Abstract
Background and aim:
Many female inflammatory bowel disease (IBD) patients report symptoms exacerbation before and during menses. Our aim was to characterize their symptoms and specific risk factors in comparison with healthy controls.
Methods:
Female IBD patients aged 18–50 years were asked to fill out a questionnaire recording their demographic and disease characteristics, menstruation history and symptoms. Disease activity was defined by Harvey–Bradshaw index (HBI) for Crohn’s disease (CD) patients and modified mayo score for ulcerative colitis (UC) patients. Healthcare providers answering an internet survey assessing bowel symptoms during menstruation served as healthy controls.
Results:
A total of 139 IBD patients, of whom 100 were CD patients, filled the questionnaire. The mean age was 30.4 [±7.7 standard deviation (SD)], mean disease duration was 7.8 (±6 SD), mean HBI was 4.7 (±3.8 SD), and mean Mayo score was 2.1 (±2.5 SD). A change in bowel habits during menstruation was reported by 72% of CD patients compared with 56% of UC patients (p = 0.07). Out of 258 healthy controls, 93% reported a change in bowel habits during menstruation compared with 68% of IBD patients (p < 0.001). However, other abdominal and constitutional symptoms were significantly more prevalent among IBD patients compared with healthy controls (p < 0.01 for most parameters). Smoking status, biologic treatment, and previous abdominal operation were found to significantly aggravate symptoms during menses in IBD patients
Conclusion:
IBD patients experience various symptoms during menses significantly more commonly than healthy women. Smoking, biologic treatment, and previous abdominal operations are risk factors for higher symptomatic burden. Following future validation and research, these results can help in patients’ risk stratification and possibly in risk reduction.
Keywords
Introduction
Changes in gastrointestinal (GI) function during menstrual cycle are a well-known phenomenon that might be attributed to the presence of sex hormones receptors along the GI tract.1,2 Although these changes affect healthy women, they are more pronounced in women with inflammatory bowel disease (IBD), as well as in women with other abdominal inflammatory diseases such as familial mediterranean fever (FMF) and Bechet’s disease.3–5
To date, only a few studies have addressed the issue of altered GI symptoms during the menstrual cycle in female IBD patients. Data from these studies suggests disease aggravation during pre-menstrual and menstrual phases.3–7 The exact mechanism for this disease exacerbation is yet to be determined, however, a possible role for prostaglandins (PG) was suggested. During menstruation there is an increase in uterine PGs, predominantly PGF2a and prostacyclin, which are known to possess a stimulatory effect on gut motor activity. 8 Moreover, PGs play a role in inflammation in IBD, and high levels of PGS released by the endometrium during menses might exacerbate disease symptoms.
An additional possible mechanism is connected to the influence of estrogen on the GI tract. Recent data shows a wide spread of estrogen receptors along the GI tract.
The estrogen receptor has three subtypes: estrogen receptor α (ERα), estrogen receptor β (ERβ), and the G protein-coupled estrogen receptor 1 (GPER). 9 Both ERα and ERβ are membrane receptors and are differently expressed along the GI tract. While the upper GI tract contains both receptors, ERβ is the predominant ER subtype expressed in colon tissues and was shown to maintain normal epithelial architecture protecting against chronic colitis.10–13
Furthermore, irritable bowel syndrome (IBS) symptoms were shown to be connected to hormonal status, and GPER-mediated estrogenic effects were involved in the regulation of visceral pain and GI motility in IBS patients.14,15 Hence, changes in estrogen levels along the menstrual cycle may have major influence on inflammation and motility in IBD patients.
Although IBD exacerbation during menses was shown in previous studies,3–7 data regarding risk factors for such a menstrual-induced clinical deterioration are scarce. Furthermore, the control groups were usually non IBD patients with other diseases, rather than healthy women, which might cause a selection bias. Therefore, in this study, we tried to characterize specific risk factors for disease exacerbation during menses, while restricting the control group to healthy women exclusively.
Patients and methods
To be included in the study the patient had to be a female IBD patient under regular follow-up of the GI clinic at the Chaim Sheba Medical Center, a tertiary center setting, and to be between the age 18–50, not pregnant, and with regular menses. IBD diagnosis was defined by conventional clinical, radiographic, endoscopic, and histological criteria. Inclusion was performed after at least 6 months of established disease. During routine visits to the clinic, patients were asked to fill out a questionnaire addressing symptoms during their pre-menses and menses period. Disease activity scores was calculated by the treating physician as part of the regular follow-up. Patients with Crohn’s disease (CD) are routinely evaluated by the Harvey–Bradshaw index (HBI) for Crohn’s disease patients, and patients with ulcerative colitis (UC) by the modified mayo score. Questionnaires were included in the analysis only when more than 95% of the items were addressed. To increase the sensitivity to temporal changes in symptoms during menses only patients in remission or with mild-moderate activity at the time of the study were included in the study reasoning that patients with moderate-severe active disease are often continuously inflicted by severe symptoms. Patients with known gynecologic disease including endometriosis were excluded for analysis.
This study has been approved by the Chaim Sheba Medical center ethics committee (protocol number 0720-13). Informed consent was obtained from all study participants.
Questionnaires addressed specific symptoms attributed to menses and pre-menses period in IBD patients according to the literature,4,7 as well as background questions concerning menstrual cycle and gynecological and obstetrical data including age of first menses, medium length of menses and of menstrual cycle, bleeding severity, use of contraceptive medications (including intra-uterine devices), and number of pregnancies and births. IBD specific questions included disease duration, medical treatment, and history of abdominal and pelvic operations. Patients were also asked about smoking and regular physical activity. The full questionnaire is shown in Supplemental document 1.
Regarding menses symptoms, patients were asked to address symptoms in their last 2 menses and grade their severity between 1 (not at all) to 5 (very much). Patients were asked to address both pre-menses (PMS) phase, which was defined as 1–5 days before menses, and the days of menses itself. List of all symptoms addressed was chosen according to the work of Kane et al. 4 Specific attention was attributed to change in bowel movements during menses and to IBD symptoms aggravation during menses.
Healthy female members in a closed Facebook group of female physicians and dieticians served as controls. They were asked to fill an internet questionnaire addressing their age, chronic disease, chronic medications, and symptoms during menstruation. Only healthy women (without any chronic disease and not taking any chronic medical treatment according to their own statement) aged 18–50 were included for analysis.
Statistical analysis
All statistical analyses were performed using SPSS program (IBM SPSS statistics VER 25, IBM CORP ARMONK, NY, USA, 2015).
A t-test was used to evaluate difference in mean values of continuous variables such as age, disease duration, age of onset of the disease, duration of the menses, and the menstrual cycle in the IBD and control groups. Mann–Whitney test for non-parametrical variables was used for assessment of relationship between health status, smoking habit, type of biological treatment, disease level, participants’ physical activity, and reported outcomes. Chi square test and Fisher’s exact test were used for assessment of relationship between health status and reported outcomes. Logistic regression was used to evaluate influence of age and health status on reported outcomes. Two-sided p values of less than 0.05 were considered statistically significant.
Results
Out of 152 female patients approached, 148 agreed to fill the questionnaire. Of them, nine patients did not meet inclusion criteria; thus 139 questionnaires were included for analysis. Of these, 39/139 (28%) were UC patients and 100/139 (72%) were CD patients. Patients’ characteristics and demographic data are shown in Table 1, and patient’s specific disease characteristics and life style habits are shown in Table 2. Mean patient age was 30.4 ± 7.7 years, and mean disease duration was 7.8 ± 6 years. Mean HBI was 4.7 ± 3.8 and mean partial Mayo score was 2.1 ± 2.5.
Patients’ demographic characteristics.

CD, Crohn’s disease; HBI, Harvey–Bradshaw index; SD, standard deviation; UC, ulcerative colitis.
Patients’ disease characteristics and lifestyle habits.

ASA, aminosalicylic acid; CD, Crohn’s disease; HBI, Harvey -Bradshaw index; UC, ulcerative colitis.
Crohn’s patients Montreal classification- 20% A2L3B2, 15% A2L3B2P, 40%- A2,L3,B1, 20%- A2L1B1, 5%- A2,L2,B1.
Results of comparisons between UC and CD patients are shown in Table 1. No significant difference was found between patient groups regarding demographics, disease background history, or menses characteristics. Most patients were either in clinical remission or suffered from mild disease (82%) at the time of questionnaire filling. However, a larger percentage of UC patients were in clinical remission (79% versus 60% of CD patients, p = 0.045), and a larger percentage of CD patients suffered from moderate disease activity (23% versus 3% in UC patients, p = 0.005).
We next assessed specific symptom differences between the two patient groups (CD versus UC; Mann–Whitney non-parametric test for two independent samples). A trend towards higher rate of change in bowel habits in CD compared with UC patients was found, whereby 72/100 (72%) of CD patients reported change in bowel habits during menses compared with 22/39 (56.4%) of UC patients (p = 0.07). Among other symptoms, abdominal pain was reported by 76 patients (54.7%). There was no difference in frequency of pain between CD (57/100, 57%) and UC patients (19/39, 48%) (p = 0.30). PMS and menses symptoms in the entire IBD group were evaluated for a possible impact by various demographic and disease characteristics, for instance, smoking, biologic treatment, previous abdominal operations, and physical activity (Table 3).
Specific symptoms aggravation in IBD patients according to risk factors.
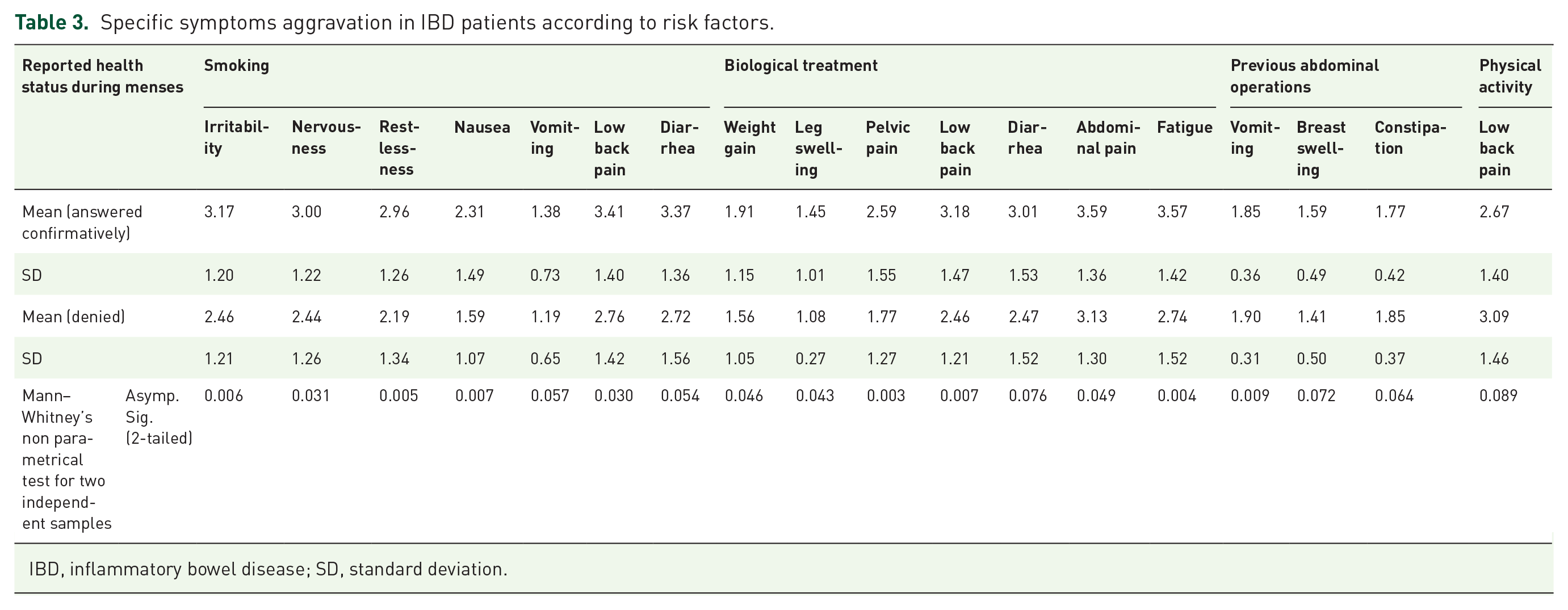
IBD, inflammatory bowel disease; SD, standard deviation.
Smoking status was found to be associated with various symptoms during menses in IBD patients. Thus, smoking patients showed a higher level of irritability (p = 0.006), nervousness (p = 0.031), restlessness (p = 0.005), nausea (p = 0.007), vomiting (p = 0.05), and low back pain (p = 0.03; Table 3).
Biologic treatment was also found to be associated with patients’ symptoms during both PMS and menses periods. During the pre-menstrual period, patients receiving biologic treatment reported significantly higher levels of irritability (p = 0.008), nervousness (p = 0.007), leg swelling (p = 0.05), pelvic pain (p = 0.02), and fatigue (p = 0.001; Table 3). During menses, patients who received biologic treatment reported pelvic pain (0.003), low back pain (0.007), abdominal pain (0.04), and fatigue (p = 0.004) at a significantly higher level than in those who did not receive biologic treatment (Table 3).
A total of 24% of patients (of them, 85% CD patients) underwent previous abdominal operations. Previous abdominal operation significantly increased the rate of vomiting during menses (p = 0.009) and tendency to constipation (p = 0.06; Table 3).
Current disease activity, assessed by HBI/partial Mayo score, age, disease duration, Montreal classification, regular physical activity, age at first menstruation, use of contraceptives, menstruation duration, and the length of menstruation cycle, did not affect patients’ symptoms during both pre-menstrual and menstrual periods.
Symptoms comparison between IBD patients and healthy controls
A total of 258 healthy female physicians and dieticians aged 20–50 (mean age 32 ± 7) served as the control group. Surprisingly, 240 women (93%) reported change in bowel habits during menstruation. Of them, 118 (49%) reported an increase in bowel movements and 108 (44%) reported frank diarrhea. Only 16 women (7%) reported constipation during menstruation.
Comparing IBD patients with healthy controls showed significantly higher frequency of changes in bowel habits during menstruation in healthy women (68.1% compared with 93%, p < 0.001). However, specific questions assessing diarrhea or worsening of diarrhea in patients group compared with controls did not differ between the two groups (46% versus 41%, respectively, p = 0.4).
In contrast, multiple other specific symptoms during menses were experienced significantly more often by IBD patients compared with healthy controls. As shown in Table 4 and graphically in Figure 1, such parameters as irritability, nervousness, restlessness, headache, anxiety, nausea, vomiting, leg swelling, low back pain, constipation, excessive urination, abdominal pain, and fatigue were all found to be significantly more common among the IBD group than in the group of healthy controls.
Menses symptoms in IBD patients compared with healthy controls.

CD, Crohn’s disease; CI, confidence interval; IBD, inflammatory bowel disease; OR, odds ratio; UC, ulcerative colitis.

Menses symptoms in IBD patients compared with healthy controls.
Discussion
Many female IBD patients report various symptoms aggravation during PMS and menses periods. Discriminating disease exacerbation from menstrual symptoms is an important and common clinical dilemma for both patients and physicians. While minor symptoms can be treated expectantly with anti-emetics or analgesics, severe symptoms may necessitate further diagnostic steps, including laboratory and imaging examinations, and can lead to change in therapeutic decisions.
Characterization of specific symptoms and risk factors for menses-related symptoms in IBD may help in both symptom recognition and categorization, in advising lifestyle modifications in order to improve patients’ symptoms, and to avoid unjustified IBD therapy changes. Thus, in cases with a high probability of menses related symptoms, patient reassurance and specific symptom-directed treatment (as pain killers) might be advised.
In our current study we evaluated female patients with established IBD (CD and UC), examined various symptoms during PMS and menses, and compared their answers with those of healthy female controls.
In agreement with a previous publication,4,16 CD patients had a tendency (although not statistically significant) towards change in bowel habits during menses (p = 0.07). However, surprisingly, and in contradiction to some previous publications4,6–7 we found that our control group reported higher rates of change in bowel habits during menses compared with IBD patients (93% vs 68%, respectively, p < 0.0001). In contrast, most other specific symptoms during menses – irritability, nervousness, restlessness, headache, anxiety, nausea, vomiting, leg swelling, low back pain, constipation, excessive urination, abdominal pain, and fatigue – were all more common among IBD patients. We speculate the reason for the lack of rate difference for bowel habits might be that IBD patients are more accustomed to alerted bowel habits, thus a minor change (such as 1–2 extra bowel movement per day) may not impact their perception of bowel habits and will be under-reported. Our results are in line with a recent prospective study 3 assessing the effect of menstrual cycle on IBD symptoms, which also found that IBD patients experience more frequent systemic symptoms than controls, but their disease specific symptoms did not change significantly during menses.
Estrogens’ effect on visceral sensitivity, motor, and sensory function of the GI tract is well established in the literature, both in animal models17,18 and in human studies,14,15,19 and all symptoms reported are in line with current data.6,7,20,21
High levels of PG might also mediate an inflammatory response characterized by abdominal pain.22,23 Along the GI tract, PG may provoke smooth muscle contractions as well as reduced absorption and increased small bowel electrolytes secretion, which may cause gastrointestinal pain and diarrhea. 23
In agreement with our results, a recent study assessing gastrointestinal symptoms before and during menses in healthy women 20 found that 73% of healthy women experienced at least one GI symptom either pre-menses or during menses, with abdominal pain (58%) and diarrhea (28%) being the most common. Fatigue was the main general symptom and was expressed by 53% of patients. Interestingly, patients experiencing emotional symptoms were more likely to suffer from multiple GI symptoms.
In addition to comparing patients’ symptoms with healthy controls, we characterized specific risk factors for higher symptomatic burden during PMS and menses. The results show that smoking is significantly associated with an increased rate of various symptoms including irritability, nervousness, restlessness, nausea, and low back pain during menses in IBD patients (Table 3). These findings are in line with a previous study assessing risk factors for pre-menstrual symptoms in the general population, 24 which similarly identified smoking as one of the major causes for severe intensity of symptoms.
Interestingly, another risk factor for symptom aggravation during both pre-menstrual and menstrual periods was biologic treatment. The specific reason for this association was not investigated in the present study but may relate to biologic treatment being a marker of a more severe underlying disease. Similarly, a previous abdominal operation correlated significantly with an increased risk of vomiting during menses. This may be related to severity of underlying IBD but may also stem from post-surgical adhesions in the abdomen and pelvis, which can aggravate symptoms.
Furthermore, current data suggest that symptoms of depression and anxiety can influence GI symptoms severity in IBD patients.25,26 Naturally, and as shown in previous research, 27 disease activity affects and enhances anxiety and depression among these patients. Therefore, it is reasonable to assume that patients suffering from severe disease (as marked by biologic treatment or previous operation) are more likely to suffer from emotional distress which in turn influence menstrual GI symptoms. Results of a recent study showed that IBD patients reporting symptom aggravation during hormonal fluctuation had a lower quality of life and were diagnosed at an earlier age 16 – risk factors that correlate with disease severity and further support our findings.
In line with previous data, 16 use of contraceptives did not correlate with GI symptoms in our patients. In a large patient cohort, 16 85% of 613 patients using hormonal contraceptives reported no change in IBD symptoms, while 3% reported improvement, and nearly 7% reported worsening. In a smaller patient cohort, a phone survey conducted on 129 women treated with hormonal contraceptives 28 showed no change in symptoms in 75% of patients, improvement in 20%, and worsening in 5% of patients.
To the best of our knowledge, this is the first study assessing specific risk factors for symptoms exacerbation during menses. If corroborated by additional studies, they may add to patient risk stratification and possibly to risk reduction and unjustified IBD treatment changes.
There are several strengths to the present study. It included a relatively large patient and control group compared with previous studies, which can increase its validity. The use of an internet questionnaire in closed groups of female healthcare professionals enabled us to reach a very large control group with high credibility.
Our study has several limitations. There was a slight but significant age difference between IBD and control groups. However, on multivariable logistic regression analysis, this difference did not independently associate with menses-related symptom reporting. A second limitation is the use of female healthcare workers as our control group, which may possibly introduce some bias. However, we reasoned that professional health workers will be more reliable regarding their medical condition and treatment.
In conclusion, in this study we found that, with the exception of altered bowel habits, various abdominal and non-specific systemic symptoms during pre-menstrual and menstrual periods afflict IBD patients significantly more often than in healthy women. Smoking, biologic treatment, and previous abdominal operations are risk factors for higher symptomatic burden. If corroborated, these results can assist in patient risk stratification and astute management.
