Abstract
Introduction:
Ulcerative colitis (UC) contributes to impaired health-related quality of life (HRQoL). Although disease activity is the most important factor, reduced HRQoL has been reported even in quiescent UC. We aimed to determine HRQoL, and identify predictors thereof, in patients with long-standing UC in remission.
Methods:
In total, 66 patients with inactive UC were included 10 years after the disease onset. Clinical assessment including rigid sigmoidoscopy was performed to ensure remission. Data on demographic, clinical, treatment-related, and psychological determinants of HRQoL were obtained with a structured interview and self-assessment questionnaires measuring gastrointestinal (GI) and psychological symptoms and fatigue. HRQoL was measured with the Short Form Health Survey (SF-36).
Results:
The SF-36 domains were comparable to the general Swedish population, except for Vitality, where UC patients scored lower. Gender, smoking, comorbidity, or disease phenotype had no impact on HRQoL. In contrast, corticosteroid use and sick leave during the previous year were independently associated with Physical Functioning and Bodily Pain domains of SF-36; persisting GI symptoms during remission with Bodily Pain; and fatigue with Role Physical, General Health and Vitality. For all other SF-36 domains reflecting mental HRQoL (Social Function, Role Emotional, Mental Health), only psychological distress contributed uniquely.
Conclusions:
Although overall HRQoL in long-standing UC in remission is comparable to the general population, previous disease activity as well as persisting GI symptoms, fatigue, and psychological distress are associated with a lower HRQoL among these patients. Improved HRQoL may allow for better UC patient health and reduced costs for health care.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory bowel disease (IBD) of unknown etiology. 1 The natural course of UC is unpredictable, characterized by alternating periods of quiescent disease and symptomatic relapses. 2 The presence of UC contributes to impaired health-related quality of life (HRQoL). UC patients report poorer HRQoL relative to healthy individuals, and similar to those with other gastrointestinal (GI) diseases.3,4
Health-related quality of life is defined as the aspects of well-being that are related to or affected by the presence of disease or its treatment, as perceived by the patient. 5 The determinants of HRQoL in IBD are considered to be socio-demographic, clinical, psychological and treatment-related. 6 However, there is a lack of consensus on the contribution of the specific determinants of HRQoL in UC, due to different measures of disease activity and definitions of remission among studies. 7 Disease activity has been shown to be the most important factor associated with reduced HRQoL in UC. 8 However, data on the degree to which HRQoL is affected in UC patients in remission have been inconclusive. Although some studies report that most HRQoL domains in patients with inactive UC are comparable to the general population, others report more compromised HRQoL in quiescent UC.9,10
The instruments used for assessment of HRQoL in UC vary among studies. While some use disease-specific instruments, which are considered to be more sensitive, others apply generic ones, which give the advantage to estimate the burden of the disease compared with other medical conditions or the general population. 11 Finally, although previous research suggests that HRQoL in UC may improve over time, 12 most studies focus on the effects of the disease on HRQoL during the first years after the diagnosis. However, since the disease is chronic, lifelong, and not curable, there is a need to further investigate HRQoL over a longer time frame.
The main objective of the present study was to determine HRQoL in UC patients in remission after 10 years’ disease duration and to compare this with the general population. Secondary objective was to identify potential predictors of the patients’ HRQoL in long-standing UC in remission.
Materials and methods
Patient cohort and study design
Patients with new onset of UC were prospectively recruited from two outpatient specialized IBD clinics (Sahlgrenska University Hospital, Gothenburg, Sweden, and Södra Älvborg Hospital, Borås, Sweden) between 2004 and 2007. The inclusion criteria were adults with newly diagnosed UC. The exclusion criteria were malignancy during the previous 5 years, alcohol or drug abuse and other significant disease, such as severe heart, kidney, neurological or psychiatric disease, that could affect the possibility to comply with the study protocol as judged by the investigators. All included patients were offered prescheduled follow-up visits at the time of diagnosis (inclusion) and 3 months, 1, 2, 3 and 10 years after the inclusion. Only data collected at the 10-year follow-up were used for the purpose of the present study. All patients received verbal and written information, and signed an informed consent form, before any study-related procedures were initiated. Approval of the study protocol was obtained by the Regional Ethical Review Board in Gothenburg prior to the start of patient inclusion (inclusion visit to 3-year control: S563-02/ January 12, 2003; 10-year follow-up: 638-15/ September 30, 2015). The reporting of this study conforms to the STROBE (Strengthening The Reporting of OBservational Studies in Epidemiology) statement 13 (see checklist in Supplementary Material)
During the 10-year study period, the patients were monitored and treated for UC according to clinical routine. At the 10-year follow-up visit, which took place in conjunction with a regular planned consultation at the unit, clinical assessment including a rigid sigmoidoscopy was performed to grade the disease activity according to the Mayo score. 14 Information about demographic characteristics (gender, age), smoking, comorbidity (other than the exclusion criteria), UC phenotype (proctitis, left-sided colitis and extensive colitis), maintenance treatment of UC and extraintestinal manifestations was collected. Fecal samples for calprotectin analysis were collected. A structured interview and a review of the medical records were conducted to retrospectively obtain information on the number of relapses of the disease during the last year (questions about self-management of mild relapses in the structured interview and documentation of relapse and receipt prescription in the medical records), medications used to treat relapses (systemic corticosteroids or other, and treatment duration), and sick leave duration due to relapse. Relapse was defined as any deterioration of the disease requiring more aggressive treatment.
Self-assessment questionnaires to measure GI symptom severity, psychological distress (anxiety/depression), fatigue, and HRQoL were completed by the patients. GI symptom severity was assessed with the Gastrointestinal Symptom Rating Scale (GSRS), 15 psychological distress with the Hospital Anxiety and Depression scale (HAD), 16 fatigue with the Multidimensional Fatigue Inventory (MFI), 17 and HRQoL with the Short Form Health Survey (SF-36). 18
Remission was defined as Mayo score ⩽ 1 with physician global assessment (PGA) = 0, rectal bleeding = 0 and endoscopic subscore = 0 (mucosal evaluation performed with rigid sigmoidoscopy). As the aim of the study was to evaluate the HRQoL during remission of long-standing UC, patients who did not fulfill the remission criteria were offered a new follow-up visit 3 months later and after treatment escalation in accordance with conventional practice. The same study procedures were performed at the following visit. In this case, data collected at the second visit were included in the further analyses, provided that remission had been achieved, otherwise the patients were excluded.
Questionnaires
The Gastrointestinal Symptom Rating Scale (GSRS) is a questionnaire assessing the severity of GI symptoms during the past week. It consists of 15 items combined into five symptom clusters depicting reflux, abdominal pain, indigestion, diarrhea, and constipation. Each item is rated on a 7-point Likert-type scale where 1 represents no discomfort and 7 very severe discomfort. The results are expressed as mean scores, and higher scores indicate increasing severity of GI symptoms. 15
The Hospital Anxiety and Depression (HAD) scale was developed to detect anxiety and depression in patients with various clinical conditions. It contains 14 items with responses being scored on a 4-grade Likert-type scale (0-3, 3 indicates more severe symptoms). It consists of two subscales, one for anxiety (seven items) and one for depression (seven items). Scores of each subscale range from 0 to 21 and are categorized as following: normal 0–7, borderline depression/ anxiety 8–10, clinically significant depression/anxiety ⩾ 11. 16
The Multidimensional Fatigue Inventory (MFI) is a 20-item self-report instrument measuring five dimensions of fatigue (general fatigue, physical fatigue, reduced activity, reduced motivation, and mental fatigue). 17 The general fatigue scale is recommended to be used as the primary indicator for fatigue, with a higher score indicating higher fatigue (range = 4–20). High fatigue is defined as a score of at least 13, a value based on the 95th percentile of the score in a healthy control group. 19
The Short Form Health Survey (SF-36) is a generic 36-item self-report instrument measuring HRQoL. It assesses eight domains of functioning and well-being: physical functioning (PF), role limitations due to physical health problems (RP), bodily pain (BP), perception of general health (GH), vitality (VT), social functioning (SF), role limitations due to emotional health problems (RE), and mental health (MH). For all domains, scores range between 0 and 100 and higher scores indicate better HRQoL. 18 The questionnaire has demonstrated high validity and reliability when used for UC patients. 9 The SF-36 has been translated and validated in Swedish. The Swedish SF-36 population sample consisted of 8930 persons representative of the Swedish normal population. 20 Normative data on overall scores and on scores adjusted for gender and age derived from this population were used for the purposes of this study.
Data analyses
SF-36 domain scores of the study population (means and z scores) were compared with the normative SF-36 data of the Swedish population. Analyses stratified by gender and age were also conducted. To identify factors affecting HRQoL, we compared the SF-36 domain scores of different subgroups of the study population. The variables considered in these analyses were gender, smoking status, presence of comorbidity, presence of extraintestinal manifestations, disease phenotype, relapses during the previous year (dichotomized as no or at least one relapse), use of systemic corticosteroids during the previous year (dichotomized as yes or no), sick leave due to UC during the previous year (dichotomized as yes or no), GI symptom severity (dichotomized as GSRS score 1–2; no/minor symptoms or GSRS score 3–4; mild/moderate symptoms), anxiety/depression (dichotomized as at least one HAD subscore > 7; borderline or clinically significant anxiety/depression or both HAD subscores ⩽ 7; no anxiety/depression), and fatigue (dichotomized as MFI general fatigue score ⩾ 13; high fatigue or MFI general score < 13; low fatigue). Correlations between SF-36 and GI symptoms (GSRS), psychological factors (HAD), fatigue (MFI general fatigue), number of flares, weeks on systemic corticosteroids, and sick leave during the previous year were analyzed as a next step. Finally, factors independently associated with SF-36 were identified.
Statistical analyses
Statistical analyses were performed with the software package IBM SPSS Statistics version 23 (IBM Corporation, Armonk, New York, USA). Categorical data are reported as absolute numbers/percentages and compared with Pearson’s chi-square or Fischer’s exact test. Continuous data are presented as means with standard deviations or 95% confidence interval when parametric, and as median with 10–90 percentile range when nonparametric. For comparisons of parametric data, we used unpaired Student’s t test, and for nonparametric data the Mann–Whitney U test or the Kruskal–Wallis test. The comparison of SF-36 scores with normal population scores was performed by calculating the standardized difference between the means [z scores = (mean patient score minus mean population score) divided by population standard deviation]. These were then evaluated according to Cohen’s effect size index, where <0.2 indicated no difference, 0.2–0.5 small difference, 0.5–0.8 moderate difference, and >0.8 large difference. 21 Correlations between variables were evaluated with Spearman’s rank correlation coefficient. To explore factors independently associated with HRQoL, multiple linear regression models (enter method) were performed after multicollinearity had been excluded (variance inflation factor (VIF) < 5). 22 Variables with statistically significant correlations with the SF-36 in the univariate analyses were included in the regression models. A significance level of 0.05 was accepted throughout the report.
Results
Demographics and clinical characteristics
Initially, 99 patients were included at the time of UC diagnosis, out of these 66 (67%) were followed up 10 years later. Table 1 demonstrates the demographic and clinical characteristics of the patients who completed the follow-up. The mean age was 47.4 years, 64% were males, and the majority reported no relapse, that is, no worsening of UC symptoms resulting in the need for treatment escalation, during the previous year (70%). Thirty-three patients were lost to follow-up totally, due to resistance to medical treatment resulting in colectomy (n = 4), death (n = 1), moving abroad (n = 3), unwillingness to participate in the 10-year follow-up (n = 23) and persistent disease activity at the 10-year follow-up (n = 2). Ten patients did not initially fulfill the remission criteria, but eight of those had achieved remission at the new follow-up visit they were offered 3 months later and after treatment escalation. No differences between the patients who completed the 10-year follow-up and those who did not were observed, except for comorbidity (44% for patients completing the 10-year follow-up versus 24% for patients lost to follow-up, p = 0.02) (Supplementary Table 1).
Demographic data and clinical characteristics of the study population at 10-year follow-up, n (%).

ASA, aminosalicylic acid; GSRS, Gastrointestinal Symptom Rating Scale; HAD, Hospital Anxiety and Depression; MFI, Multidimensional Fatigue Inventory; TNF, tumor necrosis factor.
Mean, standard deviation.
Median, 10–90 percentile range.
Only for subjects with active working live, that is, age < 65 years.
Comparison of HRQoL in long-standing UC in remission with the general population
Overall, the SF-36 domains of the patients with long-standing UC in remission were comparable to those of the general population sample. The only difference was observed in the Vitality domain, where UC patients had a 6.8 points lower mean score (p < 0.05) than the general population (Table 2A). When stratified by gender (Table 2B) or age (Table 2C), no differences in the SF-36 domain scores were found between patients and general population subgroups. Furthermore, z scores for SF-36 domains confirmed these findings by showing only small or no differences for all domains, in the whole cohort as well as when stratified by gender and age (Figure 1).
SF-36 domain scores for the study population versus the general population (A), and SF-36 domain scores stratified by gender (B) and age (C), mean (95% confidence interval).
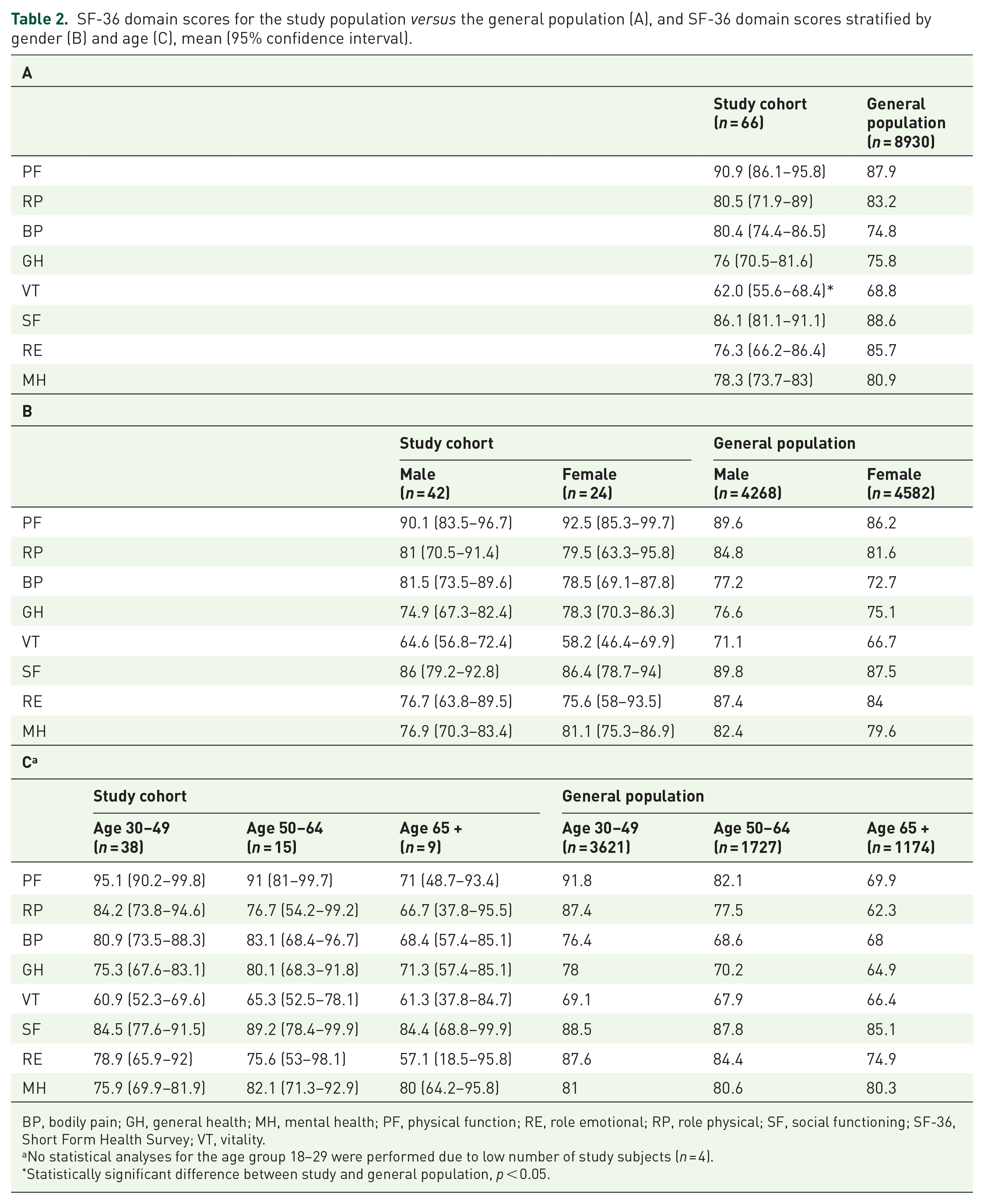
BP, bodily pain; GH, general health; MH, mental health; PF, physical function; RE, role emotional; RP, role physical; SF, social functioning; SF-36, Short Form Health Survey; VT, vitality.
No statistical analyses for the age group 18–29 were performed due to low number of study subjects (n = 4).
Statistically significant difference between study and general population, p < 0.05.

(a) Mean z-scores for SF-36 domains, (b) mean z-scores for SF-36 domains stratified by sex, and (c) mean z-scores for SF-36 domains stratified by age in patients with long-standing inactive ulcerative colitis. Z scores are equal to patient score minus the mean population score divided by the standard deviation of the population scores. Cohen’s effect size index for z scores; 0.2 no difference, 0.2 to 0.5 small difference, 0.5 to 0.8 moderate difference, 0.8 large difference.
Determinants contributing to HRQoL in long-standing UC in remission
Gender, smoking, presence of comorbidity, and disease phenotype had no impact on the SF-36 scores, while the presence of extraintestinal manifestations affected three SF-36 domains (Physical Functioning, Role Physical, Bodily Pain) among patients with long-standing UC in remission (Figure 2). Furthermore, patients who had at least one relapse during the past year had lower scores in the Role Physical domain of SF-36 when compared with those without relapses (Figure 3(a)). However, the use of systemic corticosteroids and sick leave due to UC during the previous year negatively influenced the majority of the domain scores (Figure 3(b) and (c)). Finally, the presence of at least mild GI symptoms, borderline or clinically significant anxiety/depression, and high fatigue had a negative impact on almost all SF-36 domains (Figure 3(d)–(f)).
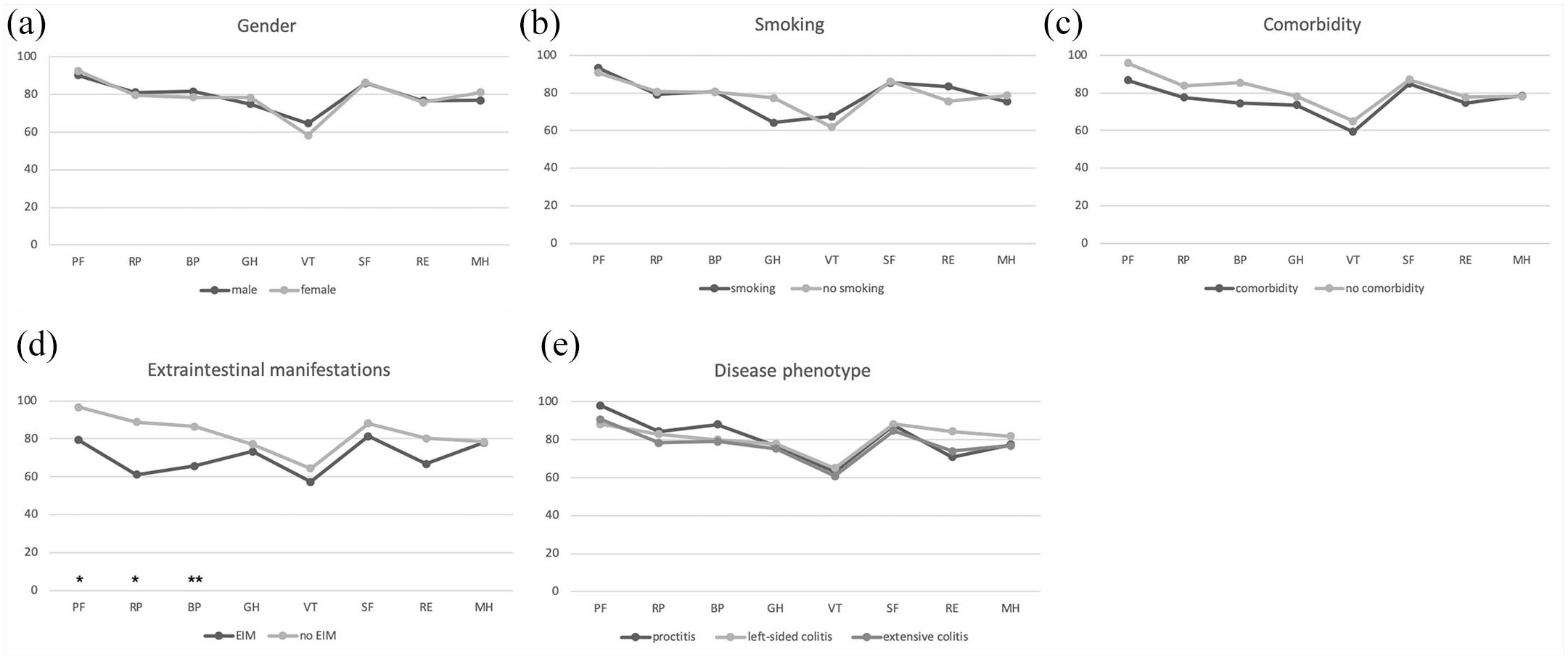
SF-36 domain score comparisons (mean values) between different subgroups according to (a) gender, (b) smoking status, (c) presence of comorbidity, (d) presence of extraintestinal manifestations (EIM), and (e) disease phenotype.

SF-36 domain score comparisons (mean values) between (a) patients reporting no relapses versus patients reporting at least one relapse during the previous years, (b) patients who used systemic corticosteroids during the previous year versus patients who did not, (c) patients with versus without sick leave due to ulcerative colitis during the previous year, (d) patients reporting no/minor gastrointestinal symptoms in remission (total GSRS score = 1–2) versus patients reporting mild/moderate symptoms (total GSRS score = 3–4), (e) patients with borderline or clinically significant anxiety/depression (at least one HAD subscore > 7) versus patients with no anxiety/depression (both HAD subscores ⩽ 7), and (f) patients with high fatigue (MFI general fatigue score ⩾ 13) versus those with low fatigue (MFI general score < 13).
Table 3 demonstrates the correlations between SF-36 domain scores and GI symptoms (GSRS), psychological factors (HAD), fatigue (MFI general fatigue) and number of flares, weeks on corticosteroids, and sick leave during the previous year. GSRS, HAD, and MFI general fatigue scores were associated with all SF-36 domains, with the strongest correlations between GSRS and Bodily Pain (r = -0.53), HAD and Mental Health (r = -0.83), and MFI general fatigue and Vitality (r = -0.91). The number of relapses during the last year was not correlated with any SF-36 domain score except for Role Physical (r = 0.29). However, weeks on corticosteroids and sick leave during the previous year, indicating more severe preceding disease activity, were significantly correlated with almost all SF-36 domain scores. The strongest correlations were found between weeks on corticosteroids and Role Physical (r = -0.44) and sick leave and General Health (r = -0.37).
Correlations between SF-36 domain scores and gastrointestinal symptoms, psychological factors, fatigue, number of flares, and weeks on systemic corticosteroids and sick leave during the previous year (r values).
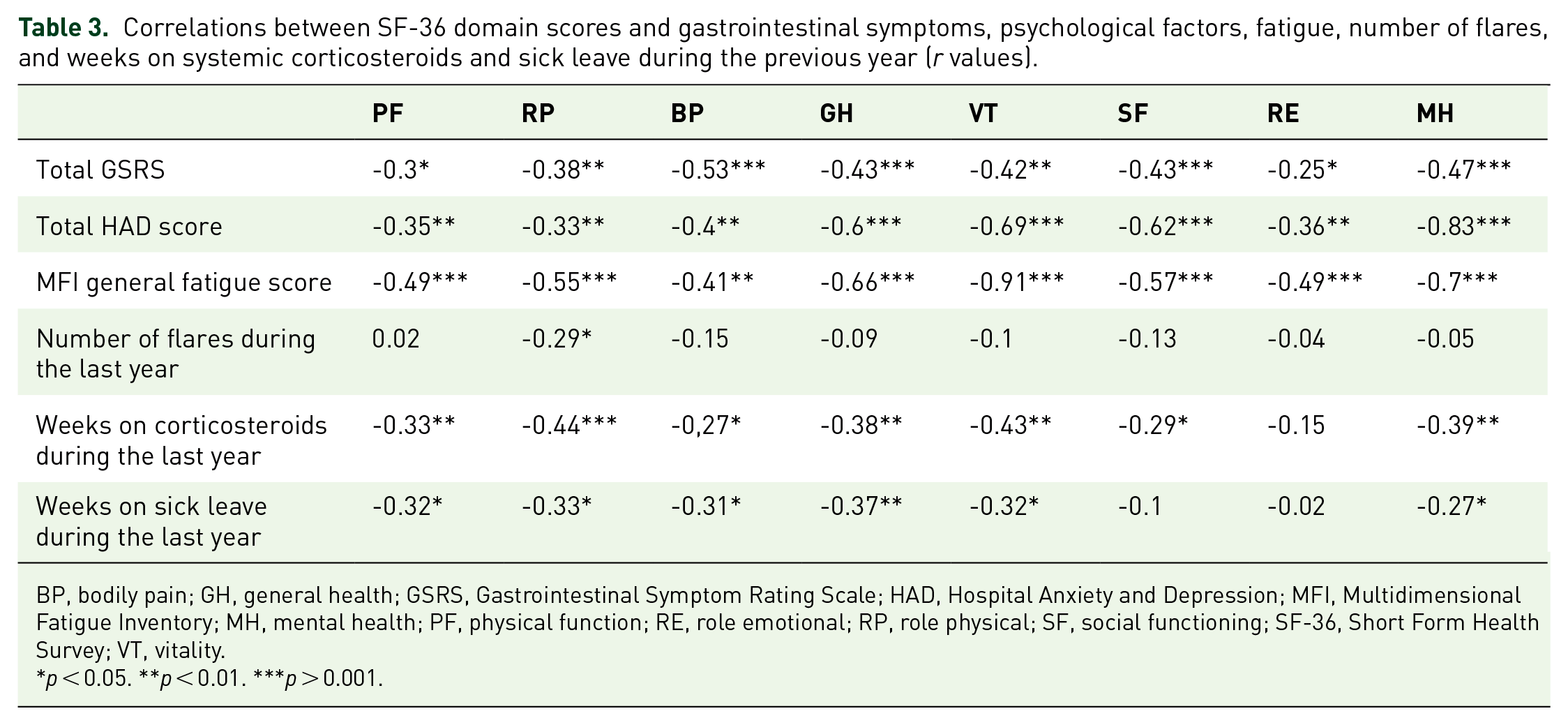
BP, bodily pain; GH, general health; GSRS, Gastrointestinal Symptom Rating Scale; HAD, Hospital Anxiety and Depression; MFI, Multidimensional Fatigue Inventory; MH, mental health; PF, physical function; RE, role emotional; RP, role physical; SF, social functioning; SF-36, Short Form Health Survey; VT, vitality.
p < 0.05. **p < 0.01. ***p > 0.001.
Factors independently associated with the SF-36 domain scores in long-standing UC in remission
We performed multiple linear regression analyses to identify factors independently associated with the SF-36 domain scores of the patients. We entered all factors (GSRS and HAD total scores, MFI general fatigue score, extraintestinal manifestations, and weeks on corticosteroids, weeks on sick leave and number of relapses during the last year) univariately associated with the SF-36 domain scores. Variables related to preceding disease activity/severity (weeks on corticosteroids and weeks on sick leave during the last year) remained significant in the multiple regression models for Physical Functioning and Bodily Pain domains. The presence of persisting GI symptoms during remission (GSRS) was also independently associated with Bodily Pain. Fatigue was the only determinant with a significant unique contribution to Role Physical, General Health, and Vitality domains. In contrast, for all domain scores reflecting mental HRQoL except for Vitality (namely Social Functioning, Role Emotional, Mental Health), the only independently significant predictor was psychological distress (HAD total score). Overall, the linear regression models explained between 28% (Role Emotional) and 85% (Vitality) of the variances in the dependent variables. Results from the linear regression models are presented in Table 4.
Linear regression models for SF-36 domain scores.

BP, bodily pain; EIM, extraintestinal manifestations; GH, general health; GSRS, Gastrointestinal Symptom Rating Scale; HAD, Hospital Anxiety and Depression; MFI, Multidimensional Fatigue Inventory; MH, mental health; PF, physical function; RE, role emotional; RP, role physical; SF, social functioning; SF-36, Short Form Health Survey; VT, vitality.
Factors making a significant unique contribution to the prediction of the dependent variable (SF-36 dimensional scores) are highlighted with *, **, ***, depending on the p value.
p < 0.05. **p < 0.01. ***p < 0.001.
Discussion
The results of the present study suggest that the overall HRQoL in long-standing UC in remission is comparable to the general population. Nevertheless, markers of at least moderate preceding disease activity, such as corticosteroid treatment and work disability due to UC during the previous year, but also fatigue and persisting GI symptoms during remission were found to have an impact on different aspects of physical HRQoL. In contrast, mental HRQoL was determined mainly by psychological factors, such as psychological distress, along with fatigue.
The finding that HRQoL in long-standing UC in remission was comparable with the HRQoL of the general population is in accordance with previous research. 12 The only SF-36 domain score that was reduced among the UC patients in our study was vitality, a measure of energy level and fatigue. In line with our results, it has been reported that fatigue contributes to reduced HRQoL in UC independently of disease activity. 23 Previous research has also demonstrated a reduction in the general health domain of SF-36 in inactive UC, which has been attributed to the patients’ experience of having a chronic disease.24,25 This is inconsistent with our findings and might be explained by the longer follow-up in our study. Thus, a longer disease duration potentially results in a patient adaptive process allowing the general health perceptions to reach normal levels over the course of time.
The presence of persistent GI symptoms during remission was identified as an independent factor associated with the bodily pain domain of SF-36. Our group has demonstrated a negative correlation between the total score of GSRS and HRQoL, measured by Psychological General Well-Being Index (PGWB). 26 Moreover, we have previously demonstrated that UC patients in remission with IBS-like symptoms or symptoms resembling other functional bowel disorders scored lower in the disease-specific HRQoL instrument IBDQ (Inflammatory Bowel Disease Questionnaire) than those without symptoms of this type.27,28 Scores on SF-36 and PGWB or IBDQ are strongly correlated.29,30 The negative impact of fatigue on HRQoL in patients with IBD in remission has also been well established.19,23,31 Our results demonstrate that general health, role physical, and vitality domains of SF-36 are independently associated with fatigue. This strengthens previous research on the effect of fatigue on HRQoL in UC, as we excluded patients with other severe comorbidities possibly explaining fatigue.
Similar to previous reports on HRQoL in UC, our study shows that preceding treatment with corticosteroids and work disability, as markers of moderate to severe disease activity, but not recent relapse rate, were associated with impaired physical HRQoL after achieving remission.25,32 This suggests that the time to achieve HRQoL normalization after a relapse is determined by the severity grade of the relapse, with patients who suffer from mild disease activity reaching normal HRQoL levels soon after entering remission. However, we did not specify the time since the last relapse during the past year. Thus, patients with relapse a year before inclusion to the study were indistinguishable from those with relapse closer to recruitment. Relevant information was also obtained by medical records review, where self-management of mild relapses might not have been documented. This highlights the need for studies with standardized definitions of these parameters in order to explore the long-term effects of relapses on HRQoL after entering remission. Finally, whether the negative association between corticosteroid treatment and HRQoL depends on corticosteroid-specific side effects cannot be concluded by the design of our study.
The importance of psychological function, together with disease activity, in determining HRQoL of IBD patients has been previously described, 33 and anxiety and depression have been suggested to constitute risk factors for reduced mental HRQoL.34,35 In our study, psychological distress, measured by HAD, was independently associated with the most of SF-36 domain scores that composite mental HRQoL. However, we could not confirm the previous reports demonstrating psychological disturbance as an independent predictor of physical HRQoL in IBD. Although there was a clear univariate association between HAD scores and all SF-36 domain scores of physical health, these associations did not remain statistically significant in the multivariate analyses. We believe that this disparity depends on the low levels of anxiety and depression in our sample. Still, low levels of psychological distress have been previously reported when evaluating HRQoL in long-standing IBD in remission. 26
Similar to our study, no differences with regard to gender, smoking status and disease phenotype in SF-36 domains were shown in a Norwegian study including UC patients with a disease duration of 10 years and the majority of patients reporting no or mild symptoms. 25 However, the effect of gender on HRQoL in UC still remains unclear, as several other reports have demonstrated a relationship, with females having reduced HRQoL.32,36 Psychological factors and a more severe rating of individual symptoms by females have been brought forward as explanations for these gender differences. 36 In our cohort, no gender differences were found in HAD and GSRS scores (Supplementary Table 2) and overall the scores were low, possibly explaining the lack of gender differences in our study. Finally, the presence of extraintestinal manifestations had a negative effect on most subscales of physical HRQoL, which is congruent with a previous report showing that daily joint pain is associated with lower HRQoL in IBD. 37 However, this association disappeared in our data when analyses were adjusted for other factors. A possible explanation might be that the presence of extraintestinal manifestations is a surrogate marker of preceding active and more severe disease.
In our sample of patients included in the study between 2015 and 2017, none of the patients were currently on biologics and only 15% used thiopurines, thus one could question if it is representative for UC patients. The use of biologics has undoubtedly increased over the last years. However, according to a Canadian study, in 2014, 6% of patients with UC had been exposed to an anti-TNFa at any time, and only 4% had received anti-TNFa in the previous 90 days. 38 Likewise, in a Swedish study, the percentage of UC patients with a drug prescription was 13% for immunomodulators and 1% for biologics in 2010. 39 Moreover, it should not be overlooked that our cohort consisted exclusively of UC patients in remission. Hence, we believe that our results are somewhat generalizable for the subgroup of patients with long-standing UC in remission, and even if our study patients had a quite mild disease course, we could still demonstrate differences in aspects of HRQoL.
The major strength of our study is the well-characterized cohort of UC patients and that we used an objective method, that is, endoscopy, to define remission. However, it has some limitations that should be addressed. The number of patients with UC in remission included was relatively modest, so minor differences between groups might have been overlooked, and the generalizability of our findings is not totally obvious. The differences in the studied determinants of HRQoL between the various subgroups were though large, so this does not seem to have influenced our main findings, which are also in line with previous publications on this topic as discussed above. Moreover, we used the generic instrument SF-36 for the measurement of HRQoL, and not a disease specific, such as the IBDQ. Although the two types of instruments are rather complementary, 40 we chose a generic one as the main objective of the study was to compare HRQoL in long-standing UC in remission with that of the general population. SF-36 is also considered to be reliable and valid for UC. 9 In addition, some data, for example number of relapses during the past year, were collected retrospectively with the risk of recall bias and the time since the last relapse was not taken into consideration, although the majority of the patients were in remission for a long period of time (at least 1 year). Future studies, possibly with digital monitoring of patients, can help overcome this issue. Finally, a large variety of possible determinants of HRQoL in UC is described in the literature and investigating all of them and their interactions in a single study is practically unattainable. We tried to minimize this problem by simultaneously evaluating different demographic, disease-related, treatment-related, and psychological factors. However, we cannot rule out that important determinants were neglected.
UC is a chronic and not curable disease. Although most aspects of HRQoL in patients with long-standing UC in remission are comparable to the general population, these patients still represent a heterogeneous group and a subset of them experience burdens on their physical and mental functioning and well-being. Our findings have important clinical implications. Factors like psychological distress, fatigue, and persisting GI symptoms in patients with UC in remission are often underrecognized and suboptimally treated in clinical practice, as clinicians primarily focus on the proper management of disease activity related to inflammation. However, our study underlines the significance of also assessing psychological distress, fatigue, and persisting GI symptoms, as these factors contribute to patient well-being and are potentially modifiable. Specifically, clinical evaluation of UC should incorporate screening and monitoring patients for psychological comorbidities and, if present, treat with psychotropic medications or refer the patient for behavioral treatment. Likewise, identifying fatigue or GI symptoms that persist in remission, and offering appropriate interventions, that often need to be multidisciplinary, such as dietary, pharmacological, and behavioral, is of great importance. After all, a holistic and strategic approach in the management of UC patients is necessary to better understand the patients’ global situation. This may facilitate in clinical decision making and ultimately improve HRQoL and decrease health care and society costs.
In conclusion, the overall HRQoL in long-standing UC in remission is comparable to the general population. Nevertheless, previous moderate or severe disease activity as well as persisting GI symptoms, fatigue, and psychological distress are all associated with a lower HRQoL among these patients. A thorough understanding of determinants influencing HRQoL may facilitate clinical decision making in patients with long-standing UC in remission, allowing improved patient health.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211062406 – Supplemental material for Health-related quality of life in patients with long-standing ulcerative colitis in remission
Supplemental material, sj-docx-1-tag-10.1177_17562848211062406 for Health-related quality of life in patients with long-standing ulcerative colitis in remission by Georgios Mavroudis, Magnus Simrén, Lena Öhman and Hans Strid in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848211062406 – Supplemental material for Health-related quality of life in patients with long-standing ulcerative colitis in remission
Supplemental material, sj-docx-2-tag-10.1177_17562848211062406 for Health-related quality of life in patients with long-standing ulcerative colitis in remission by Georgios Mavroudis, Magnus Simrén, Lena Öhman and Hans Strid in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GM has served as consultant/advisory board member for Abbvie, Janssen, Tillotts, and Pfizer, and as speaker for Ferring, Tillotts, and Takeda. MS received unrestricted research grants from Danone Nutricia Research and Glycom, and served as consultant/ advisory board member for Danone Nutricia Research, Ironwood, Menarini, Biocodex, Genetic Analysis AS, Glycom, Tillotts, Arena and Adnovate, and as speaker for Tillotts, Menarini, Kyowa Kirin, Takeda, Shire, Biocodex, Alimentary Health, AlfaSigma, Sanofi, and Falk Foundation. LÖ received unrestricted research grants from AstraZeneca, and served as consultant/advisory board member for Genetic Analysis, and as a speaker for Takeda, Ferring, and Abbvie. HS has served as consultant/ advisory board member for Abbvie, Takeda, Janssen, Pfizer, Gilead, MSD, and Tillotts, and as speaker for Abbvie, Tillotts, Takeda, Ferring, and Pfizer.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Healthcare Committee Region Västra Götaland (554651, 733981, and 929698 to GM), the Local Research and Development Council Södra Älvsborg, the Swedish Medical Research Council (2018-02566 to MS, 2015-02317 and 2019-01052 to LÖ, 727881 and 932651 to HS), and the Alice Swenzons Foundation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
