Abstract
Background:
Tofacitinib is an oral Janus kinase inhibitor for the treatment of ulcerative colitis (UC).
Objectives:
To investigate potential predictors of efficacy in RIVETING.
Design:
This post hoc analysis included patients with UC in stable remission (⩾6 months) on tofacitinib 10 mg twice daily (BID) maintenance therapy (⩾2 years of treatment), who received tofacitinib 5/10 mg BID in RIVETING.
Methods:
Achievement of modified Mayo (mMayo) remission, remission (based on total Mayo score), partial Mayo score (PMS) remission, and modified PMS (mPMS) remission at month (M)6 was analyzed by baseline characteristics, duration of PMS remission at RIVETING entry, biomarkers, and patient-reported outcomes (PROs) to identify factors associated with achieving/maintaining efficacy outcomes, including following dose reduction.
Results:
Seventy patients received tofacitinib 5 mg BID. At M6, PMS remission was maintained in 66.7%, 60.0%, 82.4%, 75.0%, and 90.0% of patients with baseline PMS remission durations of 6 to ⩽12, >12 to ⩽24, >24 to ⩽36, >36 to ⩽48, and >48 months, respectively. Patients in mMayo remission at M6 had smaller increases in PMS at M1 compared with those not in mMayo remission at M6, while numerically higher proportions of patients with a stool frequency subscore/rectal bleeding subscore/mPMS of 0 at M1/M3 achieved most efficacy endpoints at M6 compared with patients with respective subscores >0. Maintenance of mMayo remission was independent of the number of tumor necrosis factor inhibitors failed and/or prior corticosteroid use. In multivariable models (which included tofacitinib 10 mg BID data), endoscopic subscores (1 vs 0) at RIVETING baseline were significantly associated with lower odds of mMayo remission at M6 (odds ratio, 0.33; 95% confidence interval, 0.11–0.94; p = 0.0379).
Conclusion:
Prior duration of remission/baseline endoscopic subscore may help guide tofacitinib dose reduction, while PROs may be useful early indicators of efficacy. Close monitoring of patients following dose reduction could identify those unlikely to achieve/maintain efficacy.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory disease that is usually characterized by rectal bleeding and increased frequency of bowel movements, and follows a relapsing and remitting course. 1 The goal of UC therapy is to achieve long-term, steroid-free remission2,3 while minimizing the medication burden, adverse events, and cost associated with long-term treatment.4,5 Therefore, it is important for physicians managing patients with UC to understand the possible clinical consequences of dose adjustments and flexible treatment.
To better identify patients for whom dose adjustments are likely to be successful, there is a need to identify clinical factors that are predictive of maintaining response to therapy following dose adjustment, particularly dose reduction.6,7 Tofacitinib is an oral Janus kinase (JAK) inhibitor for the treatment of UC. While carefully considering the risk/benefit profile for each patient, reduction of tofacitinib dose may decrease treatment-related costs and reduce the risk of dose-dependent adverse events associated with immunosuppression in patients in remission. 8
The efficacy and safety of tofacitinib in patients with UC in stable remission (⩾6 months) on 10 mg twice daily (BID) maintenance therapy (⩾2 years of treatment) whose dose reduced to tofacitinib 5 mg BID, compared with patients who remained on tofacitinib 10 mg BID, have been evaluated in the phase IIIb/IV, double-blind, randomized RIVETING (NCT03281304) study. 8 Prior to entering the RIVETING study, patients would have been enrolled in one of two phase III induction studies (OCTAVE Induction 1 and 2, NCT01465763 and NCT01458951), 9 before progressing to the phase III maintenance study (OCTAVE Sustain, NCT01458574) 9 or enrolling directly in the open-label long-term extension (OLE) study (OCTAVE Open, NCT01470612). 10 The RIVETING study demonstrated that a higher proportion of patients whose dose was reduced to tofacitinib 5 mg BID, and were in deep endoscopic remission (an endoscopic subscore of 0 from their most recent endoscopy, prior to or at RIVETING baseline) or did not have prior tumor necrosis factor inhibitor (TNFi) failure, maintained the primary endpoint of modified Mayo (mMayo) remission at month 6 than patients with an endoscopic subscore of 1 or prior TNFi failure. 8
The exploratory post hoc analyses presented here further evaluated the association between baseline clinical characteristics, prior treatment, disease activity measures (partial Mayo score [PMS] and modified PMS [mPMS]), laboratory assessment biomarkers (fecal calprotectin [FCP] and C-reactive protein [CRP]), patient-reported outcomes (PROs; stool frequency and rectal bleeding subscores), and maintenance of efficacy outcomes in patients with UC in stable remission in the RIVETING study.
Methods
Patients and study design
RIVETING was a phase IIIb/IV, multicenter, double-blind, randomized, parallel-group study designed to evaluate the efficacy and safety of dose reduction to tofacitinib 5 mg BID compared with remaining on 10 mg BID in patients with moderately to severely active UC in stable remission (⩾6 months) on tofacitinib 10 mg BID maintenance therapy (⩾2 years of treatment in OCTAVE Open). 10 The full details of RIVETING have been published previously. 8 Briefly, patients were randomized in a 1:1 ratio to receive either tofacitinib 5 or 10 mg BID at baseline. Dose adjustments in patients treated with tofacitinib 5 mg BID to open-label 10 mg BID were permitted after the month 1 trial visit if patients met protocol-specified flare criteria. Flare was defined as an increase in Mayo rectal bleeding subscore of ⩾1 point and an increase in endoscopic subscore of ⩾1 point; or an increase in rectal bleeding subscore of ⩾2 points and an endoscopic subscore >0; or an increase in Mayo stool frequency subscore of ⩾2 points and an increase in endoscopic subscore by ⩾1 point; or an increase in endoscopic subscore of ⩾2 points. When flare was confirmed, the patient either increased their dose from 5 to 10 mg BID (provided the patient did not have any risk factors for pulmonary embolism) or remained on 10 mg BID in an open-label fashion; patients and local investigators remained blinded to their pre-flare dose. In addition, if a patient had a newly identified risk factor for pulmonary embolism, their dose was reduced to open-label 5 mg BID. Although the focus of the analyses was to explore the maintenance of remission after dose reduction from tofacitinib 10 to 5 mg BID, the tofacitinib 10 mg BID group was included to increase the sample size in some of the modeling analyses. The study protocol was approved by the Institutional Review Board or independent ethics committee at each study site; details of all study sites have been previously published. 8
The stable remission cutoff of ⩾6 months prior to enrollment for entry to RIVETING was selected partly on the timing of the endoscopy procedures in OCTAVE Open, and on prior study data showing that patients in remission for ⩾6 vs <6 months were more likely to stay in remission. 11 Concomitant corticosteroids were prohibited in RIVETING. Corticosteroid use in the induction, maintenance, and OLE studies has been described previously.9,10
Efficacy endpoints
To identify potential predictors of month 6 efficacy outcomes (with a particular focus on the group of patients whose dose was reduced to tofacitinib 5 mg BID), the following efficacy endpoints were assessed (February 20, 2020 data cutoff): remission based on mMayo score (the primary efficacy endpoint of RIVETING, defined as an endoscopic subscore of 0 or 1, a stool frequency subscore of 0 or 1, and a rectal bleeding subscore of 0); remission based on total Mayo score (defined as a total Mayo score of ⩽2 with no individual subscore >1, and a rectal bleeding subscore of 0); endoscopic improvement (defined as an endoscopic subscore of 0 or 1; defined as “mucosal healing” in the RIVETING protocol) 8 ; clinical response based on total Mayo score (defined as a decrease from induction study baseline total Mayo score of ⩾3 points and ⩾30%, plus a decrease in rectal bleeding subscore of ⩾1 point or an absolute rectal bleeding subscore of 0 or 1); remission based on mPMS (defined as a stool frequency subscore of ⩽1 and a rectal bleeding subscore of 0); and remission based on PMS (defined as a PMS of ⩽2 with no individual subscore >1, and a rectal bleeding subscore of 0). PMS, stool frequency subscores, and rectal bleeding subscores were also assessed at months 1 and 3.
Statistical analyses
In these post hoc analyses, the full analysis set included all patients who were randomized into the study and received at least one dose of tofacitinib 5 or 10 mg BID. Patients were analyzed as randomized. As patients receiving tofacitinib 5 mg BID with confirmed flare could be dose escalated to receive tofacitinib 10 mg BID, patients in the tofacitinib 5 mg BID group who escalated dose were treated as non-responders for visits after their dose escalation to determine achievement of binary efficacy endpoints. Analyses of predictors and covariates were derived based on observed case data, with non-responder imputation applied for month 6 binary efficacy endpoints.
Month 6 binary efficacy endpoints were analyzed descriptively according to the following variables: the number of prior TNFis failed (0, 1, or >1); corticosteroid use (yes/no) and corticosteroid dose (<15 or ⩾15 mg/day) at baseline of the phase III induction studies; corticosteroid use (yes/no) at OLE study baseline; and stool frequency subscore, rectal bleeding subscore, and mPMS (all 0 vs >0) at month 1 and month 3 of RIVETING. Changes from baseline in PMS, FCP, and CRP at month 1 and month 3 of RIVETING were summarized for month 6 binary efficacy endpoints.
Prior duration of PMS remission at the RIVETING baseline was analyzed categorically (6 to ⩽12, >12 to ⩽24, >24 to ⩽36, >36 to ⩽48, and >48 months), and month 6 binary efficacy outcomes were summarized according to categories of duration of remission. Duration of PMS remission was calculated in days from the onset of the most recent episode of PMS remission achieved in the studies preceding RIVETING. Per protocol, all patients were required to be in remission for ⩾6 months prior to enrollment in RIVETING.
Using univariate logistic regression analyses, the ability of the duration of PMS remission to predict each of the binary efficacy outcomes at month 6 was assessed using receiver operating characteristic (ROC) curves. The area under the curve (AUC) was calculated to assess the overall predictive value of duration of remission, as a continuous variable, in relation to efficacy outcomes. An AUC-ROC of 0.9 was used to reflect excellent discriminative ability. A predictor with an AUC-ROC value below 0.6 was considered of limited predictive ability and values greater than 0.7 were considered to be potential predictors (values 0.7–0.8 are defined as having acceptable predictive ability). 12 The Youden index (YI) defined as “sensitivity + specificity −1” was calculated for all possible threshold values of duration of remission, and the cutoff value corresponding to the maximum YI was considered of optimal predictive capacity. The optimal cutoff of duration of remission was assessed for each of the binary efficacy endpoints listed above, separately. Sensitivity and specificity corresponding to the maximum YI/optimal cutoff were also determined. Due to sample size limitations, the 10 mg BID group was included in the multivariable and additional analyses.
Associations between month 6 mMayo remission, remission (based on total Mayo score), mPMS remission, and PMS remission and each independent variable were analyzed using univariate logistic regression. Baseline explanatory variables included prior TNFi failure, prior immunosuppressant failure, prior corticosteroid use, smoking history, and sex (all at baseline of the phase III induction studies); disease duration at baseline of the induction studies and baseline of RIVETING; corticosteroid use at OLE baseline; age, body mass index, and endoscopic subscore at RIVETING baseline; and duration of PMS remission (continuous) prior to enrollment into RIVETING. Month 1/month 3 explanatory variables included the following: mPMS, stool frequency subscore, rectal bleeding subscore (all 0 vs >0), PMS, log(FCP), and log(CRP). Treatment (tofacitinib dose received during RIVETING) was included in the univariate model as a covariate for the analyses including data from both the tofacitinib 5 and 10 mg BID groups; an additional logistic regression model, that evaluated treatment by predictor interaction, was run for each predictor.
Multivariable logistic regression analyses were performed for each efficacy endpoint, with data from both the tofacitinib 5 and 10 mg BID treatment groups included in the model. Two sets of multivariable logistic regression analyses were produced for the association of each binary efficacy endpoint with the explanatory variables above. The first set of models used baseline explanatory variables, duration of remission, and month 1 explanatory variables. The second set of models used baseline explanatory variables, duration of remission, and month 3 explanatory variables. Within each of the two sets of models, there were three models for each efficacy endpoint to address the potential issue of multicollinearity. The first included rectal bleeding and stool frequency subscores, the second included PMS, and the third included mPMS. Therefore, there were a total of six multivariable models for each efficacy outcome. A backward selection procedure was used, at the 0.05 level of significance. The treatment group (tofacitinib 10 vs 5 mg BID) was included in each of the models.
Odds ratios (ORs) and 95% confidence intervals (CIs) from the logistic regression analyses were reported for variables associated with efficacy outcomes at month 6 of RIVETING, in addition to the p value for each association. No adjustments were made for multiple comparisons.
Results
Patients (tofacitinib 5 and 10 mg BID groups)
In RIVETING, 70 patients were randomized to receive tofacitinib 5 mg BID. At, or prior to, month 6, 9 patients’ dose escalated to tofacitinib 10 mg BID due to a flare. In addition, 70 patients were randomized to receive tofacitinib 10 mg BID, of whom 7 patients did not meet the primary efficacy endpoint at month 6 (all patients were included in this analysis). Baseline demographics and clinical characteristics are shown in Table S1; full details have been published previously. 8 Briefly, among patients who received tofacitinib 5 mg BID in RIVETING, the mean age was 47.8 years (standard deviation [SD] 14.1), 37.1% of patients were female, 72.9% of patients had an endoscopic subscore of 0 at baseline of RIVETING, 38.6% of patients had prior TNFi failure (17 patients had previous failure with 1 TNFi and 10 patients had previous failure with >1 TNFi), and 37.1% of patients had a prior history of corticosteroid use at induction study baseline. Among patients who received tofacitinib 10 mg BID in RIVETING, the mean age was also 47.8 years (SD 13.5), 31.4% of patients were female, and as with the 5 mg BID-treated patients, 72.9% of patients had an endoscopic subscore of 0 at baseline of RIVETING. In the tofacitinib 10 mg BID-treated group, 50.0% of patients had prior TNFi failure and 48.6% of patients had a prior history of corticosteroid use at the induction study baseline.
Effect of the number of prior TNFi failed and prior corticosteroid use on maintenance of efficacy following tofacitinib dose reduction (tofacitinib 5 mg BID group)
At month 6 of RIVETING, mMayo remission was maintained in 79.1%, 70.6%, and 80.0% of patients receiving tofacitinib 5 mg BID who had previous failure with 0, 1, and >1 TNFi, respectively (Figure 1). Similar results were observed for remission, endoscopic improvement, mPMS remission, and PMS remission. Clinical response was maintained in 86.0%, 82.4%, and 80.0% of patients receiving tofacitinib 5 mg BID who had previous failure with 0, 1, and >1 TNFi, respectively (Figure 1). Therefore, there did not appear to be a relationship between the number of prior TNFi failures and efficacy outcomes at month 6.
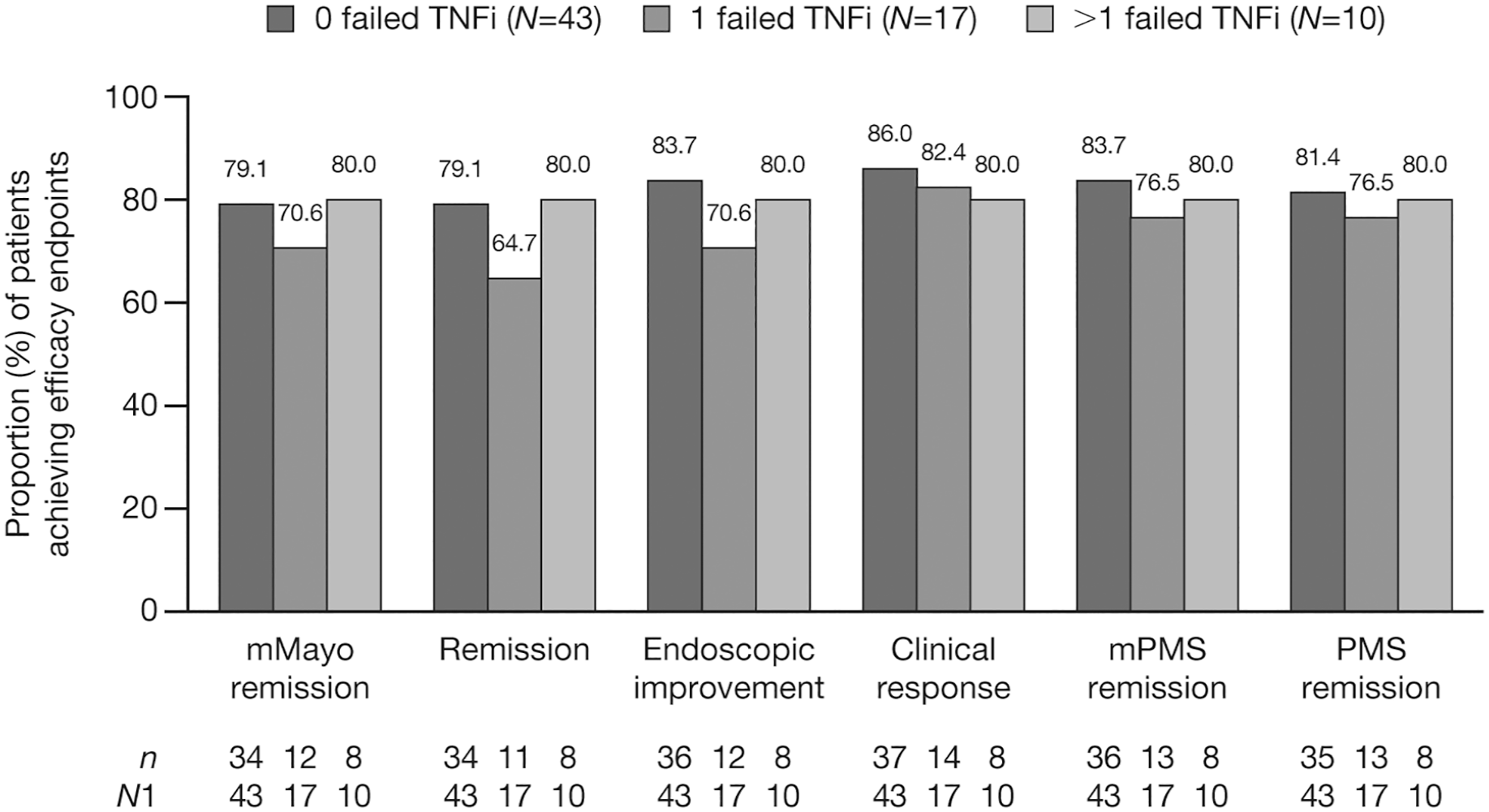
Proportion of patients receiving tofacitinib 5 mg BID who achieved efficacy endpoints at month 6, by the number of prior TNFi failed (FAS, observed).
Among patients who enrolled in RIVETING and received tofacitinib 5 mg BID, 26 patients were receiving corticosteroids (9 patients received <15 mg/day and 17 patients received ≥15 mg/day) at induction study baseline, and 7 patients were receiving corticosteroids at OLE study baseline. The median time from corticosteroid exposure to RIVETING baseline was 1288 days.
At month 6 of RIVETING, rates of mMayo remission were 84.6% among patients with corticosteroid use at the induction study baseline compared with 72.7% in those without corticosteroid use (Figure S1[a]). The month 6 changes from RIVETING baseline in mMayo score and total Mayo score were 0.7 and 1.1, respectively, in patients with corticosteroid use at induction study baseline, and 0.6 and 0.9, respectively, in patients without corticosteroid use at induction study baseline (Figure S1[a]). There was generally no difference in the achievement of efficacy endpoints when patients were stratified by corticosteroid dose at induction study baseline (Figure S1[b]), or among patients with or without corticosteroid use at OLE baseline (Figure S1[c]).
Association between the duration of PMS remission prior to enrollment into RIVETING and maintenance of efficacy following tofacitinib dose reduction (tofacitinib 5 and 10 mg BID groups)
At the RIVETING baseline, 70 of 140 patients were initially randomized to tofacitinib 5 mg BID; however, 1 patient was excluded for not meeting PMS remission criteria and was included in the study as a protocol deviation. Consequently, a total of 139 of 140 patients were in PMS remission at the RIVETING baseline, 69 of whose dose was reduced to tofacitinib 5 mg BID. At month 6, following dose reduction to tofacitinib 5 mg BID, PMS remission was maintained in 66.7%, 60.0%, 82.4%, 75.0%, and 90.0% of patients with baseline PMS remission durations of 6 to ⩽12, >12 to ⩽24, >24 to⩽36, >36 to ⩽48, and >48 months, respectively (Figure 2[a]). At month 6, in patients who continued to receive tofacitinib 10 mg BID, PMS remission was maintained in 80.0%, 88.9%, 91.7%, 100.0%, and 100.0% of patients with baseline PMS remission durations of 6 to ⩽12, >12 to ⩽24, >24 to ⩽36, >36 to ⩽48, and >48 months, respectively (Figure 2[b]).

Proportion of patients receiving (a) tofacitinib 5 mg BID or (b) tofacitinib 10 mg BID who achieved efficacy endpoints at month 6, by baseline PMS remission duration (NRIa,b).
Among patients who received tofacitinib 5 mg BID in the RIVETING study, patients who had been in PMS remission for >24 months prior to RIVETING baseline generally had a numerically smaller increase from baseline mMayo and total Mayo scores at month 6 than patients with a baseline PMS remission duration of ⩽24 months (Table S2). The mean (SD) increase from baseline to month 6 in mMayo score was 0.7 (0.6), 1.2 (2.4), 0.7 (1.1), 0.4 (1.2), and 0.9 (1.9) among patients with baseline PMS remission durations of 6 to ⩽12, >12 to ⩽24, >24 to ⩽36, >36 to ⩽48, and >48 months, respectively. Corresponding values for the mean (SD) increase from baseline to month 6 in total Mayo score were 0.7 (0.6), 1.8 (3.2), 1.1 (1.6), 0.6 (1.7), and 1.2 (2.7), respectively (Table S2).
The predictive value of duration of remission for achieving efficacy outcomes at month 6 was further investigated using AUC-ROC analysis (Table 1). With an optimal cutoff value of 775 days (25.4 months or 2.12 years), the duration of remission had an AUC-ROC of 0.62 for mMayo remission, with a sensitivity and specificity of 82.8% and 39.1%, respectively. The AUC-ROC analysis for the ability of duration of remission to predict remission, mPMS remission, PMS remission, endoscopic improvement, and clinical response is shown in Table 1.
AUC-ROC analysis for the ability of duration of PMS remission to predict maintenance of efficacy outcomes for patients receiving tofacitinib 5 or 10 mg BID at month 6 of RIVETING (FAS a ).
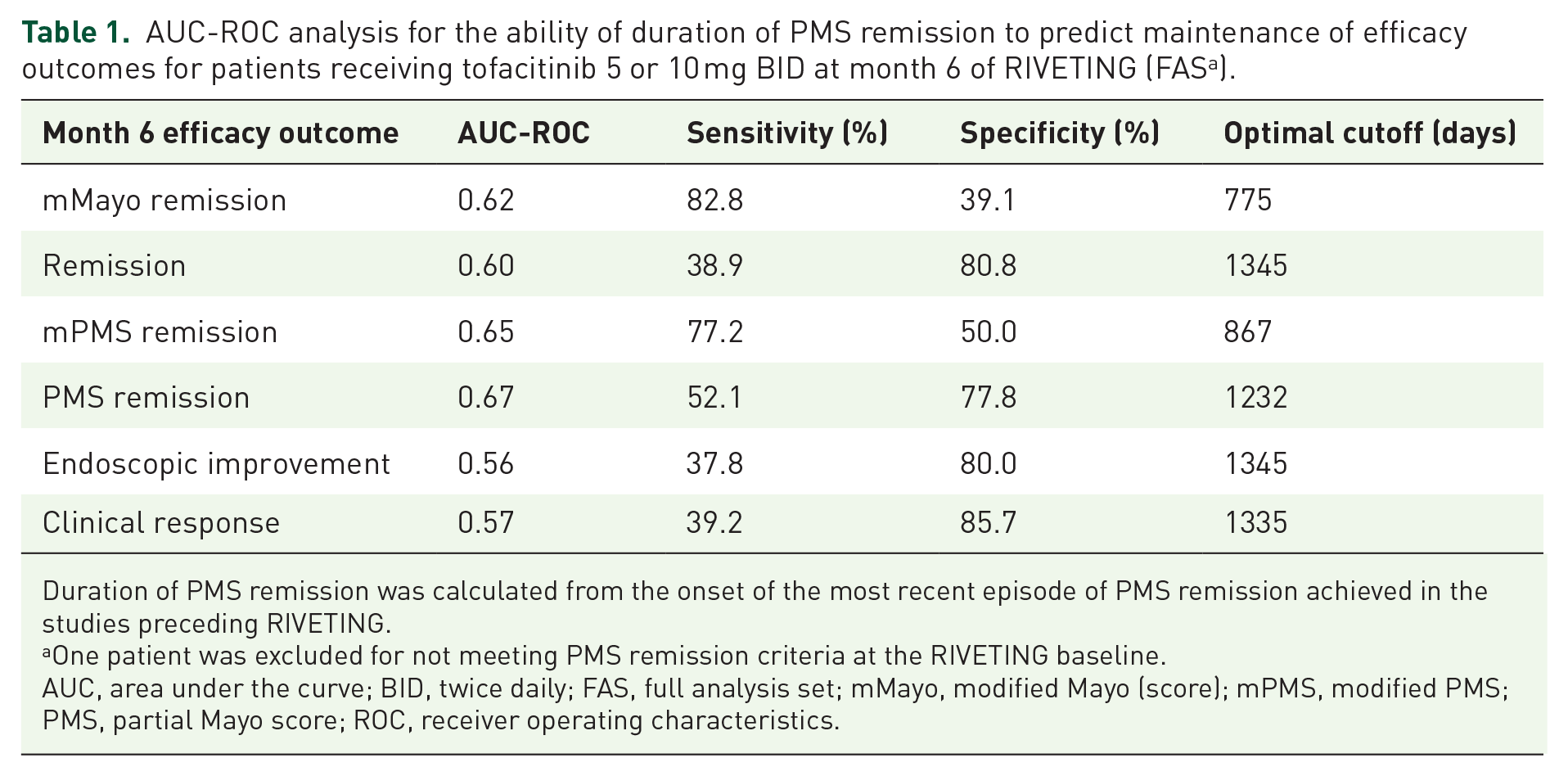
Duration of PMS remission was calculated from the onset of the most recent episode of PMS remission achieved in the studies preceding RIVETING.
One patient was excluded for not meeting PMS remission criteria at the RIVETING baseline.
AUC, area under the curve; BID, twice daily; FAS, full analysis set; mMayo, modified Mayo (score); mPMS, modified PMS; PMS, partial Mayo score; ROC, receiver operating characteristics.
Effect of early changes in noninvasive markers on maintenance of efficacy following tofacitinib dose reduction (tofacitinib 5 mg BID group)
Among patients receiving tofacitinib 5 mg BID, of those in mMayo remission at month 6, FCP level did not change from baseline to month 3 (median change, interquartile range [IQR]) 0.0 [−48.5 to 40.5]). In patients not in mMayo remission at month 6, an increase in FCP levels was observed (30.0 [−53.5 to 429.0]). With the exception of remission, this trend was also observed for the other efficacy endpoints (Table 2).
Change from baseline in PMS, FCP level, and CRP level in patients receiving tofacitinib 5 mg BID by month 6 efficacy endpoints (FAS, observed).

Binary efficacy endpoint subgroups were determined using NRI.
BID, twice daily; CI, confidence interval; CRP, C-reactive protein; FAS, full analysis set; FCP, fecal calprotectin; IQR, interquartile range; mMayo, modified Mayo (score); mPMS, modified PMS; N1, number of patients with non-missing values; NRI, non-responder imputation; OR, odds ratio; PMS, partial Mayo score; SD, standard deviation.
Among patients receiving tofacitinib 5 mg BID in RIVETING, in those who achieved efficacy outcomes at month 6, there were minimal or no changes in CRP levels from baseline at month 1 or month 3. The change from baseline in CRP level (median (IQR)) at month 1 and month 3 was similar in patients with and without mMayo remission at month 6 (0.0 [−0.7 to 0.4] and 0.1 [−0.4 to 0.7] vs 0.0 [−0.2 to 0.5] and 0.1 [−1.2 to 0.4], respectively; Table 2).
Among those patients receiving tofacitinib 5 mg BID who achieved efficacy endpoints at month 6 of RIVETING, the mean PMS was relatively stable over time. By contrast, mean PMS increased from baseline to month 1 and month 3 in patients who did not achieve efficacy endpoints at month 6 (Table 2). Mean (SD) change from baseline PMS among patients who achieved mMayo remission at month 6 was 0.2 (0.7) and 0.1 (0.8) at month 1 and month 3, respectively, compared with 0.5 (1.1) and 0.7 (1.3), respectively, among patients who did not achieve mMayo remission (Table 2).
Effect of early changes in PROs on maintenance of efficacy following tofacitinib dose reduction (tofacitinib 5 mg BID group)
The proportion of patients who achieved mMayo remission at month 6 was higher in patients with a stool frequency subscore of 0 at month 1 or month 3 compared with patients with a stool frequency subscore >0 at the same time points (83.3% and 91.1% compared with 68.4% and 68.4%, respectively). The same was true among patients with a rectal bleeding subscore of 0 compared with rectal bleeding subscore >0 (82.5% and 86.4% compared with 25.0% and 60.0%, respectively) and patients with a mPMS of 0 compared with >0 (87.0% and 92.9% compared with 61.9% and 68.2%, respectively). Similar trends were observed for remission, mPMS remission, PMS remission, endoscopic improvement, and clinical response (Figure 3).

Proportion of patients receiving tofacitinib 5 mg BID who achieved efficacy endpoints at month 6, categorized by stool frequency subscore, rectal bleeding subscore, and mPMS at month 1 and month 3 (FAS, observed).
Association between potential predictors and month 6 efficacy endpoints in patients receiving tofacitinib 5 or 10 mg BID in RIVETING (tofacitinib 5 and 10 mg BID groups)
Univariate logistic regression exploring the potential association of predictors with efficacy outcomes at month 6 is shown for tofacitinib 5 mg BID, tofacitinib 10 mg BID, and both doses (Tables S3 and S4). Factors at months 1 and 3 that were indicative of achieving all efficacy outcomes assessed at month 6 included PMS (continuous) and mPMS (0 vs >0). Stool frequency subscore 0 versus >0 at month 1 was indicative of achieving PMS remission at month 6, and stool frequency subscore 0 versus >0 at month 3 was indicative of achieving all efficacy outcomes measured at month 6. In addition, RBS 0 versus >0 at month 1 was indicative of achieving all efficacy outcomes measured at month 6.
In the multivariable models containing baseline explanatory variables, duration of PMS remission, and month 1 explanatory variables, higher endoscopic subscore at baseline of RIVETING was significantly associated with lower odds of mMayo remission at month 6 (1 vs 0; OR, 0.33; 95% CI, 0.11–0.94; p = 0.0379; Table 3). There was an association between “smoking history” (includes all categories (current smokers, ex-smokers, and never-smokers)) and mMayo remission at month 6 (p = 0.0376). Current smokers (patients who were smokers at induction study baseline) had lower odds of achieving mMayo remission than never-smokers (OR, 0.05; 95% CI, 0.00–0.49; p = 0.0104), while there was no difference between ex-smokers and never-smokers (OR, 0.74; 95% CI, 0.26–2.14; p = 0.5808). In addition, shorter disease duration at baseline of OCTAVE Induction 1 and 2 was significantly associated with higher odds of mMayo remission at month 6 (<6 vs ⩾6 years; OR, 3.13; 95% CI, 1.04–9.40; p = 0.0425). A longer prior duration of PMS remission was significantly associated with higher odds of achieving mMayo remission at month 6 (OR, 1.00; 95% CI, 1.00–1.00; p = 0.0316; Table 3).
Multivariable logistic regression analysis results for the association of explanatory variables with mMayo remission at month 6 in patients receiving tofacitinib 5 or 10 mg BID (FAS, NRI). a

ORs and 95% CIs are reported to two decimal places; values shown as 1.00 may be less than or greater than 1.
Backward selection was employed to arrive at the final models.
These three models were run separately with identical results.
Baseline of RIVETING.
Baseline of OCTAVE Induction 1 and 2.
Continuous.
BID, twice daily; CI, confidence interval; FAS, full analysis set; mMayo, modified Mayo (score); mPMS, modified PMS; NRI, non-responder imputation; ns, not selected; OR, odds ratio; PMS, partial Mayo score; RBS, rectal bleeding subscore; SFS, stool frequency subscore; TNFi, tumor necrosis factor inhibitor.
In the multivariable models containing baseline explanatory variables, duration of remission, and month 3 explanatory variables, prior corticosteroid use at induction study baseline was associated with higher odds of mMayo remission at month 6 (OR, 4.49; 95% CI, 1.02–19.75; p = 0.0470). A longer prior duration of PMS remission was significantly associated with higher odds of achieving mMayo remission at month 6 (OR, 1.00; 95% CI, 1.00–1.00; p = 0.0134; Table 3). Prior to TNFi failure, disease duration from induction baseline and smoking history were also selected in the final model containing baseline explanatory variables, duration of remission, and month 3 explanatory variables; however, none of these variables were significantly associated with mMayo remission at month 6 (Table 3).
Multivariable logistic regression analyses to determine factors associated with remission, mPMS remission, and PMS remission at month 6 are shown in Tables S5 and S6. Duration of PMS remission was selected in the final model in most cases (p < 0.05).
Discussion
This report presents post hoc analyses evaluating the association between baseline demographics, clinical characteristics, previous treatment, biomarkers, and PROs and maintenance of efficacy outcomes in patients with UC in the RIVETING study, which included those in stable remission on tofacitinib 10 mg BID whose dose was reduced to tofacitinib 5 mg BID.
These analyses showed that patients with a duration of PMS remission ⩾2 years or an endoscopic subscore of 0 at baseline of RIVETING were numerically more likely to maintain mMayo remission at month 6, while maintenance of mMayo remission was independent of the number of prior TNFis failed and/or prior corticosteroid use. Patients without mMayo remission at month 6 had a greater increase in PMS at month 1 following dose reduction than patients who achieved mMayo remission at month 6. Unsurprisingly, stool frequency subscore, rectal bleeding subscore, or an mPMS of 0 at either month 1 or month 3 and PMS (continuous) was indicative of the likelihood of maintaining efficacy with tofacitinib 5 mg BID at month 6.
The possibility of dose reduction of UC therapies once remission has been achieved is of concern for both physicians and patients, driven by safety considerations and treatment-related costs. Tofacitinib is the first JAK inhibitor approved for the treatment of moderately to severely active UC, with efficacy having been demonstrated in the tofacitinib UC clinical program and real-world studies.9,13,14 Given that the current tofacitinib labels and clinical guidelines state that the lowest effective dose needed to maintain response should be used during maintenance treatment,15–18 it is important to identify potential predictors of maintaining response/remission.
The duration of remission was evaluated as a potential predictor of achieving efficacy outcomes among patients in stable remission. Rates of mMayo remission, remission, mPMS remission, and PMS remission at month 6 of RIVETING were numerically higher in patients with a PMS remission duration of >24 months, versus patients with a PMS remission duration of ⩽24 months. However, the number of patients in the groups with shorter duration of remission was very small, limiting interpretation. Longer prior duration of PMS remission was found to be significantly associated with an increased likelihood of patients achieving efficacy outcomes in the multivariable logistic regression models (p < 0.05). However, since the duration of remission was calculated in days, and as this was at least 6 months upon entering RIVETING, an increase of 1 day in duration of remission was unlikely to have a clinically meaningful impact on the likelihood of achieving efficacy outcomes, even though the p value was significant. Moreover, the duration of remission AUC-ROC value for PMS remission was 0.67, which falls below the acceptable predictive cutoff value of 0.7. The potential association between duration of remission and sustained remission was assessed in a previous post hoc analysis of the “maintenance remission” subpopulation from the OLE study (i.e., patients in remission after 52 weeks of tofacitinib 10 mg BID maintenance therapy during OCTAVE Sustain who were assigned to receive 5 mg BID in the OLE study). This previous analysis showed that maintenance of remission through to month 24 in the OLE study was numerically more likely in patients who had been in remission for ⩾6 months compared with those who had been in remission for <6 months before dose reduction. 11 Of note, the tofacitinib label states that the lowest effective dose to maintain response should be used. These data suggest that prior duration and depth of remission may be important considerations alongside additional information when making individualized clinical decisions regarding dose reduction during maintenance therapy.15,16
Further to the descriptive findings in the primary RIVETING analysis, 8 data from the multivariable analysis showed that, when month 1 variables were included in the models, patients with an endoscopic subscore of 0 were more likely to maintain mMayo remission than patients with an endoscopic subscore of 1. This is consistent with findings from several studies in patients with UC, which found that patients with an endoscopic subscore of 0 had a reduced risk of disease recurrence compared with patients with an endoscopic subscore of 1,19–21 and further underscores the clinical importance of a patient’s endoscopic subscore, irrespective of other characteristics when considering treatment or dose. Smoking history was significantly associated with mMayo remission at month 6, with current smokers versus never-smokers (all at induction study baseline) having lower odds of achieving this efficacy endpoint. These findings contrast with other studies in patients with UC, which suggest that smoking may have a protective effect against adverse UC outcomes. 22 However, the findings presented here may be limited by the small number of smokers included in the study.
In these post hoc analyses, patients in stable remission receiving tofacitinib 10 mg BID for ⩾2 years maintained efficacy following 6 months of dose reduction to 5 mg BID, regardless of the number of prior TNFis failed. A previous analysis of the RIVETING study demonstrated that, for patients who dose reduced, a slightly higher observed proportion of patients without prior TNFi failure maintained remission or achieved other binary efficacy endpoints at month 6 compared with patients with prior TNFi failure, 8 findings that were consistent with data from the phase III maintenance study (OCTAVE Sustain). 23 Patients who have previously failed TNFi therapy, particularly multiple different therapies, are generally considered more refractory than those who have not.24,25 However, tofacitinib is still considered to be effective in patients with prior TNFi failure, 23 and our findings showed that maintenance of mMayo remission with tofacitinib following dose reduction in patients with stable remission with prior TNFi failure was not affected by the number of previously unsuccessful therapies. This is consistent with previous findings in the OCTAVE studies, which show the limited effect of prior TNFi number on efficacy of tofacitinib 5 mg BID in OCTAVE Sustain. 23 The limited difference between patients with and without prior TNFi failure in the achievement of efficacy outcomes in the tofacitinib UC clinical program is likely related to the selection of patients with clinical response or remission for study entry. Per protocol, only patients with clinical response following induction studies could enter the maintenance study, and only patients in remission could enter RIVETING; therefore, they are responding well to tofacitinib, irrespective of the number of previously failed therapies.8,9
In patients who were in stable remission for at least 6 months, efficacy endpoints were maintained following dose reduction to tofacitinib 5 mg BID, regardless of prior corticosteroid use or dose at program entry, which required mandatory taper of corticosteroids following induction. These findings are consistent with data from the phase III maintenance study, in which tofacitinib demonstrated significant maintenance efficacy compared with placebo, regardless of maintenance baseline corticosteroid use. 9 The lack of difference between patients with and without prior corticosteroid use or corticosteroid dose in the achievement of efficacy outcomes in the RIVETING study is possibly due to the length of time between program entry and enrollment in RIVETING, which for some patients could be 5–6 years. Of note, the results from the multivariable model that utilized month 3 potential predictors indicated that prior corticosteroid use was associated with higher odds of mMayo remission at month 6 which may, in part, be due to the small number of patients who did not achieve efficacy outcomes at this time point.
In terms of clinical practice, following dose reduction, it is advisable that patients be closely monitored for early changes in biomarkers and PROs that may identify those likely to maintain efficacy outcomes. 26 It should be noted that overall the median changes in CRP and FCP levels from baseline to month 3 were minor. This may be due to the relatively short time frame for these analyses. However, interpretation of these data was limited due to the small sample size, as most patients met the month 6 efficacy outcomes. Furthermore, a stool frequency subscore, rectal bleeding subscore, or mPMS of 0 at either month 1 or month 3, as well as an endoscopic subscore of 0 versus 1 at RIVETING baseline, stool frequency subscore 0 versus >0 at Month 3, and RBS 0 versus >0 at Month 1, could indicate the likelihood of maintaining efficacy with tofacitinib 5 mg BID at month 6. Taken together with other aspects of a patient’s response to tofacitinib therapy, these PROs could represent a practical early measure for identifying which patients may benefit from dose reduction, or those who are likely to maintain remission longer term; therefore, close monitoring of patients following dose reduction is recommended.
Limitations
A limitation of the current study was that these were post hoc analyses, and the RIVETING study was not designed, nor powered, to identify or evaluate predictors of response following dose reduction. Data interpretation was limited by small sample sizes, particularly in relation to the small number of patients in each duration of remission subgroup, those who did not achieve month 6 efficacy outcomes, those with subscores >0, and the subgroup sizes for prior TNFi history. As such, the implications for clinical practice are challenging to fully determine. In addition, some patients had missing data, particularly for the FCP and CRP analyses, and analyses did not control for multiple comparisons; therefore, findings should be interpreted with caution. Prior TNFi failure in this study was determined by investigators and did not specify a minimum dose or treatment duration with TNFi and, as such, the number of prior TNFi failed may not be a reliable indicator of more refractory disease. Although the study aimed to determine predictors of response in the tofacitinib 5 mg BID group (who had dose reduced), due to the small sample size, data from both treatment groups were included in the multivariable modeling and ROC-AUC analyses. The results of the analyses of treatment by predictor interaction indicated that there was no evidence of significant treatment interaction for any potential predictor; therefore, the effect of potential predictors was independent of treatment.
Conclusion
In conclusion, these data suggest that prior duration of remission and baseline endoscopic subscore may be used with additional information to guide dose reduction. Furthermore, close monitoring of patients and early assessment of PROs could help identify those likely to achieve/maintain efficacy outcomes.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848251318849 – Supplemental material for Potential predictors of efficacy outcomes following tofacitinib dose reduction in patients with ulcerative colitis in stable remission: a post hoc analysis of outcomes from the RIVETING study
Supplemental material, sj-pdf-1-tag-10.1177_17562848251318849 for Potential predictors of efficacy outcomes following tofacitinib dose reduction in patients with ulcerative colitis in stable remission: a post hoc analysis of outcomes from the RIVETING study by Joana Torres, Geert R. D’Haens, Miguel Regueiro, Genoile Santana, Julian Panés, Séverine Vermeire, Sean Gardiner, Nicole Kulisek, Irene Modesto, Chinyu Su, Nervin Lawendy, Rajiv Mundayat, Jerome Paulissen and Marla C. Dubinsky in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors would like to thank the patients, investigators, and study teams involved in the OCTAVE trials. This study was sponsored by Pfizer. Medical writing support, under the direction of the authors, was provided by Sarah Mancini, PhD, CMC Connect, a division of IPG Health Medical Communications, and was funded by Pfizer, New York, NY, USA, in accordance with Good Publication Practice (GPP 2022) guidelines (Ann Intern Med 2022; 175: 1298–304). Jerome Paulissen is an employee of Syneos Health, which was a paid contractor to Pfizer in connection with the development of this manuscript and the related statistical analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
