Abstract
Background:
We performed a post hoc secondary analysis for the effect of body mass index (BMI) on the efficacy, tolerability, and safety of ready-to-drink sodium picosulfate, magnesium oxide, and citric acid (SPMC oral solution) bowel preparation.
Methods:
A phase III, randomized, assessor-blinded, multicenter, noninferiority study was conducted comparing split-dose, low-volume SPMC oral solution with a powder formulation for oral solution. A post hoc secondary analysis assessed efficacy, safety, and tolerability of SPMC oral solution stratified by BMI. BMI was classified by Centers for Disease Control and Prevention definitions (underweight and normal weight: BMI < 25 kg/m2; overweight: BMI 25–29.9 kg/m2; class I obesity: BMI 30–34.9 kg/m2; class II obesity: BMI 35–39.9 kg/m2; class III/severe obesity: BMI ⩾40 kg/m2). Prespecified primary efficacy endpoint (‘responders’) was the proportion of participants with ‘excellent’ or ‘good’ ratings on a modified Aronchick Scale (AS). Secondary efficacy outcomes were the quality of cleansing of the right colon as assessed by the Boston Bowel Preparation Scale (BBPS); as well as selected findings from the Mayo Clinic Bowel Prep Tolerability Questionnaire. Safety assessments included adverse events (AEs) and laboratory evaluations.
Results:
Between 82.8% and 92.5% of participants in any BMI group were responders by AS, and between 91.3% and 100% were responders by BBPS in the right colon. Efficacy was consistent across BMI groups, with no clear trends. Greater than 83% of participants in any BMI group found the preparation ‘easy’ or ‘acceptable’ to ingest, and the majority (>58%) rated SPMC oral solution as ‘better’ than a prior bowel preparation. In all BMI groups, safety data were similar to the overall cohort. Commonly reported, drug-related, treatment-emergent AEs were, by ascending BMI group, nausea (1.1%, 5.3%, 1.0%, 5.7%, and 0%) and headache (1.1%, 4.1%, 1.0%, 5.7%, and 0%).
Conclusions:
Ready-to-drink SPMC oral solution had consistent, good quality colon cleansing, and favorable tolerability among participants of all BMI groups.
ClinicalTrials.gov Registration:
NCT03017235
Keywords
Introduction
Regular colorectal cancer (CRC) screening for people over 50 years old has been shown to be an important step to reduce CRC incidence and mortality.1–4 Furthermore, certain individuals are at higher risk of developing CRC and should follow their clinicians’ recommendations to undergo CRC screening at recommended intervals.5–7
Adults with obesity [body mass index (BMI) ⩾30 kg/m2] have an approximately 30% greater risk of developing CRC compared with individuals with lower BMI.8,9 Adults with obesity have worse disease-free survival and increased risk of cancer recurrence compared with individuals of normal BMI. 10 Likewise, higher BMI is associated with poorer CRC prognosis, including a 14% increased risk of cancer-specific mortality compared with adults of normal BMI. 11 Adults with CRC who have low BMI (<20 kg/m2) also have a 50% increased risk of cancer-specific mortality compared with those with normal BMI. 10
An effective bowel preparation is essential for an optimal CRC screening colonoscopy,12–15 but BMI has been shown to affect the quality of bowel preparation. Higher BMI has been cited as an independent risk factor for poorer quality of bowel preparation, with estimates of up to 46% higher risk of inadequate bowel preparation for those with BMI ⩾30 kg/m2.16–19 However, the rates reported in the literature are not uniform, and a recent prospective, observational study did not find an association between BMI and quality of bowel preparation. 20 In real-world settings, the association of high BMI with poorer quality of bowel preparation may be related to nonadherence with dietary restrictions.21,22
Most of the available data on bowel preparation and colonoscopy outcomes by BMI are from retrospective or cohort studies, which may introduce confounding factors into the outcomes. 21
Results from a phase III, randomized, assessor-blinded, multicenter study of ready-to-drink sodium picosulfate, magnesium oxide, and citric acid (SPMC oral solution) have recently been described. 6 Here, we performed a post hoc secondary analysis for the effect of BMI on the efficacy, tolerability, and safety of SPMC oral solution to determine whether the efficacy trends by BMI in the literature are consistent in the setting of a randomized, controlled trial. This analysis is a follow-up analysis to the primary analysis, where the superiority of ready-to-drink SPMC oral solution was demonstrated compared with a powder formulation of the same ingredients.
Methods
Study design
A phase III, randomized, assessor-blinded, multicenter, noninferiority study was conducted comparing split-dose, low-volume SPMC oral solution (Clenpiq®, Ferring Pharmaceuticals Inc., Parsippany, NJ) with split-dose, low-volume sodium picosulfate, magnesium oxide, and citric acid powder for oral solution (Prepopik®, Ferring Pharmaceuticals Inc., Parsippany, NJ) [ClinicalTrials.gov identifier: NCT03017235]. Details of the full study have been published previously. 23 The study was conducted in accordance with the principles set forth in the Declaration of Helsinki and in compliance with ICH-GCP standards. The study protocol was approved by Schulman IRB (protocol #000253).
Eligible participants included females and males, 18–80 years of age, who were undergoing elective colonoscopy (screening, surveillance, or diagnostic). (Full inclusion and exclusion criteria have been published previously. 23 )
Eligible participants must have had an average of at least three spontaneous bowel movements per week for 1 month prior to the colonoscopy, and been willing, able, and competent to complete the procedure and comply with study instructions. Written informed consent was obtained at screening.
Interventions
The colon-cleansing regimen was a split-dose preparation, with one dose taken the evening before and one dose taken the same day as the colonoscopy, within 5–9 h prior to the procedure.
SPMC oral solution (two 5.4-oz doses) is a ready-to-drink formulation and was consumed as supplied (without mixing, stirring, or dilution), followed by five or more 8-oz glasses of clear liquid within 5 h of the first dose, and four or more 8-oz glasses of clear liquid within 4 h of the second dose.
In both cases, participants were instructed to maintain a diet of clear liquids from 24 h before the colonoscopy and to stop taking anything by mouth 2 h before. Immediately prior to the colonoscopy, participants returned the Mayo Clinic Bowel Prep Tolerability Questionnaire, 24 and chemistry and hematology laboratory samples were obtained. Following the colonoscopy, participants returned for visits at 1–2 days, 7 days, and 4 weeks per study protocol to assess laboratory and safety measures.
Endpoints
The primary efficacy outcome was overall quality of colon cleansing as measured by the modified Aronchick Scale (AS) prior to irrigation of the colon, assessed by a treatment-blinded endoscopist. Investigators had an average of 33 years of experience each as practicing gastroenterologists. The prespecified primary efficacy endpoint (‘responders’) by AS was the proportion of participants with ‘excellent’ or ‘good’ ratings.
Secondary efficacy outcomes were the quality of cleansing of the right colon as assessed by the Boston Bowel Preparation Scale (BBPS), as well as the findings of selected questions from the Mayo Clinic Bowel Prep Tolerability Questionnaire. The prespecified key secondary efficacy rate (‘responders’) by BBPS was the proportion of participants with a segmental score of ‘3’ or ‘2’ in the right colon. The proportion of participants with a BBPS score of ⩾2 in each of the three colon segments was calculated.
Safety assessments included adverse events (AEs), laboratory evaluations, and electrocardiograms. AEs were classified according to the Medical Dictionary for Regulatory Activities (MedDRA) version 20.1.
The endoscopist noted the number of lesions found during the colonoscopy (recorded as an AE) and removed polyps when possible and appropriate. Lesion biopsies were sent for histological analysis. All malignancies found during the study period, including colonic lesions that were determined to be cancerous, were reported as a serious AE. Polyp and adenoma findings were not a key efficacy endpoint in the study.
Statistical analysis
A post hoc secondary analysis was performed to assess efficacy, safety, and tolerability of SPMC oral solution. BMI was classified according to the Centers for Disease Control and Prevention (CDC) definitions (underweight and normal weight: BMI < 25 kg/m2; overweight: BMI 25-29.9 kg/m2; class I obesity: BMI 30–34.9 kg/m2; class II obesity: BMI 35–39.9 kg/m2; severe obesity: BMI ⩾40 kg/m2). 25
The analysis included all participants who were randomized and received at least one dose of the study drug [modified intent-to-treat (mITT) population]. Baseline and demographic characteristics were descriptively summarized.
The responder rates in the primary, key secondary, and other secondary efficacy endpoints were summarized with exact 95% confidence intervals (CIs), calculated by the Clopper-Pearson method. Tolerability endpoints were descriptively summarized.
Adenoma detection rate (ADR) and polyp detection rate (PDR) were calculated as the proportion of participants who had at least one adenoma or polyp, respectively, in the treatment group.
Results
Of the 448 participants, most (61.2%; 274/448) had a BMI of 25–29.9 or 30–34.9 kg/m2 (Table 1). There were few participants with a BMI ⩾40 kg/m2. There were two individuals (2.2%; 2/93) in the BMI < 25 kg/m2 group who were ‘underweight’ by CDC definition (BMI < 18.5 kg/m2). The mean age was consistent across BMI groups (by ascending BMI group: 56.6 years, 57.7 years, 57.7 years, 57.5 years, 54.9 years). Type 2 diabetes prevalence increased by ascending BMI group.
Demographic and baseline characteristics in BMI subgroups, mITT population.

BMI, body mass index; mITT, modified intent-to-treat; SD, standard deviation.
Efficacy
By ascending BMI group, 82.8%, 90.1%, 85.4%, 92.5%, and 89.3% of participants receiving SPMC oral solution were responders (those with ‘excellent’ or ‘good’ ratings) for the primary efficacy endpoint, overall colon cleansing by modified AS (Table 2; Figure 1). There was no consistent pattern of responder rate by BMI. Rates of ‘inadequate’ rating, by ascending BMI group, were 1.1%, 0.6%, 1.9%, 0%, and 0%.
Primary efficacy endpoint, overall colon cleansing quality by modified AS, mITT population.
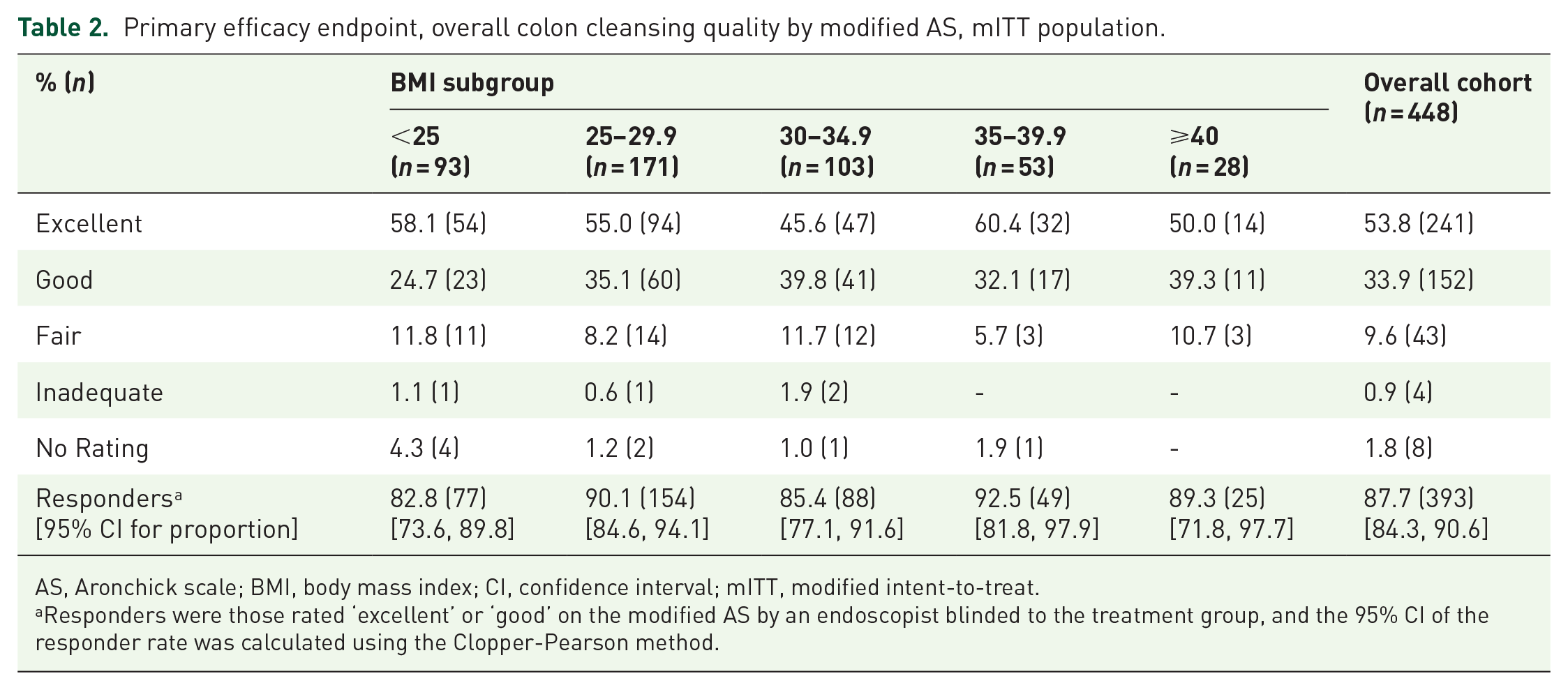
AS, Aronchick scale; BMI, body mass index; CI, confidence interval; mITT, modified intent-to-treat.
Responders were those rated ‘excellent’ or ‘good’ on the modified AS by an endoscopist blinded to the treatment group, and the 95% CI of the responder rate was calculated using the Clopper-Pearson method.

In the SPMC oral solution treatment arm, a substantial majority of participants in each BMI group were responders on the modified AS, rated by a treatment-blinded endoscopist. Likewise, at least 87% of participants had a BBPS score of 2 or better in all 3 colon segments. The efficacy ratings were consistent across BMI groups.
For the key secondary efficacy endpoint, responders by BBPS in the right colon (those with a rating of ‘3’ or ‘2’), overall 94.2% of participants receiving SPMC oral solution were responders. There was no consistent pattern in responder rate by BBPS in the right colon across BMI groups (by ascending BMI group: 92.5%, 95.3%, 91.3%, 96.2%, 100%) (Table 3). The mean total BBPS score by BMI group was similar to the overall population score of 7.7 (out of a possible 9; Table 3). At least 87% of participants in each BMI group had a BBPS score of 2 or better in each of the three colon segments (Figure 1; Table 3).
Colon cleansing quality by BBPS, mITT population.

BBPS, Boston Bowel Preparation Scale; BMI, body mass index; CI, confidence interval; mITT, modified intent-to-treat.
Responders were those rated ‘3’ or ‘2’ on the BBPS by an endoscopist blinded to the treatment group, and the 95% CI of the responder rate was calculated using the Clopper-Pearson method.
Tolerability
Depending on the BMI group, 96.4–100% of participants were able to complete the majority of the bowel preparation (at least 75% of preparation consumed). At least 83.0% of participants in each BMI group stated that SPMC oral solution was ‘easy’ or ‘acceptable’ to ingest (Figure 2). Of participants who had experience with a prior colonoscopy, the majority (>58%) of those in the SPMC oral solution arm rated the preparation as ‘better’ than a prior bowel preparation (Figure 3). There were no clear, consistent trends of tolerability by BMI in these data.

Participants were asked ‘Was the bowel preparation tolerable?’ on the Mayo Clinic Bowel Prep Tolerability Questionnaire. At least 83% of participants in each BMI group indicated that SPMC oral solution was ‘easy’ or ‘acceptable’ to ingest.
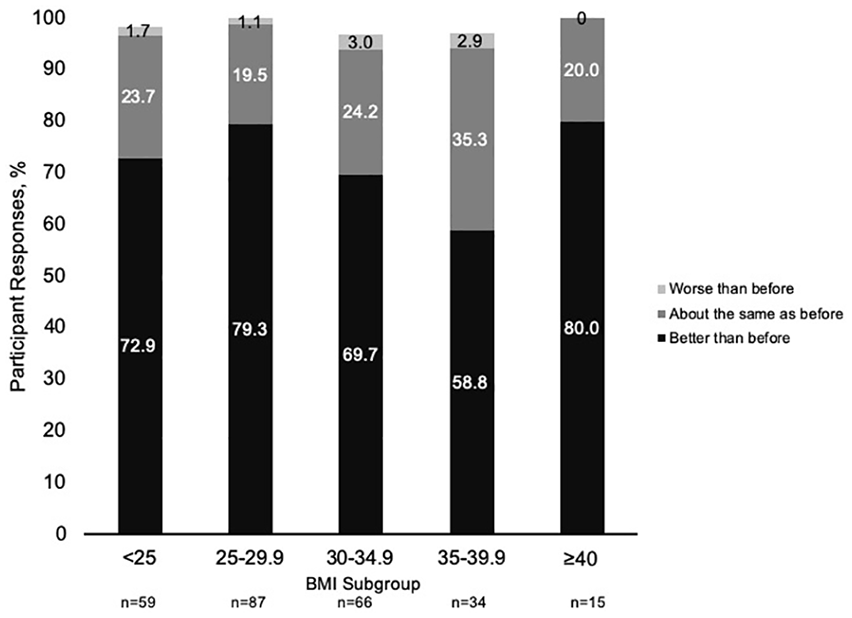
Participants who had experience with a prior colonoscopy (51–64% of each BMI cohort) were asked to rate the tolerability of the study bowel preparation compared with the previous preparation. The majority of participants rated SPMC oral solution bowel preparation as ‘better’ than a previous preparation. No consistent trend of responses was seen across BMI groups. Participants with no response are not shown on the graph and, therefore, numbers may not add to 100%.
Polyp detection
PDR varied by BMI group, without any observable trend in the data (Figure 4). PDR ranged from 39.3% for those with BMI ⩾40 kg/m2 to 62.3% for those with BMI 35–39.9 kg/m2. ADR values were similar across BMI groups, with no consistent trend (by increasing BMI group: 30.1%, 28.7%, 33.0%, 39.6%, and 32.1%).

The PDR varied by BMI, with no consistent trend. ADR was above the guideline-recommended target for all BMI groups. Any polyps found during the colonoscopy were removed, recorded as AEs, and sent for histological analysis. PDR and ADR were calculated as the percentage of any participants who had at least one polyp or adenoma, respectively.
Safety
Across all BMI groups, there were no deaths, no treatment-emergent adverse events (TEAEs) leading to study discontinuation, and no serious adverse drug reactions (Table 4). Rates of serious TEAEs were ⩽5.7% in any subgroup, with none reported for those with BMI ⩾40 kg/m2, and rates of severe TEAEs were ⩽7.5% in any subgroup. Rates of adverse drug reactions across BMI groups were largely consistent with the overall population rate of 13.2%, except for those who had BMI ⩾40 kg/m2 (3.6%).
TEAEs, safety population.
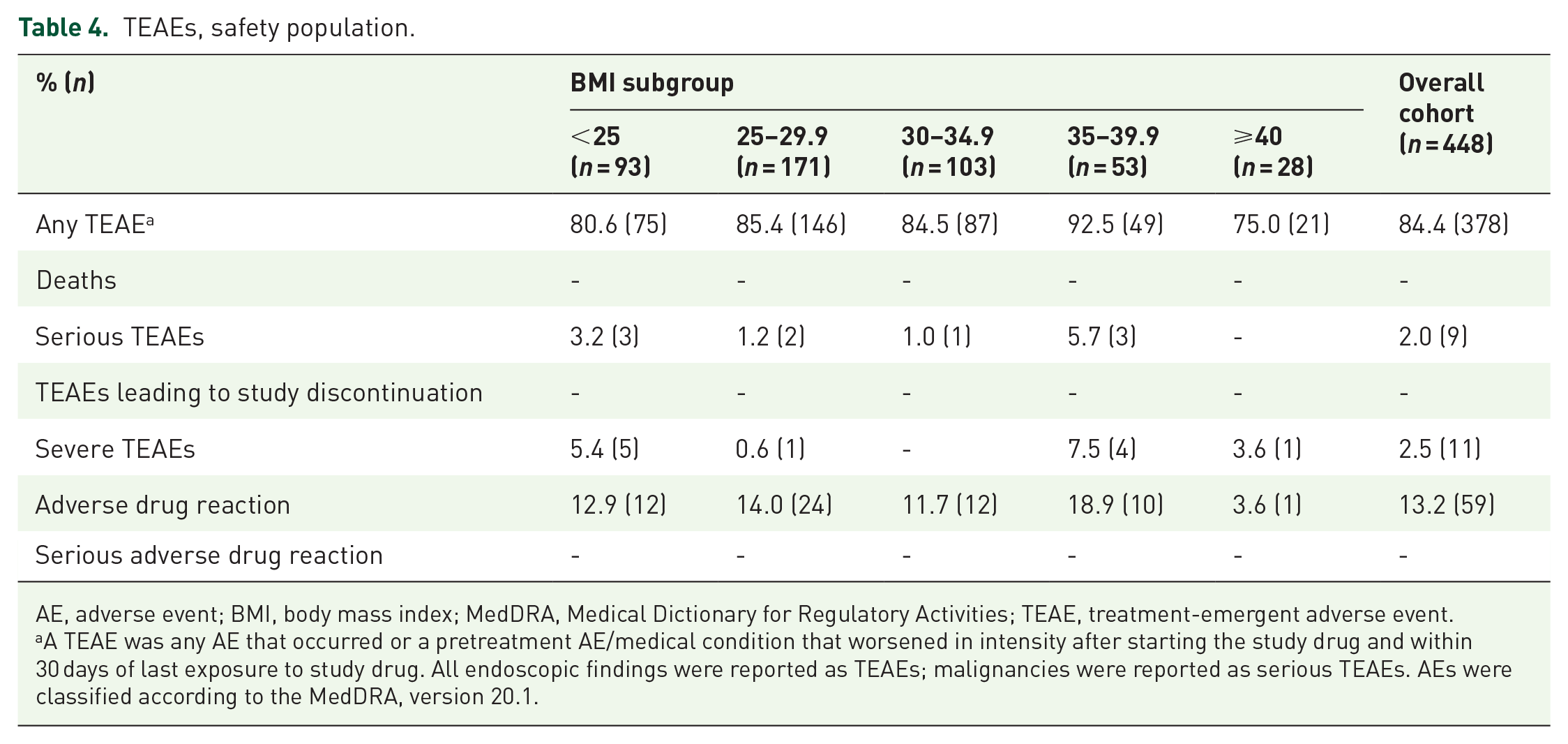
AE, adverse event; BMI, body mass index; MedDRA, Medical Dictionary for Regulatory Activities; TEAE, treatment-emergent adverse event.
A TEAE was any AE that occurred or a pretreatment AE/medical condition that worsened in intensity after starting the study drug and within 30 days of last exposure to study drug. All endoscopic findings were reported as TEAEs; malignancies were reported as serious TEAEs. AEs were classified according to the MedDRA, version 20.1.
Gastrointestinal AEs were the most frequently reported drug-related AE category in the entire study. Here, they occurred in ⩽7.5% of participants in any BMI group (Table 5). No consistent patterns of drug-related TEAEs by BMI group were evident. By ascending BMI group, rates of nausea were 1.1%, 5.3%, 1.0%, 5.7%, and 0%, and rates of headache were 1.1%, 4.1%, 1.0%, 5.7%, and 0%. Similarly, rates of hypermagnesemia were 1.1%, 2.9%, 1.0%, 3.8%, and 0%.
Treatment-emergent, drug-related AEs of interest, safety population.

AE, adverse event; BMI, body mass index; GI, gastrointestinal; MedDRA, Medical Dictionary for Regulatory Activities.
AEs were classified according to the MedDRA, version 20.1.
Discussion
CRC screening in adults with high BMI is essential due to their increased risk of developing CRC and increased risk of CRC-related mortality.8–11 A high quality bowel preparation is needed to achieve optimal CRC screening colonoscopy.12,15
Results of this subanalysis from a phase III trial of ready-to-drink SPMC oral solution displayed consistent and high efficacy of colon cleansing across all BMI groups, as measured by responder rates by AS and BBPS. There were very low rates of bowel cleansing rated ‘inadequate’ or ‘unprepared’ (score of ‘0’ by BBPS) in patients across all BMI groups. Previous studies reported an increased risk of inadequate bowel preparation by AS and BBPS for those with higher BMI16–18,20,26; however, this trend was not observed for participants receiving SPMC oral solution.
Lower BMI has also been cited as a factor in incomplete colonoscopy. 27 A retrospective review of 2000 colonoscopies revealed that 49% of women with BMI < 22 kg/m2 had a failed colonoscopy. 28 In addition to poor bowel preparation, another factor that may be related to incomplete colonoscopy in individuals with low BMI is a smaller area of adipose tissue, both the quantity and location of which may impact insertion of the colonoscope.29,30
The consistent colon cleansing efficacy of SPMC oral solution across BMI groups may be attributed to preparation factors, such as the SPMC dual mechanism of action of peristaltic stimulation and osmotic agent; dosing factors, such as split dosing and hydration instructions; or due to study design with less risk of confounding factors in the setting of a randomized, controlled trial, and a patient population that has received intense instruction and may be more motivated to adhere to dosing instructions.
Results from a recent meta-analysis suggested that individuals who receive ratings of ‘excellent’, ‘good’, or ‘fair’ by AS have adequate enough colon cleansing to follow regular intervals for CRC screening. 31 In this study, the corresponding population would be, by ascending BMI group, 94.6%, 98.2%, 97.1%, 98.1%, and 100%, which is the vast majority of each group and largely consistent across BMI. Interestingly, prior studies have reported that patients with total BBPS scores of 6–7 (‘good’ colon cleansing) had higher ADR and PDR than patients with BBPS scores of 8 or 9 (‘excellent’ colon cleansing).32,33 The BBPS allows endoscopists to influence the presence of (aspirate/wash) any residual stool in the colon prior to grading, which may affect endoscopists’ perception of the relative cleanliness of each segment. 33
Higher BMI has previously been associated with increased rates of polyp and adenoma detection. 34 Various studies have reported that individuals with high BMI have 1.2–1.8 times higher risk of adenomas than individuals with normal BMI, though the studies did not use a uniform definition of high BMI.27,35–37 Other studies have found no association between BMI and the presence of polyps or adenomas.38,39 It is unknown if the higher rates of polyps measured in individuals with higher BMI were related to biological factors that increased the absolute number of polyps, or related to earlier missed polyps because of poorer quality of bowel cleansing. 40
Our subanalysis also showed no trends in polyp detection by BMI. This could be due to high quality and consistent colon cleansing achieved in all BMI groups here, which allowed for similar visualization of polyps in all groups.38,39 ADR calculations in this study included average-risk patients undergoing screening colonoscopy and high-risk patients undergoing surveillance colonoscopy; therefore, ADR values may be slightly overestimated, given that some high-risk patients were included in the total population for calculation. As this was a post hoc secondary analysis, it was not designed to assess ADR/PDR as the primary outcome or capture confounding factors associated with ADR/PDR.
Participants receiving SPMC oral solution also reported good tolerability of the bowel preparation, with high completion rates and high rates of ‘easy’ or ‘acceptable’ ratings in all BMI groups. The majority of participants with prior colonoscopy experience also rated SPMC oral solution as ‘better’ than a prior bowel preparation. There is limited literature on the tolerability of bowel preparation by BMI, although a single retrospective review in Poland showed increasing tolerability for bowel preparation with increasing BMI. 27
The safety data were consistent across all BMI groups, with no new safety signals, indicating that SPMC oral solution is a safe bowel preparation for patients of any BMI.
A limitation of this subanalysis is the small sample size for those with BMI ⩾40 kg/m2, thus conclusions drawn from those data should be made judiciously.
The strength of this subanalysis is the use of data from a randomized, controlled trial with prespecified efficacy endpoints using validated instruments, and standardized safety reporting methods. Importantly, there are no existing large-scale studies of bowel preparation in individuals with severe obesity. Data from 2015–2016 show that 7.7% of the adult population in the United States suffers from severe obesity, and this percentage continues to rise. 41
Conclusion
Ready-to-drink SPMC oral solution had consistent and good quality colon cleansing among participants of all BMI groups, as measured by two independent and validated colon cleansing scales. In this heterogenous study population, ADR was above the ASGE/ACG Taskforce guideline-recommended targets in all BMI groups. Participants in all BMI groups reported favorable tolerability for SPMC oral solution, with most participants preferring SPMC oral solution over a prior bowel preparation. No new safety signals were seen in participants of any BMI group. SPMC oral solution should be considered for patients of any BMI who are undergoing colonoscopy.
Footnotes
Acknowledgements
Medical writing and editorial support was provided by Agnella Izzo Matic (AIM Biomedical, LLC) and was funded by Ferring Pharmaceuticals Inc. The authors would like to thank the investigators, study staff, and participants who were involved in the trial.
Author Contributions
LH, GB, KLJ, MB, MA, and DND analyzed and interpreted the data, drafted and critically revised the article for important intellectual content, and approved the article for publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Ferring Pharmaceuticals Inc., Parsippany, NJ.
Conflict of interest statement
LH has participated in the speaker’s bureau for Ferring Pharmaceuticals Inc. GB was an investigator for clinical trials sponsored by Ferring Pharmaceuticals Inc. and has served as a consultant and has participated in the speaker’s bureau for Ferring Pharmaceuticals Inc. KLJ is an investigator on clinical trials sponsored by Ferring Pharmaceuticals Inc. MB, MA, and DND are employees of Ferring Pharmaceuticals Inc.
Prior Publication
Portions of the data contained in this manuscript appeared in abstract/poster form at Digestive Disease Week 2019, May 18-21, 2019, Poster Mo1682. This manuscript has not been submitted and is not under consideration for publication anywhere else.
