Abstract
Hepatocellular carcinoma (HCC) is a global health issue with increasing incidence and high mortality rate. Depending on the tumor load and extent of underlying liver cirrhosis, aggressive surgical treatment by hepatectomy or liver transplantation (LT) may lead to cure, whereas different modalities of liver-directed locoregional or systemic tumor treatments are currently available for a noncurative approach. Apart from tumor burden and grade of liver dysfunction, assessment of prognostic relevant biological tumor aggressiveness is vitally important for establishing a promising multimodal therapeutic strategy and improving the individual treatment-related risk/benefit ratio. In recent years, an increasing body of clinical evidence has been presented that 18F-fludeoxyglucose (18F-FDG) positron emission tomography (PET), which is a standard nuclear imaging device in oncology, may serve as a powerful surrogate for tumor invasiveness and prognosis in HCC patients and, thereby, impact individual decision making on most appropriate therapy concept. This review describes the currently available data on the prognostic value of 18F-FDG PET in patients with early and advanced HCC stages and the resulting implications for treatment strategy.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the most frequent primary liver cancer, with liver cirrhosis being the major risk factor. Despite significant progress in the medical treatment of chronic viral hepatitis B and C, which are the leading causes of cirrhosis development along with alcohol, its epidemic significance is further increasing. Currently, HCC is the fifth most common cancer and the third most common reason for malignancy-related death in the world.1,2 In the United States, HCC is among the most rapidly increasing causes of cancer-associated mortality and its disease burden is expected to grow in the upcoming years. In particular, metabolic liver disorders, such as diabetes mellitus, obesity, and nonalcoholic fatty liver disease, are predicted to be the major sources of increasing HCC incidence.3,4
Well-established diagnostic algorithms, such as the European Association for the Study of the Liver (EASL) and the American Association for the Study of the Liver Diseases (AASLD) criteria have been developed, based on contrast enhanced computed tomography (CT)/magnetic resonance tomography (MRI) tumor size assessment with or without additional tumor biopsy.5,6 As demonstrated in the Barcelona Clinic Liver Cancer (BCLC) treatment algorithm, there is a wide spectrum of well-developed surgical, locally ablative, and systemic therapy options. Depending on tumor size criteria, liver function, and general physical status, therapy options range from curative approaches such as liver resection (LR), liver transplantation (LT), or radiofrequency ablation (RFA)/percutaneous ethanol injection (PEI), to noncurative locoregional tumor therapies (LRTTs), such as transarterial chemoembolization (TACE) or radioembolization (TARE), and systemic treatment by sorafenib. 7 Owing to the complex interactions between oncology and liver function, each HCC case has to be discussed extensively in a multidisciplinary hepatobiliary board. Very often, a multimodal treatment approach implementing nonsurgical and surgical interventions is thereby indicated. 8 Aggressive anticancer concepts may, however, not only impact the oncological course of disease, but also result in severe complications with significant consequences for quality of life and overall prognosis. Therefore, the suggested oncological benefit has to be balanced against treatment-related morbidity and mortality, particularly in the context of aggressive surgical approaches. 9 To further refine treatment allocation, more insight into biological tumor behavior affecting the long-term course of disease far beyond the initial post-treatment period seems to be mandatory. Whereas marked intrahepatic tumor load, tumor infiltration into large portal veins and extrahepatic tumor spread clearly reflect aggressive tumor attitude, assessment of HCC biology may be challenging in earlier tumor stages.
In recent years, poor differentiation and microvascular invasion (MVI) have been clearly identified as significant predictors of tumor invasiveness and poor outcome in liver cancer.10–12 In particular, in a difficult decision-making process between just about acceptable aggressive multimodal (surgical) treatment and noncurative palliative approaches, tumor biopsy may be inappropriate owing to tumor heterogeneity, risk of tumor cell seeding, and biopsy-related complications such as bleeding and infection.13,14 18F-fludeoxyglucose (18F-FDG) positron emission tomography (PET) emerged as a highly effective nuclear imaging tool for diagnostic setup, treatment allocation, and assessment of post-interventional tumor response in medical and surgical oncology.15–17 Initially, 18F-FDG PET did not play a clinical role in the evaluation of patients with suspected liver cancer. However, in recent years, an increasing body of clinical evidence has been presented that 18F-FDG PET may be very valuable for describing biological tumor behavior and the outcome of HCC patients.
This review summarizes current available data on the prognostic significance of 18F-FDG PET in curative and noncurative approaches for HCC, with a specific focus on implications for therapeutic recommendations.
Molecular basis of 18F-FDG PET: implications for diagnosis, staging, tumor biology, and treatment allocation
Highly simplified, 18F-FDG PET makes use of the specific cancer characteristic of enhanced glucose metabolism. Comparable to glucose, FDG is incorporated into malignant cells by glucose transporter 1 (GLUT1) activity. Subsequent intracellular accumulation of the tracer is related to phosphorylation via hexokinase (HK) rendering 18F-FDG to 18F-FDG-phosphate. In contrast, FDG concentration in the cells may be decreased by the gluconeogenesis enzyme glucose-6-phosphatase (G6Pase).18,19 Differences in metabolic uptake patterns between varying cancer entities are related to heterogeneity of enzyme activities. For example, colorectal liver cancer metastases exhibit overexpression of GLUT1 and HK and underexpression of G6Pase when compared with HCC. Thus, intracellular FDG accumulation is higher and the cancer detection rate by FDG PET imaging is better in metastatic liver tumors than in primary hepatoma.20,21 In addition, the diagnostic accuracy of 18F-FDG PET in HCC is also hampered by enzyme expression variations between different gradings. The FDG uptake in low-grade tumors is rather comparable with that of the surrounding nontumorous liver tissue, which makes them frequently invisible to 18F-FDG PET scanning. In contrast, enzymatic activities in moderately/poorly differentiated HCC lead to an increased standardized uptake value (SUV) in comparison with uninvolved liver regions, therefore allowing tumor detection. Overall, sensitivity of 18F-FDG PET for appropriate HCC diagnosis was reported to be only about 50% or even lower, which is inappropriate compared with nearly 90% provided by modern diagnostic devices including ultrasound imaging, multidetector CT, and contrast-enhanced MRI.22–25 Thus, 18F-FDG PET is currently not a recommended standard imaging modality for the diagnosis of HCC. 26
However, FDG uptake characteristics may be used for the assessment of biological tumor aggressiveness in HCC patients. As far back as in 2001, Shiomi et al. demonstrated that both tumor-volume doubling time and survival correlated significantly with SUV ratio between tumor and nontumor regions. 27 Molecular studies have shown that increased FDG accumulation is associated with overexpression of genes promoting tumor angioinvasiveness.22,28–30 In a series of 63 HCC patients that underwent LR, Kitamura et al. demonstrated that enhanced 18F-FDG uptake indicated higher tumor proliferation as expressed by Ki-67 index. 31 Recently, Lee et al. found that enhanced SUV ratio correlated with increased expression of epithelial–mesenchymal transition markers, which are suggested to play an important role in metastatic tumor progression. 32
Owing to its role as a surrogate marker for tumor aggressiveness, adding 18F-FDG PET to the standardized diagnostic algorithm was suggested to modulate final tumor staging, which may finally change treatment recommendation.33,34 A Japanese trial demonstrated therapy alterations in 25% of 64 HCC patients following 18F-FDG PET scanning. For example, 41 patients had initially been declared appropriate for curative LR or LT, whereas only 28 of them (68%) remained suitable for surgical treatment following FDG PET imaging. 34
In particular, information on possible extrahepatic tumor spread is important in treatment specification, because this excludes patients from radical surgical approaches. Although data is still limited, there is increasing clinical evidence that 18F-FDG PET provides advantages in the detection of lung metastases (>1 cm in diameter) and bone metastases compared with conventional imaging techniques, whereas diagnostic value for assessing lymph node metastases remains conflicting (Table 1).35–40
Diagnostic value of 18F-FDG PET in detecting extrahepatic HCC metastases.

CC, cholangiocarcinoma; CT, computed tomography; EHM, extrahepatic metastasis; 18F-FDG, 18F-fludeoxyglucose; HCC, hepatocellular carcinoma; MTS, metastasis; LN, lymph node; PET, positron emission tomography.
18F-FDG PET in the curative surgical treatment of HCC
The role of 18F-FDG PET for improving surgical strategy in LR for HCC
Hepatectomy is the treatment of choice in compensated liver cirrhosis without portal hypertension. However, there are several critical issues that have to be considered in individual decision making. In particular, the extent of LR has to be carefully scheduled based on accurate evaluation of patients’ general performance, liver size, functional liver capacity, and tumor load.41,42 Although data is still inconclusive, anatomic LR using wide resection margins seems to offer oncological benefit in comparison with limited atypical LR.43,44 In contrast, extended hepatectomy in cirrhotics may be associated with severe postoperative complications, such as septic problems, small-for-size syndrome, or even liver failure. In addition, there may be a substantial risk of post-LR HCC reappearance, either as local tumor relapse triggered by vascular invasion, or as de novo tumor appearance in the underlying cirrhotic liver.41,42,45,46
For an accurate assessment of the patients’ individual risk/benefit ratio in the context of extended LR for HCC, reliable data on prognostically relevant tumor biology features is essential.
Although being hampered by their retrospective character and the use of different SUV cutoff values, a large number of studies were in the past able to demonstrate that 18F-FDG PET is a valuable imaging device for evaluating the oncological risk following hepatectomy (Table 2). Enhanced FDG uptake on PET was shown to indicate the presence of unfavorable histopathologic features, such as poor differentiation and MVI, and to predict poor overall survival (OS) and recurrence-free survival (RFS).47–59
The value of 18F-FDG PET for predicting prognosis following liver resection for HCC.

AFP, alpha fetoprotein; ALBI, albumin bilirubin; AUC, area under the curve; CI, confidence interval; CT, computed tomography; 18F-FDG, 18F-fludeoxyglucose; HCC, hepatocellular carcinoma; HR, hazard ratio; LR, liver resection; MC, Milan criteria; MVI, microvascular invasion; OR, odds ratio; OS, overall survival; PET, positron emission tomography; RFS, recurrence-free survival; RR, relative risk; SUV, standard uptake value.
As demonstrated recently by Yoh et al., the prognostic power may be further improved by combining metabolic tumor aggressiveness with parameters of functional liver capacity. 59 In a retrospective analysis including 207 HCC patients, Yoh et al. have identified SUV ratio (HR = 1.743; 95% CI 1.114–2.648; p = 0.016) and albumin–bilirubin grade (HR = 1.966; 95% CI 1.349–2.884; p < 0.001), which is a novel parameter of functional hepatic reserve, as the only significant and independent prognostic factors of OS following LR. 59 The introduced novel hybrid variable demonstrated more predictive significance than other established staging systems, such as the BCLC system, the Cancer of the Liver Italian Program score and the Japan Integrated Staging score. 59
In addition, data from several investigations implicated that 18F-FDG PET may not only be useful in outcome prediction, but also for modulating the individual surgical strategy.
One study suggested that FDG uptake measurement is able to identify patients who benefit from hepatectomy when being implemented in a neoadjuvant setting prior to rescue LT (salvage LT concept). 50 In a series of 63 consecutive HCC patients, Kitamura et al. demonstrated that pre-LR SUV ratio was not only an independent prognostic factor of early HCC recurrence, but proved to be the only independent predictor of HCC recurrence pattern. It was significantly lower in patients without tumor relapse (1.3 ± 0.5) or with HCC recurrence meeting the Milan criteria (MC; 1.9 ± 1.6), still qualifying for salvage LT, compared with nontransplantable advanced HCC relapses (2.9 ± 2.6; p < 0.05) beyond MC. 50
Data from two other studies indicated that LR margin should be more extended in PET-positive patients for achieving oncological benefit.52,57
Ochi et al. identified SUVmax as the only independent predictive factor of microsatellite distance >1 cm from primary tumor lesion (HR = 1.60; 95% CI 1.23–2.26; p = 0.002) assessed on histopathologic specimen, which is a well-known promoter of HCC recurrence. 52
Another retrospective trial by Park et al. assessed PET+ status as a significant and independent prognostic factor for RFS (HR = 2.8; 95% CI 1.273–6.158; p = 0.01), along with presence of satellite nodules, MVI, and multicentric tumor occurrence. They found improved OS (p < 0.001) and trend to better RFS (p = 0.188) following >1 cm compared with <1 cm surgical resection margin in PET-positive but not in PET-negative patients. 58
18F-FDG PET to expand HCC selection criteria in liver transplant patients
The introduction of the MC in 1996 (one tumor nodule up to 5 cm; or 3 HCC nodules, each not exceeding 3 cm in diameter; no macrovascular tumor invasion) for a rigid patients’ selection contributed significantly in establishing LT as standard treatment in early stage HCC and decompensated liver cirrhosis. From now on, post-transplant 5-year RFS and OS rates above 70% have been reported. Consequently, the MC were implemented for model for end-stage liver disease (MELD)-based prioritization in various large public allocation systems, such as the United Network of Organ Sharing and Eurotransplant.60,61
However, it has been clearly demonstrated in recent years that strict adherence to the MC does not completely prevent post-LT HCC recurrence. 62 Apart from that, many advanced HCC patients beyond the MC are, thereby, excluded from potentially curative LT. Therefore, several more liberal macromorphologic selection criteria sets have been proposed, such as the University of California San Francisco score (UCSF; one single lesion ⩽ 6.5 cm; or 2–3 lesions ⩽4.5 cm each; total tumor diameter ⩽ 8 cm) or the Up to Seven (UTS; sum of size and number of lesions not exceeding 7) criteria.
In recent years, it became evident that biological tumor behavior rather than tumor size criteria determines post-LT outcome.63,64 To improve patient selection on a tumor biology basis, reliable pretransplant available surrogate marker of HCC aggressiveness have to be implemented.65–67
In fact, a large number of retrospective studies have in the last decade identified 18F-FDG PET as a powerful predictor of poor OS and RFS in the LT setting.68–85
Notably, FDG PET was able to discriminate the oncological risk in both MC In and MC Out tumors (Table 3). Overall, PET-positivity has been proven to identify a small subset of within MC patients that are on a very high oncological risk (HCC recurrence rates between 46% and 67%), which may be inacceptable in view of global donor organ shortage. In contrast, LT provided oncological cure in patients with PET-negative MC In tumors, as recurrence rates were about 0% in this specific subset of liver recipients (Table 3).68,69–72,75,76
The prognostic value of 18F-FDG PET in HCC liver transplant patients according to Milan criteria.

18F-FDG, 18F-fludeoxyglucose; HCC, hepatocellular carcinoma; MC, Milan criteria; OS, overall survival; PET, positron emission tomography; SUV, standard uptake value.
Equally important, FDG PET was shown to select advanced HCC patients that may benefit from LT although exceeding standard selection criteria. No significant outcome differences could be found between PET-negative MC Out patients and patients with MC In tumors (Table 3). Therefore, 18F-FDG PET has been proposed as useful noninvasive metabolic imaging for safely expanding the selection criteria beyond the MC burden limits.68–72,76,81–86
The extraordinary prognostic impact of 18F-FDG PET in the LT setting seems to be triggered by its capability to correlate with MVI, which in turn is one of the most important predictors of HCC recurrence (Table 4).57,68–69,72,74,76,78,81
Predictive value of 18F-FDG PET as a surrogate for the presence of MVI.
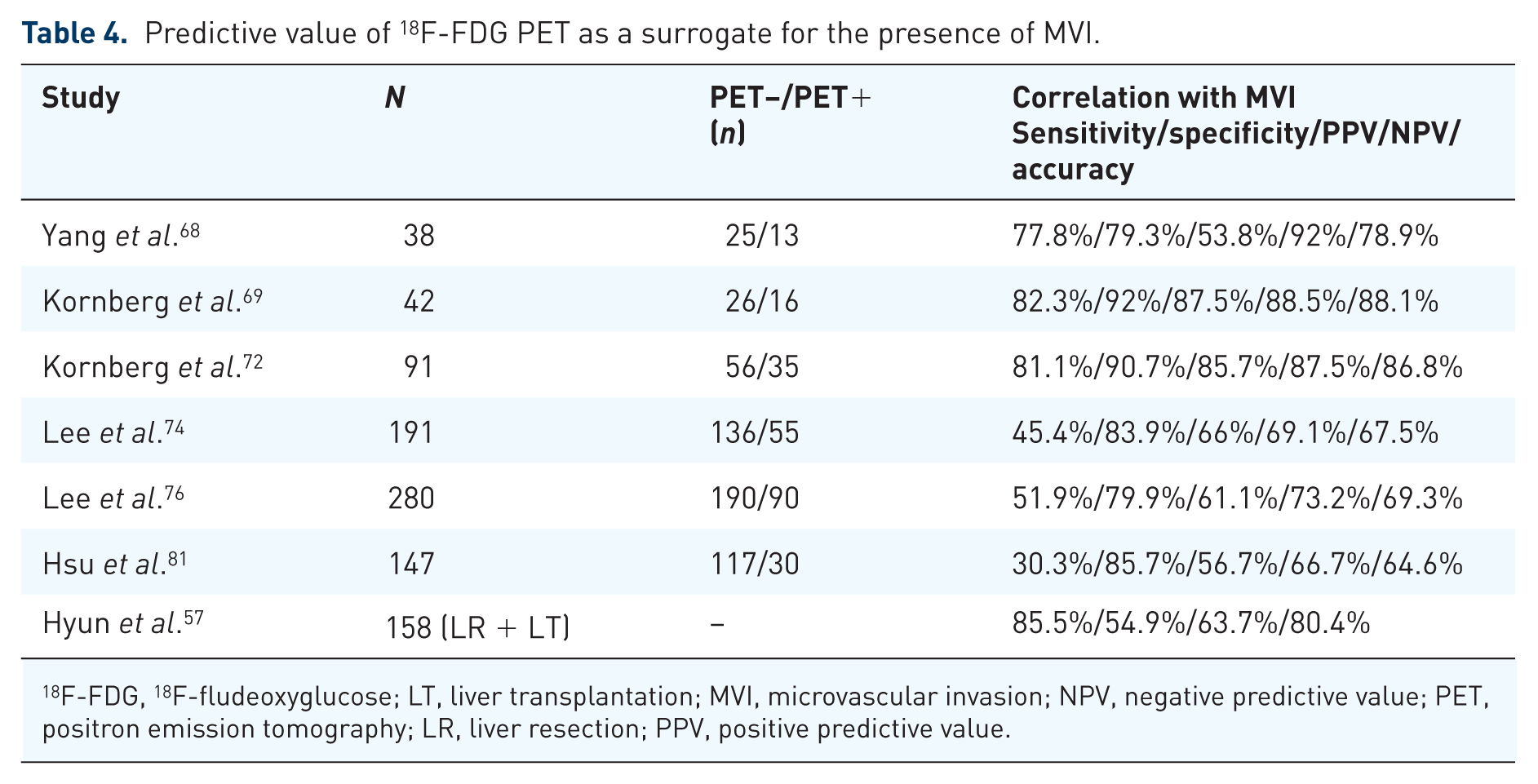
18F-FDG, 18F-fludeoxyglucose; LT, liver transplantation; MVI, microvascular invasion; NPV, negative predictive value; PET, positron emission tomography; LR, liver resection; PPV, positive predictive value.
Our transplant group identified PET-positive status as the only independent pre-LT available predictor of MVI on explant pathology (HR = 13.4; 95% CI 0.003–0.126; p = 0.001) in a study of 42 LT patients (Figure 1A and B). 69

18F-FDG PET (A) and 18F-FDG PET/CT (B) of two different LT patients with PET-positive HCC. Both of them revealed MVI on explant histopathology and were suffering from early post-LT HCC recurrence.
Results of a recently demonstrated multicenter retrospective cohort study including 158 HCC patients confirmed the paramount role of 18F-FDG PET in this context. The authors reported that SUV ratio (HR = 2.43; 95% CI 1.01–5.84; p = 0.047) was able to independently predict MVI on histopathologic specimen following LR or LT with the highest AUC. 57
Consequently, several expanded HCC transplant selection criteria implementing 18F- FDG PET have been proposed in recent years (Table 5).76,80–82,84,86
Proposals for expanded LT selection criteria implementing 18F-FDG PET.

18F-FDG, 18F-fludeoxyglucose; AFP, alpha fetoprotein; AUC, area under the curve; CI, confidence interval; HCC, hepatocellular carcinoma; HR, hazard ratio; MC, Milan criteria; NCCK, National Cancer Center Korea; OS, overall survival; PET, positron emission tomography; RFS, recurrence-free survival; SUV, standard uptake value; TNR, tumor to non-tumor standardized uptake value ratio; TTS, total tumor size; UCSF, University of California San Francisco; UTS, Up-to-Seven.
In a large retrospective analysis including 280 patients following living donor liver transplantation (LDLT), Lee et al. have identified PET-positive status, total tumor size (TTS) >10 cm, and MVI as significant and independent predictors of poor RFS in beyond MC tumors. There was no significant difference in OS and RFS between MC In patients and PET-negative MC Out patients with TTS <10 cm. 76 The newly defined so-called National Cancer Center Korea (NCCK) criteria (MC In or PET-negative MC Out with TTS <10 cm) were shown to increase the number of appropriate liver recipients without affecting tumor-specific outcome (RFS at 5-years post-LT: NCCK 80%; MC In 82%). The accuracy of pre-LT tumor staging for predicting explant pathology was 95% in NCCK-based, but only 78.9% in MC-based patient selection. 82
Hsu et al. suggested expansion of selection criteria based on UCSF and 18F-FDG PET. 81 In a series of 147 patients following LDLT, they reported on significantly better 5-year RFS rates in low-risk (UCSF In + FDG-negative) and intermediate risk (UCSF Out + FDG-negative or SUV ratio <2) patients compared with a high-risk (SUV ratio ⩾2) group (p < 0.001). However, a very low number of high-risk patients (n = 7) limited clinical significance of the data. 81
Our transplant group recently demonstrated in a retrospective study of 116 patients that combining radiographic UTS criteria with FDG PET safely expands the HCC selection criteria. Five-year RFS rates did not differ between MC In (86.2%), UTS In (81%), and PET-negative beyond UTS patients (87.1%), respectively. 85
Other study groups favored the implementation of both FDG PET and AFP to realize a tumor biology-based selection approach.83,86
One of them even proposed expansion of criteria without any tumor size limitation. 86 In a retrospective study including 123 LDLT patients by Hong et al., only PET-positivity (HR = 9.766; 95% CI 3.557–26.861; p < 0.001) and serum AFP level ⩾200 ng/ml (HR = 6.243; 95% CI 2.643–14.707; p < 0.001) were identified as independent prognostic factors. Combining them for indicating high-risk oncological status had a HR of 29.069 (95% CI 8.797–96.053; p < 0.001), whereas it was only 1.351 (95% CI 0.500–3.652; p = 0.553) for MC Out constellation. 86
Compared with a strict MC-based organ allocation, the application of these expanded selection criteria resulted finally in an increase of transplant eligibility between 16.5% and 51.5%, without increasing the oncological risk for the patients (Table 5).
18F-FDG PET in locally advanced HCC
Patients with intermediate-to-advanced-stage HCC (BCLC stage B or C) are mostly ineligible for curative surgical resection owing to enhanced tumor load, progressive liver dysfunction, tumor infiltration into major portal veins, or extrahepatic tumor manifestation. Depending on the individual tumor constellation and remaining liver function, these patients may, however, benefit from LRTT or systemic chemotherapy.7,8 Overall, data on the prognostic and therapeutic value of 18F-FDG PET in this specific subset of patients is still rather limited. However, there are some interesting studies indicating that FDG uptake pattern not only correlates with OS and progression-free survival (PFS), but may also impact individual decision making on most effective treatment in noncurative approaches (Table 6).87–94
Prognostic value of pre-treatment 18F-FDG PET in locally advanced HCC.
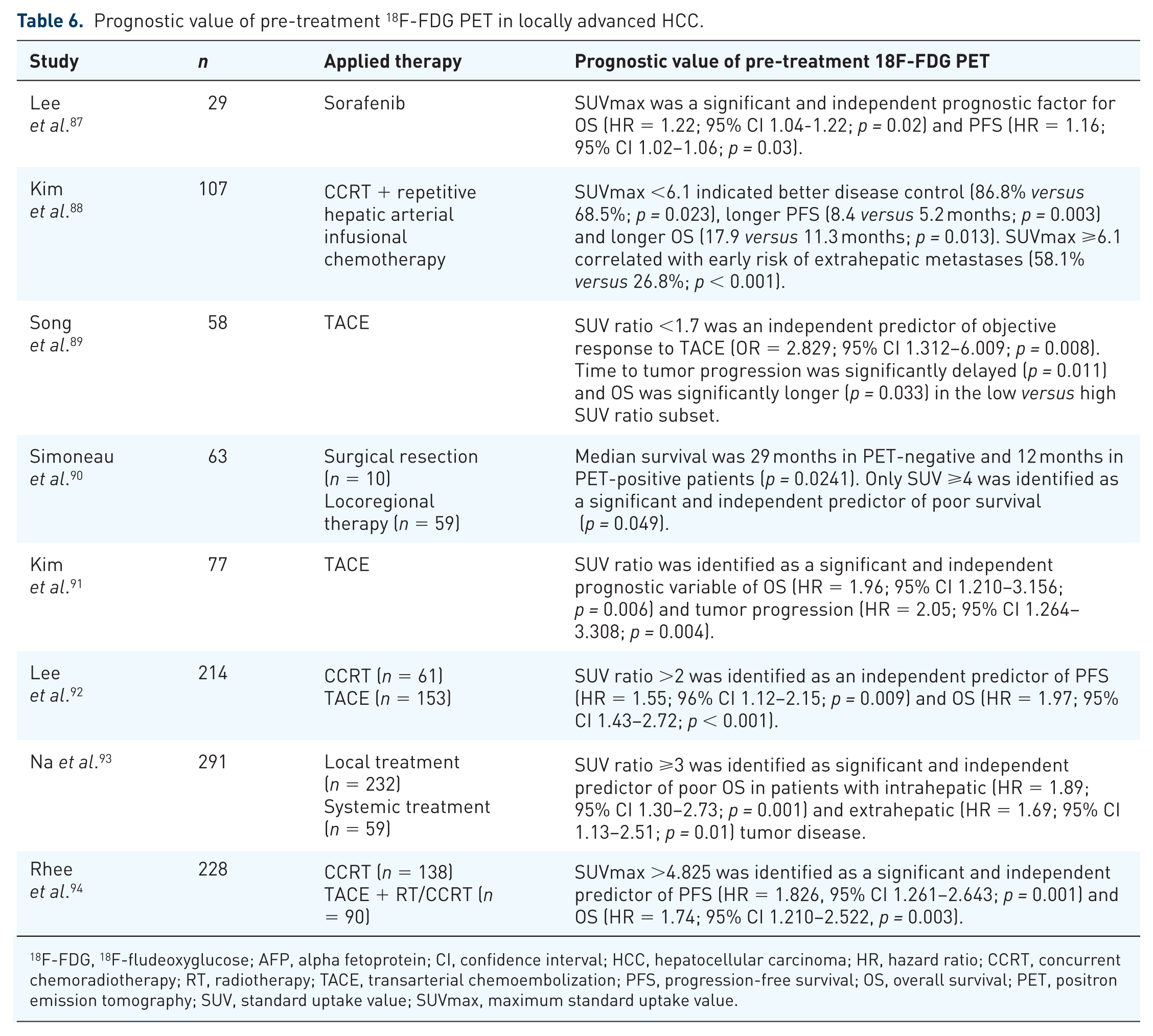
18F-FDG, 18F-fludeoxyglucose; AFP, alpha fetoprotein; CI, confidence interval; HCC, hepatocellular carcinoma; HR, hazard ratio; CCRT, concurrent chemoradiotherapy; RT, radiotherapy; TACE, transarterial chemoembolization; PFS, progression-free survival; OS, overall survival; PET, positron emission tomography; SUV, standard uptake value; SUVmax, maximum standard uptake value.
It has been suggested that systemic rather than LRTT should be applied in high SUV patients.88–94
For example, Kim et al. found significantly higher rates of early (within 6 months) extrahepatic metastases in SUVmax > versus ⩽6.1 (58.1% versus 26.8%; p < 0.001) in a series of 107 advanced HCC patients treated with concurrent chemoradiotherapy (CCRT) followed by repetitive hepatic arterial infusional chemotherapy. 88
Another retrospective multicenter study including 214 advanced tumor patients by Lee et al. reported that in a high SUV ratio (>2) subset, CCRT resulted in significantly better PFS (p = 0.018) and OS (p = 0.009) than LRTT by TACE, even when being adjusted for tumor size and number. In contrast, there was no significant difference in outcome between the treatment modalities in SUV ⩽2. 90
Based on results of a recent retrospective study including 228 locally advanced HCC patients, Rhee et al. proposed a double biomarker risk stratification by using SUVmax (> versus ⩽4.825) and serum AFP level (> versus ⩽550 ng/ml). 94 Both PFS (p < 0.001) and OS (p < 0.001) were significantly longer in low-risk patients (SUVmax ⩽4.825 + AFP ⩽550 ng/ml), compared with an intermediate-risk group (SUVmax >4.825 or AFP >550 ng/ml) and with a high-risk subset (SUVmax >4.825 + AFP >550 ng/ml), respectively. Rates of local disease control (p < 0.001) and conversion to surgery (p = 0.002) were lowest, and frequency for extrahepatic failure highest (p = 0.006) in the high-risk subset. The authors finally concluded that high-risk patients according to SUVmax and AFP level (SUVmax >4.825 + AFP >550 ng/ml) should receive systemic rather than locoregional therapy. In contrast, low-risk patients were more likely to suffer from intrahepatic ‘in-field’ failure, which supported a more aggressive local treatment in this specific subpopulation. 94
18F-FDG PET for metabolic assessment of tumor viability following LRTT
LRTT by using intraarterial or percutaneous techniques are nowadays an integrative part of a differentiated multimodal treatment approach in HCC.7,8 They may be indicated for a life-prolonging palliative intention or in a neoadjuvant fashion for achieving tumor downstaging prior to definite surgical treatment via LR or LT. 95 Appropriate assessment of tumor response following LRTT is essential for individual decision making on need for additional treatment sessions or change of therapeutic strategy. Using contrast-enhanced CT or MRI, the modified Response Evaluation criteria in Solid Tumors (mRECIST) and the EASL criteria are currently the standard for differentiating residual viable tumor tissue from post-interventional tumor necrosis. However, post-LRTT CT/MRI imaging may be misleading and, in addition, biological tumor features are not implemented in the current evaluation of treatment success. 96
Therefore, the applicability of 18F-FDG PET for metabolic assessment of post-interventional response has increasingly been investigated in recent years (Table 7).97–104
The value of 18 F FDG PET for evaluation of metabolic tumor response following LRTT.

18F-FDG, 18F-fludeoxyglucose; CI, confidence interval; HCC, hepatocellular carcinoma; LR, liver resection; LRTT, locoregional tumor therapy; LT, liver transplantation; MC, Milan criteria; NPV, negative predictive value; OS, overall survival; PEI, percutaneous ethanol injection; PET, positron emission tomography; PPV, positive predictive value; RFA, radiofrequency ablation; ROC, receiver operating characteristic; SUV, standard uptake value; TACE, transarterial chemoembolization; TAE, transcatheter arterial embolization; TAI, transcatheter arterial infusion.
In several curative intent studies, post-LRTT enhanced FDG uptake on PET prior to LR or LT was shown to correlate with tumor viability on postoperative histopathologic assessment.97–101 In comparison with contrast-enhanced CT, high diagnostic sensitivity and moderate specificity of 18F-FDG PET/CT have been demonstrated.98,101
In a study of 27 patients with intermediate stage HCC receiving noncurative intent TACE, Ma et al. demonstrated the superiority of tumor viability assessment by FDG PET over mRECIST criteria. 103 The authors identified ΔSUVmax ratio% to provide the highest discriminative prognostic power. Responders to TACE (ΔSUVmax ratio% <0.1) had a significantly better OS than nonresponders (SUVmax ratio% ⩾0.1; p = 0.025). 103
In addition, it has been suggested that 18F-FDG PET may be a valuable diagnostic approach in unexplained post-interventional increase of AFP level where contrast-enhanced CT remained inconspicuous.105–107
In a study of 100 LT candidates receiving neoadjuvant TACE, Refaat et al. have recently reported on distinctively higher sensitivity (92.8% versus 74.7%), specificity (94.1% versus 70.6%), and accuracy (93% versus 74%) of FDG PET/CT compared with contrast-enhanced CT for detecting post-interventional HCC recurrence. 107 SUVmax ratio >1.21 was identified as the best threshold (AUC = 0.935; 95% CI 0.868–0.975; p < 0.0001) for indicating intrahepatic or extrahepatic tumor manifestation. 107
Conclusion
18F-FDG PET is currently not a generally recommended imaging modality for improving diagnostic accuracy in HCC patients. However, as shown in this review, it provides very interesting data on biological tumor viability and cancer-specific prognosis that may be valuable in different prognostic and therapeutic aspects of curative and noncurative treatment approaches. In particular, in the context of aggressive surgery either by extended hepatectomy or by LT beyond current standard criteria, data on metabolic tumor aggressiveness preoperatively delivered by 18F-FDG PET/CT may be useful for a differentiated indication and therapeutic strategy. Thereby, a refined benefit/risk assessment for the patient may be achieved. However, also in the nonsurgical setting, patients seem to benefit from pre- and post-treatment metabolic tumor evaluation on FDG PET by an individually tailored multimodality approach. To more precisely define the strategic role of 18F-FDG PET in early and advanced HCC stages, prospective multicenter studies including a larger number of patients and implementing a standardized FDG PET setting would be desirable. Current available trials on this topic are mainly hampered by their predominantly retrospective character, great variabilities regarding tumor stage and underlying liver function, and by use of different FDG uptake measurements and SUV cutoff values. Nonetheless, as shown in our review, there seems to be enough clinical evidence that 18F-FDG PET may be used as a reliable pre- and post-treatment available surrogate marker of biological tumor invasiveness and outcome in HCC patients.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
