Abstract
Objective
To compare the short-term efficacy and safety profile of the S-1 + irinotecan + oxaliplatin (TIROX) and docetaxel
Methods
Patients with recurrent or metastatic gastric cancer diagnosed by pathology were randomly divided into two groups to receive six cycles of either the TIROX regimen (21-day cycle) or the DCF regimen (21-day cycle). After six chemotherapy cycles, the short-term efficacy was evaluated according to the Response Evaluation Criteria in Solid Tumors guidelines and adverse reactions were recorded according to National Cancer Institute Common Toxicity Criteria 2.0 standards.
Results
A total of 60 patients were enrolled in the study. The response rate (complete response + partial response) was significantly higher in the TIROX group (18/30 patients; 60.0%) compared with the DCF group (10/30 patients; 33.3%). The rates of grade III–IV leucopenia and neurotoxicity were significantly higher in the TIROX group than the DCF group.
Conclusion
The TIROX regimen was effective for the treatment of advanced gastric cancer, but it was associated with leucopenia and neurotoxicity.
Introduction
Although the overall incidence of gastric cancer has decreased over the past 70 years, it is still a major threat to human life.1,2 Due to the lack of effective screening methods, 60–80% of gastric cancers are already at an advanced stage when patients are first diagnosed. 3 Gastric cancer is the second highest cause of cancer mortality. 4 There have been many reports about the use of new drugs for the treatment of gastric cancer, such as paclitaxel, docetaxel, irinotecan and oxaliplatin, combined with cisplatin or fluorouracil (5-FU) for advanced gastric cancer. 5 Drugs such as paclitaxel, cisplatin and the novel oral fluoropyrimidine S-1 in combination with irinotecan are usually used in clinical practice in East Asia. 6 However, the response rate is low and the adverse reactions are severe, particularly for the combination of cisplatin with 5-FU. 7 The National Comprehensive Cancer Network guidelines suggest that the docetaxel + cisplatin + 5-FU (DCF) regimen may be used as the first-line treatment for advanced gastric cancer. 8 Therefore, this current study investigated the effects of the S-1 + irinotecan + oxaliplatin (TIROX) and DCF regimens on advanced gastric cancer in order to evaluate the short-term efficacy and safety of the TIROX regimen.
Patients and methods
Patient population
This prospective study enrolled consecutive patients with recurrent or metastatic gastric cancer in the Department of Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan Province, China between March 2011 and October 2012. The inclusion criteria were as follows: (i) gastric cancer diagnosed by pathology; (ii) patients not currently receiving chemotherapy (i.e. chemotherapy-naïve) or those who had stopped chemotherapy ≥1 month prior to enrolment; (iii) measurable lesions shown by computed tomography (CT) or magnetic resonance imaging (MRI); (iv) Eastern Cooperative Oncology Group (ECOG) score 9 ≤2; (v) expected survival time ≥3 months; (vi) normal results including routine blood, electrolytes, liver function, renal function, thrombotest and electrocardiogram; (vii) no contraindications for chemotherapy treatment; (viii) unresectable disease. Gastric cancer was diagnosed by pathology and classified according to tumour differentiation, presence of distant metastases, pathological type and tumour stage. 8
All of the study methods were approved by the Ethics Committee of the First Affiliated Hospital of Zhengzhou University (no. 2010-003854). All patients enrolled in the study gave written informed consent to participate.
Treatment regimens
The patients were divided randomly into the TIROX and DCF treatment groups using a computer-generated randomization schedule. The rationale for the dosage of the TIROX regimen was based on a previous phase II study. 10 In the TIROX group, patients received 40 mg/m2 S-1 orally twice daily after a meal on days 1–14; 150 mg/m2 irinotecan intravenously (i.v.) infused over 90 min on the first day; 85 mg/m2 oxaliplatin i.v. infused over 2 h on the first day. This treatment regimen was repeated every 21 days and a 21-day treatment period was defined as one chemotherapy cycle. In the DCF group, patients received 75 mg/m2 docetaxel i.v. and 75 mg/m2 cisplatin i.v. on the first day; 750 mg/m2 5-FU via continuous i.v. infusion once a day from the first day to the fifth day. This treatment regimen was repeated every 21 days and a 21-day treatment period was defined as one chemotherapy cycle. In both groups, 5 mg tropisetron i.v. twice daily was administered during the chemotherapy in order to prevent vomiting; and if bone marrow depression (i.e. leucopenia) occurred, granulocyte colony-stimulating factor (5 µg/kg) was given. All patients underwent a total of six chemotherapy cycles during this study.
Evaluation criteria for short-term efficacy and safety
After each set of two complete chemotherapy cycles for each group, the lesion size was monitored using CT (Brilliance CT 16-slice system; Philips Healthcare, Andover, MA, USA) and MRI (Intera 1.5 T and Achieva 1.5 T systems; Philips Healthcare) scanning with a scan slice thickness of 5 mm. The short-term efficacy was evaluated after six chemotherapy cycles according to the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines and recorded as complete response (CR), partial response (PR), stable disease, progressive disease, and response rate (RR = CR + PR). 11 Adverse reactions were assessed throughout the six chemotherapy cycles according to National Cancer Institute Common Toxicity Criteria 2.0 standards. 12
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 17.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Data are expressed as mean ± SD or n (%). The planned sample size in this study was based on a previously published paper. 13 The two treatment groups were compared using χ2-test. A P-value P < 0.05 was considered statistically significant.
Results
Clinical characteristics of patients with recurrent or metastatic gastric cancer who were recruited to a study that compared the short-term efficacy and safety of two chemotherapy regimens (n = 60).

Data presented as mean ± SD or n of patients (%).
TIROX group: patients received 40 mg/m2 S-1 orally twice daily after a meal on days 1–14; 150 mg/m2 irinotecan intravenously (i.v.) infused over 90 min on the first day; 85 mg/m2 oxaliplatin i.v. infused over 2 h on the first day. This treatment regimen was repeated every 21 days and a 21-day treatment period was defined as one chemotherapy cycle.
DCF group: patients received 75 mg/m2 docetaxel i.v. and 75 mg/m2 cisplatin i.v. on the first day; 750 mg/m2 fluorouracil via continuous i.v. infusion once a day from the first day to the fifth day. This treatment regimen was repeated every 21 days and a 21-day treatment period was defined as one chemotherapy cycle.
No statistically significant between-group differences; χ2-test (P ≥ 0.05).
ECOG, Eastern Cooperative Oncology Group.
The short-term efficacy of two chemotherapy regimens evaluated in patients with recurrent or metastatic gastric cancer following six chemotherapy cycles (n = 60).
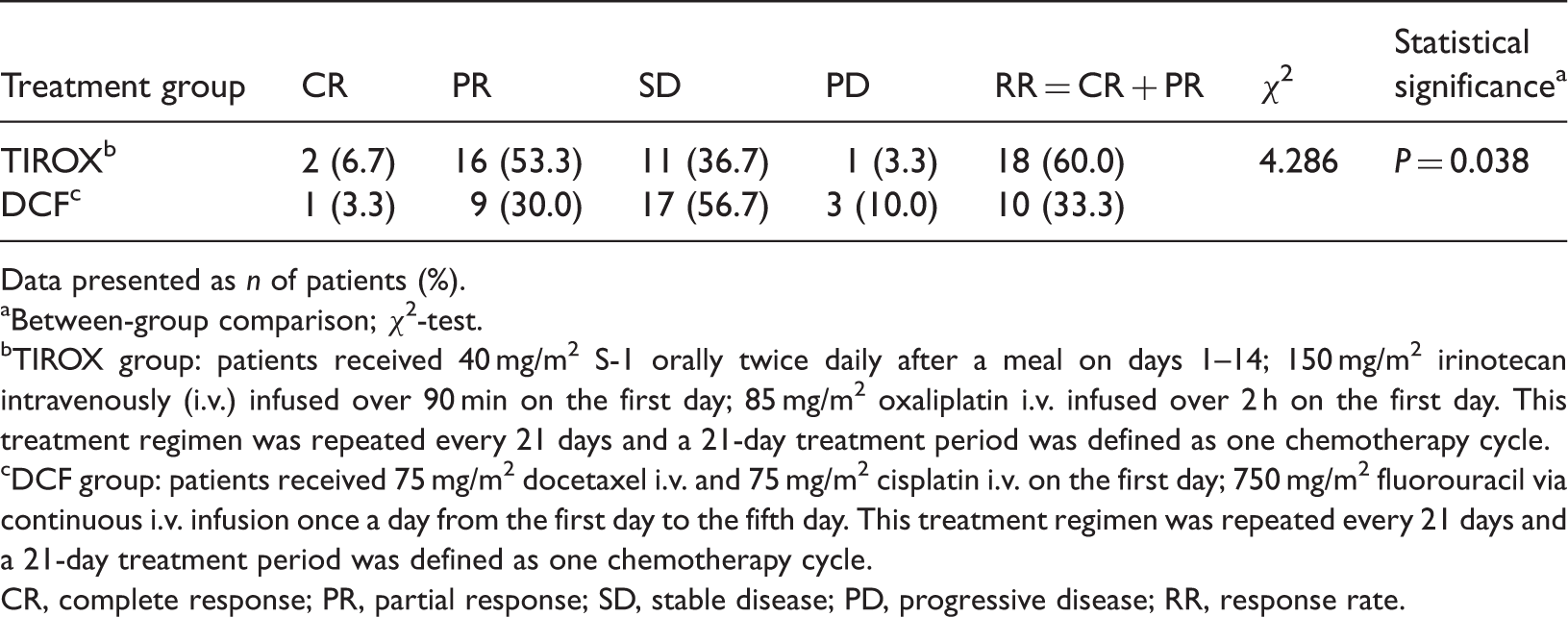
Data presented as n of patients (%).
Between-group comparison; χ2-test.
TIROX group: patients received 40 mg/m2 S-1 orally twice daily after a meal on days 1–14; 150 mg/m2 irinotecan intravenously (i.v.) infused over 90 min on the first day; 85 mg/m2 oxaliplatin i.v. infused over 2 h on the first day. This treatment regimen was repeated every 21 days and a 21-day treatment period was defined as one chemotherapy cycle.
DCF group: patients received 75 mg/m2 docetaxel i.v. and 75 mg/m2 cisplatin i.v. on the first day; 750 mg/m2 fluorouracil via continuous i.v. infusion once a day from the first day to the fifth day. This treatment regimen was repeated every 21 days and a 21-day treatment period was defined as one chemotherapy cycle.
CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; RR, response rate.
The incidence and severity of adverse reactions observed during the administration of six chemotherapy cycles of two chemotherapy regimens evaluated in patients with recurrent or metastatic gastric cancer (n = 60).

Data presented as n of patients (%).
TIROX group: patients received 40 mg/m2 S-1 orally twice daily after a meal on days 1–14; 150 mg/m2 irinotecan intravenously (i.v.) infused over 90 min on the first day; 85 mg/m2 oxaliplatin i.v. infused over 2 h on the first day. This treatment regimen was repeated every 21 days and a 21-day treatment period was defined as one chemotherapy cycle.
DCF group: patients received 75 mg/m2 docetaxel i.v. and 75 mg/m2 cisplatin i.v. on the first day; 750 mg/m2 fluorouracil via continuous i.v. infusion once a day from the first day to the fifth day. This treatment regimen was repeated every 21 days and a 21-day treatment period was defined as one chemotherapy cycle.
Between-group comparison; χ2-test.
NS, no statistically significant between-group differences; χ2-test (P ≥ 0.05).
Discussion
There is an ongoing clinical problem with the systemic chemotherapeutic treatment of advanced gastric cancer. This is because despite gastric cancer cells being sensitive to chemotherapeutic agents, they readily develop multiple drug resistance. 14 Previously used drugs such as cisplatin and 5-FU have several negative characteristics including lower therapeutic efficacy and severe toxicity. 15 The novel oral fluoropyrimidine S-1 was developed in Japan. 16 S-1 is an oral fluoropyrimidine designed to improve the antitumour activity of 5-FU whilst reducing its toxicity. It is a combination of three pharmacological compounds, tegafur, gimeracil (CDHP) and oteracil potassium, in a 1:0.4:1 molar ratio. 17 Tegafur is a prodrug of 5-FU; CDHP is an inhibitor of dihydropyrimidine dehydrogenase, which degrades 5-FU to inactive 5-fluorodihydrouracil in the liver, so it prolongs the half-life of 5-FU; and oteracil potassium inhibits the phosphorylation of 5-FU in the gastrointestinal tract, decreasing serious gastrointestinal toxicities including nausea, vomiting, stomatitis and diarrhoea. 17 S-1 maintains an effective concentration of 5-FU in the blood and tumour for a long period of time and reduces gastrointestinal adverse events. 18 In 1999, Japan approved the use of S-1 in the treatment of advanced gastric cancer. 19 In 2010, a phase III clinical trial demonstrated that the therapeutic effects of the S-1 + cisplatin and 5-FU + cisplatin regimens were similar, but the safety profile was better with the S-1 + cisplatin regimen than with the 5-FU + cisplatin regimen. 20
Oxaliplatin, a third generation platinum antitumour drug, has no cross-resistance with cisplatin and carboplatin. 21 Oxaliplatin, when used in combination with 5-FU, is known to cause higher frequencies of peripheral neuropathy than when 5-FU is used alone. 22 Compared with cisplatin, oxaliplatin has better therapeutic effects and milder adverse reactions, and its use can avoid the severe renal toxicity caused by cisplatin in the treatment of advanced gastric cancer. 23 Irinotecan, an inhibitor of topoisomerase I, and its active metabolite, SN-38, have antitumour effects. 24 It is likely that irinotecan is activated to SN-38 by liver carboxylesterase, so liver carboxylesterase is an important enzyme in patients given this drug. 25 Irinotecan is effective against gastric, colorectal and lung cancer.26–28 A phase II clinical trial demonstrated that a fortnightly triple therapeutic regimen consisting of 5-FU + calcium folinate + oxaliplatin + irinotecan had an overall response rate of 63.3% in patients with advanced gastric cancer. 29 Based on these previous results, 29 this present study used the TIROX regimen of S-1 + irinotecan + oxaliplatin in patients with advanced gastric cancer, obtaining an overall response rate of 60%. Although the TIROX regimen demonstrated better short-term efficacy in patients with advanced gastric cancer than the DCF regimen in this present study, it was associated with significantly higher rates of grade III–IV leucopenia and grade III–IV neurotoxicity, which might limit its large-scale clinical application.
This present study had a number of limitations. First, it was not able to explore different doses and timings of administration for the constituents of the TIROX regimen due to the small number of patients. Secondly, the rates of long-term progression-free survival and overall survival were not measured.
In conclusion, this present preliminary study demonstrated that the TIROX regimen was effective for the treatment of advanced gastric cancer, but its safety profile remains to be further investigated.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
