Patients with spondyloarthritis related diseases have a stronger association with IBD.
Introduction
Psoriasis, a prevalent systemic inflammatory condition, exhibits a heightened likelihood of specific comorbidities when compared to both the general population and individuals with alternative skin disorders, particularly in its severe manifestations. 1 Despite notable progress, numerous aspects related to the comprehension of psoriasis’ pathogenesis still lack resolution. 2 Nonetheless, it is widely accepted that psoriasis arises from an intricate interaction among genetics, environmental stimuli, and the immune system. 2
Crohn’s disease (CD) and ulcerative colitis (UC) are the two predominant types of chronic relapsing inflammatory disorders of the gastrointestinal (GI) tract, collectively referred to as inflammatory bowel disease (IBD). IBD is believed to occur as a host response to intestinal microbes in genetically predisposed individuals. 3
Several lines of evidence suggest a possible association between the gut–joint–skin axis, with some epidemiological studies showing an association between IBD and psoriasis while others negate such a relationship.4,5 The coexistence of psoriasis and IBD can potentially be explained by common genetic background, 6 similar pathogenic pathways,6,7 or specific interaction between the immune system8,9 and the microbiota. 10 The prevailing hypothesis postulates that the pathogenesis of psoriasis and/or IBD arises from the interplay of environmental and immunological factors in individuals with a genetic predisposition. Epigenetic mechanisms, including DNA methylation, histone modifications, and noncoding RNA-mediated gene regulation, also play a significant role. Interestingly, many disease-associated variants in both IBD and psoriasis are found in noncoding regions, hinting at the importance of epigenetic regulation. 11 There is evidence of extensive alterations in DNA methylation patterns in psoriatic skin, even in unaffected areas.12,13
Treatment approaches for both conditions exhibit significant overlap, with immunosuppressants serving as the fundamental therapeutic agents.
The primary objective of this study was to elucidate the relationship between psoriasis and IBD. Specifically, we tried to determine the magnitude of this association with respect to the severity of psoriasis. An additional objective of this study was to identify specific predictive factors in patients with psoriasis that were associated with an elevated risk of developing IBD. To accomplish this, we analyze data derived from a large population-based registry.
Methods
Study design and population
In this retrospective study, we aimed to examine the correlation between psoriasis and IBD within a specified population. The study utilized the electronic database of one of the four health maintenance organizations in Israel as the source of data for analysis. The data search included all individuals aged 18 or older who were diagnosed with psoriasis, identified by International Classification of Diseases-9 (ICD-9) codes 696, 696.0, and 696.1, between 1 January 2000 and 31 December 2020. The dataset used for analysis contained information available until 10 February 2022.
Data collection and definitions
This study utilized data sourced from the Meuhedet Health Maintenance Organization (MHMO), which serves a vast population of 1.2 million patients throughout Israel. Operating under the Israeli National Health Insurance Act implemented in 1994, MHMO ensures comprehensive healthcare coverage accessible to all citizens. Enrolment in one of the four HMOs is obligatory for every Israeli, guaranteeing direct healthcare services and insurance coverage, irrespective of gender, age, health status, or other factors. MHMO’s extensive database consolidates patient medical records from various healthcare providers, encompassing diagnostic visits, pharmaceutical interventions, in-office procedures, laboratory test results, imaging studies, and comprehensive summaries of hospital encounters such as outpatient clinic visits, emergency department visits, and in-patient discharge records. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 14
Variables and measures
Various parameters, such as age, gender, and ethnicity (classified as Arab or Jewish), were taken into consideration in this study. The primary objective was to investigate the occurrence of IBD, identified through recorded diagnoses using ICD-9 codes 555. This category was further divided into CD (ICD-9 codes 555.0, 555.1, 555.9) and UC (ICD-9 codes 556.9). Similarly, other psoriasis-related conditions, including psoriatic arthritis (PsA, ICD-9 codes 696.0, 713.3) and spondyloarthropathies such as ankylosing spondylitis (AS, ICD-9 codes 720), were identified using a similar methodology. Diagnoses were validated by having at least two records with similar diagnoses from specialists. Body mass index (BMI) was calculated based on height and weight measurements taken closest to the date of psoriasis diagnosis and categorized as either ⩾30 (obese) or <30 kg/m². Information regarding chronic comorbidities such as type 2 diabetes, ischemic heart disease (IHD), stroke, chronic obstructive pulmonary disease (COPD), congestive heart failure (CHF), and cancer was obtained from both the MHMO and the national chronic diseases registry, which underwent prior validation.
Treatment methods, including phototherapy, methotrexate (MTX), anti-tumor necrosis factor-α agents (anti-TNF), anti-interleukin (IL)-12/23 agents, Janus kinase inhibitors (JAKi), and anti-IL-17 agents, were identified based on the recorded dispensation of these agents after the diagnosis of psoriasis. Treatment was then classified as either ‘ever used’ or ‘never used’. Psoriasis was categorized as severe if any systemic agent or phototherapy was administered, while it was classified as mild if only topical agents were used.
Statistical analysis
Prior to data analysis, each psoriasis patient was meticulously matched with four controls who did not have a psoriasis diagnosis, considering their date of birth, gender, and ethnicity. A comparative analysis was then conducted between the two groups to assess the occurrence of IBD, including UC or CD. The diagnoses were determined by a gastroenterologist using the ICD-9 criteria.
Continuous variables were reported as mean ± standard deviation, and the Student’s t-test was employed to compare these variables. Categorical variables were presented as percentages and analyzed using Pearson’s chi-square test. To examine the association between psoriasis and IBD, both univariate and multivariate logistic regression models were utilized, with adjustments made for potential confounding factors. The multivariate logistic regression model, using the ‘Enter’ method, was employed to identify specific parameters that were associated with a higher likelihood of developing IBD within the psoriasis cohort. Only variables that demonstrated significance in the univariate model were included in this analysis. All p values were considered two-tailed, and the null hypothesis was accepted if p ⩾ 0.05. The statistical analysis was performed using IBM Corp’s SPSS software, version 26 (SPSS, Armonk, NY, USA).
Ethical considerations
The Ethical Committee of MHMO approved the study, which was exempted from obtaining informed consent.
Results
Study population
The study included a total of 61,003 patients diagnosed with psoriasis, and they were compared to 244,012 controls who were matched in terms of age, gender, and ethnicity. Table 1 presents the characteristics of each group and Figure 1 represents the distribution of patients in this study. Both study and control groups had a mean age of 36.1 years [standard deviation (SD) ±19.3 years], with an equal distribution of 49.2% males and 11.5% individuals of Arab ethnicity. The prevalence of chronic comorbidities was significantly higher among psoriasis patients compared to the control group. Notably, psoriasis patients exhibited higher rates of obesity (26.3% versus 22.4%; p < 0.001), diabetes (16.3% versus 14.0%; p < 0.001), IHD (10.2% versus 9.0%; p < 0.001), stroke (5.9% versus 5.4%; p < 0.001), COPD (9.5% versus 7.8%; p < 0.001), CHF (3.0% versus 2.6%; p < 0.001), and cancer (11.9% versus 10.5%; p < 0.001). Furthermore, psoriasis patients had a significantly higher prevalence of spondylarthritis-related comorbidities, specifically PsA (6.1% versus 0.0%; p < 0.001) and axial spondyloarthropathies (3.2% versus 2.3%; p < 0.001).
Characteristics of the study population.

ANA, antinuclear antibody; anti-IL, anti-interleukin; anti-TNF, anti-tumor necrosis factor; ASCA, anti-Saccharomyces cerevisiae antibodies; COPD, chronic obstructive pulmonary disease; IHD, ischemic heart disease; JAKi, Janus kinase inhibitors; p-ANCA, perinuclear anti-neutrophil cytoplasmic antibodies; SD, standard deviation.
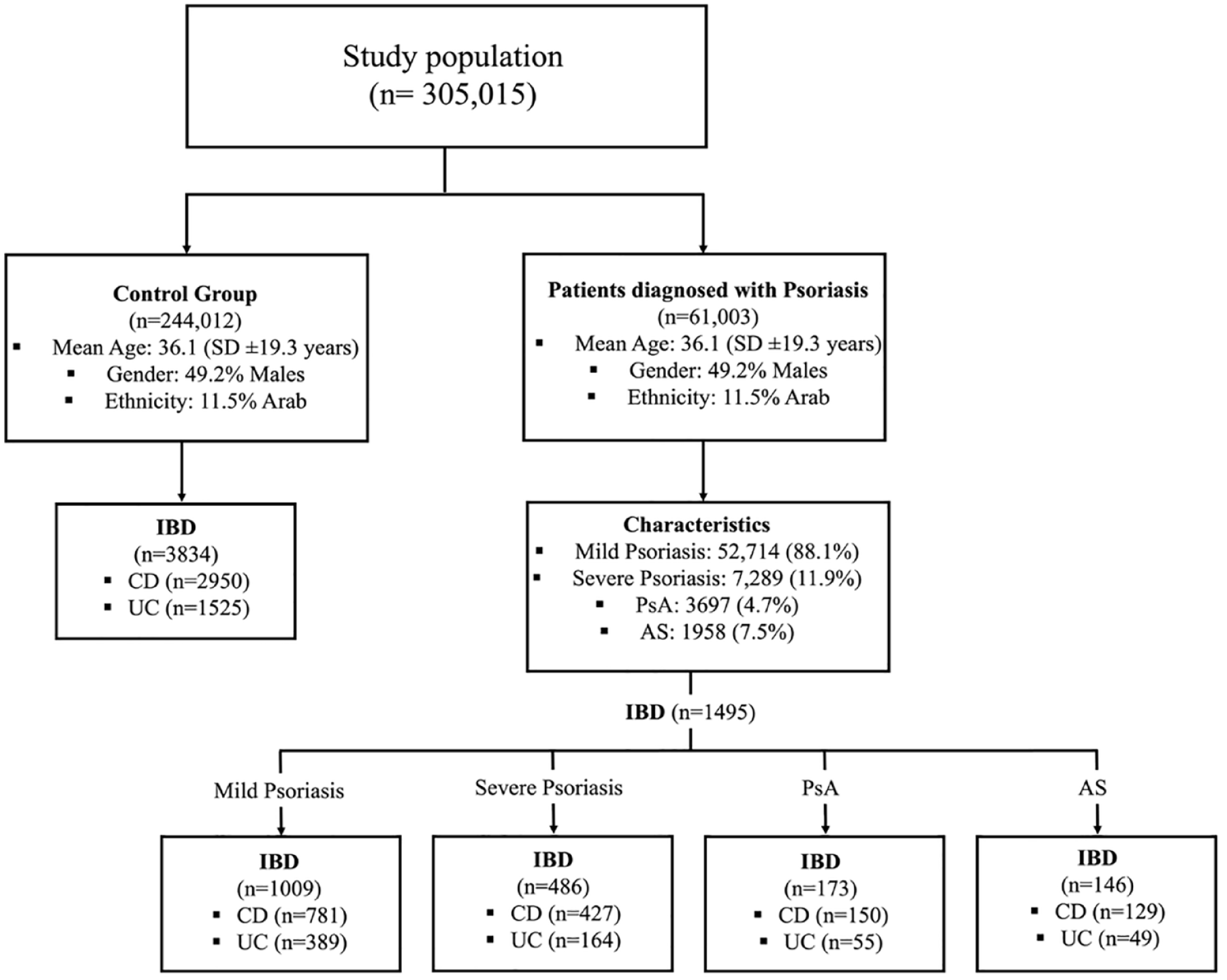
Patients’ distribution in the study.
Regarding the characteristics of psoriasis within the overall population, our analysis revealed that among the individuals included, mild psoriasis was present in 52,714/61,003 individuals (88.1%), while severe psoriasis was observed in 7289/61,003 patients (11.9%). Various treatment approaches were employed, with phototherapy being the most commonly used (5039/61,003 patients, 8.3%), followed by MTX (2289/61,003 patients, 3.1%), anti-TNF agents (1385/61,003 patients, 2.3%), anti-IL-12/23 agents (247/61,003, 0.4%), anti-IL-17 agents (128/61,003 patients, 0.2%), and JAKi (24/61,003, 0.04%). In addition, a small proportion of patients (525/61,003, 0.9%) received multiple lines of biological treatment as part of their therapeutic regimen.
Characteristics of patients with psoriasis and IBD
Out of the 61,003 psoriasis patients enrolled in this study, a total of 1495 individuals (2.4%) were diagnosed with IBD. Among these patients, 553/61,003 patients (0.9%) had UC, and 1208/61,003 patients (1.9%) were diagnosed with CD. The average age at the time of psoriasis diagnosis was 38.3 years (SD ± 17.3 years), while the average age at the time of IBD diagnosis was 39.1 years (SD ± 17.7 years). Table 2 presents demographic and clinical characteristics of psoriasis patients with IBD. Within the psoriasis patients with concurrent IBD, approximately half of the patients were male (760/1495 patients, 50.8%), and 87/1495 patients (5.8%) were of Arab ethnicity. In terms of psoriasis severity, 1009/1495 patients (67.5%) with concurrent IBD had mild psoriasis, while 486/1495 patients (32.5%) had severe psoriasis. PsA and AS were present in 173/1495 patients (11.6%) and 146 patients (9.8%), respectively.
Demographic and clinical characteristics of psoriasis population with IBD.

Anti-IL, anti-interleukin; anti-TNF, anti-tumor necrosis factor; CD, Crohn’s disease; IBD, inflammatory bowel diseases; IQR, interquartile range; JAKi, Janus kinase inhibitors; SD, standard deviation; UC, ulcerative colitis.
Regarding the sequence of diagnoses, among the patients with concurrent IBD and psoriasis, 776/1495 patients (51.9%) received their IBD diagnosis before being identified with psoriasis, while the remaining 720/1495 patients (48.1%) were diagnosed with IBD after psoriasis. In terms of treatment approaches, within the cohort of patients with concurrent IBD and psoriasis, 163/1495 patients (10.9%) received phototherapy, 135/1495 patients (9%) were treated with MTX, and 343/1495 patients (22.9%) underwent anti-TNF therapy, which was the leading therapeutic modalities in the study.
Risk of IBD in psoriasis patients
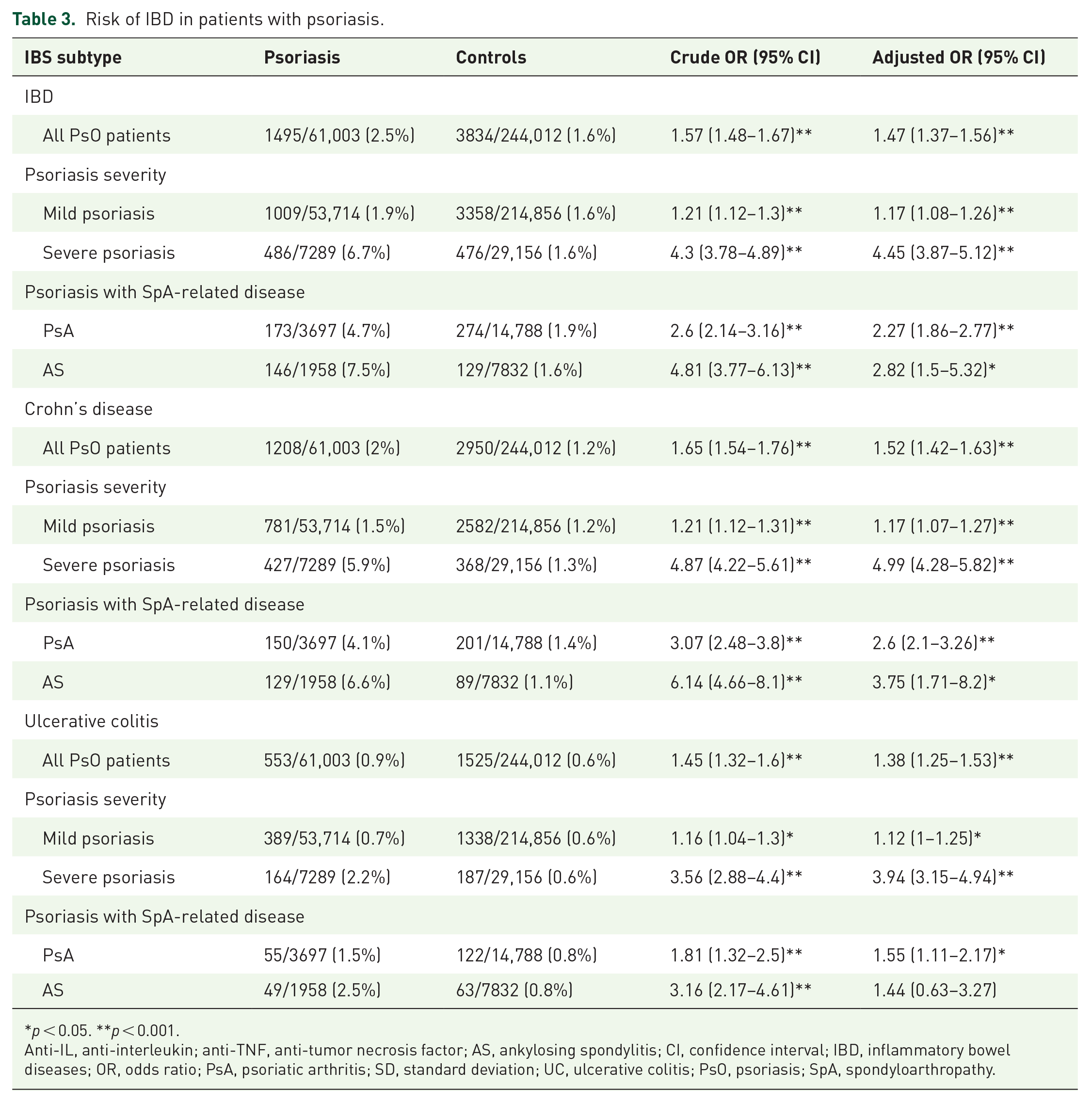
Regarding the association of psoriasis and IBD, we observed that 1495/61,003 patients (2.5%) with psoriasis were diagnosed with IBD, whereas in the control group, the number was 3834/244,012 (1.6%). The crude odds ratio (OR) for this association was 1.57 (95% CI: 1.48–1.67, p < 0.001), and after adjusting for potential confounders such as age, gender, and various comorbidities, the adjusted OR (aOR) was 1.47 (95% CI: 1.37–1.56). When stratifying the data based on psoriasis severity, both mild and severe psoriasis were significantly associated with an increased risk of IBD. The crude OR for mild psoriasis was 1.21 (95% CI: 1.12–1.30; p < 0.001), and after adjustment, the OR remained significant at 1.17 (95% CI: 1.08–1.26; p < 0.001). For severe psoriasis, the crude OR was 4.3 (95% CI: 3.78–4.89; p < 0.001), and after adjustment, the OR remained substantially elevated at 4.45 (95% CI: 3.87–4.12; p < 0.001). In addition, when examining different spondyloarthropathies, both PsA and AS showed significant associations with IBD; for PsA, the crude OR was 2.6 (95% CI: 2.14–3.16; p < 0.001), and the aOR was 2.27 (95% CI: 1.86–2.77; p < 0.001). For AS, crude OR was statistically significant 3.16 (95% CI: 2.17–4.61) while after adjustment it was found to be non-significant (aOR: 1.44; 95% CI: 0.63–3.27).
CD- and UC-specific risk
We conducted further analysis to examine the risk associated with specific subtypes of IBD using our dataset. Table 3 presents a risk of IBD in patients with psoriasis with sub-analysis for CD and UC. Among all patients with psoriasis, 1208/61,003 patients (2%) were concurrently diagnosed with CD, and 553/61,003 patients (0.9%) were diagnosed with UC. Both associations were found to be significant when compared to the control group, with an aOR of 1.52 (95% CI: 1.42–1.63; p < 0.001) for CD and 1.38 (95% CI: 1.25–1.53; p < 0.001) for UC.
Risk of IBD in patients with psoriasis.
p < 0.05. **p < 0.001.
Anti-IL, anti-interleukin; anti-TNF, anti-tumor necrosis factor; AS, ankylosing spondylitis; CI, confidence interval; IBD, inflammatory bowel diseases; OR, odds ratio; PsA, psoriatic arthritis; SD, standard deviation; UC, ulcerative colitis; PsO, psoriasis; SpA, spondyloarthropathy.
Furthermore, we tried to characterize the relationship between the IBD subtypes and psoriasis severity. Mild psoriasis was significantly associated with both CD and UC, with an aOR of 1.17 (95% CI: 1.07–1.27; p < 0.001) for CD and 1.12 (95% CI: 1.00–1.25; p < 0.05) for UC. In addition, the presence of severe psoriasis was strongly associated with CD (aOR: 4.99; 95% CI: 4.28–5.82; p < 0.001) and UC (aOR: 3.94; 95% CI: 3.15–4.94; p < 0.001).
Nevertheless, we investigated the association between spondyloarthropathies and IBD subtypes. PsA was significantly associated with both CD (aOR: 2.6; 95% CI: 2.1–3.26; p < 0.001) and UC (aOR: 1.55; 95% CI: 1.11–2.17; p < 0.05). However, AS was found to be associated only with CD (aOR: 3.75; 95% CI: 1.71–8.2; p < 0.05).
IBD predictors within patients with psoriasis
The adjusted multivariate logistic regression analysis revealed significant associations with IBD across various factors which are fully presented in Table 4. Increased age (OR: 1.01; 95% CI: 1.01–1.02; p < 0.001 per 1-year increment), male gender (OR: 1.22; 95% CI: 1.03–1.45; p = 0.024), and Jewish ethnicity (OR: 2.5; 95% CI: 1.2–4.13; p < 0.001) were identified as significant risk factors for IBD. Figure 2 shows a forest plot demonstrating the various parameters that were analyzed as potential predictors associated with IBD in the psoriasis cohort. Spondyloarthropathies, including PsA (OR: 2.27; 95% CI: 1.86–2.77; p < 0.001) and AS (OR: 2.82; 95% CI: 1.5–5.32; p < 0.05), were also associated with a higher prevalence of IBD. Interestingly, obese patients (BMI > 30) exhibited a lower risk of IBD comorbidity (OR: 0.76; 95% CI: 0.62–0.94; p = 0.01).
Predictors for IBD within the psoriasis cohort, age, and sex-adjusted logistic regression analysis.

ANA, antinuclear antibody; ASCA, anti-Saccharomyces cerevisiae antibodies; CI, confidence interval; COPD, chronic obstructive pulmonary disease; IBD, inflammatory bowel diseases; OR, odds ratio; p-ANCA, perinuclear anti-neutrophil cytoplasmic antibodies; SE, standard error.

The forest plot represents the odds ratios of various predictors for IBD in patients with psoriasis.
In terms of psoriasis-related factors, severe disease was significantly associated with a higher likelihood of IBD (OR: 16.03; 95% CI: 11.02–23.34; p < 0.001), while treatment with phototherapy was inversely associated with the risk of IBD (OR: 0.09; 95% CI: 0.06–0.15; p < 0.001). In addition, the presence of uveitis was correlated with an increased risk of IBD (OR: 3.13; 95% CI: 2.08–4.71; p < 0.001). Serological markers, such as anti-Saccharomyces cerevisiae antibodies (ASCA) and perinuclear anti-neutrophil cytoplasmic antibodies (p-ANCA), were found to be associated with IBD risk (aOR: 10.71; 95% CI: 7.38–15.55; p < 0.001 for ASCA; aOR: 7.71; 95% CI: 2.04–29.04; p = 0.003 for p-ANCA).
Discussion
Our study shows an increased association between psoriasis and IBD, a finding that remains significant after adjusting for the various confounders. This association was discernible not only in the context of IBD as an entity but also in its distinct clinical subtypes, CD and UC. Moreover, our study illustrates that patients presenting with severe psoriasis exhibit an intensified correlation with IBD broadly and more specifically, with CD and UC. A similar trend of augmented association was noted among psoriasis patients with additional spondyloarthropathy-related comorbidities. The relationship was found to be more pronounced with CD compared to UC.
In our analysis to identify potential predictors of IBD within the psoriasis cohort, several significant factors were revealed; male gender, axial spondyloarthropathies, and uveitis were all found to be positively associated with an increased likelihood of developing IBD. Moreover, serological parameters, such as ASCA and p-ANCA, were also shown to be potentially correlated with IBD development. Contrarily, antinuclear antibody positivity was not linked to an increased IBD risk among patients with psoriasis.
While trying to investigate the causal relationship between IBD and psoriasis, several studies have utilized the Mendelian randomization (MR) approach. One such study used genetic instruments for IBD and its subtypes, CD and UC found that a genetic predisposition to IBD was linked to an increased risk of psoriasis (OR: 1.1268; 95% CI: 1.0662–1.1908). 15 The same study also discovered a significant association between total IBD and PsA (OR: 1.1217; 95% CI: 1.0047–1.2043) but found no such association for psoriasis vulgaris or UC. 15
On the other hand, a systematic review and meta-analysis of case–control, cross-sectional, and cohort studies found a strong association between psoriasis and both CD (OR: 1.70; 95% CI: 1.20–2.40) and UC (OR: 1.75; 95% CI: 1.49–2.05). 16 Patients with psoriasis showed a 2.53-fold increased risk of developing CD (risk ratio: 2.53; 95% CI: 1.65–3.89) and a 1.71-fold increased risk of developing UC (risk ratio: 1.71; 95% CI: 1.55–1.89) when compared to controls. 16 Notably, patients with PsA had a similar pattern of increased risk for developing CD and UC. 16
Moreover, another MR study, using a bidirectional two-sample approach with data from genome-wide association studies (GWAS), found that genetically predicted IBD was associated with a higher risk of psoriasis (pooled OR: 1.10; 95% CI: 1.05–1.15; p < 0.001) and PsA (pooled OR: 1.10; 95% CI: 1.04–1.18; p = 0.003). 3 Remarkably, this association was stronger with the CD subentity, in relation to both psoriases (OR: 1.16; 95% CI: 1.12–1.20; p < 0.001) and PsA (OR: 1.13; 95% CI: 1.06–1.20; p < 0.001). 17 These findings collectively suggest a significant link between IBD, particularly CD, and psoriasis or PsA. However, the relationships seem to be complex and multifactorial, warranting further investigation to fully understand the mechanistic and genetic interplay between these conditions.
Our study adds to the current literature by showing the intriguing result that indicates an association between the severe manifestations of psoriasis – those requiring systemic therapy – and an augmented association with both UC and CD. Upon examining the predictors for IBD within the psoriasis cohort, those patients presenting with severe, as opposed to mild psoriasis, demonstrated a multivariate OR of 16.03 (95% CI: 11.02–23.34; p < 0.001). Furthermore, trends within the predictors for IBD in the psoriasis cohort were quite noteworthy. More specifically, individuals with psoriasis, who concurrently have axial spondyloarthropathy and uveitis, exhibit a significantly elevated risk for IBD development compared to controls, with ORs of 1.75 (95% CI: 1.29–2.45) and 3.13 (95% CI: 2.08–4.71), respectively. This may reflect the propensity for patients with multiple diseases within the spondyloarthropathy spectrum to acquire additional conditions.
The observation that obese patients (BMI >30) demonstrated a reduced risk of comorbid IBD could potentially be attributed to the inherent characteristics of IBD, such as diarrhea, abdominal pain, and loss of appetite, all of which may contribute to weight loss.
IBD and psoriasis, both multifactorial chronic inflammatory diseases, are influenced by a combination of genetics, environmental factors, and epigenetic mechanisms.18,19 Genetic involvement in these conditions is evidenced by twin studies demonstrating a higher concordance rate in monozygotic twins compared to dizygotic ones. 20 Furthermore, the risk of these diseases is increased in individuals with a first-degree relative affected by either IBD or psoriasis.21,22 Over the years, GWAS have made significant contributions toward unraveling the genetic basis of both IBD and psoriasis.23–25 These studies have identified multiple susceptibility genes and shared genetic correlations between the two diseases. The overlap extends to chromosomal loci such as 1p31, 5q33, 6p22, and 16q,23–25 indicating common genetic underpinnings.
Severe psoriasis and CD share genetic predispositions that influence inflammatory pathways, potentially exacerbating both conditions concurrently. 22 Unlike UC, which is restricted to the colon and involves only the mucosal layer, CD affects any part of the GI tract with transmural inflammation, 3 possibly leading to more systemic inflammatory responses that could amplify psoriasis severity. This intersection of genetic, immunological, and physiological factors could explain the stronger linkage between severe psoriasis and CD compared to UC.
Our investigation holds numerous advantages, such as the possibility for broad application due to the substantial number of patients sourced from a verified database. However, there are certain drawbacks. These limitations consist of dependence on registry diagnoses, and the study’s design that does not allow for establishing a causative link between the examined elements. Furthermore, we lacked access to data concerning inflammatory load and disease duration. Nevertheless, the diagnostic accuracy within the Meuhedet registry is recognized to be of high quality, given the ongoing systemic validation and logistical checks carried out through cross-verification from multiple data sources. Moreover, while paradoxical psoriasis is recognized as a known adverse effect of anti-TNF treatment. 26 Our research design did not allow us to determine the proportion of cases where this treatment might have contributed to the development of psoriasis in patients with preexisting IBD treated with anti-TNF. Furthermore, while our study provides valuable epidemiological insights into the association between psoriasis and IBDs within a specific country, its applicability to other populations is limited. Our analysis categorized smoking status in a binary manner; however, the granularity of data regarding smoking cessation was limited due to inconsistent reporting, which may have influenced the precision of our findings. To address this, we incorporated the presence of COPD in our multivariate analysis as an indirect measure of smoking-related damage, acknowledging that this approach is a surrogate marker and not a direct measure of smoking habits.
In conclusion, our research shows a significant association between psoriasis and IBD, including its clinical subtypes CD and UC. This link may depend on disease severity with such association more pronounced in patients with severe psoriasis and those with additional SpA-related comorbidities. The result of this study implies a potential need for closer monitoring of patients with severe psoriasis. However, additional investigation is warranted to further test our findings while aiming to enhance patient treatment and care.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848241227037 – Supplemental material for The association between psoriasis, psoriasis severity, and inflammatory bowel disease: a population-based analysis
Supplemental material, sj-pdf-1-tag-10.1177_17562848241227037 for The association between psoriasis, psoriasis severity, and inflammatory bowel disease: a population-based analysis by Uria Shani, Niv Ben-Shabat, Roula Qassem, Adi Lahat, Mahmud Omar, Einat Savin, Arad Dotan, Yonatan Shneor Patt, Lior Fisher, Galia Zacay, Howard Amital, Abdulla Watad and Kassem Sharif in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
