Abstract
Background:
Biological therapies have revolutionized the treatment of patients with inflammatory bowel disease (IBD). Infliximab (IFX) has been shown to be effective in inducing and maintaining remission in patients with Crohn’s disease and ulcerative colitis. However, about one-third of the patients are primary non-responders, and up to half can lose response over time. Hence, it is important to assess which factors are related to treatment failure.
Objectives:
We aimed to identify factors predicting clinical and endoscopic remission with IFX treatment during maintenance therapy in a Brazilian IBD referral center.
Design:
We conducted a cross-sectional study to describe demographic, clinical, and IBD therapy-related characteristics of IBD patients treated with IFX for at least 6 months in a Brazilian referral center. Subsequently, we evaluated factors associated with clinical and endoscopic remission (primary and secondary outcomes, respectively).
Methods:
We used descriptive statistics to summarize the essential demographic and clinical characteristics of the population. The association of sociodemographic and clinical variables with outcomes was analyzed using multivariable logistic regression.
Results:
A total of 131 IBD patients (the mean age 41.7 years) were enrolled in this study. Clinical and endoscopic remission were observed in 79.4% and 58.2% of the patients, respectively. In the multivariable analysis, IFX therapy duration and higher albumin levels increased the likelihood of clinical remission, while previous surgery decreased its chance. Prior use of adalimumab and higher C-reactive protein levels reduced the likelihood of endoscopic remission.
Conclusion:
In summary, this study has enhanced our understanding of the predictive factors of treatment response to IFX in a well-characterized Brazilian IBD population.
Trial registration:
4.254.501 and 2.903.748.
Introduction
Ulcerative colitis (UC) and Crohn’s disease (CD) are idiopathic inflammatory disorders characterized by a relapsing and remitting course. Although the geographic prevalence of inflammatory bowel disease (IBD) varies considerably, higher prevalence rates are traditionally reported in Western countries. Despite the lack of data, newly industrialized countries have swiftly reported increases in IBD incidence and prevalence, probably due to industrialization, urbanization, and westernization of culture and diet.1–3 Interestingly, a recent large Brazilian study reported a significant increase in estimated prevalence rates and stable incidence rates of IBD from 2012 to 2020, likely resulting in a continued high burden of the disease in the region. 3
The use of biological therapies has substantially improved the management of patients with IBD over the last two decades. Infliximab (IFX), the first approved biologic drug, is considered an effective therapy for IBD, leading to a better life quality and reduction in complications, such as surgeries and hospitalizations.4,5 Its efficacy in inducing and maintaining clinical and endoscopic remission has been supported by clinical trials and real-life studies for both UC and CD. However, roughly one-third of patients are primary non-responders, and 23%–46% develop secondary loss of response. 6 Thus, it is crucial to define which patients will have no response and which factors are related. Some data suggest that mechanisms underlying treatment failure are multifactorial and include the characteristics of the patients and the disease, drug-related factors, genetics, and immunopharmacological aspects.7,8 Although several European and North American studies have reported factors associated with increased response to IFX treatment, the IBD phenotype differs among various ethnic groups, perhaps due to different genetic backgrounds and distinct responses to treatment. Hence, it is essential to have data from diverse populations because of differences in demographic, socioeconomic, and disease-related factors between the Brazilian and the North American or European populations. Such variations limit the extrapolation from currently available data. In this study, we aimed to identify factors predicting clinical and endoscopic remission with IFX treatment during maintenance therapy in a Brazilian referral hospital for IBD.
Methods
Study design and population
We consecutively enrolled adult Brazilian patients diagnosed with IBD (either CD or UC) treated with scheduled IFX for at least 6 months (maintenance therapy) at the tertiary IBD referral hospital at the University of São Paulo from January 2019 to February 2021. This study had cross-sectional design and a consecutive recruitment of patients. They were treated with IFX either alone or in combination with immunomodulators (azathioprine, 6-mercaptopurine, methotrexate). In our study, the standard IFX dose was defined as 5 mg/kg every 8 weeks, and the IFX dose escalation was defined as 5 mg/kg every 4 weeks, 5 mg/kg every 6 weeks, or 10 mg/kg every 8 weeks.9–12
The exclusion criteria were patients under 18 years of age, primary non-responders, and those who did not accept participating and having their IFX trough levels measured. Primary non-response was defined as lack of improvement in clinical signs and symptoms during induction therapy. Patients with an ostomy or a total colectomy, or those using oral systemic and local corticosteroids were not eligible either.
The IBD diagnosis was confirmed by clinical, endoscopic, imaging, and histological tests. Information on sex, body mass index (BMI), age, diagnosis date, disease duration, location as well as behavior, smoking history, presence of comorbidities, duration of current biological therapy, the dose of IFX (defined in mg/kg), the interval between doses, IFX standard or escalation dose, use of concomitant immunomodulators, previous IBD treatments, and surgical procedures (including enterectomy, right hemicolectomy, and perianal approaches) was collected from electronic medical records at the moment of measuring IFX trough levels. Immunomodulators included 6-mercaptopurine, azathioprine, and methotrexate. Laboratory tests data, such as those from albumin and C-reactive protein (CRP) tests, were also extracted from medical records. They were measured locally. We also monitored the patients for any clinical changes or complications during the IFX treatment. We considered the IFX level test and other laboratory tests carried out within 3 months of assessment of clinical and endoscopic remission.
IFX levels
The IFX level test was neither available in Brazil’s public health system, nor at the hospital of the University of São Paulo. We had 131 tests available to us as a donation. This was clearly stated in the project approved by the local ethics committee. All IFX level measurements were performed proactively at variable time points during maintenance treatment with IFX. All participants had a blood sample collected to measure the IFX level. This measurement was performed just before the subsequent IFX infusion on a scheduled date, that of the trough IFX level. Serum samples for IFX concentrations were measured at the University of São Paulo, using the Quantum Blue test (Bühlmann Laboratories, Schönenbuch, Switzerland), which is an in vitro diagnostic lateral flow immunoassay for the quantitative determination of trough levels of IFX in serum samples. The measurement range is between 0.4 and 20.0 μg/mL. Data above this range were not analyzed. It was not possible to measure the anti-IFX antibody due to the unavailability of this test in our hospital because of financial limitations.
Definitions of clinical and endoscopic remission
Clinical remission was assessed at the same moment of the IFX measurement, and the endoscopic assessment was performed within 3 months before and after the IFX measurements. As an evaluation of clinical remission, we used the Harvey Bradshaw Index (HBI) for CD 13 and the partial Mayo score (PMS) for UC. 14 An HBI score lower than 5 and a PMS lower than 2 were considered as a clinical remission.
Endoscopic data were used to assess endoscopic healing. For UC, we used the endoscopic Mayo score of 0 and 1 for endoscopic remission. 5 For CD, we used the Simple Endoscopic Score for Crohn’s Disease equal to or lower than 2. 15 We only used Rutgeerts’ score 16 for patients with a previous history of hemicolectomy (i0 or i1). For this study, we considered endoscopic remission equivalent to endoscopic healing.
Outcomes
The primary outcome was the presence of clinical remission during maintenance treatment with IFX and the secondary outcome was the occurrence of endoscopic healing.
Ethical considerations
The study protocol numbers 4.254.501 and 2.903.748 were approved by the Research Ethics Committee of the Clinics Hospital of the School of Medicine of the University of São Paulo. Before enrollment in the study, all patients provided a written informed consent for their participation and measurement of the IFX trough levels. This study was conducted in compliance with the Declaration of Helsinki. The reporting of this study conforms to the STROBE statement. 17
Statistical analysis
We first used descriptive statistics to summarize the essential demographic and clinical characteristics of the population. The qualitative characteristics were described using absolute and relative frequencies, and association was verified with chi-square tests or exact tests (Fisher’s exact test or likelihood ratio test). Categorical variables were described using frequencies and percentages and continuous variables using means and standard deviations.
The association of sociodemographic and clinical variables with outcomes was analyzed using multivariable logistic regression. The odds ratios (ORs) were estimated with 95% confidence intervals (CIs) for each characteristic with its outcomes.
Logistic regression was used to estimate the effect of potential explanatory variables. For a multiple logistic regression model, we adjusted the variables that presented a descriptive level in the analyses of bivariate values lower than 0.10 (p < 0.10), using the backward stepwise selection method with entry and exit criteria for the models of 5%. All analyses were performed for the total number of patients and separately by disease.
All statistical analyses were conducted using the IBM-SPSS version 22.0 software for Windows and tabulated using Microsoft-Excel 2010. The tests were performed with a significance level of 5%.
Results
At the time of the study, 386 IBD patients were regularly undergoing maintenance therapy with IFX. Among the eligible patients, we were able to consecutively measure IFX levels and enroll 131 IBD (95 CD and 36 UC) patients as shown in Figure 1. A male predominance (57.3%) was observed in the population. In UC patients, the majority (69.4%) had pancolitis. In CD patients, 53.7% were ileocolonic, about half of them (50.5%) had structuring behavior and 23.2% had penetrating behavior. Perianal disease was present in 43.2% of the CD patients. Demographic, clinical, and IBD treatment-related characteristics are listed in Table 1.

Selection of patients.
Baseline characteristics of study population and stratified by clinical and endoscopic remission.
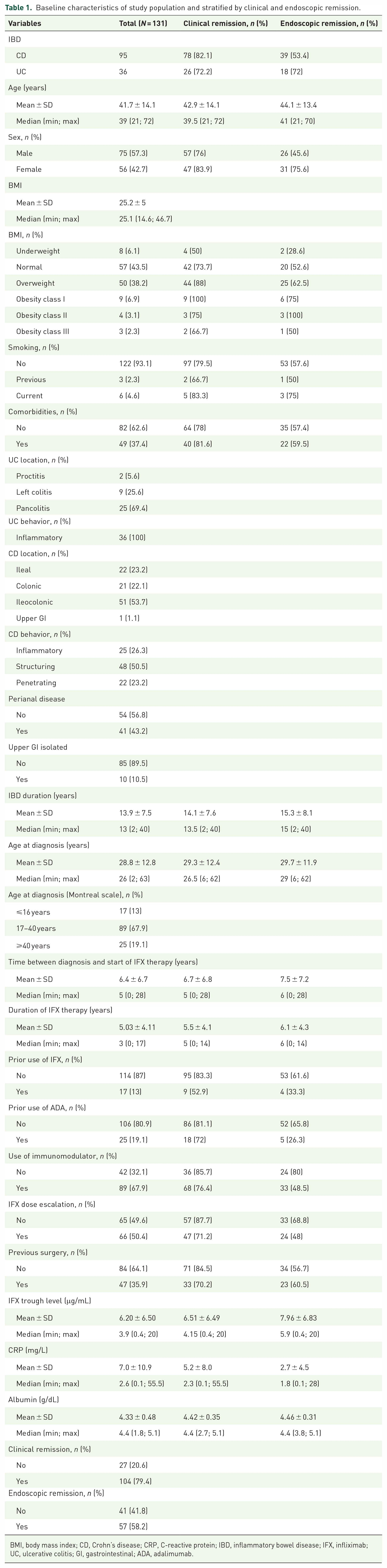
BMI, body mass index; CD, Crohn’s disease; CRP, C-reactive protein; IBD, inflammatory bowel disease; IFX, infliximab; UC, ulcerative colitis; GI, gastrointestinal; ADA, adalimumab.
The mean age of the patients was 41.7 (SD ±14.1), and the median disease duration was 13 years (range: 2–40). The median time between diagnosis and the start of IFX therapy was 5 years (range: 0–28). The median duration of current IFX therapy was 3 years (range: 0–17). An immunosuppressor was concomitantly administered to 67.9% of the IBD patients. The UC patients had a lower frequency of concomitant use of immunosuppressants (p = 0.002). Half of the participants (±50.4%) received the IFX dose escalation. The UC patients showed a lower rate of IFX dose escalation than CD patients (p = 0.044). Seventeen patients had been previously treated with IFX. The median time between IFX interruption and the current treatment was 3 years (range: 1–8).
Among the IBD patients on combination therapy with an immunomodulator, the median IFX level was 3.1 μg/mL (range: 0.4–20), and for those on IFX monotherapy, the median IFX level was 5.2 μg/mL (range: 0.4–20), with no significant difference between them (p = 0.19). Among the IBD patients with dose escalation, the median IFX level was 5.7 μg/mL (range: 0.4–20), and for those on the standard dose, the median IFX level was 3 μg/mL (range: 0.4–20), with a statistical difference between them (p = 0.01).
The primary outcome was observed in 104 (79.4%) patients. Outcome data of the cohort by demographic, clinical, and treatment characteristics are summarized in Table 1. Age, sex, BMI, smoking, and comorbidities showed no statistical difference (p < 0.05).
When analyzed separately, 82.1% (n = 78) of the CD patients and 72.2% (n = 26) of the UC patients were in clinical remission. Outcome data by each characteristic for CD and UC are described in Supplemental Tables 1 to 4.
For all patients in clinical remission, the median time of IFX therapy was 5 years (range: 0–14 years; p = 0.009). As described in Table 1, most patients (83.3%) on IFX as the first biologic therapy experienced the primary outcome. In addition, most of the patients (87.7%) on standard IFX therapy met the primary outcome (p = 0.02). The median CRP level was 2.3 mg/L (range: 0.1–55.5) for the primary outcome, whereas for those who did not achieve clinical remission, the median CRP level was 6.7 mg/L (range: 0.1–46) with statistical difference (p = 0.042) (Table 2).
Univariable analysis and multivariable regression for clinical remission from our cohort.

Chi-square test.
Fisher’s exact test.
Likelihood ratio test.
Student’s t-test.
Mann–Whitney test.
There are no cases to estimate.
BMI, body mass index; CRP, C-reactive protein; IBD, inflammatory bowel disease; IFX, infliximab.
Among the CD patients, those who had received IFX dose escalation showed a lesser chance of achieving clinical remission. Overall, 47 CD patients underwent 52 surgeries. The most prevalent surgery (23/52) was hemicolectomy. In addition, prior surgery (OR: 0.16; 95% CI: 0.04–0.59) was negatively associated with clinical remission in CD patients (p = 0.003). In the case of CD patients with perianal disease, no significant difference was observed in clinical remission. In UC patients, the use of immunomodulators (OR: 0.13; 95% CI: 0.02–0.76) was the only factor associated with a lower chance of clinical remission (p = 0.025).
For the secondary outcome, 98 IBD patients were analyzed, 58.2% of whom were in endoscopic remission. Most cases (75.6%) were female (p = 0.003). The median time of IFX treatment of the patients in endoscopic remission was 6 years (range: 0–14). As described in Table 3, only 26.3% of the IBD patients who previously used adalimumab presented endoscopic healing (OR: 0.19; 95% CI: 0.06–0.57; p = 0.002), and 80% of the IBD patients in IFX monotherapy were in endoscopic remission (OR: 0.24; 95% CI: 0.09–0.65; p = 0.004). When analyzed individually, 53.4% and 72% of the CD and the UC patients, respectively, were in endoscopic remission.
Univariable analysis and multivariable regression for endoscopic remission from our cohort.

Chi-square test.
Student’s t-test.
Likelihood ratio test
There are no cases to estimate.
Mann –Whitney test.
BMI, body mass index; CRP, C-reactive protein; IBD, inflammatory bowel disease; IFX, infliximab.
Most patients (68.8%) on standard IFX dose experienced the secondary outcome, while this was true for only 48% of those with IFX dose escalation (OR: 0.42; 95% CI: 0.18–0.96; p = 0.037). For the patients who achieved endoscopic healing, the median IFX trough level was 5.9 μg/mL (range: 0.4–20; p = 0.002), whereas for those who did not achieve it, the median IFX trough level was 1.4 μg/mL (range: 0.4–20). As described in Table 3, the median CRP level was 1.8 mg/L (range: 0.1–28) for the secondary outcome, and for patients who did not meet the secondary outcome, the median CRP level was 8.6 mg/L (range: 0.3–46), with statistical difference (p < 0.001). In the case of CD patients with perianal disease, no significant difference was observed in endoscopic remission.
For all the patients in the univariable analysis, the previous use of IFX was negatively associated with clinical remission. As described in Table 3, gender (female) and higher IFX trough levels were positively associated with endoscopic remission, whereas the previous use of adalimumab and the concomitant use of immunomodulators had a negative association. Finally, the duration of IFX therapy, IFX dose escalation, and CRP and albumin levels showed statistical significance in primary and secondary outcomes.
For CD, the factors associated with both outcomes are presented in Supplemental Tables 1 and 2. They were very similar compared to the univariable analysis for the entire cohort. For UC, the only factor associated with a lesser chance of endoscopic remission was the concomitant administration of the immunomodulator, as shown in Supplemental Tables 3 and 4.
For the entire cohort in the multivariable analysis (Tables 2 and 3), the duration of IFX therapy (OR: 1.27; 95% CI: 1.06–1.52) and higher albumin levels (OR: 4.08; 95% CI: 1.06–15.70) were significantly associated with clinical remission. The prior use of adalimumab (OR: 0.06; 95% CI: 0.00–0.77) and low CRP levels (OR: 0.75; 95% CI: 0.61–0.93) were associated with endoscopic remission.
In the adjusted multivariable model (Tables 2 and 3), the study showed a 27% increase in the likelihood of a clinical remission with each additional year of IFX treatment. Furthermore, for each 1 mg/L increase in albumin level, there was a threefold higher chance of clinical remission. We also observed that the previous use of adalimumab reduced the probability of endoscopic remission by 94%. The likelihood of endoscopic remission was reduced by 25% with each increase of 1 mg/L in the CRP level.
When considering CD patients alone, the analysis showed that the chance of clinical remission was reduced by 8% with each increase of 1 mg/L in the CRP level. Moreover, the probability of clinical remission in CD patients with IFX dose escalation was 89% lower. The study demonstrated a 49% rise in the likelihood of endoscopic remission with each additional year of IFX treatment. For UC, the concomitant use of immunosuppressive agents was the only factor statistically associated with a lower chance of both outcomes.
Discussion
This study reports the characteristics and outcomes of IBD patients treated with IFX in a tertiary hospital in Brazil. The results from 131 patients show a clinical remission rate of 79.4%, while endoscopic healing occurred in 57 of 98 IBD patients (58.2%). In short, for all participants, the duration of IFX therapy and higher albumin levels were predictive factors for the primary outcome. The prior use of adalimumab and CRP levels were associated with the secondary outcome.
In general, remission rates ranging from 39% to 53% between weeks 30 and 44 have been reported by randomized controlled trials conducted with patients treated with IFX.18,19 Accordingly, the majority of real-world data have confirmed the effectiveness of IFX therapy, and observational studies in the real-world scenario have demonstrated remission rates ranging from 39% to 70% after 12 months of treatment.20–26 In our population, with a mean duration of IFX therapy of 5.03 years, we observed a clinical remission rate of 79.4% and an endoscopic remission rate of 58.2%. The remission rates higher than previously reported of our cohort are most likely attributable to treatment continuation among responders.
Some studies have demonstrated that, when a tumor necrosis factor antagonist (anti-TNF) therapy is followed by another such therapy, the latter is associated with failure.27–29 Gonczi et al. 27 showed that prior exposure to an anti-TNF agent was inversely associated with clinical remission in weeks 14, 30, and 54 for CD patients. A systematic review and meta-analysis by Gisbert et al. 30 found that the effectiveness of a second anti-TNF therapy for CD patients mostly depends on the reason for switching. Such being the case, the remission chance is higher when the reason to withdraw the first anti-TNF medication is intolerance (61%) when compared to secondary (45%) or primary non-response (30%). 30 Besides, favorable responses with IFX were observed in biologic-naïve patients compared to biologic-experienced patients. 31 These results are consistent with our data, as we observed that IBD patients who previously used IFX or adalimumab had a lower chance of clinical and endoscopic remission. Although we have 5-ASA derivatives, immunomodulators, and corticosteroids, it is important to emphasize that anti-TNF agents were the only advanced therapies available for IBD treatment in the Brazilian public health system during this study.
The landmark SONIC and SUCCESS trials demonstrated that concomitant treatment with immunosuppressants was associated with better responses to IFX treatment.32,33 Nevertheless, it is worth highlighting that, in the SONIC trial, only clinical remission at week 26 presented a significant statistical difference between patients treated with IFX in combination with azathioprine and patients undergoing IFX monotherapy. Interestingly, recent post hoc data from the SONIC trial have shown that, when patients are stratified by interquartile of trough concentrations of IFX, the corticosteroid-free clinical remission rates are similar among the patients in the same interquartile range group, irrespective of the association with thiopurines. 34 These data suggest that the role of thiopurines is linked to improvements in the pharmacokinetic profile of IFX. By contrast, in our cohort, the use of immunomodulators was associated with a lower chance of endoscopic healing. However, these findings should be interpreted with caution, since combination therapy with IFX and immunomodulators is associated with lower endoscopic remission rates possibly as a result of unadjusted confounding factors. These in turn would reflect more severe diseases at baseline, given that physicians tend to select more severely ill patients for concomitant administration of immunomodulators. In addition, in our study, combination therapy with immunomodulators was not associated with significant differences in pharmacokinetics, because the median IFX levels were 3.1 and 5.2 μg/mL (p = 0.192) in patients using combination therapy and IFX monotherapy respectively.
In our cohort, prior surgery was negatively associated with clinical remission of CD, which might reflect a more refractory disease as described in Supplemental Table 1. Furthermore, the median time between diagnosis and the start of IFX therapy was 5 years, which underscores the fact that at least part of our population was likely affected by irreversible bowel damage at baseline owing to delayed initiation of IFX treatment. Several studies with CD patients have demonstrated that previous bowel resection is a negative predictive factor of response to anti-TNF therapy.8,35–37 In an Italian multicentric study, Orlando et al. 35 reported that a previous resection was predictive of a worse response in luminal CD. Also, a Belgian cohort study showed that previous surgery was inversely associated with responsiveness in 240 CD patients. 36
It has been observed that patients with lower albumin concentrations have lower remission rates with IFX treatment.8,38 Also, some studies suggest that low albumin levels are a predictive factor of increased IFX clearance in both UC and CD.39,40 These data are further supported by the findings of our study, for we noted an association between albumin levels and primary outcome that was probably influenced by IFX pharmacokinetics.
We observed that lower CRP serum levels were positively associated with endoscopic healing in the present study. CRP levels play a significant role as an inflammatory biomarker in patients with IBD. Some studies have suggested an association between CRP levels and responses to anti-TNF therapy in UC and CD patients. In a follow-up with CD patients by Jürgens et al., 41 early normalization of CRP levels was associated with sustained long-term response. In addition, a post hoc analysis from the ACCENT I trial showed that CRP normalization during IFX therapy resulted in a higher likelihood of sustained response or remission. 42 Besides, a retrospective cohort study with UC patients found that a baseline CRP level of equal to or greater than 5 mg/L was an independent predictor of colectomy. 43 Oussalah et al. 44 reported that CRP at IFX initiation greater than 10 mg/L was also a predictor of colectomy. Furthermore, a retrospective cohort study with UC patients showed that an elevated CRP level (higher than 5 mg/L) was a significant predictive factor for poor outcomes. 25
Several studies discuss the relationship between anti-TNF levels and favorable treatment outcomes.45–48 For example, post hoc analyses from the ACT-1 and ACT-2 data found that higher serum IFX concentrations were associated with higher rates of endoscopic healing in UC patients. 49 Also, a multicenter retrospective cohort study, which included moderate-to-severe UC patients on IFX maintenance therapy, showed that IFX trough concentrations were expressively higher in patients with endoscopic healing than in non-healed patients. 50 Finally, a recent prospective observational study, the PANTS study, which enrolled biologic-naïve CD patients who had started treatment with IFX or adalimumab, identified that low drug concentrations at week 14 were associated with non-remission both at week 14 and at week. 51 Accordingly, in our study, we observed that higher IFX levels were positively associated with endoscopic healing.
This study has a few limitations. First, the small sample size probably contributed toward some discrepancies in the analyses and results with insufficient statistical power. Due to cohort heterogeneity, it is impossible to draw definitive conclusions regarding specific phenotypes of the disease neither extrapolate the findings of the whole IBD cohort to subgroups of patients (UC and CD). This situation reflects the real-world experience of IBD referral hospitals. Additionally, given the cross-sectional design of this study, it was not possible to estimate the performance of IFX treatment at specific time points. It is important to highlight that our cohort included patients during the maintenance phase of treatment. Therefore, the findings should not be extended to primary non-responders. Finally, given the impossibility of purchasing and performing anti-IFX antibody and fecal calprotectin tests, we had no pertinent data to include in our study.
Despite the limitations, this comprehensive analysis describing characteristics and outcomes of IBD patients treated with IFX in Brazil has shed some light on important challenges IBD physicians may confront during IBD care in less developed countries. The baseline characteristics of our population consisted primarily of IBD patients with long disease duration, treatment delay (time between diagnosis and start of IFX therapy), and a complicated disease. This is in accordance with the few available epidemiological data from Brazilian cohorts.52–55 In contrast, data from European cohorts reflect a different scenario possibly caused by the current epidemiological stage of the disease in this region and/or income status in the region. 56 An IBD Swiss cohort study reported that 26%–50% of CD patients had inflammatory behavior with perianal disease ranging from 3.7% to 26%57,58 and that 37.5% of the UC patients had pancolitis.57,59 In another European cohort study, van den Heuvel et al. 60 showed that 77% of the CD patients had inflammatory behavior. Of these, a minority of 15% had structuring behavior and an even smaller minority of 7% had penetrating behavior. On surgery rates, Witte et al. 61 demonstrated that 12.9% of the IBD patients underwent surgery during a 4-year follow-up. Moreover, Burisch 62 showed that the median time for immunobiological treatment in Western countries was 3–5 months (0–15 months). We believe these differences highlight difficulties in the public health system in Brazil, such as diagnostic delays, misdiagnoses, and the lack of access to biologics with novel action mechanisms.
The data presented here fill a vital knowledge gap, given that real-world data originating from Latin America on biological treatments are scarce. Notably, a systematic review by Quaresma et al. showed that biological penetration of anti-TNF agents in Latin America varied from 1.51% up to 46.9% for CD and that the use of anti-TNF in UC was even lower, reaching a maximum of 16.2% in Mexico. 63
Even though the aforementioned limitations might have influenced the data presented here, they have enhanced our understanding of the predictive factors of response to IFX treatment in a well-characterized Brazilian IBD population. Thus, we hope this piece of research will stimulate further investigation into this critical topic in Latin America.
Supplemental Material
sj-doc-1-tag-10.1177_17562848231210053 – Supplemental material for Predictive factors of response to infliximab therapy in Brazilian inflammatory bowel disease patients
Supplemental material, sj-doc-1-tag-10.1177_17562848231210053 for Predictive factors of response to infliximab therapy in Brazilian inflammatory bowel disease patients by Camilla de Almeida Martins, Matheus Freitas Cardoso de Azevedo, Alexandre Sousa Carlos, Aderson Omar Mourão Cintra Damião, Carlos Walter Sobrado Junior, Sergio Carlos Nahas and Natália Sousa Freitas Queiroz in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
