Abstract
Aims:
Early gastric cardiac cancer (EGCC) has a low risk of lymph node metastasis with the potential for endoscopic therapy. We aimed to evaluate the short- and long-term outcomes of endoscopic submucosal dissection (ESD)-resected EGCCs in a large cohort of Chinese patients and compare endoscopic and clinicopathologic features between EGCC and early gastric non-cardiac cancer (EGNC).
Methods:
We retrospectively studied 512 EGCCs in 499 consecutive patients and 621 EGNCs in 555 consecutive patients between January 2011 and March 2018 at our center. We investigated clinicopathological characteristics of EGCC tumors, ESD treatment results, adverse events, and postresection patient survival.
Results:
Compared with EGNC patients, EGCC patients were significantly older (average age: 66 years versus 62 years, p < 0.001). The percentage of the gross 0–IIc pattern was higher in EGCCs (46.1%) than in EGNCs (41.5%), while the frequency of the 0–IIa pattern was lower in EGCCs (14.9%) than in EGNCs (22.4%) (p = 0.001). Compared with EGNCs, EGCCs showed smaller size, deeper invasion, fewer ulcerated or poorly differentiated tumors, but more cases with gastritis cystica profunda. The prevalence of ESD-related complications was higher in EGCCs (6.1%) than in EGNCs (2.3%) (p = 0.001). In EGCCs, the disease-specific survival rate was significantly higher in patients of the noncurative resection group with surgery (100%), compared with that (93.9%) without surgery (p < 0.001).
Conclusion:
Clinicopathological characteristics were significantly different between EGCCs and EGNCs. ESD is a safe and effective treatment option with favorable outcomes for patients with EGCC. Additional surgery improved survival in patients with noncurative ESD resection.
Introduction
According to the 2015 World Health Organization (WHO) global cancer statistical data, gastric cancer remains the fifth most common malignancy and the third leading cause of cancer-related deaths worldwide, with over 1,000,000 new cases and estimated 783,000 deaths annually. 1 This cancer varies widely in incidence geographically, as evidenced by a predominance of Chinese patients in about half new gastric cancer cases.2,3 Gastric cancer can be divided into cardiac and noncardiac groups. Compared with Japanese and most other ethnic populations, gastric cardiac cancer in Chinese patients is much more prevalent,4,5 accounting for 23.2% of gastric cancer resection cases in a single center study. 6 Gastric cardiac carcinoma is an aggressive disease, with a 5-year survival rate of 16%–32.3% after standard therapy.7–9 Early gastric carcinoma (EGC) is defined as a tumor restricted to the mucosa or to the mucosa and submucosa, regardless of lymph node metastasis. In patients with early gastric cardiac cancer (EGCC), the 5-year survival rate is more than 90%. 10 Because of the significantly lower risk of lymph node metastasis in EGCCs than in EGNCs, 11 endoscopic resection is beginning to play an increasing role in the treatment of EGCC. 10 This is because endoscopic therapy, especially endoscopic submucosal dissection (ESD), is widely accepted as a minimally invasive alternative to surgical resection with a high curative rate for early carcinomas of the esophagus, stomach, and colon. 12 ESD allows for a high en bloc complete resection rate of early carcinomas with minimal injury and rapid recovery.13,14 However, ESD for EGCC is technically demanding because of the interference of contractions of the lower esophageal sphincter and the need for endoscopic retroflexion over the entire procedure within a confined operative space. While several previous studies have demonstrated clinical results of ESD for EGCC resection, those studies had limited numbers of patients and short follow-up periods.15–17 As such, many key issues related to ESD-resected EGCC, such as adverse events and long-term prognosis, remain uncertain. Therefore, the aim of this study was to investigate the safety, feasibility, effectiveness, and outcomes of ESD for EGCC in a large cohort from a single high-volume center in China.
Methods
Patient selection
This retrospective study included consecutive patients with ESD-resected EGCC tumors at the Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School from January 2011 to March 2018. Patients were identified through a search in a prospectively maintained electronic endoscopic database and chart review. EGCC was defined as early gastric carcinoma with the epicenter located in a narrow region of approximately 3 cm below the gastroesophageal junction line, defined as the proximal end of gastric longitudinal folds, as previously described.10,11 A detailed flow chart on patient selection is shown in Figure 1. The thoracic and abdominal computed tomography (CT) of all patients was performed before the ESD procedure to ensure the absence of lymph node and distant metastases. The study protocol was approved by the Medical Ethics Committee of the Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School (approval number: 2019-050-01). All patients in this study have signed the written informed consent.

Flow diagram for the patients in this study.
Pre-ESD endoscopic examination
Once a suspicious lesion in the gastric cardia was identified endoscopically and diagnosed histopathologically on a biopsy material as high-grade intraepithelial neoplasia or intramucosal carcinoma, the patient was recommended for resection. Before the ESD procedure, a routine esophagogastroduodenoscopy was performed in every patient to evaluate the lateral and vertical margins of resection and the invasion depth of an EGCC tumor. The initial assessment of the tumor surface and lateral borders was performed with white light and magnifying endoscopy (GIF-H260Z: Olympus Medical Systems, Tokyo, Japan). Narrow-band imaging was used to assess the tumor lateral bounder. Endoscopic ultrasonography with a high-frequency microprobe (UM-DP20-25R, Olympus Medical Systems) was performed by experienced endoscopists to assess the lesion location, extent, and depth. The endoscopic morphology of all lesions was categorized, according to the Paris Classification of early gastric cancer. 18 Infiltrative (INF) patterns were subgrouped as follows: INFa, expanding growth with a distinct border from the surrounding stroma; INFc, infiltrative growth without a defined border with the surrounding stroma; and INFb, an intermediate pattern between INFa and INFc. 19
ESD procedure
The ESD procedure was performed by five experienced endoscopists in our center with a standard endoscopic therapy protocol (Figure 2). All patients were placed under conscious sedation with intravenous anesthesia using midazolam and propofol. In brief, several mucosal spots were endoscopically marked outside the tumor lateral margin using the Dual Knife (KD-650L, Olympus Medical Systems). Then, a mixture of normal saline and indigo carmine with diluted epinephrine (1:100,000) was injected to lift the submucosal layer. Subsequently, a circumferential mucosal resection was made around the marking spots, and the submucosal layer was dissected using the Dual Knife (KD-650L, Olympus Medical Systems). Finally, the resected specimen was retrieved with grasping forceps (FD-410LR, Olympus Medical Systems). All visible active bleeding vessels on the resection bed were coagulated to prevent delayed bleeding (Figure 3).

The endoscopic characteristics of one early gastric cardiac carcinoma (0–IIc) located in the posterior wall of the gastric cardia. The tumor shows a depressed growth pattern under white-light (A). The tumor border is clearly demarcated in ME-NBI at low magnification (B). The destructive microstructure and fine network microvascular patterns are highlighted in ME-NBI at high magnification (C).
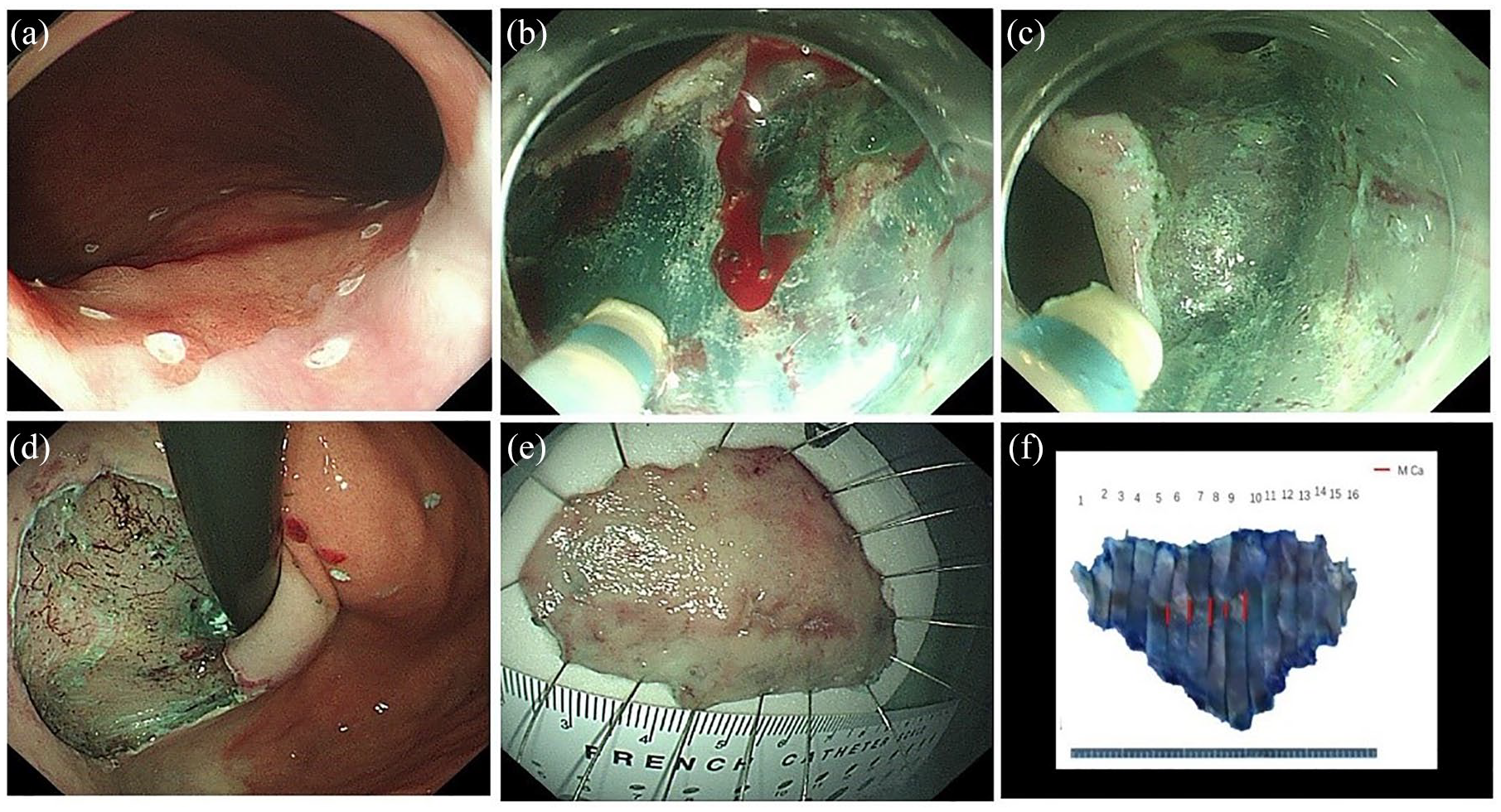
The endoscopic submucosal dissection of one early gastric cardiac carcinoma. (A) Marking outside the tumor margin. (B) A circumferential mucosal resection around the marking spots. (C) Submucosal dissection. (D) The resection bed. (E) The resected specimen. (F) Using the gross photograph to map and rebuild the lesion.
The standard procedure of endoscopic submucosal tunnel resection (ESTD) is: submucosal injection; creation of tunnel entry (anal and then oral); creation of submucosal tunnel; lateral resection. ESD using a pocket-creation method (PCM) can be regarded as a submucosal tunnel, the only difference being that there is only one “tunnel entry;” thus, ESD using PCM can be regarded as a modification of ESTD. A traction-assisted (TA) strategy has been recently developed for ESD. TA-ESD involves the provision of countertraction after the mucosal cut of the lesion has been performed. This enables the submucosal layer to be visualized, which reduces the complication rate. More methods have recently been developed for TA-ESD, such as the clip with-thread method, clip-flap method, magnetic anchor-guided ESD, clip-band technique, and double channel scope.
En bloc resection was defined as the endoscopic resection of a lesion in one piece. Complete resection referred to the endoscopic en bloc resection in a single procedure without histopathological evidence of tumor involvement of the lateral and vertical margins of resection. According to the Japanese gastric cancer treatment guidelines from 2010 (version 3), the following was curative ESD for the absolute indications: en bloc resection, tumor size 2 cm or less, differentiated-type histological appearance, pT1a, negative HM (HM 0), negative vertical margin (VM 0), no lymphatic invasion, and no venous invasion. 20 In addition, the resection was considered as curative ESD for the expanded indications when all of the following conditions were fulfilled: en bloc resection, HM 0, VM 0, no lymphatic invasion, and no venous invasion, with (1) tumor size greater than 2 cm, differentiated-type cancer, pT1a (M), no ulcerative findings; (2) tumor size 3 cm or smaller, differentiated-type cancer, pT1a, ulcerative findings; (3) tumor size 2 cm or smaller, undifferentiated-type cancer, pT1a, no ulcerative findings; (4) tumor size 3 cm or smaller, differentiated-type cancer, pT1b (SM1). Noncurative endoscopic resection was defined as endoscopic resection with multiple fragments, positive resection margins, lymphovascular invasion, or lesions that did not meet the expanded indications for ESD.
Pathological examination of the ESD-resected specimen
Endoscopically resected fresh specimens were oriented by the ESD operator and pinned down on a dental wax plate, photographed, measured, and then fixed in 10% neutral buffered formalin solution overnight. With a standard surgical pathology specimen processing protocol, all resection margins were inked. After gross examination and recording of the tumor size, shape, surface color, and the distance from the tumor edge to the lateral margins of resection, the specimen was transversely sectioned at the interval of 2 mm and embedded sequentially in paraffin in its entirety. Tissue sections (5 µm) were cut and stained routinely with hematoxylin and eosin (H&E). Two experienced pathologists independently evaluated each case blindly without the knowledge of clinical and endoscopic information. The international diagnostic criteria for chronic gastritis and the WHO diagnostic criteria for intraepithelial neoplasia (low- and high-grade) and intramucosal carcinoma were followed (Figure 4A). 21
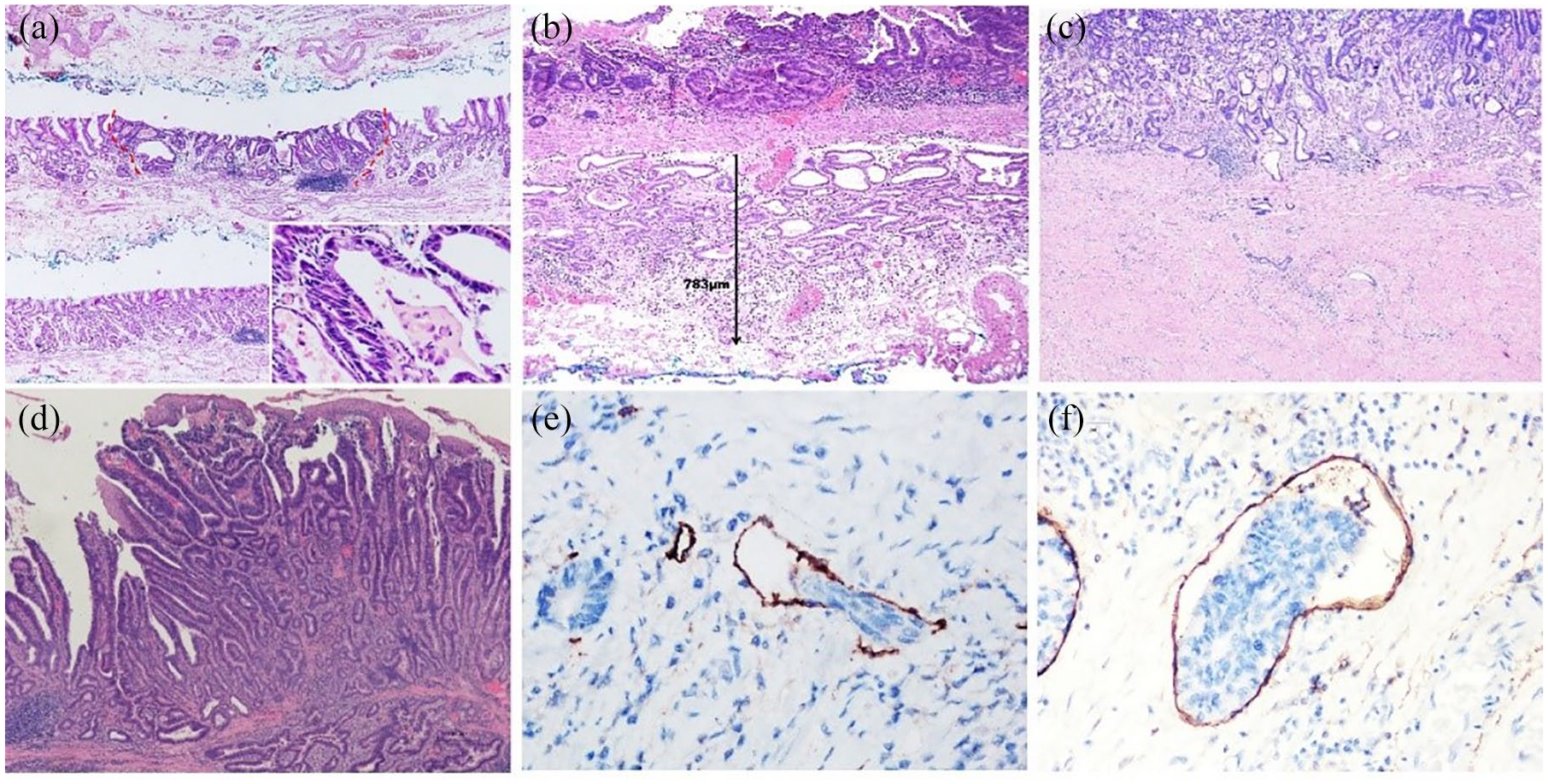
Histological evaluation of endoscopic submucosal dissection-resected specimens. (A) An intramucosal well differentiated adenocarcinoma shows crowded neoplastic glands at low magnification (H&E ×40); the inset illustrates neoplastic epithelium and intraluminal necrotic debris (inset, H&E ×400). (B) The submucosal cancerous invasion depth was measured from the lower-most edge of the muscularis mucosa to the deepest invasive front of the tumor (H&E ×100). (C) Ulcer-healing scar is present within a submucosal invasive carcinoma (H&E ×40). (D) An early papillary adenocarcinoma centered in the gastric cardia extends upwards into the distal esophagus underneath the benign squamous epithelium without the evidence of Barrett’s esophagus (H&E ×100). (E) Venous invasion is highlighted by brown immunostaining of CD31 in the venous endothelium (immunohistochemistry stain ×400). (F) Lymphatic invasion is demonstrated by brown immunostaining of D2-40 for the lymphatic channel lining cells (immunohistochemistry stain ×400).
Tumor size, depth of invasion (Figure 4B), the presence or absence of ulceration (Figure 4C), the status of lateral and vertical margins, histological types, the presence or absence of lymphovascular invasion, the extent of atrophic gastritis, and the presence or absence of esophageal invasion (Figure 4D) were evaluated microscopically in each case. 22 Gross tumor characteristics were subgrouped into five patterns as follows: polypoid/protruding (type 0–I), slightly elevated with rough surface (type 0–IIa), flat (type 0–IIb), slightly depressed with erosion (type 0–IIc), and (5) depressed area in an elevated lesion (type 0–IIa+IIc)(18). 23
When submucosal invasion was present, the depth of tumor invasion was measured microscopically from the lowest edge of the muscularis mucosae to the deepest point of invasive glands. The depth of tumor penetration was divided into four categories: (1) M2, tumors limited to the lamina propria without the involvement of the muscularis mucosae; (2) M3, tumors involving the muscularis mucosae; (3) SM1, tumor invasion into the superficial submucosa (<500 μm penetration into the submucosa from the muscularis mucosae); and (4) SM2, tumor involvement of the deep submucosa (⩾500 μm in submucosa). 23 Immunohistochemical staining for CD31 and D2-40, histochemical staining and Elastica van Gieson staining were used for the identification of lymphovascular invasion (Figure 4E–F).
The decision for an ESD procedure was based on the criteria of the 2004 Japanese Gastric Cancer Association Classification. 22 The absolute indication for ESD referred to intramucosal tubular/papillary adenocarcinoma with a tumor size smaller than 2 cm without ulceration. The expanded indications included the following: (1) intramucosal tubular/papillary adenocarcinoma greater than 2 cm in size without lymphovascular invasion or ulceration; (2) intramucosal tubular/papillary adenocarcinoma smaller than 3 cm in size with ulcers but without lymphovascular invasion; (3) poorly differentiated intramucosal carcinoma smaller than 2 cm in size without ulcer and lymphovascular invasion; and (4) tubular/papillary adenocarcinoma with superficial submucosal invasion (SM1) smaller than 3 cm in size without ulcer and lymphovascular invasion.
ESD outcome assessments
Intra- and peri-operative ESD primary adverse events were bleeding, perforation, and stenosis. Early delayed bleeding was defined as endoscopically visible bleeding discovered within 48 h after the ESD procedure and manifested clinically as melena, hematemesis, or a marked drop in serum hemoglobin levels of over 2 g/dl, requiring emergency endoscopy or surgery to stanch the bleeding. Bleeding events treated by hemostatic forceps during the ESD procedure were excluded. Late delayed bleeding was defined as bleeding discovered more than 48 h after the ESD operation. A diagnosis of perforation required direct endoscopic observation of a hole in the gastric wall during the ESD procedure or the finding of free air in the abdomen on chest radiography. Stenosis was diagnosed whenever marked narrowing of the gastric cardiac lumen was discovered at upper endoscopy to the extent that an endoscope tube was not able to easily passing through the distal esophagus into the cardia.
Local recurrence was considered once a new carcinoma tumor was discovered at the prior ESD site. Tumors that were detected at other sites within 1 year after the ESD procedure were interpreted as synchronous tumors, while tumors that were revealed at other sites more than 1 year after the ESD procedure were termed metachronous tumors.
Follow up
Patients who underwent curative resection after the ESD procedure were required to repeat endoscopy with biopsy at the interval of 3, 6, 12 months, and annually thereafter. Radiological surveillance using CT of the abdomen and chest was performed at 6, 12 months, and annually thereafter to detect lymph node and distant metastases. The patients who underwent a noncurative ESD resection with a positive horizontal margin were required to have an additional ESD procedure, or total gastrectomy or proximal gastrectomy with distal esophagectomy along with lymph node dissection in selected cases. If the patients refused additional resection procedures, they were suggested to have follow-up upper endoscopy and the thoracic and abdominal CT 3–6 months later.
Statistical analysis
Statistical analyses were performed using SPSS 22.0 (IBM, Armonk, New York, USA). Differences between clinical characteristics were assessed using the two-tailed Student’s t-test or the chi-square test. Statistical significance was evaluated using the chi-square test for categorical variables. Overall and disease-specific survival rates were estimated by the Kaplan–Meier method with a log-rank test. A p value of <0.05 was considered statistically significant.
Results
Among 570 ESD-treated EGCC cases over the study period, 71 (12.5%) were excluded, and 512 lesions in 499 patients met the selection criteria for this study.
Demographic and clinical features
As shown in Table 1, there was a predominance of male patients in two groups, with a male:female ratio of 4.6 and 2.9 in EGCC and EGNC group, respectively. The proportion of male patients was much higher in EGCCs (82.2%) than in EGNCs (74.4%) (p = 0.002). Compared with EGNC patients, EGCC patients were significantly older (average age: 66 years versus average age: 62 years, p < 0.001). The most common comorbidities in the two groups included hypertension and diabetes mellitus. In the EGCC group, only 14 patients (2.8%, 14/499) had reflux esophagitis, and only one patient (0.2%) had Barrett’s esophagus.
Demographic and clinical characteristics of EGCC and EGNC.

EGCC, early gastric cardiac cancer; EGNC, early gastric non-cardiac cancer.
Endoscopic and pathological characteristics
The comparison of endoscopic and pathological characteristics between EGCC and EGNC groups is shown in Table 2. The mean specimen size was 48 mm (range, 12–120) in EGCCs and 48mm (range, 10–170) in EGNCs (p = 0.101). In the EGCC group, the most common location of EGCC tumors was the posterior wall of the cardia (50.0%, 256/512), followed by the lesser curvature (43.0%, 220/512). The most common location of EGNCs tumors was the antrum (44.9%, 279/621), followed by the angularis (28.2%, 175/621). In the EGCC group, the most common gross endoscopic pattern, in a descending order, was 0–IIc (46.1%, 236/512), 0–IIa+IIc (27.3%, 140/512), 0–IIa (14.9%, 76/512), 0–IIb (9.8%, 50/512), 0–I (0.8%, 4/512), and 0–III (1.1%, 6/512). The gross 0–IIc pattern was seen more commonly in EGCCs (46.1%, 236/512) than in EGNCs (41.5%, 258/621), while the percentage of the 0–IIa pattern were lower in EGCCs (14.9%, 76/512) than in EGNCs (22.4%, 139/621) (p = 0.001). Ulceration was present more frequently in EGNCs (9.0%, 56/621) than in EGCCs (4.7%, 24/512) (p = 0.005). The mean tumor size was 17 mm (range 2–66) and 18mm (range 2–154) in EGCCs and EGNCs, respectively. Tumor size >2 cm occurred more commonly in EGNCs (33.8%, 210/621) than in EGCCs (26.2%, 134/521) (p = 0.005). Compared with EGNCs, EGCCs invaded deeper with a higher frequency of submucosal invasion (28.7%, 147/512 versus 11.0%, 69/621, p < 0.001). Histopathologically, the vast majority of EGCCs (98.4%, 504/512) were well- to moderately differentiated tubular or papillary adenocarcinomas. Only eight (1.6%) tumors were poorly differentiated carcinomas (four mucinous adenocarcinomas, two signet ring cell carcinomas, two poorly differentiated adenocarcinoma), while the proportion of poorly differentiated carcinomas was significantly more common in EGNCs (4.7%, 29/621) than in EGCCs (1.6%, 8/512) (p = 0.03). There was no significant difference in lymphovascular invasion between EGCCs (2.9%, 15/512) and EGNCs (2.4%, 15/621) (p = 0.592). Atrophic gastric carditis was found in 90% (461/512) EGCCs and 87.0% (606/621) EGNCs (p = 0.107). Helicobacter pylori infection was 57.5% (287/512) in EGCCs. Gastritis cystica profunda occurred significantly more frequently in EGCCs (24.8%, 127/512) than in EGNCs (7.4%, 46/621) (p < 0.001). Focal distal esophageal involvement was detected in 11.7% (60/512) in EGCCs. In EGCC group, diagnosis at initial EFB classified 324 lesions (63.3%) as HGN and 51 lesions (10.0%) as carcinoma. For lesions with a biopsy diagnosis of HGN on EFB, 2.8% (9/324) had a concordant final diagnosis of HGN, 315 cases (97.2%) were finally diagnosed as a higher grade neoplasia such as well-differentiated carcinoma (95.9%, 311/324) or poorly differentiated carcinoma (1.2%, 4/324). All cases of carcinoma on EFB were diagnosed as carcinoma in ESD specimens [well-differentiated carcinoma (96.1%, 49/51) or poorly differentiated carcinoma (3.9%, 2/51)].
Pathological characteristics of EGCC and EGNC.

0–I, protruding; 0–IIa, superficial elevated; 0–IIb, superficial flat; 0–IIc, superficial depressed; 0–IIa+IIc, mixed type; 0–III, excavated.
Included poorly differentiated adenocarcinoma, poorly cohesive/signet ring cell carcinoma, mucinous adenocarcinoma, carcinoma with lymphoid stroma, neuroendocrine carcinoma, and adenosquamous carcinoma.
EGCC, early gastric cardiac cancer; EGNC, early gastric non-cardiac cancer.
Intra-/peri-operative findings
As shown in Table 3, the proportion of cases fulfilling the Japanese absolute and expanded indications for ESD in EGCCs (81.4%) was lower than that in EGNCs (91.3%) (p < 0.001). Overall, the median ESD procedure time was 65 min (range, 10–353) in EGCCs and 63 min (range, 12–300) in EGNCs (p = 0.586). The en bloc, complete, and curative resection rates were 99.8% (511/512), 94.3% (483/512), 80.5% (412/512) in EGCCs and 99.8% (620/621), 98.6% (612/621), 90.5% (562/621) in EGNCs, respectively. Compared with EGNCs, the proportion of complete resection (94.3% versus 98.6%, p = 0.021) and curative resection (80.5% versus 90.5%, p < 0.001) was lower in EGCCs. Among 31 (6.1%, 31/512) cases with ESD-associated adverse events, ESD procedure-related bleeding was observed in 2.3% (12/512) of cases, 8 of which were early delayed bleeding and 4 were late delayed bleeding. All bleeding events were successfully managed using endoscopic hemostasis, and none of the patients required blood transfusions. Perforation occurred in only one case (0.2%, 1/512) and was immediately closed endoscopically without any sequelae. The proportion of complication in EGCCs (6.1%, 31/512) was higher than in EGNCs (2.3%, 14/621) (p = 0.001). The median hospital stay of patients was 6 days (range 2–19) in EGCCs and 6 days (range 3–31) in EGNCs.
Intra-/peri-operative findings of endoscopic dissection of EGCC and EGNC.

Gastric cancer treatment guidelines. 22
EGCC, early gastric cardiac cancer; EGNC, early gastric non-cardiac cancer.
Causes of noncurative resection of EGCC
Table 4 lists causes of noncurative ESD resection of EGCC lesions. Five lesions (5%, 5/100) showed positive resection margins in two cases with absolute indication (2%, 2/100) and three with expanded indication (3%, 3/100). In the study, 95 cases (18.6%) met the Japanese beyond indications for ESD. A diagnosis of cT1a was made for five lesions (5%, 5/100), as intramucosal EGCC in size of more than 3 cm with ulceration. The most common cause of noncurative ESD resection was SM2 invasion (72%, 72/100), followed by SM1 with EGCC > 3 cm in size (16%, 16/100), SM1 with undifferentiated type (1%, 1/100) and SM1 with lymphovascular invasion (1%, 1/100).
Cases with noncurative resection of EGCC.

EGCC, early gastric cardiac cancer.
Post-ESD outcomes
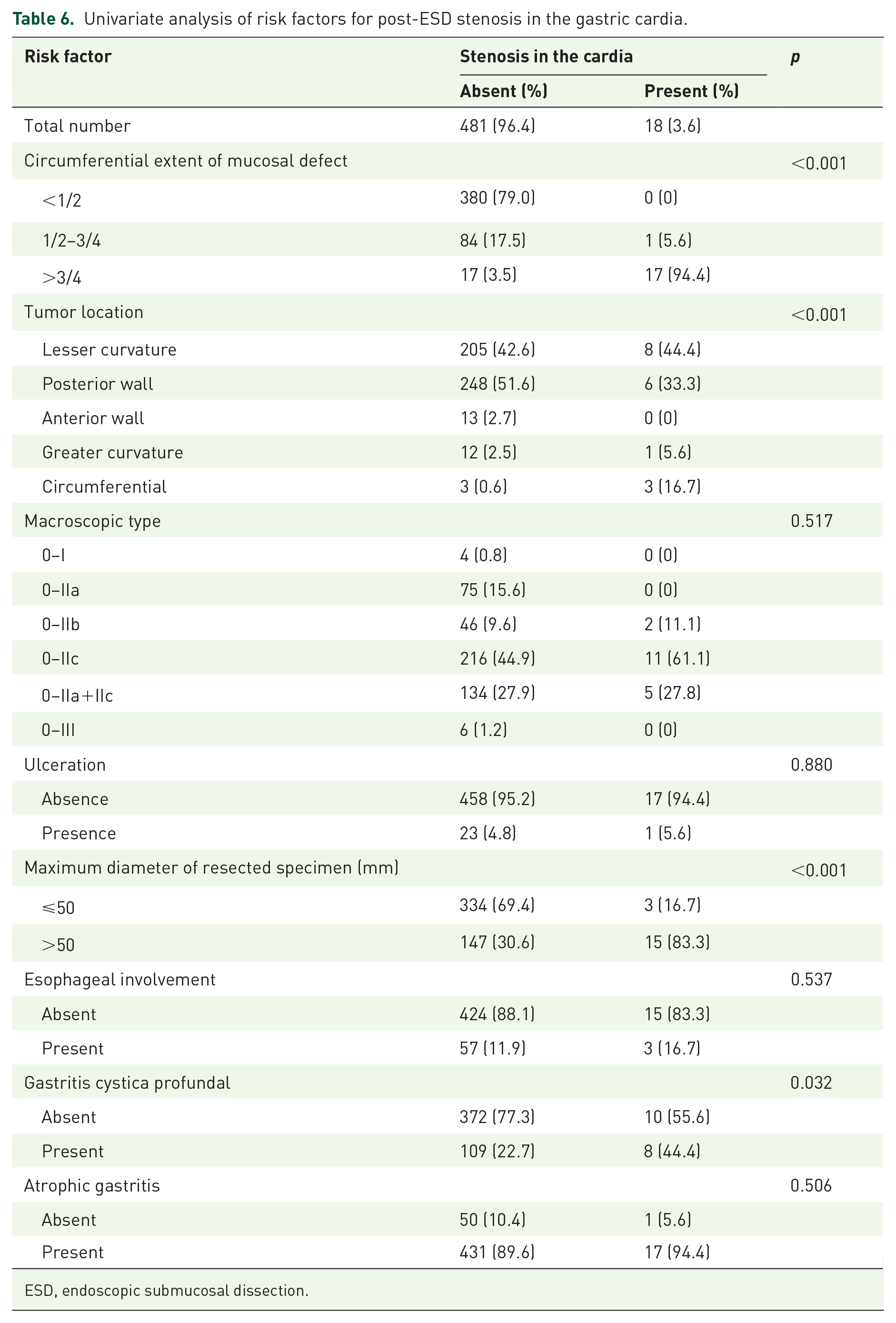
The mean number of follow-up months was 48 (range 2–101). A total of 44 patients (8.8%, 44/499) were lost to follow up (36 with curative resection and 8 with noncurative resection). Table 5 shows the clinicopathologic characteristics of the patients who developed stenosis after ESD resection for EGCC. Stenosis was discovered in 18 (3.6%, 18/499) patients during the follow-up period, with an average of 45 days (range 22–111). The mean specimen size was 74 mm (range 41–100). The circumferential extent of an ESD-related mucosal defects was more than 75% in 17 cases with stenosis, which was significantly larger than that of cases without stenosis (p = 0.000) (Tables 6 and 7). All patients with stenosis were successfully managed endoscopically with balloon dilation (average: 3 sessions; range 1–10) (Figures 5 and 6).
Clinical characteristics of patients with stenosis after ESD of early gastric cardiac carcinomas.

ESD, endoscopic submucosal dissection.
Univariate analysis of risk factors for post-ESD stenosis in the gastric cardia.
ESD, endoscopic submucosal dissection.
Multivariate analysis of risk factors for post-ESD stenosis in the cardia.

CIm confidence interval; ESD, endoscopic submucosal dissection.

Cardiac stenosis after ESD with a circumferential resection of an early gastric cardiac carcinoma. (A) WLE images of a depressed early gastric cardiac adenocarcinoma. (B) The clear demarcation line of the lesion in ME-NBI at low magnification. (C) Marking outside the tumor margin. (D) The lesion was completely removed. (E) Cardiac stenosis discovered 34 days after the ESD procedure. (F) The stenosis was successfully relieved with endoscopic balloon dilation in three sessions without adverse effects.

Kaplan–Meier plots of survival among patients after curative or noncurative ESD resection with or without additional surgery. (A) Overall survival. (B) Disease-specific survival.
Additional surgical resection was carried out in 43 (46.7%, 43/92) patients with a noncurative resection and in 19 (5.2%, 19/363) patients with a curative resection. A residual tumor, lymph node metastasis, or both were revealed in surgical resection specimens of 43 patients with a noncurative resection, but not in any patients with curative ESD resections.
During the follow-up period, as shown in Table 8, metachronous tumors were discovered in six patients with a curative resection; all were managed successfully with additional endoscopic resections and achieved complete remission. Local or distant recurrent tumors were found in four patients with a noncurative resection. In the group of noncurative resection without additional surgery, death occurred in four patients (8.2%, 4/49), three of whom were gastric cancer-related and one died of other causes. In contrast, in the group of noncurative resection with additional surgery, three patients died of other causes and none died of gastric cancer.
Post-endoscopic dissection outcomes in patients with EGCC.

Group 1, Curative resection with surgery; Group 2, Curative resection without surgery; Group 3, Noncurative resection with surgery; Group 4, Noncurative resection without surgery.
EGCC, early gastric cardiac cancer.
In this cohort, the 5-year overall survival rate was 89.6% (Table 8). The difference in the 5-year overall survival rate was not significant between the curative and noncurative resection groups with or without surgery. However, the 5-year disease-specific survival rate was significantly lower (93.9%) in the patients with a noncurative resection than in the patients (100%) with a curative resection (p < 0.001).
Discussion
Most previously published studies indicated that Barrett’s esophagus is uncommon in the Chinese population. The prevalence rates of Barrett’s esophagus are only 0.76% and 0.06% in southern China and Taiwan, respectively.24,25 Our previous study compared the demographic and clinical characteristics of gastro-esophageal junction cancers in Chinese and American patients. We found almost all gastric cardia cancers in Chinese arise in the proximal stomach and show the features of proximal gastric cancer. Of 43 Chinese patients, 100% (43/43) had the epicentre of their tumor within the proximal stomach, compared with 4 (11%, 4/37) American patients. A total of 26 (87%) American patients showed distal oesophageal columnar metaplasia, as compared with only 6 (18%) Chinese patients (p = 0.001). In contrast, most Chinese patients had chronic or chronic active gastritis (81% versus 24%, p = 0.01), and they had significantly higher frequencies of H. pylori infection (35% versus 19%, p = 0.01), gastric intestinal metaplasia (47 versus 19%, p = 0.009), and gastric dysplasia (58% versus 5%, p = 0.001) than American patients.26–28 These results illustrate significant differences between cancers arising above or below the EGJ. EGCC is the result of progression from H. pylori superficial gastritis to atrophic gastritis and hypochlorhydria to dysplasia and finally to cancer. In our study, the epicenter of all tumors was within 3 cm below the gastroesophageal junction.
Although surgical resection of EGCC with or without distal esophagectomy is a conventional treatment strategy, 7 endoscopic therapy, especially ESD, has emerged as an alternative option in qualified EGCC patients with no or extremely low risk of lymph node metastasis.29,30 Based on a multicenter clinicopathologic study of 2101 radical gastrectomies for early gastric carcinoma, the risk of lymph node metastasis was found to be significantly lower in EGCC than in EGNC, especially for intramucosal EGCC, thus supporting endoscopic therapy, such as ESD, for qualified EGCC patients. 11 Compared with EMR, the main advantage of ESD is the high rate of en bloc resection, especially for large lesions, so that accurate pathologic evaluations and appropriate staging become possible. Several reports have demonstrated the clinical outcomes of ESD for EGCC but with limited numbers of patients.15–17 Their conclusions require validations and verification with large samples. For instance, Yoshinaga et al. reported 25 superficial adenocarcinomas located at the esophagogastric junction, including only eight EGCCs; their ESD curative resection rate was 72%. 15 Jang et al. showed the outcomes for ESD of gastric cardiac neoplasms with 33 adenomas and 49 cancers, in which the proportions of en bloc, complete, and curative resection rates were 87%, 79%, and 66%, respectively. 16 Osumi et al. demonstrated that the curative resection rate was higher in 87 gastric cardiac adenocarcinoma lesions (81%) than in 55 Barrett’s adenocarcinomas (66%). 17 Although their study results for outcomes of ESD resection of EGCC are informative, the numbers of their cases with EGCC are small, fewer than 100, and several key issues related to this ESD procedure, such as the long-term outcomes in EGCC patients from high-risk regions, remain unsettled. In our present study of 499 patients with 512 ESD-resected EGCC lesions, the en bloc resection rate was 99.8%. The most common location of EGCC tumors was the posterior wall of the cardia (50.0%), followed by the lesser curvature (43.0%). Ulceration (4.7%) was uncommon. In contrast, the most common gross endoscopic pattern was 0–IIc (46.7%). Most EGCC tumors (71.3%) were intramucosal, and submucosal EGCCs accounted for 28.7% of the cohort. By histology, well- to moderately differentiated tubular or papillary adenocarcinomas comprised the absolute majority (98.4%). Only eight (1.6%) tumors were poorly differentiated carcinoma (four mucinous adenocarcinomas, two signet ring cell carcinomas, two poorly differentiated tubular adenocarcinomas). Gastritis cystica profunda was found in the area adjacent to the EGCC in over 24% of cases. Although the pathogenesis mechanisms are unclear, this lesion may be related to chronic carditis, as we previously reported. 11 Focal distal esophageal involvement was detected in 11.7% of cases, but evidence for esophageal adenocarcinoma was lacking because the epicenter of these tumors was below the gastroesophageal junction line and the absence of the evidence of long segment Barrett’s esophagus. Lymphovascular invasion was identified in only 2.9% of all cases, suggesting a low risk of nodal metastasis in this cohort. Despite difficulty operating endoscopically with the ESD procedure in the gastric cardia, we succeeded in our efforts in EGCC resections, and our results demonstrate that the ESD resection of EGCC indeed achieved high complete resection (94.3%) and curative resection (80.5%) rates, which are similar to those of surgical resections of early gastric cancer.31–35
In our study, adverse events occurred in 6.1% of lesions, including one case of perforation, 12 cases with bleeding, and 18 cases of cardiac stenosis, which were easily managed endoscopically. None of the patients required surgical intervention. Stenosis is a common complication in patients undergoing endoscopic resection for gastric cardiac tumors, especially for large lesions with a circumferential mucosal defect of over 75%. 4 As reported by others, cardiac stenosis could be successfully treated by endoscopic balloon dilation.4,36 Most patients require only one session, and a few patients require multiple sessions. After endoscopic dilation, stenosis-associated symptoms disappeared without additional surgical intervention. Steroid applications were reported to be effective for the prevention of stricture formation. Steroid treatment for stenosis prevention was administered to two patients (5.9%, 2/34) with at least 75% of circumferential resection. One patient developed stenosis and successfully managed endoscopically with balloon dilation in three sessions; another was not present for dysphagia and a standard 10-mm diameter endoscope could be passed through. In this cohort, none of our patients experienced dilation-related adverse events such as bleeding or perforation. 37
In our study, the noncurative resection rate was 19.5% (100/512). The most common reason for noncurative resection of EGCC was SM2 invasion (72/100, 72%); the risk of noncurative resection of intramucosal carcinoma was 9.0% (9/100) and that of submucosal carcinoma was significantly increased to 91.0% (91/100) (p < 0.001). Therefore, it is important to accurately predict the invasion depth of the EGCC preoperatively. However, the gastric cardia is a particularly constricted region located at the most proximal part of the stomach. Therefore, accurate targeted biopsy can be difficult because of the sharp angle and narrow lumen. Diagnosing very early lesions in this location is difficult. The precision of evaluation for the invasion depth by endoscopic ultrasonography (EUS) is low for lesions located in the cardia. When the lesions were located near the heart, it is difficult to position the ultrasound transducer optimally, with resultant pseudo-thickening and a poor visualization of the gastric wall layers. These limitations result in an inability to accurately assess the invasion depth of the EGCC by EUS. Magnifying endoscopy (ME) with narrow band imaging (ME-NBI) is a useful modality for detailed visualization of microstructures and microvessels within the superficial layer of the gastric mucosa. However, ME could not accurately assess the infiltration depth. Although the depth of invasion was deep at the time of diagnosis, we found that the lymph node metastasis rate of early gastric cardia cancer was lower than that of non-cardiac early cancer. Our previous study found the risk of lymph node metastasis was significantly lower in early gastric cardiac cancers (6.7%, 33/495), compared with early gastric non-cardiac cancers (17.1%, 275/1606) (p < 0.0001). The frequency of lymph node metastasis was significantly lower for 33 early gastric cardiac cancer cases with 10/115 (8.7%) for SM1 and 23/187 (12.3%) for SM2, compared with early gastric non-cardiac cancers (26.1%, 213/817) with 58/316 (18.4%) for SM1 (p < 0.05) and 155/501 (30.9%) for SM2 (p < 0.0001), respectively. 11 Although the depth of invasion was deep in early gastric cardiac cancer, these results lend support to the role of endoscopic therapy in the treatment of patients.
In this large cohort of EGCCs, we showed several different clinical, endoscopic, and pathologic features, compared with EGNCs, such as older age, more gross pattern of 0–IIc, fewer ulcerated cases, smaller tumor size, deeper invasion, and better tumor differentiation, which may help design better diagnostic and management strategy in the future. The rates for complete and curative resections were slightly lower in the EGCC group than EGNC group. In our cohort, most patients showed H. pylori infection with atrophic gastric carditis, similar to those in EGNC patients. The results suggest pathogenesis mechanisms of EGCC in our patient population may be different from those reported in the Western countries, in which gastroesophageal reflux disease with a high gastric acid levels and atrophic carditis plays an important role. 38
In the present study, 19 patients with curative resection were treated by additional surgery due to psychological burden. In China, many patients lack medical knowledge. They are worried about tumor recurrence and lymph node metastasis. More than half of patients with a noncurative resection (53.3%, 49/92) refused additional surgery for a variety of reasons, such as poor quality of life after surgery, advanced age, and severe comorbidities. However, the disease-specific survival benefits of those patients were found to be significantly inferior to those of the patients with additional surgery after a noncurative resection, as previously reported. 39 Apparently, additional surgery in qualified cases remains necessary to achieve a better survival outcome.
In this study, metachronous cancer was detected as early gastric carcinoma in six (1.2%, 6/499) patients approximately 23 months, on average, after curative ESD resections; it was successfully resected endoscopically. Although the incidence of metachronous tumors after EGCC resection is considerably lower than that (21.2%) of Barrett’s neoplasia therapy, 40 all patients with EGCC should be monitored carefully in the follow-up endoscopy after curative ESD resections because chronic carditis-associated intestinal metaplasia and atrophy are widespread in our patient populations. This is especially important for those with noncurative resections, although local recurrence in our study was detected during the follow-up surveillance in only one patient with noncurative resection without surgery because of the short follow-up period. Some studies have shown that local recurrence and distant metastasis are not detected in patients with curative ESD resection during follow-up surveillance.36,41 However, Jang et al. showed local recurrence in two patients after curative ESD resection. 16 Therefore, close follow-up endoscopy remains one of the essential clinical management strategies to detect local recurrence and metachronous cancer in EGCC patients after curative or noncurative ESD resection.
Our study had several limitations. First, this retrospective single-center study is subject to potential selection bias. However, we executed the investigation with a set of strict selection criteria for consecutive qualified cases, which minimized potential selection bias. Second, this study was conducted in a major tertiary medical center with extensive experience in the ESD procedure for EGCC; thus, our results may not be generalizable to all centers for every ethnic patient population. Despite these limitations, a strength of this study was the inclusion of the largest number of ESD-treated EGCC cases with a strict investigation protocol, and we showed for the first time the excellent data on safety and long-term outcomes in 499 EGCC patients.
In conclusion, our study showed a high curative resection rate for ESD-resected EGCC tumors, with very few ESD procedure-related adverse events. Additional surgical resection with lymph node dissection should be performed after a noncurative resection to improve patient long-term survival. Close follow-up surveillance should be conducted in all patients after the ESD procedure to detect local recurrence and metachronous cancer.
Footnotes
Author contributions
Study concept and design: Qin Huang, Lei Wang, Xiaoping Zou, Guifang Xu; acquisition of data: all authors; performed endoscopic submucosal dissection: Xiaoqi Zhang, Tingsheng Ling, Lei Wang, Xiaoping Zou, Guifang Xu; pathological examination: Qi Sun, Tianyun Liu, Xiangshan Fan; analysis and interpretation of data: Yi Wang, Bin Zhang, Ying Lv; the manuscript: Shouli Cao, Ting Fan, Qin Yin, Jingwei Jiang, Dekusaah Raymond; critical revision of the manuscript for important intellectual content: all authors; statistical analysis:, Yuzheng Zhuge, Shouli Cao.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval
The study protocol was approved by the Medical Ethics Committee of the Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School (the approval number: 2019-050-01).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (Grant Nos. 81572338 and 81672380, 81201909, 81602089), the Nanjing Medical Science and Technology Development Program (Nos. YKK12072, YKK15061 and YKK16078). This work was also part of a C-class sponsored research project of the Jiangsu Provincial Six Talent Peaks (WSN-078). Jiangsu Province “333 High-level Talents Training Project (2016-III-0126).
Guarantors
Qin Huang, Lei Wang, Xiaoping Zou, Guifang Xu.
