Abstract
It is increasingly recognized that gastric cancer is a heterogeneous disease which may be divided into subgroups based on histological, anatomical, epidemiological and molecular classifications. Distinct molecular drivers and tumor biology, and thus different treatment targets and predictive biomarkers, may be implicated in each subtype. However, there is little evidence in the literature regarding the correlation among these different classifications, and particularly the molecular aberrations present in each subtype. In this review, we approach advanced gastric cancer (AGC) by presenting aberrant molecular pathways and their potential therapeutic targets in gastric cancer according to histological and anatomical classification, dividing gastric cancer into proximal nondiffuse, distal nondiffuse and diffuse disease. Several pathways are involved predominantly, although not exclusively, in different subtypes. This may help to explain the disappointing results of many published AGC trials in which study populations were heterogeneous regardless of clinicopathological characteristics of the primary tumor. Histological and anatomical classification may provide insights into tumor biology and facilitate selection of an enriched patient population for targeted agents in future studies and in the clinic. However, some molecular pathways implicated in gastric cancer have not been studied in correlation with histological or anatomical subtypes. Further studies are necessary to confirm the suggestion that such classification may predict tumor biology and facilitate selection of an enriched patient population for targeted agents in future studies and in the clinic.
Keywords
Introduction
Modest efficacy and considerable toxicities associated with chemotherapy in advanced gastric cancer (AGC) has prompted the pursuit of novel systemic treatment strategies. Aberrant genetic and molecular alterations in gastric carcinogenesis represent logical treatment targets. However, the enthusiasm in developing such therapy has not been met with great success thus far. Trastuzumab is currently the only approved targeted agent for the subgroup of human epidermal growth factor receptor (HER)-2-positive AGC, based on results of the phase III trastuzumab for gastric cancer (ToGA) trial [Bang et al. 2010]. Although an overall survival (OS) advantage was demonstrated, the majority of patients did not respond to the combination of chemotherapy and trastuzumab in the first-line setting despite having HER2-positive disease. However, the addition of bevacizumab to chemotherapy did not result in significant OS benefit in the phase III Avastin in Gastric Cancer (AVAGAST) trial [Kang et al. 2010]; more recently the results of the Randomized EOX for advanced and locally advanced esophagogastric cancer 3 (REAL-3) study in abstract form reported an inferior OS with the addition of panitumumab to chemotherapy [Wadell et al. 2012]. Data on other agents are still awaited.
The difficulties encountered in the development of targeted therapy in AGC are caused by the lack of biomarkers to guide patient management. In the clinic to date, except for HER2, there are no established biomarkers predictive of tumor response to targeted agents. Few potential biomarkers are pending clinical validation, including amplification of MET [Lennerz et al. 2011] and fibroblast growth factor receptor 2 (FGFR2) [Deng et al. 2012], while others are more controversial. Moreover, the process of gastric carcinogenesis is complex [Wu et al. 2009; Yin, et al. 2009]. Rather than predominantly addicted to a particular oncogene and its associated signaling pathway, gastric cancer may be driven by multiple essential pathways with their cross talks poorly understood, preventing effective targeting by single agents. It is also increasingly recognized that gastric cancer is a heterogeneous disease which may be divided into subgroups based on histological [Lauren, 1965], anatomical [Blot et al. 1991], epidemiological [Crew and Neugut, 2006], and more recently, genomic or molecular classifications [Tay et al. 2003; Ooi et al. 2009; Shah et al. 2011; Deng et al. 2012]. Distinct molecular drivers and tumor biology, and thus different treatment targets and predictive biomarkers, may be implicated in each subtype. However, there is little evidence in the literature regarding the correlation between these different classifications, and particularly the molecular aberrations present in each subtype. Also, the classifications currently have a limited role in the prospective selection of patients with AGC in the clinic or trials, and no consensus classification exists.
In this review, we approach AGC by describing molecular aberrations predominantly implicated in according histological-anatomical subtypes of gastric cancer, namely proximal nondiffuse, distal nondiffuse and diffuse disease (Figure 1). While providing a brief account of clinical trials of targeted agents in AGC, which have been summarized in detail elsewhere [Wong and Yau, 2012], we also focus on preclinical developments.

Subtypes of gastric cancer based on anatomical and histological classification, and important molecular targets implicated in each subtype. EGFR, epidermal growth factor receptor; FGFR, fibroblast growth factor receptor; GEJ, gastroesophageal junction; HER, human epidermal growth factor receptor; MMP, matrix metalloproteinase; mTOR, mammalian target of rapamycin; PI3K, phosphatidylinositol 3 kinase; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor. (Illustration courtesy of Alessandro Baliani, Copyright © 2012.)
Proximal nondiffuse gastric cancer
Proximal nondiffuse gastric cancer is defined as tumor located in the gastric cardia which may extend up to the gastroesophageal junction (GEJ), where histopathology shows evidence of precursor glandular dysplasia or in situ carcinoma in the setting of chronic inflammation usually without atrophy [Shah et al. 2011]. In contrast to distal nondiffuse disease when chronic inflammation is more associated with Helicobacter pylori, in proximal tumors carcinogenic inflammation is often caused by gastric acid reflux [Blot et al. 1991; Crew and Neugut, 2006].
The overexpression of HER2 is more prevalent, though not exclusive, in intestinal and proximal gastric cancer compared with diffuse or distal disease [Koeppen et al. 2001; Tanner et al. 2005; Gravalos et al. 2007; Leon-Chong et al. 2007; Zhang et al. 2009]. HER2 overexpression is observed in 10–38% of gastric cancer tumor samples overall, in 16–34% of intestinal tumors and 2–7% of diffuse tumors [Jaehne et al. 1992; Koeppen et al. 2001; Tanner et al. 2005; Yano et al. 2006; Gravalos et al. 2007; Leon-Chong et al. 2007], and in 24–73% in the subgroup of GEJ and esophageal adenocarcinoma [Polkowski et al. 1999; Ross and McKenna, 2001; Zhang et al. 2009] (Table 1). HER2, a transmembrane tyrosine kinase receptor encoded by the ErbB2 gene, is a member of the HER family. Epidermal growth factor receptor (EGFR/HER1), HER3 and HER4 are all activated by ligand binding, while the HER2 receptor has no known ligand. Activation of these receptors leads to homo- or hetero-dimerization, which in turn initiates phosphorylation cascades and subsequent signaling pathways, including RAS– RAF–MEK–mitogen-activated protein kinase and phosphatidylinositol 3 (PI3K)–AKT–mammalian target of rapamycin (mTOR) pathways, for cancer cell proliferation and survival [Schlessinger, 2004; Dhanasekaran and Johnson, 2007] (Figure 2).
Comparison of molecular characteristics between intestinal and diffuse subtypes of gastric cancer.

Note: The bold values signify the higher prevalence of particular molecular characteristics when comparing gastric subtypes. HER2-, EGFR-, MET- and VEGF-related signaling are predominantly implicated in the intestinal subtype, while loss of E-cadherin, FGFR2-, mTOR-, HER3- and MMP-related pathways are more frequently involved in the diffuse subtype. EGFR, epidermal growth factor receptor; FGFR, fibroblast growth factor receptor; FISH, fluorescence in situ hybridization; HER, human epidermal growth factor receptor; IHC, immunohistochemistry; MMP, matrix metalloproteinase; mTOR, mammalian target of rapamycin; TKI, tyrosine kinase inhibitor; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor.
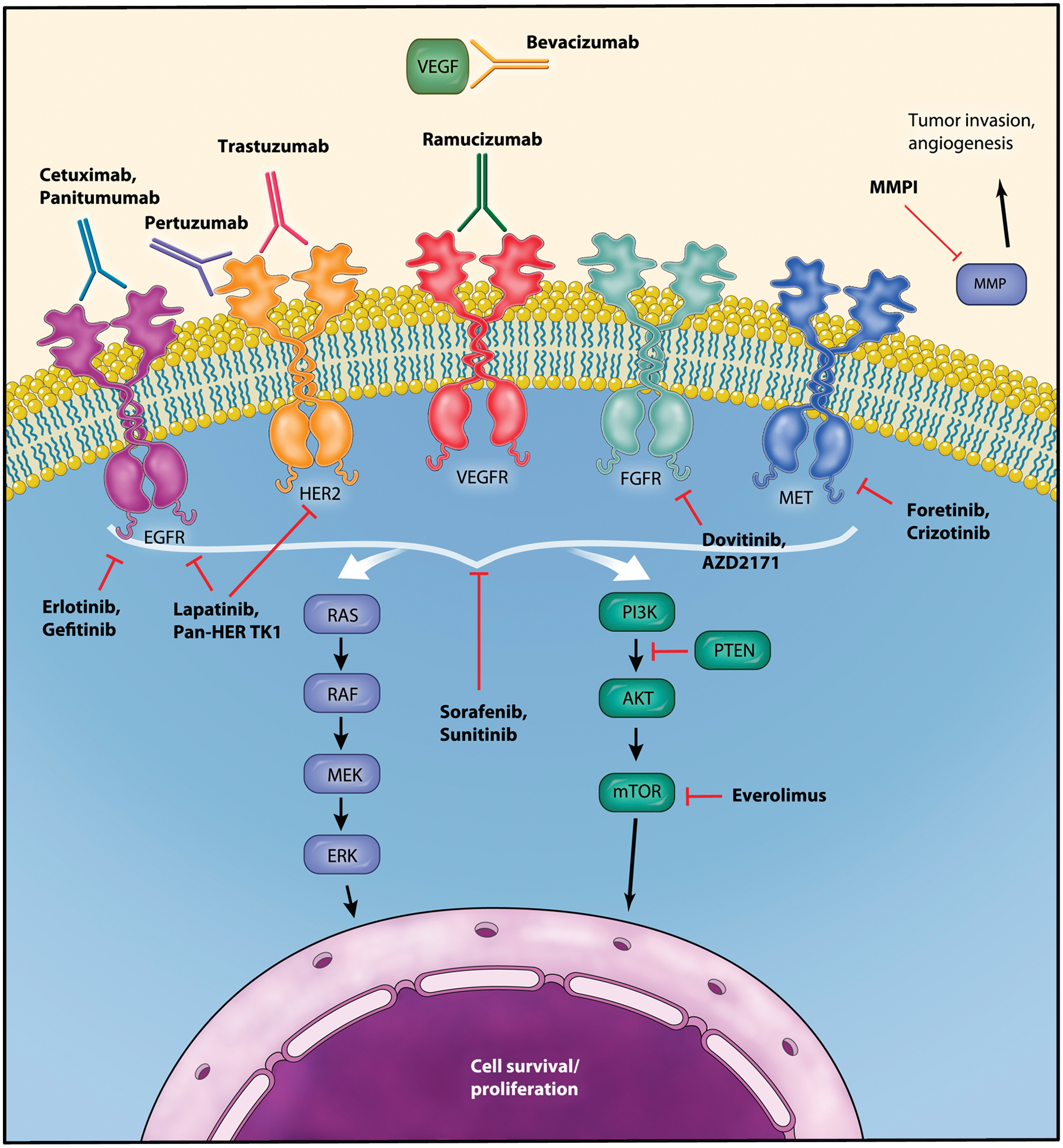
Schematic diagram of signaling pathways in gastric cancer and targets for molecular therapy. EGFR, epidermal growth factor receptor; ERK, extracellular signal-regulated kinase; FGFR, fibroblast growth factor receptor; HER, human epidermal growth factor receptor; MMP, matrix metalloproteinase; mTOR, mammalian target of rapamycin; PI3K, phosphatidylinositol 3 kinase; PTEN, phosphatase and tensin homolog; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor. (Illustration courtesy of Alessandro Baliani, Copyright © 2012.)
While EGFR overexpression is observed in 27–44% of all gastric cancer [Gamboa-Dominguez et al. 2004; Kim et al. 2008; Lieto et al. 2008; Matsubara et al. 2008], it is reported in 30–60% of proximal or esophageal adenocarcinoma [Al-Kasspooles et al. 1993; Yacoub et al. 1997; Wilkinson et al. 2004; Isinger-Ekstrand et al. 2010]. It was also observed that on EGFR inhibition with tyrosine kinase inhibitors (TKIs), response tended to occur in GEJ rather than gastric cancers [Dragovich et al. 2006; Rojo et al. 2006]. However, HER3 overexpression is more prevalent in diffuse gastric cancer (see later text), and little is known about HER4 in AGC.
In addition to HER2 and EGFR, amplification of the proto-oncogene MET by fluorescence in situ hybridization (FISH), although overall rare, occurs more frequently in GEJ (3%) compared with gastric tumors (0–2%), and more in intestinal than diffuse histology [Janjigian et al. 2011; Lee et al. 2011; Lennerz et al. 2011; Guo et al. 2012]. Increase in MET copy number was reported in up to 20% of all patients with gastric cancer [Lee et al. 2011], and MET mutation was also described [Lee et al. 2000]. Activation of receptor tyrosine kinase MET, whose ligand is hepatocyte growth factor, leads to proliferation and antiapoptotic signals [Migliore and Giordano, 2008].
HER2, EGFR and MET signaling are therefore of particular importance, especially in proximal nondiffuse tumors, and constitute logical targets for molecular therapy.
Targeting HER2
HER2 overexpression has been shown to predict response to trastuzumab, a humanized recombinant monoclonal antibody which selectively binds to the extracellular domain of HER2, thereby blocking its downstream signaling. The addition of trastuzumab to cisplatin plus capecitabine/fluorouracil was shown in the large phase III ToGA trial to significantly improve objective response rate (ORR) from 35% to 47% (p = 0.0017), progression free survival (PFS) from 5.5 to 6.7 months (p = 0.0002) and OS from 11.1 to 13.8 months (p = 0.0046) [Bang et al. 2010] in patients whose tumors were HER2 positive by FISH and 2–3+ by immunohistochemistry (IHC) in the first-line setting. Trastuzumab was well tolerated. The analysis of HER2 positivity in relationship to anatomical locations of primary tumors in the ToGA trial, in concordance with previous findings, showed a higher HER2-positive rate in GEJ than gastric cancers (33.2% and 20.9% respectively), and in the intestinal subtype than mixed and diffuse types (32.2%, 20.4% and 6.1% respectively) [Bang et al. 2009]. This observation again highlights disease heterogeneity in AGC and the potential role of incorporating histological and anatomical classifications into selecting patients for targeted therapy. Of note, as the level of HER2 overexpression may be associated with the magnitude of benefit from trastuzumab [Bang et al. 2009], its correlation with histological gastric cancer subtypes warrants further investigation.
HER2 dimerization inhibitor
Pertuzumab is a monoclonal antibody which binds to HER2 to prevent dimerization of HER2 with other HER receptors. In a HER2-positive, but not HER-negative, human gastric cancer xenograft model, pertuzumab in combination with trastuzumab showed significant antitumor activity compared with monotherapy. HER2-EGFR and HER2-HER3 heterodimerization and downstream signaling activation were inhibited [Yamashita-Kashima et al. 2011]. A phase II study is ongoing to investigate the combination of pertuzumab and trastuzumab in patients with HER2-positive AGC [ClinicalTrials.gov identifier: NCT01461057].
Targeting epidermal growth factor receptor
Unlike HER2 in HER2-positive gastric cancer, there are no established biomarkers to predict response to EGFR inhibitors. The predictive value of EGFR mutation, increased EGFR copy number, K-ras mutation status, and patients’ development of skin rash are controversial [Han et al. 2009; Zhang et al. 2009; Lordick et al. 2010].
Cetuximab is a recombinant human/mouse chimeric monoclonal antibody against EGFR. Many first-line phase II trials have evaluated cetuximab in combination with various chemotherapy regimens [Pinto et al. 2007, 2009; Woell et al. 2008; Han et al. 2009; Kanzler et al. 2009; Kim et al. 2009; Zhang et al. 2009; Lordick et al. 2010; Moehler et al. 2010] showing an ORR in the range of 40–60%, time to progression 5.5–8 months, and OS 9.5–16 months. However, preliminary results of a randomized phase II study showed no clinically significant benefit when cetuximab was added to docetaxel plus oxaliplatin [Richards et al. 2011]. A randomized phase III trial, EXPAND (Erbitux in Combination With Xeloda and Cisplatin in Advanced Esophago-gastric Cancer) [ClinicalTrials.gov identifier: NCT00678535] is ongoing to investigate first-line capecitabine and cisplatin with or without cetuximab. However, there are limited and conflicting data in the literature regarding the use of cetuximab-based therapy as salvage for pretreated patients with AGC [Stein et al. 2007; Tebbutt et al. 2008; Li et al. 2010].
In contrast to cetuximab, panitumumab is a fully humanized monoclonal antibody targeting EGFR. The REAL-3 trial aimed to explore the role of panitumumab in combination with epirubicin, oxaliplatin and capecitabine (EOC). Although in the phase II section of the study ORR of EOC plus panitumumab was promising at 52% [Chau et al. 2011], the phase III results showed a significantly inferior OS in the panitumumab-containing arm [8.8 months versus 11.3 months, hazard ratio (HR) 1.37, p = 0.013] [Wadell et al. 2012]. The negative results may be caused by the reduced dose intensity of chemotherapy in the panitumumab-containing arm, and by the recruitment of an unselected population. Similarly, the results of randomized phase II studies of other EGFR monoclonal antibodies, namely matuzumab and nimotuzumab, were both negative [Rao et al. 2010; Kim et al. 2011].
EGFR-TKIs have shown modest activity, mainly in GEJ rather than more distal cancers. In a phase II trial of 70 patients, erlotinib monotherapy resulted in an ORR of 9% (including one complete response) in patients with GEJ cancer but none in the gastric cancer subgroup [Dragovich et al. 2006]. In contrast, gefitinib was associated with a lack of efficacy in patients with AGC mostly with distal gastric cancer [Doi et al. 2003].
Dual targeting HER2 and epidermal growth factor receptor
Lapatinib is a TKI with dual action against HER2 and EGFR. While phase II studies of lapatinib in patients with HER2-unselected AGC showed disappointing results [Iqbal et al. 2007; Hecht et al. 2008], two phase III trials targeting HER2-positive disease are currently ongoing. The Lapatinib Optimization Study in ErbB2 (HER2) Positive Gastric Cancer (LoGIC) is investigating first-line treatment with capecitabine and oxaliplatin with or without lapatinib [ClinicalTrials.gov identifier: NCT00680901]; Tykerb with taxol in Asian HrbB2+ gastric cancer (TYTAN) is investigating second-line paclitaxel with or without lapatinib in Asian patients [Satoh et al. 2010].
The combination of lapatinib and trastuzumab was shown in a preclinical study to be synergistic in inhibiting the cell growth of the HER2-amplified human upper gastrointestinal cancer cell lines [Wainberg et al. 2010]; these data provide a rationale for testing this interesting combination in early phase clinical trials.
Pan-HER inhibition
In HER2-positive gastric cancer in vitro and in vivo models, a few pan-HER TKIs have demonstrated activity against tumor growth and synergistic effects in combination with chemotherapeutic or molecular agents. These include BMS-599626 [Wong et al. 2006], HM781-36B [Nam et al. 2011] and PF00299804 [Nam et al. 2012]. A phase II study of PF00299804 monotherapy in patients with HER2-positive AGC is currently ongoing [ClinicalTrials.gov identifier: NCT01152853].
Role of downstream components of HER pathway
In gastric cancer, KRAS mutation was observed in 2–20% [Nanus et al. 1990; Hongyo et al. 1995; Lee et al. 1995, 2003; Hiyama et al. 2002; Kim et al. 2003] and BRAF in 0–2.7% [Kim et al. 2003; Lee et al. 2003]. These mutations lead to constitutively activated signaling proteins, and are linked to resistance to monoclonal antibodies targeting upstream receptors in colorectal cancer [Normanno et al. 2009; Bardelli and Siena, 2010]. In gastric cancer, the predictive ability of KRAS and BRAF has not been extensively studied, but small reports did not demonstrate such characteristics [Lordick et al. 2010; Park et al. 2010]. There is also little evidence whether these biomarkers are different across histological-anatomical subtypes of gastric cancer.
Targeting MET
Amplification of MET predicts response to MET inhibition in vitro [Smolen et al. 2006; Kataoka et al. 2011]. The interim results of a phase II study of foretinib (GSK1363089, GSK089, formerly XL880), a MET TKI, showed minimal activity in a MET-unselected AGC cohort while it was well tolerated with toxicities including liver function abnormalities, fatigue and venous thromboembolism [Jhawer et al. 2009]. Another MET inhibitor, crizotinib (PF02341066), achieved tumor shrinkage in two out of four patients with MET-amplified esophagogastric adenocarcinoma; in both the tumor was located at the GEJ [Lennerz et al. 2011].
Distal nondiffuse gastric cancer
Distal nondiffuse tumors are located between the body of the stomach and pylorus. On histopathology, chronic gastritis with a spectrum of intestinal metaplasia and dysplasia is present. Intestinal gastric cancer, especially that arising from the antrum, is often a consequence of chronic Helicobacter pylori infection [Peek and Blaser, 2002], which also occurs in diffuse gastric tumors and normal controls but at a significantly lower frequency [Asaka et al. 1994].
H. pylori infection promotes angiogenesis in gastric cancer, as reflected by greater tumor vascularity in patients with H. pylori-infected compared with H. pylori-eradicated gastric cancer [Sasaki et al. 2003]. Moreover, H. pylori activates nuclear factor κB [Sharma et al. 1998], and induces the expression of a number of angiogenic factors in gastric cancer cells, including vascular endothelial growth factor (VEGF), matrix metalloproteinase (MMP)-9, and interleukin 8 [Crabtree et al. 1994; Kitadai et al. 2003; Wu et al. 2005]. This in turn is concordant with the observation that intestinal with reference to diffuse subtype expresses significantly higher VEGF levels (mean VEGF staining intensity 2.2 versus 1.4), vessel counts (mean 37 versus 26) and VEGF receptor (VEGFR) expression (39% versus 15%), suggesting that the former is more dependent on angiogenesis [Takahashi et al. 1996].
Targeting vascular endothelial growth factor
Bevacizumab is a monoclonal antibody which binds to VEGF. In the phase III AVAGAST trial, patients with AGC were randomized to receive capecitabine and cisplatin with or without bevacizumab as first-line treatment. Capecitabine and bevacizumab or placebo were given until progression or unmanageable toxicity. Although the study did not meet its primary endpoint of OS, ORR was significantly better in the bevacizumab arm (46% versus 37%, p = 0.0315), and PFS improved from 5.3 to 6.7 months (HR 0.8, p = 0.0037) [Ohtsu et al. 2011]. The most commonly encountered grade 3–5 bevacizumab-related adverse events were thromboembolic events, hypertension, bleeding and proteinuria. Interestingly, patients in pan-America, but not those in Asia or Europe, derived survival benefit from the addition of bevacizumab. Although the results of correlative biomarker analyses are still pending, a recent unplanned exploratory analysis of AVAGAST showed that bevacizumab appeared to improve outcomes in non-Asian subjects with distal nondiffuse disease, as opposed to proximal nondiffuse cancer [Shah et al. 2012]. While awaiting prospective confirmation, this supports the particular role of angiogenesis and its targeted therapy in the distal nondiffuse subtype of AGC.
Targeting vascular endothelial growth factor receptor
Multitargeted tyrosine kinase inhibitors (MTIs) suppress angiogenesis by simultaneously targeting VEGFR and other signaling pathways. In AGC, MTIs are in phase I/II of clinical development. Sunitinib inhibits platelet-derived growth factor receptor (PDGFR), kit, RET and Flt3 together with VEGFR. In phase II studies, single agent sunitinib has limited activity as salvage therapy for patients with chemotherapy-refractory AGC [Moehler et al. 2009; Bang et al. 2011]. However, sorafenib, another inhibitor of multiple kinases including VEGFR-2, VEGFR-3 and PDGFR, has been evaluated in combination with chemotherapy in the first-line phase II setting [Sun et al. 2010]. The results showed ORR of 41%, PFS of 5.8 months and OS of 13.6 months, with significant toxicities. In pretreated patients, single agent sorafenib was studied in another phase II trial; preliminary analysis of 16 evaluable patients included one durable complete response and another protracted stable disease of over 19 months [Ilson et al. 2011]. Other MTIs under early-phase clinical investigation include telatinib [Alsina et al. 2011], axitinib [ClinicalTrials.gov identifier: NCT00842244] and apatinib [Li et al. 2011].
Ramucirumab (IMC-1121B), a fully human IgG1 monoclonal antibody targeting VEGFR-2, was evaluated in a phase I study of previously treated advanced solid tumors, and achieved responses in 4 out of 37 patients, including one with refractory gastric cancer [Spratlin et al. 2010]. It is currently in phase III investigation in combination with weekly paclitaxel in patients with AGC progressing after first-line chemotherapy [ClinicalTrials.gov identifier: NCT01170663].
Diffuse advanced gastric cancer
Diffuse gastric cancer is characterized by a diffuse pattern of infiltration and poorly differentiated signet ring cell type without apparent gastritis. In contrast to intestinal gastric cancer resulting from a multistep carcinogenic process, diffuse gastric cancer is believed to arise de novo and is associated with CDH1 downregulation [Becker et al. 1994; Carneiro et al. 2004].
CDH1 is a tumor suppressor gene that encodes E-cadherin, a cell adhesion protein that maintains cell polarity [Cavallaro and Christofori, 2004]. Downregulation of CDH1 plays a role in gastric tumorigenesis, invasion and metastases [Guilford et al. 1998; Perl et al. 1998]. Germline mutation in CDH1 is responsible in hereditary diffuse gastric cancer [Humar and Guilford, 2009], while somatic CDH1 mutations are found in over 50% of sporadic diffuse gastric cancers, much more often than in the intestinal subtype [Becker et al. 1994; Tamura et al. 1996]. Inactivation of the second CDH1 allele may be caused by mechanisms including loss of heterozygosity and DNA hypermethylation of the promoter CpG islands [Tamura et al. 2000; Liu et al. 2006]. In concordance, E-cadherin by IHC is expressed at a lower rate in diffuse (11%) compared with intestinal (31%) gastric cancer [Zhou et al. 2010].
Other molecular aberrations preferentially amplified or expressed in diffuse type gastric cancer include FGFR2 signaling and PI3K signaling; HER3 is also implicated. The FGFR2/K-sam gene, encoding the FGFR2 receptor protein, was shown to be amplified predominantly in signet ring cell stomach cancer cell lines and poorly differentiated human gastric cancer xenografts and surgical specimens [Hattori et al. 1990; Nakatani et al. 1990], although a minority of evidence did not confirm significant association between FGFR2 amplification and diffuse histology [Deng et al. 2012; Guo et al. 2012]. On IHC analysis, FGFR2 positivity was found in 20 out of 38 (53%) advanced diffuse type gastric cancer specimens but none of the intestinal type specimens [Hattori et al. 1996].
PI3K activity was higher in cell lines derived from signet ring cell gastric carcinoma than other adenocarcinomas, and was associated with the formation of poorly differentiated cancer in nude mice [Kobayashi et al. 1999, 2003]. In gastric cancer specimens unspecified for histology, PIK3CA, encoding the p110 catalytic subunit of PI3K, was reported to be amplified in 36% [Byun et al. 2003], PI3KCA activating mutation in 4–36% [Byun et al. 2003; Li et al. 2005], and phosphatase and tensin homolog loss in 20–36% [Kang et al. 2002; Byun et al. 2003]. Moreover, the PI3K–AKT–mTOR pathway is frequently activated in gastric cancer as suggested by the prevalent expression of phospho-AKT (29–86%) [Bellacosa et al. 2005; Murayama et al. 2009] and phospho-mTOR (47–64%) [Lang et al. 2007; Murayama et al. 2009; Yu et al. 2009]. In particular, the expression of phospho-mTOR, a negative prognosticator [Yu et al. 2009; An et al. 2010; Xu et al. 2010], is seen more frequently in diffuse (58–64%) than in intestinal (47–60%) gastric cancer samples [Lang et al. 2007; Feng et al. 2008], although in both subtypes it occurs in a significant proportion of tumors. Thus targeted treatment on this pathway should not be developed exclusively for the diffuse subtype.
PI3K is also activated by upstream HER3, which is selectively phosphorylated in undifferentiated gastric cancer cells [Kobayashi et al. 1999, 2003]. In addition, HER3 signaling is implicated in cell growth and survival in FGFR2-amplified gastric cancer cell lines [Kunii et al. 2008]. Concordantly, the rate of HER3 overexpression is higher in human gastric cancer specimens of the diffuse subtype compared with the intestinal subtype (26% versus 5%, p < 0.01) [Zhang et al. 2009].
Finally, expression of some MMPs are significantly more frequent in diffuse than in intestinal gastric cancer subtypes and contribute to tumor aggressiveness; for example, positivity of MMP-7 was reported in 62% and 32% in diffuse and intestinal tumors respectively [Kitoh et al. 2004], and MMP-1 in 90% and 70% respectively [Zhou et al. 2010]. In other studies evaluating various MMPs, the difference did not reach statistical significance [Gerstein et al. 2009; Kemik et al. 2011; Zhang et al. 2011]. With regard to tumor location, MMP expression was similar between proximal and distal tumors [Kitoh et al. 2004; Woolley et al. 2004]. MMPs are a family of zinc-dependent enzymes involved in degradation of extracellular matrix. They play a key role in metastasis, and represent potential targets for therapy, possibly more frequently in the diffuse subtype.
Targeting β catenin
The invasion suppressor signal of E-cadherin is mediated through binding β catenin [Wong and Gumbiner, 2003]. In normal cells, β catenin connects E-cadherin through α catenin to the actin cytoskeleton [Mareel et al. 1997], but when not bound to E-cadherin, it associates with T-cell factor (Tcf) to activate in the nucleus the transcription of genes implicated in tumor formation and progression [Mann et al. 1999; Tetsu and McCormick, 1999]. In gastric cancer cells with an activated β catenin/Tcf pathway, a recombinant adenovirus carrying a lethal gene under the control of a β catenin/Tcf-responsive promoter selectively inhibited cell growth in a time- and dose-dependent manner [Dvory-Sobol et al. 2007]. Such strategies are however still in the preclinical stage of development.
Targeting fibroblast growth factor receptor 2
Small molecule FGFR2 inhibitors such as PD173074 and AZD2171 inhibited FGFR2 phosphorylation and cell growth in FGFR2-amplified gastric cancer cell lines [Takeda et al. 2007; Kunii et al. 2008] and xenografts [Takeda et al. 2007]. Similarly, in a murine model of scirrhous gastric cancer (a unique phenotype of diffuse gastric cancer) but not nonscirrhous gastric cancer, Ki23057, another novel tyrosine kinase FGFR2 inhibitor, significantly decreased FGFR2 phosphorylation, inhibited tumor proliferation and demonstrated synergistic antitumor effects in combination with 5-fluorouracil [Nakamura et al. 2006; Yashiro et al. 2010]. More recently, dovitinib (TKI258) demonstrated growth inhibitory activity in FGFR2-amplified gastric cancer cell lines and xenografts [Deng et al. 2012]. To various extent, these agents inhibit other tyrosine kinase receptors in addition to FGFR2, including FGFR1, FGFR3, VEGFR-1, VEGFR-2, VEGFR-3, and PDGF-Rβ. However, monoclonal antibodies specific to FGFR2 have been shown to effectively inhibit tumor growth in FGFR2-overexpressing gastric cancer xenografts in mice [Zhao et al. 2010]. Ongoing phase II studies may clarify the role of dovitinib and AZD2171 in patients with FGFR-amplified AGC [ClinicalTrials.gov identifiers: NCT01576380 and NCT01457846].
Targeting PI3K–AKT–mTOR pathway
A number of PI3K inhibitors have demonstrated preclinical activity and are being investigated in phase I studies of solid tumors (reviewed by Markman and colleagues) [Markman et al. 2010]. As for gastric cancer, the PI3K inhibitor LY294002 inhibited growth of implanted tumors of human gastric carcinoma cells in nude mice, and reduced tumor expression of a number of angiogenic factors [Xing et al. 2009]. Similarly, the PI3K/mTOR inhibitor BEZ235 and PI3K inhibitor BKM120 were shown to have pro-apoptotic effects for human gastric and colon cancer cell lines [Moehler et al. 2012]. In another study, BEZ235 resulted in decreased cell viability of gastric cancer cell lines and dephosphorylation of downstream effector proteins in all xenograft models [Fuereder et al. 2011]. However, an in vivo antitumor effect was only observed in one of the xenografts, correlated with downregulation of the proliferation marker thymidine kinase 1 and reduced [(18)F]FLT uptake by animal positron emission tomography. Molecular mechanisms involved in the sensitivity to PI3K inhibitors are yet to be clarified to translate preclinical activity to clinical benefit, and to date, the development of PI3K inhibitors in AGC is still in the preclinical stage.
The PI3K–AKT–mTOR pathway can also be targeted at the mTOR level. In a scirrhous gastric cancer cell line and its related cell line with propensity for peritoneal metastases, the mTOR inhibitor everolimus demonstrated growth-inhibitory activity [Taguchi et al. 2011]. Similarly, mTOR inhibitors with 5-fluorouracil showed a synergistic antiproliferative effect in scirrhous but not in nonscirrhous gastric cancer cell lines [Matsuzaki et al. 2009]. In other preclinical studies not specific for diffuse histology, antitumor effects of everolimus alone or in combination with cytotoxic chemotherapy were also reported [Cejka et al. 2008; Lee et al. 2010; Xu et al. 2010]. Clinically, a phase II trial of everolimus as salvage therapy for patients with pretreated AGC showed a disease control rate of 55%, although no objective response was noted [Doi et al. 2010]. The median PFS and OS were 2.7 and 10.1 months respectively. Based on these provocative results, a phase III randomized multicenter trial is ongoing to compare everolimus plus best supportive care with placebo plus best supportive care in patients with progressive disease after one or two prior lines of chemotherapy [ClinicalTrials.gov identifier: NCT00879333].
Matrix metalloproteinase inhibition
Marimastat, an oral MMP inhibitor, demonstrated preclinical activity in xenograft models of human gastric cancer [Wada et al. 2003] and acceptable safety in a phase I study [Tierney et al. 1999]. Despite an improved 2-year OS from 3 to 9%, the phase III trial of marimastat did not meet its primary endpoint to show a significantly different OS (p = 0.07), and was complicated by poor tolerability with musculoskeletal inflammation and pain [Bramhall et al. 2002]. Similarly, other MMP inhibitors such as prinomastat are limited by their cytostatic rather than cytotoxic action, and unfavorable toxicity profiles [Zucker et al. 2000].
Conclusion
In this review, we have presented aberrant molecular pathways and their potential therapeutic targets in gastric cancer according to histological-anatomical classification (Table 2). Several pathways are involved predominantly in different subtypes: HER2-, EGFR- and possibly MET-dependent signaling in proximal nondiffuse cancers, angiogenesis and related pathways in distal nondiffuse cancers, and β catenin, FGFR2, PI3K and HER3 activity in diffuse gastric cancers. The increased recognition that distinct molecular aberrations prevail preferentially in different gastric cancer subtypes may help to explain the disappointing results of many published AGC trials in which study populations were heterogeneous regardless of clinicopathological characteristics of the primary tumor. Despite the associations, the occurrence of the pathways in the histological-anatomical subtypes described is not exclusive; in particular, both proximal and distal nondiffuse disease belong to the intestinal histology, thus evidence may overlap. Some may also occur as resistance mechanisms in other subtypes.
Targeted agents reaching clinical development, summarized according to the histological subtype in which the molecular target is most frequently present.

EGFR, epidermal growth factor receptor; FGFR, fibroblast growth factor receptor; MMP, matrix metalloproteinase; MTI, multitargeted tyrosine kinase inhibitor; mTOR, mammalian target of rapamycin; TKI, tyrosine kinase inhibitor; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor.
To date, knowledge on the prevalence, coexistence and carcinogenic role of molecular aberrancies with respect to gastric cancer subtypes is still preliminary. Data are mostly derived from preclinical studies or small patient series, rather than dedicated investigations. Other molecular pathways, such as Wnt, ubiquitin proteosome, and hedgehog pathways, are implicated in gastric cancer but have not been studied in correlation with histological or anatomical subtypes; these are out of the scope of this review.
In conclusion, while some molecular pathways are predominantly associated with particular histological and anatomical subtypes based on available evidence, further studies are necessary to confirm the suggestion that such classification may predict tumor biology. Currently, in clinical trial design and practical treatment decision making in AGC, the molecular aberrations to target are unclear in most cases. Although molecular classification is emerging, until more biomarkers are validated and molecular tools readily available, histological-anatomical classification may provide insights into tumor biology and thus facilitate selection of an enriched patient population for targeted agents in future studies and in the clinic.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
