Abstract
Background:
The incidence and mortality of colorectal cancer (CRC) increase with age and, therefore, it is recommended that adults undergo regular CRC screening, ideally by colonoscopy, with some new guidelines recommending screening begin at 45 years. Effective bowel preparation is a critical step to a successful colonoscopy. Of concern is that older adults may have poorer quality of bowel preparation or reduced tolerability for the bowel preparation. Here, we performed a
Methods:
A phase III, randomized, assessor-blinded, multicenter, non-inferiority study was conducted comparing split-dose, low-volume SPMC oral solution with split-dose, low-volume sodium picosulfate, magnesium oxide, and citric acid powder for oral solution. A
Results:
Within age groups, at least 83.9% of participants were responders by the AS, and at least 91.1% of participants were responders by the BBPS in the right colon. On both scales, responder rates were highest in the youngest age group and decreased with increasing age. Greater than 88% of participants in any age group found the preparation ‘easy’ or ‘acceptable’ to ingest, with rates of ‘easy’ being highest in the oldest age group. No new safety signals were seen in any age group. The most commonly reported drug-related, treatment-emergent AEs were, by ascending age group, nausea (7.0%, 3.2%, 0.8%), headache (4.2%, 2.8%, 1.6%) and vomiting (2.8%, 1.2%, 0.8%).
Conclusion:
Ready-to-drink SPMC oral solution showed good efficacy of overall colon cleansing and tolerability in adults across different age groups, including those ⩾65 years.
ClinicalTrials.gov identifier: NCT03017235.
Keywords
Introduction
Regular screening for colorectal cancer (CRC) reduces both the incidence and mortality of the disease.1–4 As certain populations show increased risk of developing CRC, it is important that these individuals undergo CRC screening at recommended intervals.5–7
The incidence of CRC increases sharply with age, with 62 cases/100,000 persons for those 50–54 years, increasing to 182 cases/100,000 persons by the ages of 75–79 years. 8 While absolute rates of CRC-related mortality have been declining over time, rates still increase with patient age, with more than a third of all CRC-related deaths occurring in those 80 years or older. 9
In a recent update to clinical practice guidelines, the American Cancer Society lowered the recommended age to begin CRC screening for average-risk individuals from 50 to 45 years, in part due to the rising incidence of CRC in adults younger than 50 years over the last 2 decades, and the documented decrease in CRC incidence after implementation of previously published screening recommendations.10–12 The younger age for screening onset has not yet been endorsed by gastroenterological societies, and there remains debate as to the most appropriate method of screening to offer. 13
In the United States, colonoscopy is the standard of care for CRC screening, as well as a tool used to visualize the colon after suspicious results are obtained from other preliminary tests for CRC. An effective bowel preparation is essential for optimal colonoscopy, including adequate detection of adenomas and sessile serrated polyps.14–17 However, in earlier studies, age has been shown to affect the quality of bowel preparation. Older adults (⩾65 years) show higher rates of inadequate bowel preparation when compared with younger adults, which may be attributed to a variety of physiological, cognitive, and functional factors.18–20
Tolerability and patient compliance have also been shown to affect the quality of bowel preparation. A prospective cohort study of consecutive adults undergoing colonoscopy demonstrated significantly lower polyp detection rates in patients who reported a less tolerable bowel preparation experience. 21 Patients who did not complete a recommended screening colonoscopy cited fear of pain and disagreeable preparation, among other factors, as major reasons for noncompletion. 22 Furthermore, data show reduced bowel preparation tolerability and compliance in older adults compared with younger adults. 23
Results from a recent phase III, randomized, assessor-blinded, multicenter study of ready-to-drink sodium picosulfate, magnesium oxide, and citric acid (SPMC oral solution)
Methods
Study design
A phase III, randomized, assessor-blinded, multicenter, non-inferiority study was conducted comparing split-dose, low-volume SPMC oral solution (Clenpiq®, Ferring Pharmaceuticals Inc., Parsippany, NJ) with split-dose, low-volume sodium picosulfate, magnesium oxide, and citric acid powder for oral solution (Prepopik®, Ferring Pharmaceuticals Inc., Parsippany, NJ) [ClinicalTrials.gov identifier: NCT03017235]. Details of the full study have been published previously. 24 The study was conducted in accordance with the principles set forth in the Declaration of Helsinki and in compliance with ICH-GCP standards. The study protocol was approved by Schulman IRB (protocol #000253).
Eligible participants included females and males, 18–80 years of age, who were undergoing elective colonoscopy (screening, surveillance, or diagnostic). Full inclusion and exclusion criteria have been published previously. 24
Eligible participants must have had an average of at least three spontaneous bowel movements per week for 1 month prior to the colonoscopy, and have been willing, able, and competent to complete the procedure and comply with study instructions. Written informed consent was obtained at screening.
Interventions
For both treatment arms, the colon cleansing regimen was a split-dose preparation with one dose the evening before and one dose the same day as colonoscopy, within 5–9 h prior to the procedure. SPMC oral solution (two 5.4-oz doses) is a ready-to-drink formulation and was consumed as supplied (without mixing, stirring, or dilution), followed by five or more 8-oz glasses of clear liquid within 5 h of the first dose, and four or more 8-oz glasses of clear liquid within 4 h of the second dose.
All participants were instructed to maintain a diet of clear liquids from 24 h before the colonoscopy and to stop taking anything by mouth 2 h before the procedure. Immediately prior to the colonoscopy, participants returned the Mayo Clinic Bowel Prep Tolerability Questionnaire, 25 and chemistry and hematology laboratory samples were obtained. Following the colonoscopy, participants returned for visits at 1–2 days, 7 days, and 4 weeks for measurements, including laboratory evaluations, physical examination, vital signs, and adverse events (AEs).
Endpoints
The primary efficacy outcome was overall quality of colon cleansing as measured by the validated Aronchick Scale (AS) prior to irrigation of the colon, assessed by the treatment-blinded endoscopist. The prespecified primary efficacy endpoint (‘responders’) by AS was the proportion of participants with ‘excellent’ or ‘good’ ratings.
Secondary efficacy outcomes were the quality of cleansing of the right colon as assessed by the Boston Bowel Preparation Scale (BBPS); as well as selected findings from the Mayo Clinic Bowel Prep Tolerability Questionnaire. The prespecified key secondary efficacy endpoint (‘responders’) by BBPS was the proportion of participants with a segmental score of ‘3’ or ‘2’ in the right colon. The proportion of participants with a BBPS score ⩾ 2 in each of the three colon segments was also calculated.
Safety assessments included AEs and laboratory evaluations. AEs were classified according to the Medical Dictionary for Regulatory Activities (MedDRA), version 20.1.
The endoscopist noted the number of lesions found during the colonoscopy (recorded as an AE) and removed polyps when possible and appropriate. Lesion biopsies were sent for histological analysis. All malignancies found during the study period, including colonic lesions that were determined to be cancerous, were reported as a serious AE. Polyp and adenoma findings were not a key efficacy outcome in the study.
Statistical analysis
A
The analysis included all participants who were randomized and received at least one dose of the study drug (modified intention to treat; mITT). Baseline and demographic characteristics were descriptively summarized.
The responder rates in the primary and key secondary endpoints were summarized with exact 95% confidence intervals (CIs) calculated by the Clopper–Pearson method. Tolerability endpoints were descriptively summarized.
Adenoma detection rate (ADR) and polyp detection rate (PDR) were calculated as the proportion of participants who had at least one adenoma or polyp, respectively, in each treatment group.
Results
A total of 448 participants receiving SPMC oral solution were included (Table 1). By ascending age group, the mean [standard deviation (SD)] ages were 38.6 (7.9) years, 56.5 (4.4) years, and 69.4 (4.1) years.
Demographic and baseline characteristics in age subgroups, mITT population.
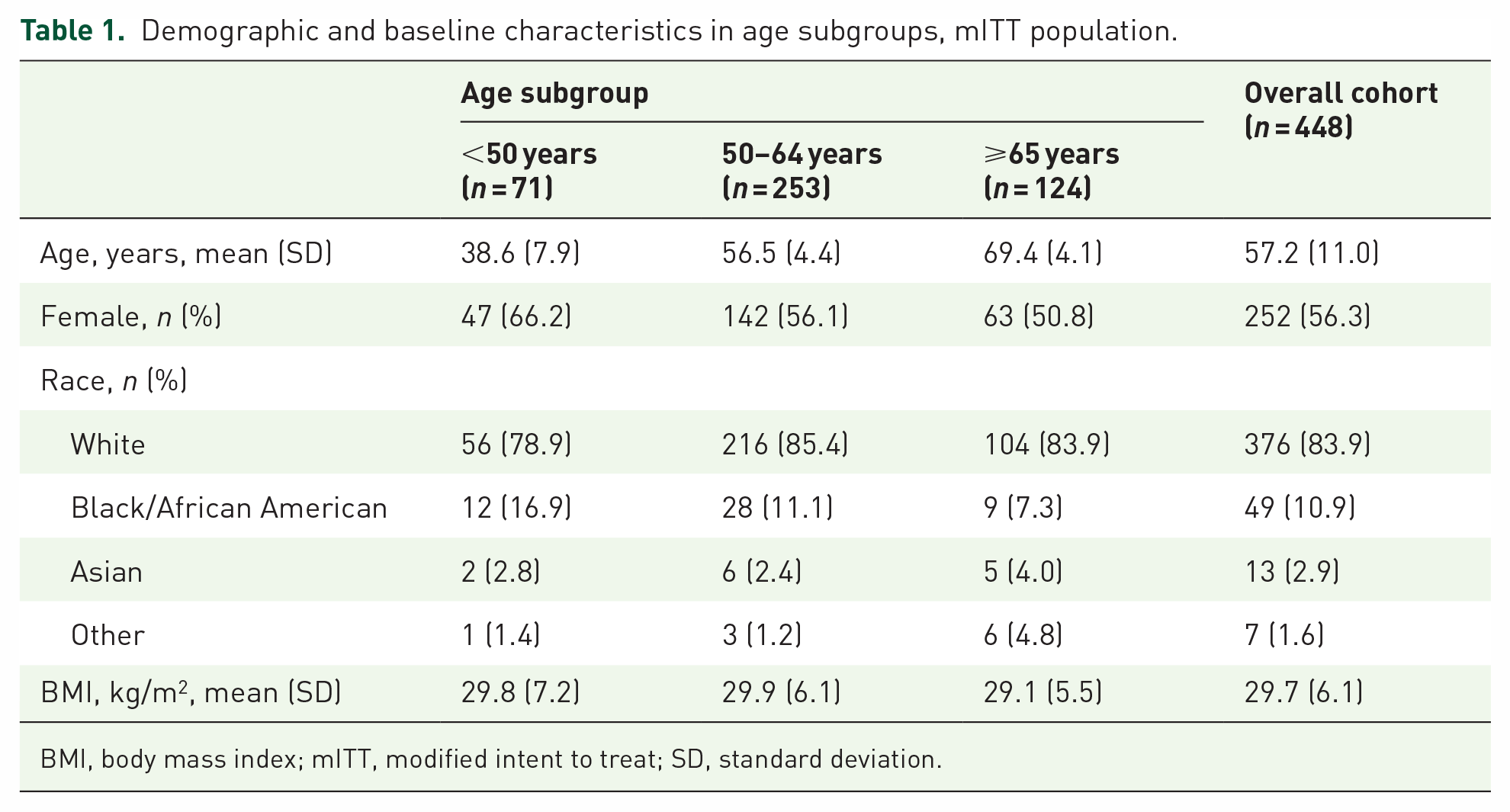
BMI, body mass index; mITT, modified intent to treat; SD, standard deviation.
Efficacy
Within age groups, at least 83.9% of participants receiving SPMC oral solution were responders (with ‘excellent’ or ‘good’ ratings) for the primary efficacy endpoint, overall colon cleansing by modified AS (Table 2, Figure 1). The responder rate was highest for those aged less than 50 years and decreased with increasing age (91.5%, 88.5%, and 83.9%, respectively). Rates of ‘inadequate’ rating by AS, by ascending age group, were 2.8%, 0.8%, and 0%, respectively; rates of ‘fair’ ratings increased with increasing age group.
Primary efficacy endpoint, overall colon-cleansing quality by modified AS, mITT population.

Responders were those rated ‘excellent’ or ‘good’ on the modified AS by an endoscopist blinded to the treatment group, and the 95% CI of the responder rate was calculated using the Clopper–Pearson method.
AS, Aronchick scale; CI, confidence interval; mITT, modified intent to treat.

Rates of participants who were responders by the modified AS or had a BBPS score of ‘2’ or ‘3’ in all three colon segments.
The key secondary efficacy endpoint was right-colon cleansing quality assessed by BBPS. Overall, 94.2% of participants receiving SPMC oral solution were responders (with a rating of ‘3’ or ‘2’). The highest responder rate, 95.8%, was observed in patients aged < 50 years, followed by the intermediate age group at 95.3%, and 91.1% for those aged 65 years and over (Table 3). When setting a threshold of a BBPS score of at least 6, with a score of 2 or more in each colon segment, 92.0% of patients in the entire cohort met this criterion. The proportion of patients who met this threshold increased as the patient age population decreased.
BBPS findings, mITT population.

Responders were those rated ‘3’ or ‘2’ on the Boston Bowel Preparation Scale by an endoscopist blinded to the treatment group, and the 95% CI of the responder rate was calculated using the Clopper–Pearson method.
BBPS, Boston Bowel Preparation Scale; CI, confidence interval; mITT, modified intent to treat; SD, standard deviation.
Tolerability
Depending on the age group, between 97.6% and 100% of participants were able to complete the majority of the SPMC oral solution (at least 75% of preparation consumed). At least 88.9% of participants in each age group found taking SPMC oral solution ‘easy’ or ‘acceptable’ (Figure 2). A greater number of older participants rated the preparation as ‘easy’ to ingest compared with younger participants. Of the participants who had experience with a prior colonoscopy, a significant majority (>70%) of those receiving SPMC oral solution rated the preparation as ‘better’ than the prior bowel preparation(s) (Figure 3).
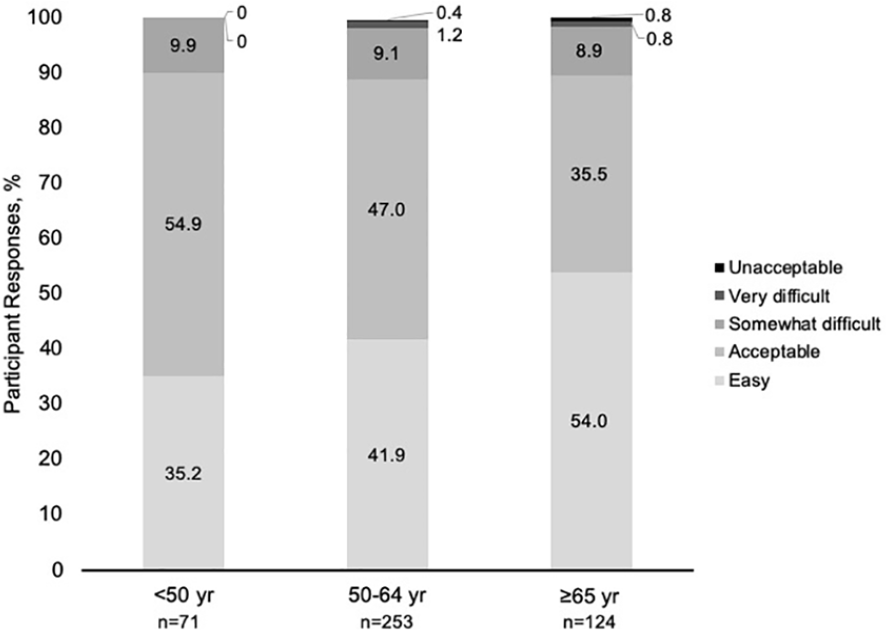
Participant tolerability by age group.

Tolerability of SPMC oral solution compared with a previous preparation.
Polyp detection
As expected, PDR and ADR increased with increasing age group (Figure 4). By ascending age group, PDR was 29.6%, 44.3%, and 56.5%. Likewise, ADR was 16.9% for those aged less than 50 years, 30.8% for those 50–64 years, and 41.1% for those 65 years and over.

Rates of polyp detection and adenoma detection
Safety
The rates of treatment-emergent AEs (TEAEs) were similar across age groups (Table 4). There were no deaths, no TEAEs leading to study discontinuation, and no serious adverse drug reactions in any group. Rates of serious TEAEs were no more than 2.4% in any age group, and rates of severe TEAEs were no more than 2.8%, with no substantial patterns by age. Adverse drug reactions occurred in 12.7–13.7% of participants by age group.
Treatment-emergent adverse events, safety population.

A TEAE was any AE that occurred or a pretreatment AE/medical condition that worsened in intensity after starting the study drug and within 30 days of last exposure to study drug. All endoscopic findings were reported as TEAEs; malignancies were reported as serious TEAEs. AEs were classified according to the Medical Dictionary for Regulatory Activities (MedDRA), version 20.1.
AE, adverse event; TEAE, treatment-emergent adverse event.
Gastrointestinal AEs were the most frequently reported drug-related AE category in the entire study. The youngest group reported these AEs most frequently (7.0%; Table 5). Rates of nausea were highest for the youngest age group (7.0%) and decreased with increasing age (0.8% for ⩾65 years). Rates of headache were also highest for the youngest age group (4.2%) and decreased with increasing age (1.6% for ⩾65 years). Rates of hypermagnesemia, by ascending age group, were 1.4%, 2.0%, and 2.4%. Upon assessing pertinent laboratory values, few participants exhibited a shift in potassium or sodium from baseline; however, shifts were transient in nature and deemed not clinically significant. No participant exhibited a severe or serious AE associated with a reduction in serum potassium (hypokalemia).
Treatment-emergent, drug-related adverse events of interest, safety population.

AEs were classified according to the Medical Dictionary for Regulatory Activities (MedDRA), version 20.1.
AE, adverse event; GI, gastrointestinal.
Discussion
To reduce CRC incidence and mortality, regular CRC screening is recommended. Currently, colonoscopy is the standard of care for CRC screening. To be effective, a colonoscopy must be preceded by high-quality bowel preparation. Characteristics of a good bowel preparation include effective colon cleansing, good tolerability, and favorable safety.
The efficacy of SPMC oral solution was robust within age groups in this subgroup analysis, with over 83% responder rate by AS and over 91% responder rate by BBPS in the right colon. The responder rate by AS or BBPS was highest for those aged less than 50 years and decreased with increasing age group. Previous studies have shown that older adults have higher rates of poor bowel preparation compared with younger adults, though this subanalysis did not find the same trends.18,20 Only 4 (0.9%) participants had colon cleansing rated ‘inadequate’ and none were rated ‘unprepared’ (score of ‘0’ on BBPS); the proportion of participants with a ‘fair’ rating increased with age. While those patients with a ‘fair’ rating did not meet the prespecified definition of responder for this study, a recent analysis has demonstrated that this group has adequate bowel preparation and can follow standard guideline-recommended CRC screening intervals. 26 For the BBPS data, within each age group, a greater proportion of patients were responders in the right colon than had a score of 6 or greater across all three colon segments, likely indicating that those few patients not meeting the BBPS score threshold of 6 had lower scores in the transverse or descending colon.
Bowel preparation continues to be a barrier for patients to complete a screening colonoscopy, with tolerability and fear of the preparation being significant factors. 27 SPMC oral solution showed favorable tolerability in this study, with at least 89% of participants in each age group reporting it was ‘easy’ or ‘acceptable’ to ingest, and at least 70% of those with prior colonoscopy experience rating it as ‘better’ than the prior bowel preparation agent. While some studies show reduced tolerability for bowel preparation in older adults,18,23 in this study, the majority (54%) of adults ⩾65 years rated SPMC oral solution as ‘easy’ to ingest, and an additional 35.5% rated it as ‘acceptable’, indicating good tolerability for SPMC oral solution in older adults.
The overall safety of SPMC oral solution was consistent across age groups, and was similar to data for the entire population. 24 Rates of headache and nausea were highest in the youngest group and lowest in the oldest group. Most cases of hypermagnesemia were transient in nature and did not result in any sequelae.
For those receiving SPMC oral solution, ADR was 30.8% for those aged 50–64 years, and 41.1% for those aged 65 years and over, well above the US Multi Society Taskforce (USMSTF) guideline-directed target of 25% or greater for a mixed cohort of males and females. 27 These data reinforce previous literature showing an increased risk of adenomas and CRC in older adults.6,8,9 It should be noted that ADR calculations in this study included average-risk patients undergoing screening colonoscopy and high-risk patients undergoing surveillance colonoscopy. Therefore, the ADRs presented may be slightly overestimated, given that some high-risk patients were included in the total population for calculation.
With the rising incidence of CRC among adults younger than 50 years during the last 2 decades, the 2018 American Cancer Society colon cancer screening guidelines recommended regular CRC screening for average-risk individuals begin at 45 years.10,13 To date, these guidelines are yet to be adopted by the gastroenterological societies. The hope and expectation is that, with these new recommendations, the incidence of CRC in younger adults will begin to decrease with increased screening.4,11,28
In this subgroup analysis, participants younger than 50 years (with a mean age of 38.6 years) had a PDR of approximately 30% and ADR of 16%. A previous study showed similar PDR and ADR in patients younger than 50 years. 29 Though these numbers are lower than PDR and ADR for older adults in our study, they are not insignificant in terms of clinical consequences. Therefore, it is important that individuals younger than 50 years follow the clinician’s recommendation for CRC screening colonoscopy.
In general, older adults show higher completion rates of recommended CRC screening compared with younger adults.22,30,31 Schedule and availability were hypothesized to be barriers for younger people to complete screening colonoscopies, given the greater likelihood for employment and childcare duties in this group compared with those 65 years or over. 22 In the United States, cost may also be a factor related to the higher completion rates of recommended screening colonoscopy for adults aged 65 years and over compared with younger adults, as the cost of the procedure is completely covered by Medicare. 32 Several studies have shown an increased diagnostic yield in screening and diagnostic colonoscopy in elderly adults, with rates of CRC diagnosis ranging from 6% to 20%, depending on the study. 33 An analysis of Medicare claims showed an increased risk for CRC in patients up to 70–74 years of age who do not complete a colonoscopy compared with those who do [0.42% absolute risk difference (confidence interval 0.24–0.63%)]. 34
Conclusion
Ready-to-drink SPMC oral solution demonstrated good efficacy of overall colon cleansing in adults across different age groups, including those aged 65 years and over. The tolerability of SPMC oral solution was favorable, with most participants in any age group, including older adults, preferring SPMC oral solution over a prior bowel preparation. ADR was above guideline-recommended targets in the appropriate age groups. No new safety signals were seen in any age group receiving SPMC oral solution. SPMC oral solution should be considered as a bowel preparation for most adults undergoing colonoscopy, including older adults.
Footnotes
Acknowledgements
Medical writing and editorial support was provided by Agnella Izzo Matic, PhD, CMPP (AIM Biomedical, LLC) and was funded by Ferring Pharmaceuticals Inc. The authors would like to thank the investigators, study staff, and participants who were involved in the trial.
LH, GB, KLJ, MB, MA, and DND analyzed and interpreted the data, drafted, and critically revised the article for important intellectual content, and approved the article for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was funded by Ferring Pharmaceuticals Inc., Parsippany, NJ.
Conflict of interest statement
Dr Hookey has participated in the speaker’s bureau for Ferring Pharmaceuticals Inc. Dr Bertiger was an investigator for clinical trials sponsored by Ferring Pharmaceuticals Inc. and has served as a consultant and has participated in the speaker’s bureau for Ferring Pharmaceuticals Inc. Dr Johnson was an investigator on clinical trials sponsored by Ferring Pharmaceuticals Inc. Drs Boules, Ando, and Dahdal are employees of Ferring Pharmaceuticals Inc.
Prior publication
Portions of the data contained in this manuscript appeared in abstract/poster form at Digestive Disease Week 2019, 18–21 May 2019, Poster Mo1673, and at American College of Gastroenterology 2019 Annual Scientific Meeting, 25–30 October 2019, Poster P2040. This manuscript has not been submitted and is not under consideration for publication anywhere else.
