Abstract
Introduction:
Patients treated with biologics for inflammatory bowel disease (IBD) have an increased risk of severe infections. Real-life vaccination coverage in this population remains low despite international vaccination guidelines. The aim of this study was to evaluate the impact of a dedicated vaccination visit on vaccination coverage.
Methods:
A dedicated vaccination visit was offered to all patients admitted for an infusion of a biologic in a tertiary IBD center during a 4-week period. At baseline, vaccination status was collected. Patients received specific information on recommended vaccinations. Perceived utility of both vaccination and the dedicated visit were assessed by visual analogue scale (VAS). Vaccination coverage was reassessed 6 months later by phone call.
Results:
Among the 207 patients analyzed (1 patient declined), rates of vaccination at baseline and 6 months later against diphtheria were 52.7% and 68.6% (p < 0.001), tetanus 55.1% and 70% (p < 0.001), poliomyelitis 51.7% and 68.6% (p < 0.001), pertussis 33.3% and 51.2% (p < 0.001), hepatitis B virus (HBV) 61.4% and 66.7% (p < 0.01), pneumococcus 15.5% and 42.0% (p < 0.001), influenza 29.5% and 36.2% (p < 0.01), and meningococcus C 11.6% and 13.0% (p = 0.083), respectively. A quarter of the patients declined at least one recommended vaccination after the visit. The main reason for this refusal was distrust toward one or more vaccines.
Conclusion:
A single visit dedicated to vaccination significantly increases rates of vaccination in patients with IBD treated with biologics.
Introduction
The widespread use of immunosuppressants and biologics has dramatically improved the course of inflammatory bowel disease (IBD). 1 However, these therapies expose patients to side effects, notably an increased risk of severe and opportunistic infections.2,3 Infection-related hospitalizations are associated with increased mortality in patients with IBD. 4 This risk is limited by vaccination for preventable diseases and respect of contraindications.5–7 The European Crohn’s and Colitis Organisation (ECCO) published vaccination guidelines in 2021. 8 As recommended in the general population, patients with IBD have to be vaccinated against tetanus, diphtheria, poliomyelitis, pertussis, and meningococcus C. When receiving immunomodulators they should also be vaccinated against the hepatitis B virus, varicella zoster virus (with vaccination completed at least 3 weeks prior to initiation of immunomodulators), influenza (annual vaccination), and pneumococcus [13-valent pneumococcal conjugate vaccine (PCV13) followed by 23-valent pneumococcal polysaccharide vaccine (PPSV23) 8 weeks later, then PPSV23 5 years after the first dose].
Despite a well-known increased infectious risk and clear guidelines about vaccination, real-life studies have reported low vaccination rates in IBD populations.9–12 The main identified patients’ barriers are concerns about vaccine side effects and forgetfulness. 13 Poor involvement of gastroenterologists in prevention leads to lack of information for patients, which maintains patients’ distrust.14–16 Poor knowledge about vaccination and lack of time have been shown to represent the most relevant limits for gastroenterologists’ involvement in this process.17,18 However, patient surveys found that incentive from their physician plays a key role in their motivation to get vaccinated. 19
The present study aimed to evaluate the impact of a single medical visit dedicated to vaccination on vaccination coverage of patients with IBD receiving intravenous biologics. Main patient-reported barriers for vaccination and their perceived utility of the visit and vaccination were also assessed.
Methods
Study design
This prospective interventional study was conducted in the IBD center of the Bordeaux University Hospital, a tertiary care medical center. Patients were informed by a phone call 2 days before their visit that a medical visit dedicated to vaccination would be offered on the day of infusion. In the event of acceptance, they were asked to bring their vaccination records to the visit. Vaccinations were registered at baseline and 6 months later.
Ethical considerations
The study was conducted in accordance with the principles of good clinical practice and the Declaration of Helsinki at all times. The use of all the data collected is in accordance with the modified French Data Protection Act of 06/01/1998 and the General Data Protection Regulation (GDPR). Written, informed consent was obtained from each patient included in the study. The study design was approved by the local Institutional Review Board, and the intervention considered at minimal risk to human subjects (Reference CE-GP-2021-26). The reporting of this study conforms to the STROBE statement. 20 The STROBE checklist is available in supplementary material.
Study population
All IBD adults hospitalized in the IBD infusion day unit between 24 September and 19 October 2018 were consecutively screened for participation when they were admitted to the outpatient clinic. Diagnosis of Crohn’s disease, ulcerative colitis, and indeterminate colitis were in accordance with the latest ECCO guidelines. 21 Exclusion criteria were unwillingness or inability to give informed consent.
Dedicated vaccination visit
Dedicated vaccination visits were carried out by a general practitioner fellow (L.G.). He had received training in vaccination, including safety data, during his med school education. Concerning IBD-specific matters, he was trained by the IBD specialists of our unit. The scheduled duration of the visit was 30 minutes. Each visit was individual and always began with the evaluation of patients’ perceived utility of vaccination by visual analogue scale (VAS – from 0 to 10) and their knowledge of their own vaccination coverage. Their documents recording previous vaccinations were checked: status was recorded on the basis of certificates of vaccination if available or of any other document related to their vaccination coverage; when missing, the information was searched in the patient’s medical record in our center or established through declaration. When vaccination status was uncertain or unknown, patients were considered as non-vaccinated. Information on vaccine types was given: contraindications to live-attenuated vaccines were explained and medical guidelines on recombinant vaccines for patients with IBD were given, together with a short explanation on each pathogen (transmission mode, symptoms, and potential complications). Patients were invited to ask questions about vaccination. For patients who wanted to update their vaccination status, vaccinations were offered the same day or by a prescription (allowing the patients to be vaccinated by their GP, nurse, or pharmacist). The reasons behind refusal for those patients who still refused vaccination after the visit were recorded. Finally, patients were asked to evaluate their perceived utility of the visit. A summary letter with vaccination guidelines and safety data intended for their general practitioner was given to the patients at the end of the visit.
All the patients who had given their informed consent were called 6 months later to collect their new vaccination status. In absence of a response after five calls, patients were considered as lost to follow-up and non-responders (no vaccination update). All the patients that had the inclusion visit were included in the final analysis.
Data collection
At baseline, the following characteristics of patients were recorded: age, gender, type of IBD, current and previous IBD therapies (immunosuppressants, biologics), and comorbidities according to the Charlson classification. 22 Vaccination against the following diseases were collected: diphtheria, tetanus, poliomyelitis, pertussis, hepatitis B virus, meningococcus C, pneumococcus, influenza, measles, mumps, rubella, and yellow fever. Adequate vaccination status corresponded to patients who had received all recommended vaccinations based on French and ECCO guidelines.23,24 The vaccination refusal reasons were collected during the visit. Patients’ perceived utility of the dedicated visit and utility of vaccination were evaluated with a visual scale from 0 (useless) to 10 (essential) (Supplementary Figures 1 and 2).
Primary and secondary objectives
The primary objective was to compare rates of vaccination against diphtheria, tetanus, poliomyelitis, pertussis, hepatitis B virus, meningococcus C, pneumococcus, and influenza at baseline and at 6 months. Secondary objectives were to evaluate the patients’ perceived utility of vaccination and the dedicated visit, to collect vaccination refusal reasons and to identify factors associated with updating at least one vaccination at 6 months.
Statistical analysis
Categorical variables were indicated as proportions and percentages and comparison was drawn by a McNemar’s test. Continuous variables were reported as medians and ranges. Two-sided statistical tests were used for all analyses and a p value <0.05 was considered significant. The identification of factors associated with updating at least one vaccination was performed by bivariate Cox proportional hazards models. Parameters with a p-value less than 0.1 in bivariate analyses were introduced into a logistic regression model. Results are expressed as odds ratio (OR) with 95% confidence intervals (CIs). The statistical analyses were performed with the PSPP software (version 1.4.1).
Results
Patient characteristics
Among the 208 IBD adults admitted in the infusion day unit during the study period, 207 patients agreed to have a dedicated vaccination visit, including 43 who were considered as lost to follow-up (including eight refusing the call at 6 months). The flowchart is presented in Figure 1. Their main characteristics are presented in Table 1. To summarize, the median age of the patients was 40 years, and 105 were women (50.7%). The most prescribed treatment was infliximab (187 patients; 87.4%). Sixty-five patients (36.2%) were treated with combotherapy (55 with thiopurines and 10 with methotrexate). The median duration of the current treatment was 4 years [interquartile range (IQR): 2–7].
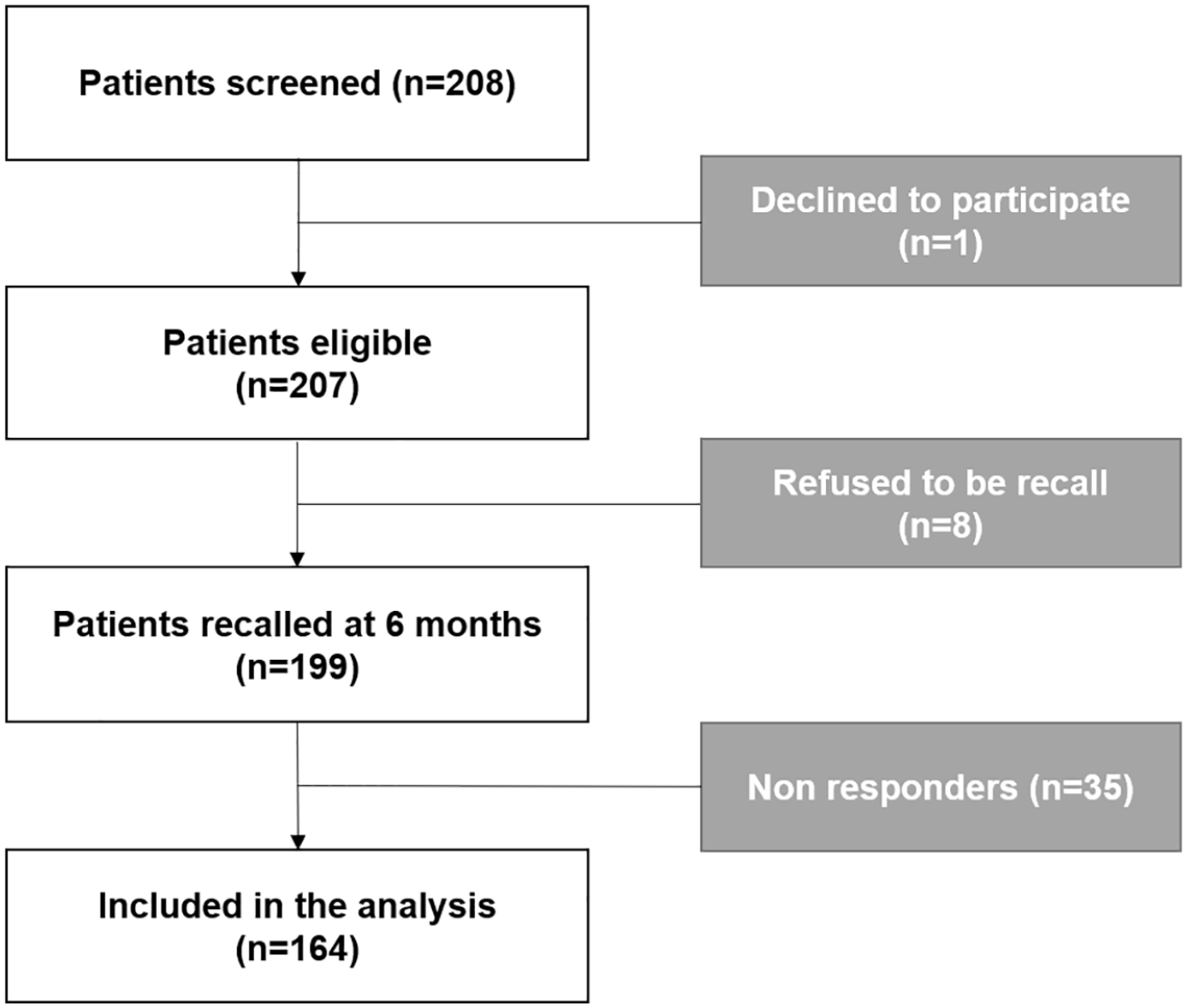
Flowchart.
Patients’ characteristics.

IBD, inflammatory bowel diseases; IQR, interquartile range.
Dedicated vaccination visit
Seventy-three (35.3%) patients knew their vaccination status at baseline and 108 brought their vaccination certificate (52.2%). Rates of live-attenuated and recombinant vaccinations at baseline are presented in Table 2 and Figure 2, respectively. Only six patients had an adequate vaccination status at the baseline against diphtheria, tetanus, poliomyelitis, pertussis, hepatitis B virus (HBV), pneumococcus, and influenza. When asked about the perceived utility of vaccination on VAS, 37 (17.9%) did not answer; the mean value was 7.42 (1–10).
Live-attenuated vaccination coverage.


Vaccination coverage evolution at 6 months.
At the end of the visit, 42 patients (20.2%) still declined at least one recommended vaccination. Only five (2.4%) of them declined all vaccines. The main reasons given to decline to be vaccinated were as follows: ‘these vaccinations aren’t mandatory’ (six patients); ‘I never get sick’ (two patients); ‘I have needle phobia’ (two patients); and ‘I don’t see any benefit of prevention’ (two patients).
The influenza and hepatitis B vaccines were refused by 31 (15%) and 19 (9.2%) patients, respectively. The most cited reasons to refuse vaccination against influenza were ‘a fear to get sick after vaccination’ (12 patients) and ‘the use of protective measures or homeopathic vaccination’ (6 patients). Regarding hepatitis B, seven patients feared contracting multiple sclerosis with the vaccination and five did not feel at risk of sexually transmitted infections.
The mean given value to the perceived utility of the dedicated vaccination visit was 8.7 (1–10). Only six (2.9%) patients preferred not to answer.
Vaccination coverage at 6 months
A significant increase of all vaccination rates at 6 months was observed, except for meningococcus C. Results are presented in Figure 2. Among unvaccinated patients against diphtheria, 33 (33.7%) were vaccinated at 6 months. Among unvaccinated patients against tetanus, 32 (34.4%) were vaccinated at 6 months. Among unvaccinated patients against poliomyelitis, 35 (35%) were vaccinated at 6 months. Among unvaccinated patients against pertussis, 37 (26.8%) were vaccinated at 6 months. Among unvaccinated patients against the hepatitis B virus, 11 (13.8%) were vaccinated at 6 months. Among unvaccinated patients against pneumococcus, 55 (31.4%) were vaccinated at 6 months. Among unvaccinated patients against influenza, 14 (7.5%) were vaccinated at 6 months. Among unvaccinated patients against meningococcus C, three (1.6%) were vaccinated at 6 months.
Two patients had only received partial vaccination against pneumococcus due to an unavailability of the 23-valent pneumococcal vaccine. No post-vaccinal reaction was observed. The non-vaccination reasons at 6 months of patients who had not expressed their opposition to update their vaccination status at the initial visit were collected: the main reason was either being too busy or an oversight in 37.7% (46/122 patients); two (1.2%) patients declared that their general practitioner was unwilling to vaccinate them under immunosuppressants.
Eighty-eight patients had updated at least one vaccination at 6 months (42.5%). In univariate analysis, the following factors were associated with updating at least one vaccination: a previous history of intestinal resection, a previous treatment with vedolizumab or methotrexate, a mean disease duration above 6 years, having brought a vaccination certificate to the visit, and a perceived utility of vaccination of 10. Skin associated manifestations were negatively associated with updating vaccination. A mean disease duration of more than 6 years and having brought a vaccination certificate to the initial visit were the only two factors significantly associated with multivariate analysis (Table 3).
Factors associated with updating at least one vaccination.

CI, confidence interval; OR, odds ratio.
Discussion
In this single-center interventional study, we found that a single dedicated visit increases vaccination rates against nearly all agents. This study also confirms, in real life, the poor vaccination coverage of IBD patients receiving immunomodulators. Our results show that rates of vaccination against diphtheria, tetanus, poliomyelitis, and pertussis are low, even if mandatory in France. 24 The present results are in accordance with the 50.5% rate of vaccination coverage against diphtheria, tetanus, and poliomyelitis in the French adult general population. 25 Our results for influenza vaccination coverage (36.2% at the end of the study) are slightly better than the usual rate observed in France for patients at risk (between 16% and 29% depending on conditions in 2015/2016). 26 The improvement of those low rates illustrates that patients lack information about vaccination even if they are treated with biologics for several years, which seems to justify the need for educational approaches (also confirmed by the high perceived utility of vaccination). Other encouraging strategies were tested to increase vaccination coverage of patients with IBD: 27 Reich et al. 28 have reported a significant increase of influenza and pneumococcal vaccinations after giving a written educational form to patients; Parker et al. 29 underlined the interest of a simple questionnaire allowing an update of vaccines; Christensen et al. 14 have shown that systematic information (oral and written) to health care professionals improves patients’ adherence to vaccination guidelines; Fleurier et al. 30 obtained similar results for children with IBD after giving a written educational form to them, their parents and general practitioner; and finally an ambitious educational program strategy by an IBD nurse has also shown benefits. 31 All these results thus suggest the need for educational interventions to consider improving vaccination coverage and the importance of the involvement of caregivers in this process.
However, a quarter of the patients still refused at least one recommended vaccination after the visit. We hypothesize that adherence might increase if regular information about the importance of vaccination is given by all the health care providers of the patient (nurse, gastroenterologist, and general practitioner). As recommended by the recently published ECCO guidelines, this strategy should start before initiation of treatment. 8 To give a summary letter with vaccination guidelines could be a first step, as we proposed in this work.
Our results for vaccination against Hepatitis B virus are low in the context of biological therapy (61.4% at inclusion). There are multiple explanations for this result. First, the result might be underestimated as it was partially based on a declarative self-questionnaire. Indeed, we did not have the serological results for the whole cohort as some of the patients had started their biological therapy in other medical centers. The second explanation resides in the 9.2% of patients who still refused to be vaccinated against HBV despite medical advice and recommendations. Finally, this result is in accordance with previously published data: 52% in the pediatric study by Fleurier et al., 30 45% in the Danish cohort reported by Christensen et al., 14 and 53% of the Belgian cohort described by Coenen et al. 31
Our study has several limitations. First, declarative self-questionnaires are exposed to response bias. It explains that the number of patients updating their vaccine against tetanus, diphtheria, and pertussis may differ while there is only one available compound containing all these vaccines. We have tried to minimize it using medical records and certificates of vaccination if they were available. Second, we had no control group of patients receiving no educational visit. However, we considered the odds of increased vaccination coverage without intervention as very low as our patients were diagnosed many years ago. Third, our results are poor for meningococcus C and do not include data on papilloma virus, measles, mumps, rubella, yellow fever, and varicella zoster vaccinations. Coverage evolution of live-attenuated vaccines (Measles, Mumps, Rubella, Yellow fever, Varicella Zoster) was not studied as their use is contraindicated under immunosuppressive treatment. Vaccination against papilloma virus is offered in France to boys and girls aged from 11 to 14 years old (with a possibility to get vaccinated from 15 to 19 if not) and until 26 years old for men having sex with men. 32 It represents six patients in our cohort. Regarding meningococcus C vaccination in France, administering a single dose is recommended at 12–24 months of life (with a possibility to get vaccinated until 24 years old if it was not done before). That explains in part the weak increase of meningococcus C vaccination coverage that we found.
We acknowledge that the generalization of our results might be limited by the evaluation of a wide range of vaccines. The reasons behind vaccination refusal may vary depending on the type of vaccine (single versus multiple doses, yearly or 10-yearly booster). However, there are common brakes for vaccination which can be partially but efficiently lifted with a single dedicated visit. Finally, our study only lasted 6 months. Despite this short follow-up, we proved that a single intervention has a rapid effect on vaccination coverage.
A survey by the American College of Gastroenterology found that most gastroenterologists would prefer that the responsibility of vaccination of patients with IBD be borne by general practitioners. 17 However, these results contrast with a recent survey of non-IBD expert gastroenterologists and general practitioners (with less than 10% of patients with IBD) 33 showing that the majority of them agreed that both should be responsible for prescribing; however, only 30% of primary care physicians felt comfortable with the topic of vaccination of patients with IBD, in another published survey. 34 This emphasizes the key role that both gastroenterologists and practitioners should play in their patients’ vaccination but also that therapeutic education and involvement of paramedical staff – as IBD nurses – can become levers to improve vaccination coverage. In order to reach this goal, IBD experts have a key role to play by disseminating the vaccination guidelines to their colleagues (gastroenterologists and general practitioners), paramedical staff, students, and patients.
To conclude, our study was conducted before the COVID-19 pandemic and it would be interesting in the future to assess if vaccination adherence has changed in this cohort of patients since this unprecedented health episode. Concerning SARS-Cov-2 vaccination, recent guidelines from the International Organization for the Study of Inflammatory Bowel Disease (IOIBD) stated that every patient with IBD should be vaccinated irrespective of their treatment status (absence, immunosuppressants, biologics, induction/maintenance). 35 Data from an international survey showed that COVID-19 vaccine acceptance was high with 71.5% of patients willing to be vaccinated among patients with IBD if it were proven safe and effective. 36 This potential vaccination coverage is similar to our vaccination rates against tetanus, diphtheria, poliomyelitis, and hepatitis B. Yet, reasons of hesitancy may vary. Indeed, an American and an Italian survey showed that the main concerns of patients were safety and a fear of side effects.37,38
Conclusion
Real-life vaccination coverage of patients with IBD under biotherapy is low despite clear guidelines. The main patient-reported reasons for poor vaccination adherence are fear of side effects and forgetfulness. A single 30-minute medical visit dedicated to vaccination can effectively increase vaccination rates.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221082879 – Supplemental material for A single dedicated vaccination visit improves vaccination coverage of patients with inflammatory bowel disease
Supplemental material, sj-docx-1-tag-10.1177_17562848221082879 for A single dedicated vaccination visit improves vaccination coverage of patients with inflammatory bowel disease by Guillaume Le Cosquer, Lionel Grangeon, Pauline Rivière, Arthur Berger, Frank Zerbib, David Laharie and Florian Poullenot in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-1-tag-10.1177_17562848221082879 – Supplemental material for A single dedicated vaccination visit improves vaccination coverage of patients with inflammatory bowel disease
Supplemental material, sj-tif-1-tag-10.1177_17562848221082879 for A single dedicated vaccination visit improves vaccination coverage of patients with inflammatory bowel disease by Guillaume Le Cosquer, Lionel Grangeon, Pauline Rivière, Arthur Berger, Frank Zerbib, David Laharie and Florian Poullenot in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-2-tag-10.1177_17562848221082879 – Supplemental material for A single dedicated vaccination visit improves vaccination coverage of patients with inflammatory bowel disease
Supplemental material, sj-tif-2-tag-10.1177_17562848221082879 for A single dedicated vaccination visit improves vaccination coverage of patients with inflammatory bowel disease by Guillaume Le Cosquer, Lionel Grangeon, Pauline Rivière, Arthur Berger, Frank Zerbib, David Laharie and Florian Poullenot in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.L.C. and L.G. have no conflict of interest. P.R. declares consultancy fees from Amgen and Janssen. A.B. has no conflict of interest. F.Z. speaker for Janssen. D.L. declares counseling, boards or transport fees from AbbVie, Biogaran, Biogen, Celltrion, Ferring, Fresenius-Kabi, Janssen, MSD, Novartis, Pfizer, Roche, Sandoz, Takeda, Theradiag, and Tillots-Pharma. F.P. declares fees from AbbVie, MSD, Takeda, Ferring, Janssen, and Pfizer.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
