Abstract
Background:
Crohn’s disease (CD) and ulcerative colitis (UC) commonly affect women in their childbearing years. Vedolizumab (VDZ) is approved for treatment of moderate-to-severe CD and UC, but there is a knowledge gap regarding its use during pregnancy. This targeted literature review describes available evidence on safety of VDZ in pregnant patients in order to offer physicians a detailed and balanced view on persistent data during their decision-making process for an individualized treatment concept.
Methods:
The search included literature from the MEDLINE database and abstracts of five gastroenterological conferences published until November 2019. Publications were included if pregnancy outcomes in women receiving VDZ or neonatal outcomes in newborns of women previously exposed to VDZ were reported.
Results:
Out of 196 initially identified records, 18 publications reporting results of five different studies were identified. In total, for 213 of 284 VDZ-exposed documented pregnancies the following pregnancy outcomes were reported: 167 live births (172 infants due to twin births), 1 stillbirth, 35 miscarriages, 10 elective terminations (1 due to detected Down syndrome). Furthermore, during pregnancy, the following complications were observed: seven cases of (pre) eclampsia, three cases of premature rupture of membranes and one case each of placenta previa, chorioamnionitis, pneumonia, first-trimester bleeding, cholestasis, sepsis, or neonatal intraventricular hemorrhage. Based on 172 infants, 30 preterm deliveries (17.4%), 9 cases of low birth weight (5.2%), 5 infections (2.9%), and 6 cases (3.8%) with congenital anomalies were reported.
Conclusion:
There was no evidence for safety concerns regarding pregnancy outcomes associated with VDZ therapy. Due to the limited scope of included records, further research is needed to understand the safety profile regarding the use of VDZ during pregnancy.
Introduction
Inflammatory bowel disease (IBD), comprising Crohn’s disease (CD) and ulcerative colitis (UC), is characterized by chronic inflammation of the gastrointestinal tract. The annual incidence in Europe is reported to be 12.7 per 100,000 person-years for CD and 24.3 per 100,000 person-years for UC. 1 In women, the peak onset of CD is at the age 15–24 years and for UC detection is at ages 25–34. 2 Thus, CD and UC commonly affect women in their childbearing years.
Women suffering from IBD are often concerned whether the disease itself and the required therapy might have detrimental effects on the fertility and pregnancy course, as well as postpartum development of the newborn. 2 Many patients suffering from CD/UC, require maintenance therapy during pregnancy to control the disease.3–5 Consequently, current clinical guidelines recommend a continuous CD/UC treatment in case of pregnancy. The European Crohn’s and Colitis Organization (ECCO) guideline, 5 the Toronto Consensus Statements, and the American Gastroenterological Association (AGA) report from the IBD Parenthood Working Group 6 for the management of IBD in pregnancy 7 outline that the exposure to most IBD medications is considered of low risk to the child, except for methotrexate. In general, women on 5-ASA, or thiopurine for maintenance therapy should continue treatment throughout pregnancy (Julsgaard, Christensen and Gibson, 2016). Continuation of anti-tumor necrosis factor (anti-TNF) therapy during pregnancy is suggested for patients with active disease or a high risk of relapse, while those with inactive disease who wish to discontinue therapy might be advised to stop anti-TNF therapy in the beginning of the third trimester. 8
Vedolizumab (VDZ) represents one of the new drugs that is approved for the treatment of moderate-to-severe CD/UC.9,10 Several clinical and observational studies confirmed the efficacy and safety of VDZ in the treatment of patients with CD/UC, especially in the maintenance of long-term remission.9–15 However, as pregnancy is typically an exclusion criterion in most CD/UC trials, 16 there is a significant knowledge gap regarding VDZ in pregnant patients. Consequently, the main aim of this targeted literature review (TLR) was to collect and describe the currently available evidence on the safety of VDZ use in pregnant patients.
Methods
A TLR was performed by conducting (a) a PubMed search, based on a pre-defined search syntax (Supplemental Table 1), (b) a manual screening of conference abstracts of five annual IBD-focused conferences [Congress of ECCO, United European Gastroenterology Week (UEGW), Digestive Disease Week (DDW), American College of Gastroenterology Annual Meeting (ACG), Advances in Inflammatory Bowel Diseases (AIBD)] and (c) a cross-check for additional literature based on the already identified references.
Generally, studies published in English or German language until 15 November 2019 were considered in the screening process. The PICOS criteria for selection of eligible publications were defined as follows:
Each identified reference went through a title, abstract and full-text review considering the previously mentioned inclusion criteria. After final inclusion of the publications, all relevant data were extracted based on a pre-defined extraction table by one reviewer. In addition to general information describing type of publication, baseline characteristics of observed patients (sample description, sample size, disease, intervention, age of patients, disease status and duration, comorbidities, clinical parameters), reported pregnancy outcomes [live births, stillbirths, preterm deliveries, miscarriages, elective terminations, delivery mode, infections, (pre)eclampsia, premature membrane ruptures, placenta previa, chorioamnionitis, other complications] and neonatal outcomes [neonatal complications, intra-uterine growth retardation (IUGR), low birth weight, small for gestational age, congenital anomalies, serious infections/malignancies during the first life year, other complications] were extracted from the publications whenever available.
Quality assessment of publications was done based on a questionnaire as published by the ISPOR-AMCP-NPC Good Practice Task Force report. 17
All extracted data were descriptively analyzed using MS Excel software (Microsoft, Redmond, USA).
Results
Selected publications
The PubMed database search yielded a total of 48 citations and the manual search of conference proceedings identified an additional 148 citations (Figure 1). Based on these 196 hits, 152 publications were excluded after the title and abstract screening (PubMed: 13, manual search/conference abstracts: 139). Another 28 publications were excluded after full-text screening resulting in 16 publications of interest. Within the PubMed search, also five literature reviews were identified.16,18–21 These were cross-checked for further relevant references. This resulted in the inclusion of two additional publications in the TLR. Finally, 18 publications were included 22–39 in this review. The characteristics of the 18 included publications are described in Table 1.

PRISMA chart.
Characteristics of included studies (original research and editorials/letters).

ACG, American College of Gastroenterology; AGA, American Gastroenterological Association; CD, Crohn’s disease; ECCO, European Crohn’s and Colitis Organization; IBD, inflammatory bowel disease; PMID, PubMed; TNF, tumor necrosis factor; UC, ulcerative colitis; UEG, United European Gastroenterology.
Three publications were full-text publications, nine were conference abstracts und six were letters/editorials. The publications reported results of five different studies/investigations:
(1) Case series – Flanagan et al.; 26
(2) Prospective observational cohort study (OTIS): Chambers et al.; 39
(3) Analysis of safety data from clinical studies and post-marketing setting: Mahadevan et al.; 24
(4) Retrospective, multicenter observational study (CONCEIVE): Moens et al.; 35
(5) Retrospective, multicenter observational study: Wils et al. 32
Figure 2 describes distribution of the 18 publications among these five studies. We assume that 21 cases with VDZ-exposed pregnancies, which were previously reported by Bar-Gil Shitrit et al. [ClinicalTrials.gov identifier: NCT02617927]31,40 have also been included in the retrospective CONCEIVE study.
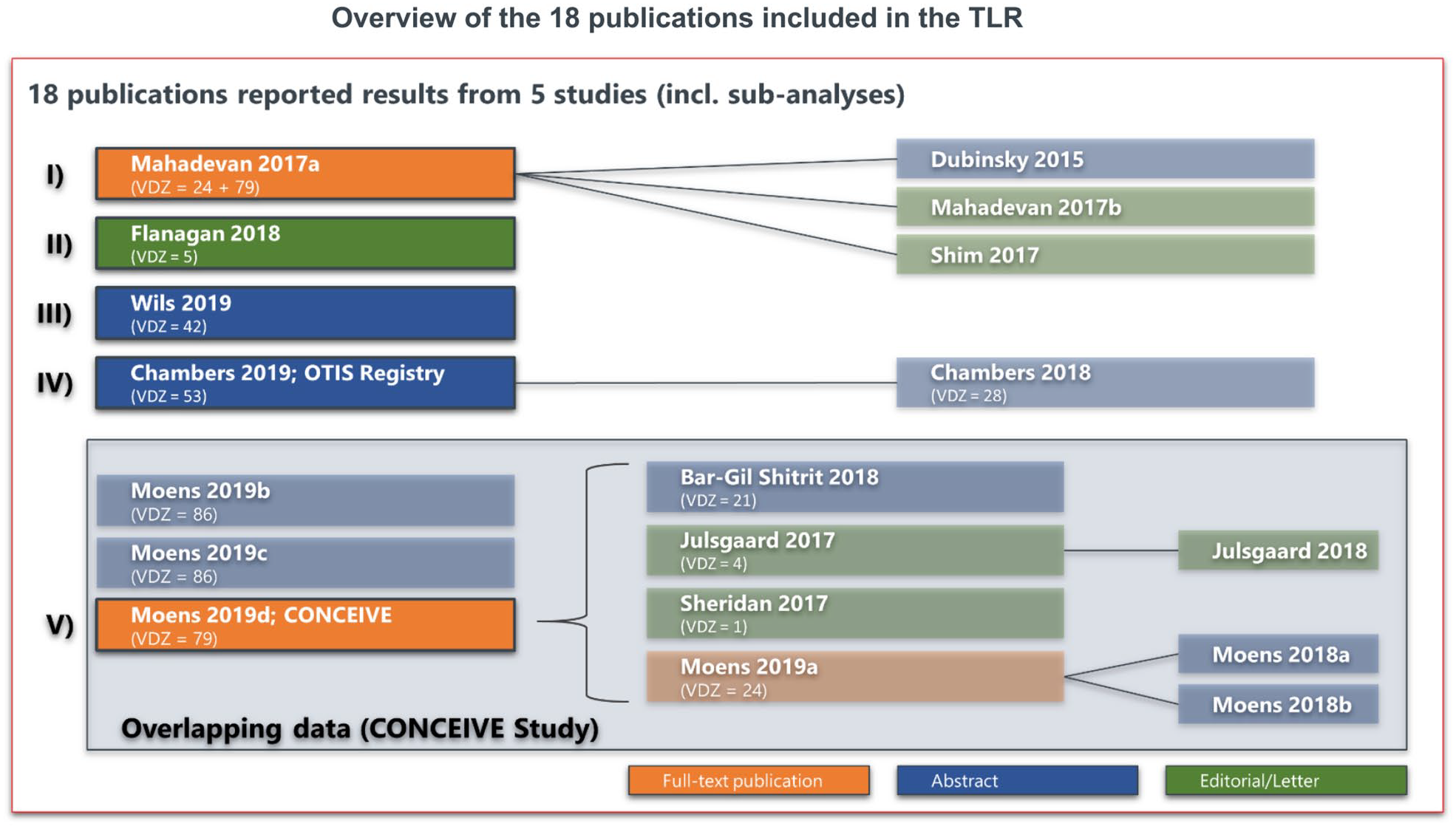
Assignment of publications to identified studies.
Quality assessment, based on the information reported in the respective publications, showed that all studies dealt with relevant populations and interventions and also observed relevant outcomes, even if not all studies completely reported pregnancy and neonatal outcomes (Supplemental Table 2). However, sample size of pregnant women treated with VDZ was generally small. Two of the five studies were comparative studies. One of these studies reported differences in the characteristics of compared patient groups. In the other comparative study, it is unclear whether the compared groups had similar characteristics.
Generally, if a full-text publication reporting results of a study was available, the outcomes of the latter were included in the TLR. Any deviations of reporting between a full-text publication and any previously published conference abstracts were assessed. In case no full-text publication was available for a study, the most recent published conference abstract was used as basis.
Reported pregnancies and observable births
In the five included studies, 284 pregnancies from 276 pregnant women receiving VDZ and 2 untreated women, whose male partners were treated with VDZ, could be observed. The age of the observed pregnant women ranged between 19 and 40 years. The disease duration of the investigated pregnant women was reported between 3 and 20 years. In one study, the pregnancy course of only five women was reported. 26 The other studies were based on 53 VDZ users in the research by Chambers et al. 39 A total of 105 VDZ-exposed women (24 pregnancies from 6 clinical trials and 81 cases based on post-marketing data, including two indirect VDZ-exposed pregnant women, whose male partners were treated with VDZ) in the work by Mahadevan et al. 24 There was a total of 79 pregnancies of 73 VDZ users in the study by Moens et al., 35 and 42 pregnancies of women who received at least one VDZ infusion during pregnancy in the study by Wils et al. 32 Detailed information on the VDZ dosing regimen over the course of observed pregnancies for each study is outlined in Table 2, if available. Only two studies were identified reporting either pregnancy or neonatal outcomes in comparison with a control group: disease-matched patients (DM) and healthy controls (HCs) in the study by Chambers et al., 39 and anti-TNF patients, as well as immunomodulator/biologic-naïve patients in the work of Moens et al. 35
Information on VDZ dosing regimen as reported in identified studies.

number of pregnancies with available information around VDZ exposure.
5-ASA, 5-aminosalicylic acid; IQR, interquartile range; MP, mercaptopurine; MT, maintenance therapy; tri, trimester; VDZ, vedolizumab.
Pregnancy outcomes
In the previously mentioned and in the TLR-included studies, for 71 out of 284 observed VDZ-exposed pregnancies, no pregnancy outcome was described. In the remaining 213 cases, the following outcomes were reported (see also Table 3): 167 live births (172 infants due to twin births), 1 stillbirth (defined as fetal loss after 20 weeks post-conception), 35 miscarriages, and 10 elective terminations. Complications can be summarized as follows: seven cases of (pre)eclampsia (in one case, observed along with gestational diabetes), three cases of premature rupture of the membranes, one case of placenta previa, one case of chorioamnionitis (leading to stillbirth), one case of community-acquired pneumonia with hypoxia, one case of first-trimester vaginal bleed, one case of cholestasis, one case of maternal catheter-related sepsis, and one case of intraventricular neonatal bleeding. One patient might have been affected by more than one of the mentioned complications. All reviewed studies concluded that there was no specific evidence for safety concerns regarding pregnancy outcomes associated with VDZ therapy.
Pregnancy outcomes reported in identified studies.

Including two pregnancies of VDZ-naïve women, whose male partners were exposed to VDZ.
DMs, disease-matched patients; HCs, healthy controls; IBD, inflammatory bowel disease; IM, immunomodulators; TNF, tumor necrosis factor; VDZ, vedolizumab.
As outlined, two of the larger studies compared the observed pregnancy outcomes with comparator groups. Chambers et al. described 53 pregnant women with IBD who received VDZ during at least a part of their first pregnancy trimester with a disease-matched control group (n = 88) and one additional HC group (n = 82). No major structural birth defects were reported in the VDZ group, compared with five and four cases in the DM and HC groups, respectively. Liveborn infant quotas in the three groups were 94.3%, 95.5% and 91.5%, preterm births occurred in 14.6%, 8.4% and 8.3% of the patients, respectively. In a retrospective, multicenter study published by Moens et al., 35 449 pregnant women with IBD, who were exposed to VDZ (n = 79, VDZ-exposed) or anti-TNFs (n = 186, TNF-exposed), as well as one cohort unexposed to immunomodulatory and biologic treatments (n = 184, CON-IBD) were observed. At conception, 92 (50%) of these women were treated with 5-aminosalicylic acid (mesalazine) monotherapy, 12 (7%) with monotherapy of steroids, 12 (7%) with a combination of mesalazine and steroids, while the remaining 67 patients (36%) did not receive any IBD therapy. A higher number of miscarriages was observed in the VDZ group (16%) in comparison with anti-TNFs (13%) and immunomodulatory/biologic-naïve patients (10%). However, non-VDZ patients had less active disease, that possibly have impact on the overall result. After subgrouping by excluding patients with active disease, quotas of miscarriages were similar in 16% (VDZ-exp.), 17% (TNF-exp.) and 15% (CON-IBD), respectively. Compared with previously published data based on 31 study sites, Moens et al.33,35,36 reported slightly lower patient numbers, since their analysis was based on only 29 study sites.
Neonatal and first-year outcomes
Based on 172 infants (167 live births) of VDZ-exposed patients in the included studies, 30 preterm deliveries, 9 cases of low birth weight of less than 2500 g/small for gestational age (SGA; newborns with weight below the 10th percentile for the gestational age), 5 infants with serious infections during first year of life, and 6 cases with congenital anomalies [2 hip dysplasia, 2 agenesis of the corpus callosum (ACC), 1 congenital pulmonary valve stenosis and 1 Hirschsprung’s disease] were reported (Table 4). In total, five twin births from VDZ-exposed women were observed. Furthermore, Down syndrome was reported in one of the previously reported elective terminations.
Neonatal and first year outcomes reported in identified studies.

IBD, inflammatory bowel disease; IM, immunomodulators; IUGR, intrauterine growth restriction; SGA, small for gestational age; TNF, tumor necrosis factor; VDZ, vedolizumab.
It should be noted that several of the previously reported neonatal outcomes may have occurred in the same infant.
With respect to reported prematurely newborns, which were defined as delivery before 37 weeks of gestation, three cases with IUGR (including one pair of twins) were observed, and one death occurred after labor was induced after 26 weeks due to fetal growth restriction and decreased amniotic fluid volume. No autopsy was performed, but histology showed placental insufficiency. Of the five identified infections that occurred in infants during the first year of life after VDZ-exposed pregnancies, three were reported as serious infections, including pyelonephritis, fever of unknown origin, and Kawasaki disease.
In the two studies including a comparator group, Chambers et al. reported that birth weight of full-term infants did not differ between VDZ-exposed, DMs, and HCs. The number of infants with major birth defects was 0.0% (VDZ-exposed), 5.7% (DM), and 5.3% (HC) in the three groups. 39 Serious infections in liveborn infants were observed in 0.0% (VDZ-exposed), 1.2% (DM), and 1.3% (HC) of the cases. In the work by Moens et al., median birth weight did not differ between the groups (VDZ, anti-TNF, immunomodulatory/biologic-naïve). 35 Congenital anomalies were reported for 5% (VDZ-exposed), 2% (TNF-exposed), and 2% (CON-IBD) of the observed cases, whereas serious infections during the first year of life were documented in 5% (VDZ-exposed), 10% (TNF-exposed), and 12% (CON-IBD) of the cases.
Discussion
The main purpose of this TLR was to collect all available clinical and observational evidence regarding the safety of VDZ in the treatment of pregnant women with IBD. The number of studies identified, and the associated patient numbers were small. However, 284 pregnancies of women treated with VDZ were identified. Of these, pregnancy outcome was reported in 213 cases. Generally, none of the studies reviewed could provide evidence for safety concerns with respect to usage of VDZ in pregnant women. Reported pregnancy and neonatal outcomes were generally in line with expected numbers in an IBD population. In the two comparative studies included in our review,24,35 outcomes of VDZ-exposed pregnancies did not differ from outcomes observed in comparator groups (HCs, DMs, IBD patients treated with anti-TNFs, and IBD patients naïve to immunomodulatory/biologic therapies). It should be highlighted that the number of identified studies and the associated patient numbers were found to be small compared with the general population. For comparison, a large Norwegian database study evaluating 421,201 registered pregnancies estimated the overall risk of miscarriage at 12.8% and confirmed a substantial recurrence risk. 41 Furthermore, a recent systematic analysis has estimated a worldwide incidence of 11.1 preterm deliveries per 100 live births in 2010, 42 while the number of cases detected in several European countries was much lower (5%). The prevalence of major congenital anomalies was recorded at 23.9 per 1000 births for 2003–2007 based on European registry data coming from EUROCAT (European Surveillance of Congenital Anomalies). 43
Our results are in line with the five literature reviews published earlier on this topic, which indicated no safety concerns using VDZ in pregnant women.16,18–21 However, only data based on very limited numbers of pregnant patients exposed to VDZ were previously presented. Due to this scarcity of available safety data in pregnancy, VDZ should be used during pregnancy only if the benefits to the mother outweigh the risks to the mother/unborn child.
We acknowledge some limitations of our study. First, we conducted a TLR that meant that our search was based on PubMed and five selected conferences only. Thus, some publications only available through other electronic literature databases or conference websites were not covered by the TLR. Second, in our overall reported patient number, we considered that some studies included patients already reported in earlier publications. Nevertheless, due to the fact a substantial number of identified publications were conference abstracts only, we cannot exclude the possibility that our number of 284 reported VDZ-exposed pregnancies includes some double-counting. Third, most studies did not report outcomes in comparison with a representative control group. Only two studies followed a cohort comparison approach, and in these studies, differences in characteristics of patients in the compared groups might have influenced the results. Therefore, in a sensitivity analysis done by Moens et al., 35 all patients with active disease during pregnancy were excluded. Thus, no difference in the number of miscarriages was detected, when looking only at women with disease remission throughout pregnancy. Moreover, as noted, a substantial number of studies reported their results as conference abstract only, among them, one study that included control groups (OTIS), whereby it should be noted that a second comparative analysis conducted by Moens et al. 35 is available as full text, but only including premature data from the CONCEIVE study. This was associated with a lack of details in terms of provided information. Furthermore, it needs to be stated that available data regarding neonatal and first-year outcomes are quite limited, which has an impact on the general conclusion. Also, regarding the cases where the partner of the pregnant woman was treated with VDZ, only two observations could be identified in this TLR, and thus, no conclusion in this respect can be made. Finally, it needs to be mentioned that there is a potential bias regarding the centers reporting VDZ-exposed pregnancies. There is a need for larger national and international prospective registries, which can provide more representative data based on a broader number of study centers.
Given the limited data on the use of VDZ in pregnancy, VDZ therapy should be only be considered for treatment in pregnant patients after a detailed review of the patient’s medical history, in particular, the control of their IBD and their previous response to other therapeutic options. Other options such as anti-TNF treatment should be considered, since currently there is more evidence available on their use in pregnant patients. For example, a large retrospective multicenter cohort study (TEDDY) compared outcomes of 388 children exposed in utero to anti-TNF drugs with 453 unexposed children. 44 With exception of the proportion of CD diagnoses and previous surgery, the relevant characteristics were similar between both compared groups. The proportion of pregnancy complications, as well as the incidence rate of severe infections, were similar in both groups. Thus, the investigators of the TEDDY study concluded that an in utero exposure to anti-TNFα drugs does not seem to be associated with increased short-term or long-term risk of severe infections in children. However, as an active disease is known to significantly impact pregnancy and neonatal outcomes, continuation or starting VDZ treatment in pregnancy must be individually evaluated for every single patient. In the case of conception under VDZ treatment, maintenance with VDZ should be discussed with the patient on an individual basis, taking especially into account the previous clinical course of disease, current disease activity, comorbidities, previous pregnancy outcomes and the patient’s will. It must be considered what weights more: a potentially undertreated maternal disease activity, the risk of a maternal disease flare, or the potential risk of an undefined unknown side effect of VDZ on the fetus. Therefore, we do not recommend switching biologics during pregnancy because there are no experiences on the risk of a disease flare. Most importantly, the patient and the partner should be informed in detail about all possible risks, and a signed patient information, based on shared decision making, should be obtained. In that respect, it should be highlighted that pregnant CD/UC patients, in general, should be treated with special vigilance, and therefore, a treatment in interdisciplinary centers with specific experiences in biological treatment of pregnant women is strongly recommended.
The use of VDZ during lactation was not a focus within this TLR. Nevertheless, it represents an important topic for pregnant patients. Available information indicates that maternal VDZ use appears to produce low levels in breastmilk.45–47 Because VDZ is a large protein molecule (molecular weight of about 147,000), absorption is unlikely, since it is probably destroyed in the infant’s gastrointestinal tract.6,48 The most recent guideline of the British Society of Gastroenterology stated that while low levels of infliximab, adalimumab, certolizumab, natalizumab, and ustekinumab can be detected in breastmilk from mothers receiving these biologics, breastfed infants of mothers receiving biologics, immunosuppressants, or combination therapy have similar risks of infection and similar milestone achievement at 12 months compared with non-breastfed infants or infants unexposed to these drugs. 8 Although the aforementioned guideline did not explicitly include VDZ, the conclusions related to biologic use might also be applicable to VDZ. Until more data become available, VDZ should be used with caution during breastfeeding, especially while nursing a newborn or preterm infant.
Generally, available information to date suggests that there is no evidence of safety concerns regarding pregnancy outcomes associated with VDZ therapy. Due to the low number of studies identified and the limited scope of included records, more data are needed to understand the safety profile regarding the use of VDZ during pregnancy.
Supplemental Material
Supplemental_Table_1_Search_Syntax – Supplemental material for Safety of vedolizumab in the treatment of pregnant women with inflammatory bowel disease: a targeted literature review
Supplemental material, Supplemental_Table_1_Search_Syntax for Safety of vedolizumab in the treatment of pregnant women with inflammatory bowel disease: a targeted literature review by Birgit Terjung, Renate Schmelz, Robert Ehehalt, Jochen Klaus, Jana Knop, Sabine Schwind, Thomas Wilke and Andreas Stallmach in Therapeutic Advances in Gastroenterology
Supplemental Material
Supplemental_Table_2_Quality_Assessment_of_identified_5_studies – Supplemental material for Safety of vedolizumab in the treatment of pregnant women with inflammatory bowel disease: a targeted literature review
Supplemental material, Supplemental_Table_2_Quality_Assessment_of_identified_5_studies for Safety of vedolizumab in the treatment of pregnant women with inflammatory bowel disease: a targeted literature review by Birgit Terjung, Renate Schmelz, Robert Ehehalt, Jochen Klaus, Jana Knop, Sabine Schwind, Thomas Wilke and Andreas Stallmach in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
The authors are solely responsible for the design and conduct of this study; all study analyses, the drafting and editing of the manuscript, and its final contents. All authors read and approved the final manuscript. T Wilke performed the literature review and writing up of the first draft of the paper. B Terjung, R Schmelz, R Ehehalt, J Klaus, J Knop, S Schwind, and A Stallmach revised the final paper and provided further input.
Conflict of interest statement
Renate Schmelz reports personal fees from AbbVie, Falk Foundation, Janssen, MSD, Pharmacosmos, Pfizer, and Takeda.
Birgit Terjung reports personal fees from AbbVie, Falk Foundation, Janssen, MSD, Microbiotica, Pfizer, and Takeda.
Robert Ehehalt reports personal fees from Falk, Janssen, Amgen, AbbVie, MSD, Ferring, Norgine, Takeda, Shield, Mundipharma, Pfizer, Vifor, Ardeypharm, Novartis, Microbiotika, Biogen, Ferring, Norgine, Celgene, Recordati, Fresenius, Tillotts, Diasorin, and Ethicon.
Jochen Klaus reports personal fees and grants from AbbVie, Amgen, Biogen, CED Service GmbH, Falk-Foundation, Janssen, MSD, Pharmacosmos, Pfizer, Takeda, Thieme, Tillotts Pharma and Vifor Pharma.
Thomas Wilke reports personal fees from several pharmaceutical/consultancy companies, for example, AbbVie, Astra Zeneca, Bayer, BMS, Boehringer Ingelheim, GSK, LEO Pharma, Merck, Novo Nordisk, Pfizer, and Takeda.
Andreas Stallmach reports personal fees and grants from AbbVie, Amgen, AllergoSan, Astellas, Biogen, Falk Foundation, Janssen, MSD, Pfizer, and Takeda.
Jana Knop and Sabine Schwind are employees of Takeda Pharma Vertrieb GmbH & Co. KG.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by Takeda Pharma Vertrieb GmbH & Co. KG.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
