Abstract
Inflammatory bowel disease (IBD) frequently affects women of childbearing age and can have implications in pregnancy. Most women with IBD have comparable fertility with women in the general population. Fertility is reduced in women with active disease or previous ileal-pouch–anal anastomosis (IPAA) surgery and is temporarily reduced in men taking sulfasalazine. Women with IBD have an increased risk of preterm delivery, low birth weight, small-for-gestational-age infants and Cesarean section (CS) delivery, however, no increased risk of congenital abnormalities. These adverse outcomes are particularly prevalent for women with active IBD compared with those with quiescent disease. Conception should occur during disease remission to optimize maternal and fetal outcomes and reduce the risk of disease exacerbations during pregnancy. Pre-conception counseling is therefore pertinent to provide patient education, medication review for risk of teratogenicity and objective disease assessment. Most medications are safe during pregnancy and breastfeeding, with the exception of methotrexate, ciclosporin, allopurinol and tofacitinib. Delivery modality should be guided by obstetric factors in most cases; however, CS is recommended for women with active perianal disease and can be considered for women with inactive perianal disease or IPAA. In conclusion, most women with IBD have uncomplicated pregnancies. Active IBD is the predominant predictor of poor outcomes and disease exacerbations; therefore, maintenance of disease remission during and before pregnancy is crucial.
Introduction
The prevalence of inflammatory bowel disease (IBD) in Western countries is approximately 0.7%, with the peak age of diagnosis occurring during reproductive years.1,2 There is a bi-directional interaction between IBD and pregnancy, with IBD affecting fertility and pregnancy outcomes, and pregnancy having implications for IBD activity. Management of pregnancy in women with IBD is challenging, complicated by lack of data and heightened concerns about medication toxicity. This review article summarizes these important themes without emphasizing medication safety, which has been thoroughly covered recently. 3
Fertility
Many studies have reported fertility rates in women with IBD comparable with the general population, except for women with active disease or ileal-pouch–anal anastomosis (IPAA) surgery.4–8 Slightly lower fertility rates have been reported in some studies, particularly for Crohn’s disease (CD), without a proven reduction in fecundity (physiologic ability to produce live offspring).9,10 Many epidemiology studies have neglected to distinguish voluntary and involuntary infertility, creating confusion in the literature. A Scottish postal survey documented a significantly higher rate of voluntary infertility in women with IBD compared with the general population [CD: 30%; ulcerative colitis (UC): 25%; controls: 7%], with no increased prevalence of involuntary infertility (CD: 14%; UC: 15%; controls: 14%). 5 Studies report involuntary infertility rates of 3–14% in CD and 1.7–15% in UC, which are comparable with 2.5–14% in women without IBD.4–7 This is supported by data demonstrating similar levels of serum anti-Mullerian hormone (AMH) in women with IBD compared with the general population, indicating no reduction in ovarian reserve.11,12 However, AMH levels are significantly lower during active disease and inversely correlate with the Crohn’s Disease Activity Index, 13 supporting the observation that fertility rates are reduced in active disease.9,14 A nationwide Korean study found a 21% reduced live birth rate in women with moderate-to-severe IBD activity, while birth rates in women with quiescent or mildly active IBD were comparable with women without IBD. 14 IPAA surgery may also impair fertility, with infertility rates rising from 15 to 20% pre-IPAA to 48–63% post-IPAA.15,16 This is attributed to reproductive organ damage from deep pelvic dissection, formation of scar tissue and adhesions, and the increased prevalence of dyspareunia post-operatively.17,18 Medications used to treat IBD do not affect female fertility. 19
In addition to physiologic factors affecting fertility, voluntary childlessness (VC) occurs more frequently in patients with IBD (13–19%) compared with the general population (6%).7,14,20,21 There is significant geographic heterogeneity in VC, with rates highest in the Middle East (26%), followed by Asia (19%) and then Western countries (4%), inversely correlating with pregnancy-specific IBD knowledge. 22 VC is contributed to by poor pregnancy knowledge, misconceptions about infertility, and fear of maternal and fetal complications, most of which are unfounded.21,22
Fecundity in males with IBD is comparable with the general population, with reduced pregnancy rates identified by some studies likely attributed to VC.23–26 Although one study found marked reductions in sperm count and motility in men with CD (in absence of sulfasalazine), 27 a recent cohort study found no difference in sperm count, motility, vitality, or morphology in men with IBD compared with the general population. 24 However, the sulfapyridine moiety of sulfasalazine has been implicated in impairing sperm maturation, causing reduced sperm motility and sperm count, and an increased number of abnormal sperm forms.28,29 Sperm recovery takes 3 months after sulfasalazine cessation. 29 There has been one case report of reversible oligospermia associated with mesalazine; however, no other 5-aminosalicylic acid formulations have been implicated. 30 Methotrexate has also been implicated in causing reversible oligospermia and reduced sperm integrity, effects which resolve within 3 months of cessation.31–34
In summary, excluding those with active disease and certain surgical procedures, most women and men with IBD have fertility rates comparable with the general population. However, misconceptions about infertility are common in this patient population and contribute toward VC, a phenomenon that is significantly more prevalent than true infertility.
Assisted reproductive technology
Assisted reproductive technology (ART), including in vitro fertilization (IVF), is frequently required for women with IBD unable to conceive naturally. 35 Women with UC and prior IPAA surgery are 3.2-fold more likely to require IVF compared with women with medically managed UC, reflecting the reduced fertility conferred from IPAA surgery.36,37 A recent meta-analysis found 33% lower live birth rates after ART in women with CD compared with the general population, and 51% lower in women with previous CD-related surgery.38–40 Women with UC have comparable pregnancy and live birth rates after ART with women without IBD.38,40–42 The efficacy of ART is not reduced in women with IPAA; however, IPAA failure confers a 64% reduced live birth rate after ART.36–40,43 Women with IBD have no greater risk of adverse outcomes from ART compared with women in the general population; however, data are limited.39,41,44 Referral for fertility therapy should be considered earlier in women with IBD compared with the general population (Figure 1).

Fertility therapy referral pathway.
Heritability
Offspring born to parents with IBD have up to 10.4% lifetime risk of developing IBD, representing a 14-times greater risk than offspring of unaffected parents. 45 This risk increases to 33–36% when both parents are affected.46,47 Heritability is greater in CD than UC, with monozygotic twin concordance rates 20–58% in CD and 6–19% in UC.48–51 Genomic imprinting has been implicated in CD, with affected mothers having a significantly increased risk of transmission to their offspring than affected fathers, and mothers significantly more likely to transmit to daughters than to sons.45,52,53 This risk can be partially mitigated by breastfeeding, with breastfed infants having up to 30% reduced risk of developing IBD. 54
Effect of pregnancy on IBD activity
Periconception disease activity is predictive of disease activity during pregnancy.6,55–62 Patients with quiescent CD and UC have 14–22% and 26–35% chance of experiencing a flare during pregnancy, respectively, which is comparable with the risk of flare in non-pregnant women.6,55,57–61 However, 26–65% of CD and 33–79% of UC patients with active disease at conception will continue to experience active disease throughout gestation.6,55–58,60,61,63,64 A meta-analysis of 227 women with active UC at conception identified that at least 24% of women continued to have active disease during pregnancy, 45% experienced worsening of disease activity and at least 27% improved. 57 Of 93 women with active CD at conception, 33% experienced worsening of disease, 32% had ongoing disease activity and 34% improved during pregnancy. The risk of flare in patients with quiescent disease at conception (CD: 27%; UC: 34%) was no greater than the risk in non-pregnant women. 57 A more recent meta-analysis of 14 studies found that women with active IBD at conception had twice the risk of ongoing active disease during pregnancy compared with women in remission at conception [CD: 46% versus 23%; odds ratio (OR) 2.0; 95% confidence interval (95%CI) 1.2–3.4; UC: 55% versus 29%; OR 2.0; 95%CI 1.5–3.0]. 65 The risk of active disease during gestation is also increased in women who experienced disease flares during previous pregnancies, reaffirming the pertinence of maintaining disease remission. 62
Relapses of UC are more common in the first and second trimesters and post-partum period than the third trimester.57,61 A recent prospective study found a significantly greater rate of disease flares in women with UC during the post-partum period compared with non-pregnant UC controls [40% versus 19%; relative risk (RR) 6.22; 95%CI 2.05–79.3]. 61 Women with CD had no increased risk of post-partum disease flares in this study. Post-partum flares can be predicted by disease activity at term, occurring in 53% of women with active IBD compared with 13% of women with quiescent IBD in one study. 58 In the long term, pregnancy has been associated with fewer IBD relapses, which may be attributed to intensified efforts to optimize disease control in pregnant women, smoking cessation during pregnancy, or an immunological effect of the pregnancy.66–68
The reasons underlying changes in disease activity during pregnancy remain elusive. In the 1960s, when use of pharmacotherapy during pregnancy was largely discouraged, 58 rising 17-hydroxycorticosteroid levels were thought to account for the declining proportion of patients with active disease throughout gestation. 69 Post-partum disease flares were similarly accounted for by the rapid fall in plasma 17-hydroxycorticosteroid after delivery. 69 More recently, smoking cessation during pregnancy was shown to significantly reduce disease activity in smokers with CD. 70 There is increasing evidence that hormonal changes during pregnancy may also affect autoimmune and inflammatory conditions, including IBD.71–73 Estrogen and progesterone are both thought to strengthen the intestinal epithelial barrier and reduce ‘leaky gut.’ These hormones progressively increase throughout pregnancy, potentially reducing bacterial translocation and IBD activity towards the end of gestation.72,73
In summary, disease activity at the time of conception is a useful predictor of disease activity for the remainder of gestation. Ensuring disease remission preconception is an effective strategy to reduce the risk of IBD flares during pregnancy and adverse maternofetal outcomes.
Effect of IBD on the pregnancy and child
Multiple studies have shown that women with IBD have an increased risk of preterm delivery, low birth weight (LBW), small-for-gestational-age (SGA) infants and Caesarean section (CS) delivery.57,64,74–85 Most studies have demonstrated this in both disease subtypes, while some found these risks to be greater for CD than UC,14,74,85,86 and a few found no increased risk of adverse outcomes compared with the general population.55,56,87
Preterm delivery occurs in 9–18% of women with IBD, compared with 5–9% of women in the general population.83,88 The risk of preterm delivery is increased with both maternal IBD (OR 2.15; 95%CI 1.36–3.39) and paternal IBD (OR 3.02; 95%CI: 1.82–5.01), and further increased in parents who also have a first-degree relative with IBD (OR 4.29; 95%CI 1.59–11.63). 74 The proposed mechanism for this relates to a shared genetic susceptibility between IBD and preterm birth. 74 SGA (birthweight <10th centile for gestational age) has been detected in 5.0–6.7% of infants born to women with IBD, compared with 0.9–3.8% in the general population.64,82,83 The risk of CS delivery is also increased in women with IBD (29–33%) compared with the general population (16–22%), for both elective and emergency indications.64,68,76,78,81–83,87–90 Limited data suggest that compared with women without IBD, women with IBD also have an increased risk of spontaneous abortions (13% versus 6.5%), 68 gestational diabetes (7% versus 2%),90,91 neonatal intensive care unit (NICU) admissions (10% versus 4%), 64 intrauterine growth restriction (IUGR) in women with CD (3% versus 1%), 14 and neonatal death in women with UC (0.4% versus 0.2%). 83 However, multiple other studies have demonstrated no increased risk of early pregnancy loss, perinatal mortality, NICU admissions, low Apgar scores, or placental diseases.82,83,87,88,90 The risk of gestational diabetes is 4.5-fold greater in women with IBD taking corticosteroids compared with the general population, and numerically but not statistically increased in women with IBD not taking corticosteroids (p = 0.09). 91
Active disease during pregnancy further increases the risk of preterm delivery, LBW, IUGR, spontaneous abortion and CS delivery.5,6,14,59,60,80,83,85,87,92,93 A recent Korean study of 2058 women with IBD found that women with quiescent disease had comparable risks of adverse outcomes with women without IBD, while risks were significantly greater for women with moderate-to-severe disease activity. 14 Similarly, another study found that women with active IBD had significantly greater rates of preterm delivery (27% versus 8%) and babies with LBW (32% versus 3%) compared with women with inactive IBD. 87 Active disease has also been implicated in increasing the risk of childhood illnesses; 79 however, no increased risk of congenital abnormalities has been detected.92,93
Multiple large studies, including two Scandinavian population-based studies,82,83 cohort and case-control studies,55,78,79,88,89 have not detected any increased risk of congenital abnormalities in offspring of women with IBD. A handful of studies did find an increased risk of congenital abnormalities in offspring of women with IBD (3.4–7.9%) compared with controls (0–2%); however, this must be interpreted with caution due to methodological flaws, including small sample sizes and lack of data on medication exposure.56,86,94,95 These studies found limb, urological, and neurological defects to be most prominent,56,94,95 and UC to confer a greater risk than CD.76,86
In summary, women with IBD have an increased risk of preterm delivery, babies with LBW, SGA, and CS delivery, particularly in the context of active disease. Fortunately, no increased risk of congenital abnormalities is suggested by data.
Management of IBD during pregnancy
Preconception management
Preconception counseling is an important component of IBD management (Figure 2). Gastroenterologists should proactively initiate preconception counseling conversations with all men and women of childbearing age. In addition to providing standard antenatal education, counseling should address patient misconceptions about infertility, medication safety, maternofetal complications, and IBD heritability, which are common concerns in IBD patients and contribute toward VC.96–99 This is associated with improved medication adherence, folic acid supplementation, smoking cessation, reduced disease flares during pregnancy, and improved neonatal outcomes.100–102 In women actively contemplating pregnancy, medication review for risk of teratogenicity and objective assessment of disease activity should be performed prior to conception, aiming to confirm disease remission to optimize pregnancy outcomes. Patients should be encouraged to join IBD support organizations, with membership predictive of significantly higher pregnancy-specific IBD knowledge.22,103,104
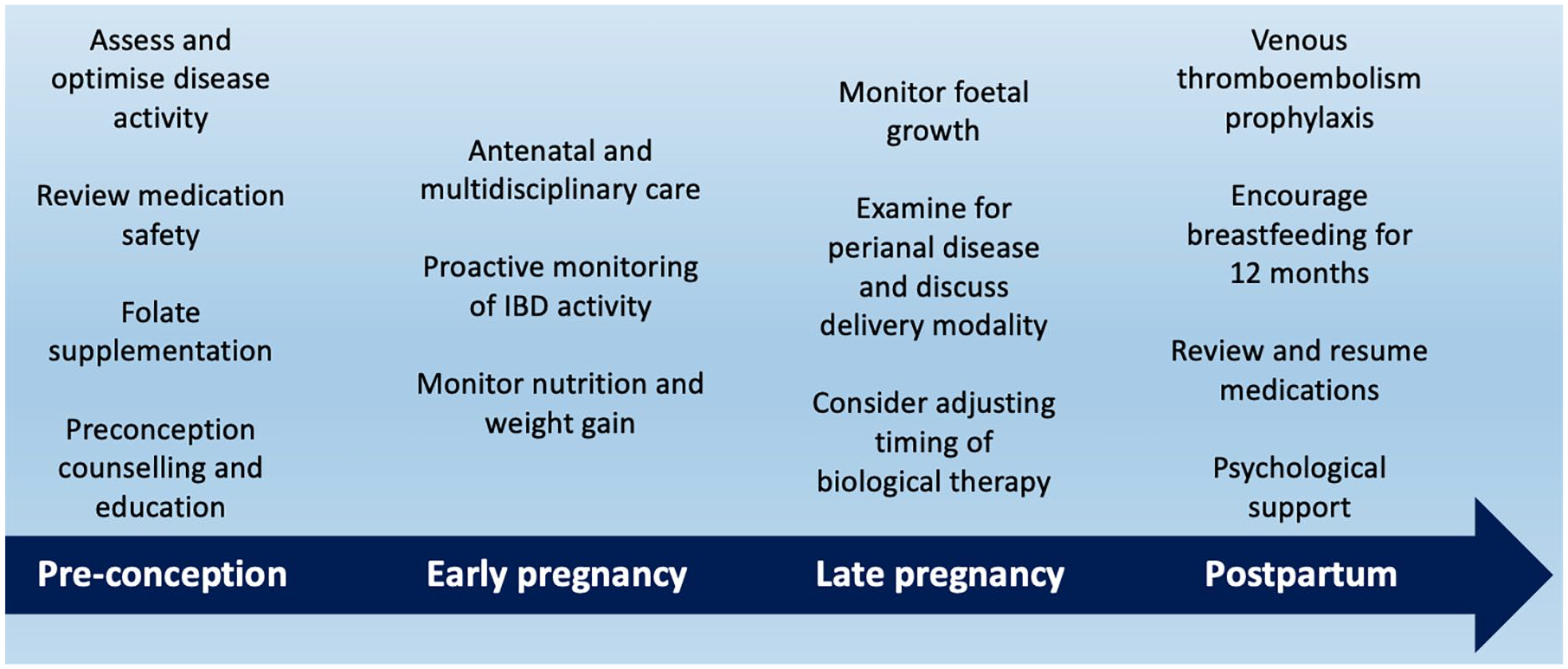
Considerations for pregnancy management in women with inflammatory bowel disease.
Folic acid can reduce the incidence of neural tube defects by 72%, without adverse neonatal effects; therefore, supplementation should be provided to all pregnant women and women contemplating conception.105,106 The recommended dose is 400–800 µg/day in combination with adequate dietary intake, commencing at least 1 month prior to conception and continuing at least throughout the first trimester. 105 Higher doses of 2 mg/day are recommended for patients taking sulfasalazine, which inhibits dihydrofolate reductase, thus impairing absorption and metabolism of folic acid.107–111 Other congenital defects associated with dihydrofolate reductase inhibitors, such as cardiovascular and oral cleft deformities, may also be mitigated by folic acid supplementation. 107
Management during pregnancy
A care pathway for pregnant women with IBD is outlined in Figure 3. All pregnant IBD patients should receive standard antenatal care, including monitoring of nutritional intake, weight gain, and fetal growth indices. Women with IBD are at risk of inadequate weight gain during pregnancy, particularly with active IBD. 112 This is associated with worse pregnancy outcomes, including twice the risk of SGA, compared with women without IBD also having inadequate weight gain. 112 Women should be followed up regularly for disease monitoring via clinical examination and biochemical markers, including fecal calprotectin, and to encourage medication adherence. Fecal calprotectin is not affected by the physiologic changes of pregnancy and significantly correlates with disease activity during all stages of gestation.113,114 Adherence to medications often reduces during pregnancy, particularly for 5-aminosalicylates, followed by immunomodulators and biologic agents. 115 This may increase the risk of disease flares and subsequently worsen pregnancy outcomes.115–118 Non-adherence is attributed to patient misconceptions and concerns about teratogenicity,98,115,119 and is more frequent in women not linked to IBD support organizations.98,119 Gastroenterologists should explain to patients about medication safety, including evidence from human studies and areas where data are lacking. The importance of maintaining disease control to optimize pregnancy outcomes should be emphasized. Reassuringly, a recent large prospective study found no increased risk of adverse maternal or fetal outcomes with biologic, thiopurine, or combination therapy exposure during pregnancy compared with unexposed women with IBD. 120 Evidence for medication safety during pregnancy and breastfeeding is summarized in Table 1. 3
Summary of evidence for medication use in pregnancy and breastfeeding.
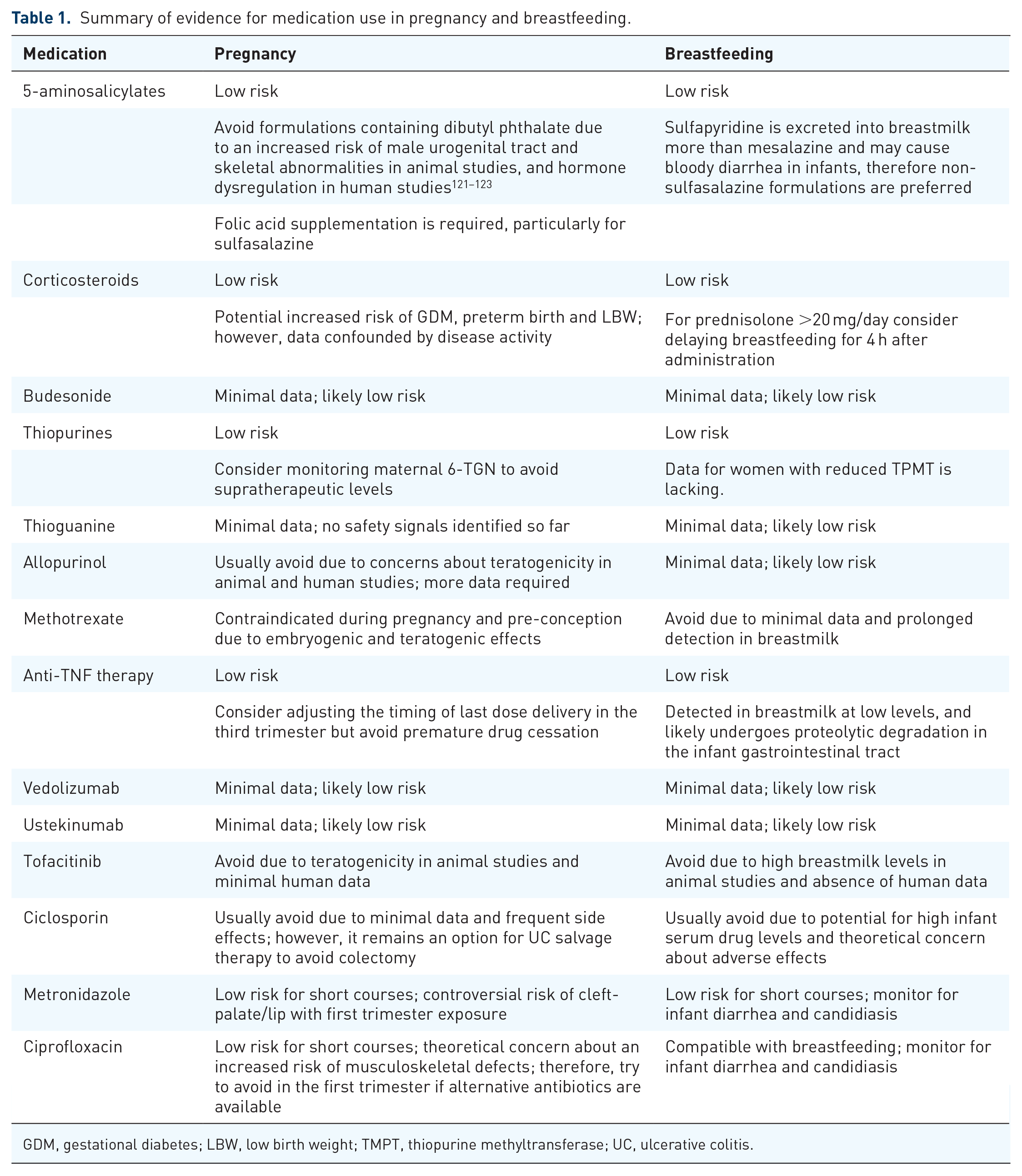
GDM, gestational diabetes; LBW, low birth weight; TMPT, thiopurine methyltransferase; UC, ulcerative colitis.
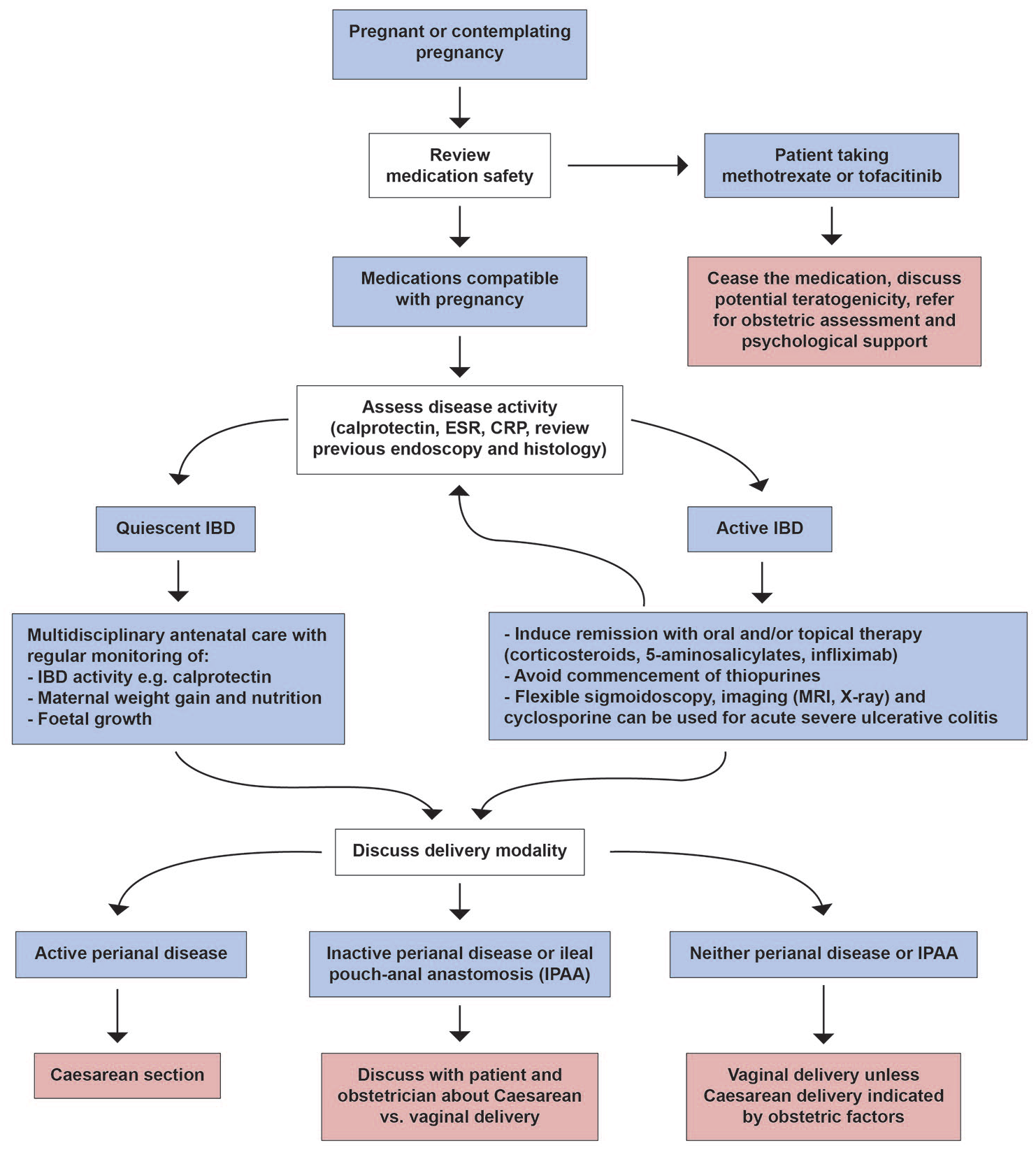
Care pathway for pregnant women with inflammatory bowel disease.
Delivery
Discussions with patients about mode of delivery should commence early during pregnancy and should address the risks and benefits to the mother and child of natural vaginal delivery (NVD) and CS. Most women with IBD can have an NVD, with the mode of delivery being governed by obstetric factors rather than IBD factors. Relevant obstetric factors include multiparity, large for gestational age, breech presentation, and prolonged labor. 124 NVD is considered to incur fewer maternal complications than CS in the general population, 125 and women with IBD may also benefit from avoiding additional abdominal surgery. Although not confirmed by a recent meta-analysis, 126 some data also suggest that children born via CS may have an increased risk of subsequent IBD diagnosis, possibly reflecting the influence of delivery modality on infant-gut microbiome due to exposure to maternal vaginal flora. 127 Nevertheless, CS is generally advised for women with active perianal disease (including fistulae, abscesses, and sphincter damage), and can be considered for women with inactive perianal disease or IPAA.19,128 Referral to high-risk pregnancy clinics should be considered for women with active disease or previous surgery. 129
There are concerns that an NVD, particularly in combination with an episiotomy, performed in the setting of active perianal disease, may worsen the perianal disease or provoke fistula formation. 130 Several studies have found that women with active perianal CD at the time of delivery have a significantly greater risk (up to 100%) of perianal disease flares after NVD.124,131 However, data on patients with quiescent perianal disease are lacking.131–134 Some studies found that NVD posed no greater risk of perianal disease flares or de novo perianal disease development compared with CS or perianal disease in non-pregnant women.132,134 A review of 18 studies found non-significantly increased rates of new [mean 6.5% (interquartile range (IQR) 0.0–19.7) versus 3.0% (IQR 1.15–11.50)] and recurrent [mean 45.0% (IQR 0–58.0) versus 13.5% (IQR 3.2–32.7)] perianal disease with NVD compared with CS in women with CD. 124 Another study found a threefold greater incidence of perianal fistulae after childbirth in women with versus without perianal CD; however, there was no greater risk with NVD versus CS or with episiotomy. 133 Women with perianal CD also have significantly greater rates of fourth-degree tears after NVD compared with women with CD without perianal disease (12.3% versus 1.4%). 135
Delivery decisions in women with IPAA are complex, and women frequently receive conflicting advice from various specialists, 136 which partly reflects the inconclusive data in the literature. During pregnancy, women with IPAA may experience an increased stool frequency which returns to baseline 3 months post-partum, irrespective of delivery modality. 137 Multiple studies have not detected any deterioration in pouch function with NVD compared with CS, including stool frequency and incontinence.137–142 However, some data suggest that women with an IPAA are significantly more likely to sustain anal-sphincter damage (50% versus 13%) and functional impairment from NVD versus CS, 143 particularly if it was a complicated NVD.124,144 This can worsen fecal incontinence, which women with IPAA are already prone to. 145 Study results may be confounded by selection bias, with higher-risk patients prioritized for CS rather than NVD, as well as short follow-up durations precluding detection of long-term outcomes. Additionally, pouch function can change over time, regardless of childbirth, including increased stool frequency and reduced sensation for gas versus liquid, which may also influence study findings. 139
There are minimal data on delivery modality in women without an IPAA or perianal CD. Although NVD can cause anal-sphincter damage, a study including >2000 women with IBD found no increased risk of subsequent fecal incontinence with NVD compared with CS. 146 There is mixed evidence on whether women with IBD have a greater risk of fecal incontinence after NVD compared with NVD in women without IBD.147,148 In the absence of convincing data, delivery decisions for women without a clear indication for CS should be based on obstetric factors.
Post-partum management
The maternal priorities in the post-partum period include commencing breastfeeding, venous thromboembolism prophylaxis, encouraging medication adherence, maintaining control of IBD, and mental health monitoring. Most medications are safe for use during breastfeeding, with the exception of methotrexate, tofacitinib, ciclosporin, and allopurinol (Table 1).3,19,108,149–152 Biologic agents can be recommenced 24 h after NVD or 48 h after CS, in the absence of infection. 153 Many women with IBD have concerns about breastfeeding, particularly regarding medication safety for the infant, which leads to unnecessary avoidance of breastfeeding. 154 In addition to reducing the incidence of respiratory and gastrointestinal infections, 155 asthma, 156 and type 1 diabetes, 157 breastfeeding reduces the development of early-onset IBD by up to 30%. 54 This risk reduction is dose dependent, with 12 months of breastfeeding conferring greater protection than 3 months or 6 months.158,159 When adjusting for medication cessation, breastfeeding does not increase the risk of post-partum IBD flares.154,160 Therefore, women with IBD should be encouraged to breastfeed for a minimum of 6–12 months where possible, in accordance with global guidelines.108,161–163
Vaccinations
Non-live vaccines, including inactivated, polysaccharide, and toxoid vaccines, do not confer an increased risk of reactivation and therefore can be safely given to infants exposed to either biologic or non-biologic therapy in utero (Table 2).153,164–167 The infant immune system is thought to adequately respond to these vaccines, with appropriate serum antibody responses documented in biologic-exposed infants receiving pneumococcus, Hemophilus influenzae B and tetanus toxoid vaccinations.164,168,169
Inactivated and live vaccinations.

Live vaccinations can also be given on schedule to infants exposed in utero to non-biologic therapy or certolizumab, which has minimal transplacental transfer.153,165,166,170 However, infants exposed to anti-tumor necrosis factor (anti-TNF) agents other than certolizumab in utero are advised to delay live vaccinations for 6–12 months, or until serum drug levels are undetectable, to reduce the risk of active viral replication or vaccine failure.153,171 This concern arises in part due to a fatal case of disseminated Bacillus Calmette-Guérin (BCG) infection after a BCG vaccination was administered at 3 months of age to a child exposed to 10 mg/kg infliximab monotherapy in utero. 172 There have been other reports of over 100 anti-TNF-exposed infants receiving the BCG vaccination and over 400 receiving the Measles–Mumps–Rubella vaccination without adverse effects;167,173 however, significant concern regarding live vaccinations prevails. The rotavirus vaccine is not approved for administration beyond 12 months of age, primarily due to concerns about intussusception; therefore, it should be forgone rather than delayed in anti-TNF-exposed infants.174,175 Of 40 infants exposed to biologic therapy in utero who received the rotavirus vaccination on schedule, 17.5% experienced mild reactions (fever or diarrhea); however, no serious adverse events were reported. 164
There are limited data regarding vaccination safety in infants exposed to the newer biologic agents; therefore, they are frequently managed similarly to anti-TNF agents by delaying live vaccinations for 6–12 months.108,176 The gut specificity of vedolizumab means vaccinations may theoretically be safer than with anti-TNF agents, which confer more systemic immunosuppression; however, the efficacy of oral vaccines is likely reduced. 177 Reassuringly, no adverse outcomes were reported in 14 infants exposed to vedolizumab in utero who received the rotavirus vaccine and 3 who received the BCG vaccine. 178 The literature on infants exposed to ustekinumab in utero is limited to case reports, which have not identified any adverse vaccination reactions, including from the BCG vaccine.179,180 As tofacitinib is contraindicated during pregnancy, no data exist on childhood vaccination outcomes in tofacitinib-exposed infants.
Conclusion
Active IBD is the main predictor of poor pregnancy outcomes, including impaired fertility, adverse neonatal and maternal outcomes, and disease exacerbations. Management of pregnancy in women with IBD can be challenging, and mismanagement can put women at risk of disease flares and adverse outcomes. Many patients also have concerns and misperceptions about fertility and pregnancy, topics which should be proactively addressed during preconception counseling and throughout gestation to optimize medication adherence and pregnancy outcomes.
