Abstract
The human epidermal receptor-2 (HER-2) is amplified in up to 25% of patients with gastroesophageal adenocarcinomas. Although the presence of this amplification does not appear to confer a poor prognosis, it provides a valuable novel therapeutic target for this group of patients. Trastuzumab is a fully humanized monoclonal antibody directed at HER-2 which binds the external domain of the receptor and exerts its action via a combination of antibody-dependent cytotoxicity, reduced shedding of the extracellular domain, inhibition of dimerization and possibly receptor downregulation. The ToGA trial was an international multicentre randomized phase III study which evaluated the addition of trastuzumab to a cisplatin plus fluoropyrimidine chemotherapy doublet in 594 patients with HER-2-positive advanced gastric or oesophagogastric junction adenocarcinoma. The combination of the antibody with chemotherapy significantly improved response rate, median progression-free survival and median overall survival without additional toxicity or adversely affecting quality of life. Accordingly, trastuzumab plus chemotherapy is now a standard first-line treatment option for patients with advanced HER-2-positive gastroesophageal cancer. Unfortunately, many patients with HER-2-positive cancer exhibit primary resistance to trastuzumab and the remainder will acquire resistance to the antibody; therefore, urgent investigation into novel agents which may circumvent resistance mechanisms is warranted. Small molecule inhibitors of HER-2, which commonly also target other members of the HER family of receptors, such as EGFR and HER-3, are currently undergoing evaluation in gastroesophageal cancer as first-line alternatives to trastuzumab and second-line salvage treatments for trastuzumab-resistant disease. Extrapolating the successful use of trastuzumab in the advanced disease setting, clinical trials are underway to assess the role of this antibody in the perioperative and adjuvant settings, where it is hoped that it will have a meaningful impact upon the currently poor survival rates.
Keywords
Background
Worldwide, gastric cancer is the fourth most commonly diagnosed cancer and the second most common cause of cancer death; accounting for almost 10% of all cancer deaths in 2008 [Ferlay et al. 2010]. The majority of patients present with advanced, inoperable disease, where treatment is palliative and the median survival is just 3 months with supportive care alone [Murad et al. 1993; Pyrhonen et al. 1995]. Chemotherapy improves survival in patients with advanced gastric cancer and combination regimens are superior in this respect to monotherapy [Wagner et al. 2010]. Whilst there is no globally accepted standard first-line regimen, a platinum-fluoropyrimidine doublet [Kang et al. 2009], with or without epirubicin [Cunningham et al. 2008] or docetaxel [Tebbutt et al. 2010; Van Cutsem et al. 2006], is most commonly used. However, even with optimal combination chemotherapy, the median survival in Western studies remains less than 1 year [Cunningham et al. 2010; Van Cutsem et al. 2006]. A longer median survival has been reported in a Japanese phase III study comparing fluoropyrimidine monotherapy, with the oral agent, S-1, with a S-1/cisplatin doublet. However, this is likely to reflect the use of second- and third-line chemotherapy, evidenced by the relatively short median progression-free survival (PFS), and possibly more patients with a lower burden of disease at study entry, rather than the efficacy of the S-1+/- cisplatin regimen itself [Koizumi et al. 2008]. Until recently, there were no supporting phase III trial data for second-line chemotherapy for patients with advanced gastric cancer, however, a survival benefit for irinotecan or docetaxel monotherapy has now been reported in patients previously treated with a platinum and fluoropyrimidine [Kang et al. 2012], confirming the efficacy of second-line irinotecan that had been reported in a small German study, which had closed prior to completing planned accrual [Thuss-Patience et al. 2011]. Although modest increments in survival may be possible from further refinements of first- and second-line chemotherapy regimens, attention has turned to potential molecular targets in oesophagogastric cancer. Like advanced breast cancers, 15–25% of advanced gastroesophageal cancers overexpress the human epidermal receptor 2 (HER-2). Trastuzumab, a monoclonal antibody targeting HER-2, improves survival in HER-2-positive advanced [Slamon et al. 2001] and early breast cancer [Slamon et al. 2011; Smith et al. 2007] and has been a key component of breast cancer treatment of for over a decade. Following the successful phase III evaluation of the antibody in HER-2-positive advanced gastric and oesophagogastric junction (OGJ) cancers, demonstrating improved radiological response rate, PFS and overall survival with the addition of trastuzumab to a cisplatin/fluoropyrimidine doublet [Bang et al. 2010], trastuzumab has become a standard treatment option for patients with this disease also. Whether a similar benefit will be attained from HER-2 inhibition with trastuzumab in patients with operable gastroesophageal cancers is currently unknown.
This review will discuss the current and future roles of trastuzumab in the treatment of gastroesophageal cancer, resistance mechanisms to the antibody, strategies to overcome trastuzumab resistance and novel anti-HER-2 targeted agents.
The HER-2 receptor
HER-2 (also known as erbB-2) is one of a family of four identified human epidermal receptors, which also includes the epidermal growth factor receptor (EGFR, HER-1), the HER-3 and HER-4 receptors. The four receptors and their respective ligands form a complex, key signalling network for cell proliferation and survival [Yarden, 2001]. The oncogene encoding the HER-2 receptor (HER-2/neu or erbB2) was first described in 1984 [Schechter et al. 1984] and mapped to q21 of chromosome 17 [Coussens et al. 1985; Schechter et al. 1985]. The gene product is a 185 kDa tyrosine kinase receptor [Akiyama et al. 1986; Toyoshima et al. 1986]. The first description of HER-2 amplification in gastric cancer was in the MKN-7 gastric cancer cell line [Fukushige et al. 1986], with a subsequent report in two of nine gastric cancer resections [Yokota et al. 1986], with amplification most commonly detected in tubular (intestinal) cancers [Yokota et al. 1988].
The HER-2 receptor protein consists of four extracellular domains, one transmembrane domain and a cytoplasmic tyrosine kinase domain [Cho et al. 2003]. HER-2 differs from the other three HER family members in that no ligand has been identified. Activation of the receptor can be either ligand-independent with the formation of HER-2 homodimers in the presence of tumour HER-2 overexpression [Worthylake et al. 1999] or ligand-dependent heterodimerization with EGFR, HER-3 or HER-4 following receptor-specific ligand binding. Heregulin is a key ligand for the HER-3 receptor [Carraway et al. 1994] and co-expression of HER-2 and HER-3 leads to high-affinity heregulin binding and tyrosine phosphorylation. Despite the HER-3 receptor possessing no functional tyrosine kinase activity itself [Guy et al. 1994], the heterodimer comprising HER-2/HER-3 is thought to produce the most potent mitogenic signal [Pinkas-Kramarski et al. 1996], stimulating cell proliferation and inhibiting apoptosis via the mitogen-activated protein kinase (MAPK) and phosphatidylinositol-3-kinase (PI3K) pathways [Daly et al. 1999]. A simplified version of HER-2 dimers and the intracellular signalling pathway is depicted in Figure 1.

Simplified diagram of the HER family signalling network. Ligand binding to HER-3 stimulates heterodimerization with HER-2 and transphosphorylation of HER-3, initiating the intracellular signalling cascade, resulting in cell growth and survival. (Illustration courtesy of Alessandro Baliani, Copyright © 2012.)
Prognostic effect of HER-2 overexpression and amplification
The presence of HER-2 overexpression or HER-2/neu amplification is associated with an aggressive phenotype in breast cancer, conferring a poor prognosis [Slamon et al. 1987, 1989]. Although initial studies in gastric cancer suggested a similarly negative impact on prognosis [Yonemura et al. 1991], more recent studies have reported no significant prognostic effect [Shah et al. 2011; Terashima et al. 2011; Yoon et al. 2011].
These studies are summarized in Table 1. The reason for these conflicting data is most likely the previous lack of standardized testing and scoring of HER-2 in gastric cancer, leading to marked heterogeneity in the definition of HER-2-positive cases. Overall, it appears likely that in contrast to breast cancer, HER-2-positive gastric cancer is not associated with an adverse prognosis. This suggests that HER-2 amplification exerts distinct biological effects according to the primary tumour site, which at present are not fully understood. However, as inhibition of HER-2 signalling is beneficial in patients with HER-2 amplified gastric and OGJ cancers, at least partial dependence upon this oncogenic signalling through HER-2 can be reasonably assumed. High levels of concordance between primary tumours and paired metastatic sites suggest that HER-2 amplification is an earlier event and not simply acquired by cells with metastatic potential [Bozzetti et al. 2011]. Disconcordance, where observed, is largely due to primary tumour heterogeneity rather than a true difference in HER-2 gene copy number between the primary and metastatic site [Kim et al. 2011].
Recent large studies examining HER-2 as a prognostic marker in gastric cancer.
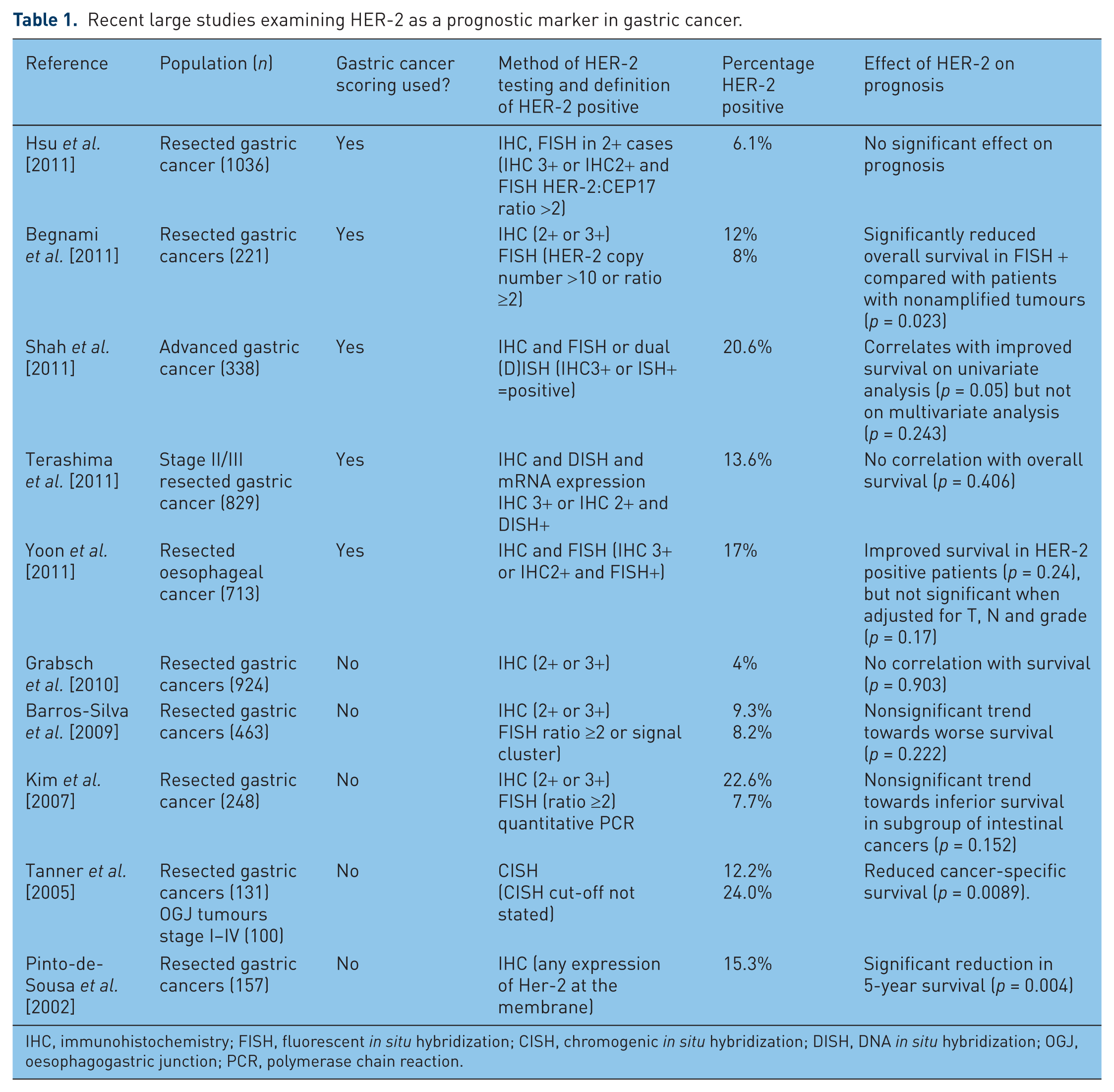
IHC, immunohistochemistry; FISH, fluorescent in situ hybridization; CISH, chromogenic in situ hybridization; DISH, DNA in situ hybridization; OGJ, oesophagogastric junction; PCR, polymerase chain reaction.
HER-2 testing in gastric cancer
A separate gastric cancer HER-2 immunohistochemistry (IHC) scoring system has been developed due to key differences in HER-2 staining patterns in gastric compared with breast cancer, noted in a study designed to validate HER-2 testing for the phase III trial of trastuzumab in gastric cancer. In particular, an increased frequency of tumour heterogeneity and incomplete basolateral reactivity led the authors to recommend that incomplete strong staining could be IHC 3+ and for biopsy samples, the 10% required cut-off used in breast cancer should be replaced by a minimum of 5 cohesive cells staining with 3+ intensity [Hofmann et al. 2008]. These guidelines were further validated in a larger study [Ruschoff et al. 2010] and have been accepted into routine clinical practice. The gastric cancer HER-2 scoring is summarized in Table 2.
Recommended scoring for HER-2 in gastric cancer.

IHC, immunohistochemistry; ISH, in situ hybridization.
Tumours of the OGJ (15–33%) [Bang et al. 2009; Boers et al. 2011; Terashima et al. 2011; Yoon et al. 2011] and oesophagus (16–21%) [Thompson et al. 2010; Yoon et al. 2011] are most frequently HER-2 positive, with the lowest rates reported in distal gastric cancers (7%) [Boers et al. 2011]. This may in part be due to the association with intestinal histology, where HER-2-positive diffuse cancers are uncommon [Bang et al. 2009].
Development of trastuzumab
Following the development of specific anti-HER-2 monoclonal antibodies [Fendly et al. 1990], antiproliferative effects in both HER-2-positive breast and gastric cancer cell lines and xenografts were reported, with synergy with chemotherapy agents including cisplatin noted [Hancock et al. 1991; Kasprzyk et al. 1992]. A humanized antibody directed at the extracellular domain of HER-2 was developed, with increased affinity to the HER-2 receptor, improved antibody-dependent cytotoxicity (ADCC) and equal antiproliferative potency in HER-2-positive breast cancer cell lines to the original murine antibody [Carter et al. 1992]. Similar efficacy of trastuzumab has since been reported in HER-2-positive gastric cancer cells lines and xenografts [Matsui et al. 2005; Tanner et al. 2005]. Trastuzumab binds the HER-2 receptor on the C-terminal of domain IV, the juxtamembrane region, facilitating endocytosis of the receptor-antibody complex and blocking constituent activation of HER-2, which can occur via proteolysis of the extracellular domain, leaving a truncated HER-2 receptor known as p95 HER-2 [Cho et al. 2003]. Trastuzumab is also believed to exert its anticancer effects by direct blockade of dimerization, thereby preventing tyrosine kinase activation, ADCC via recruitment of host myeloid cells [Clynes et al. 2000] and possibly via anti-angiogenic effects [Petit et al. 1997]. Figure 2 is a diagrammatic representation of the HER-2 receptor.
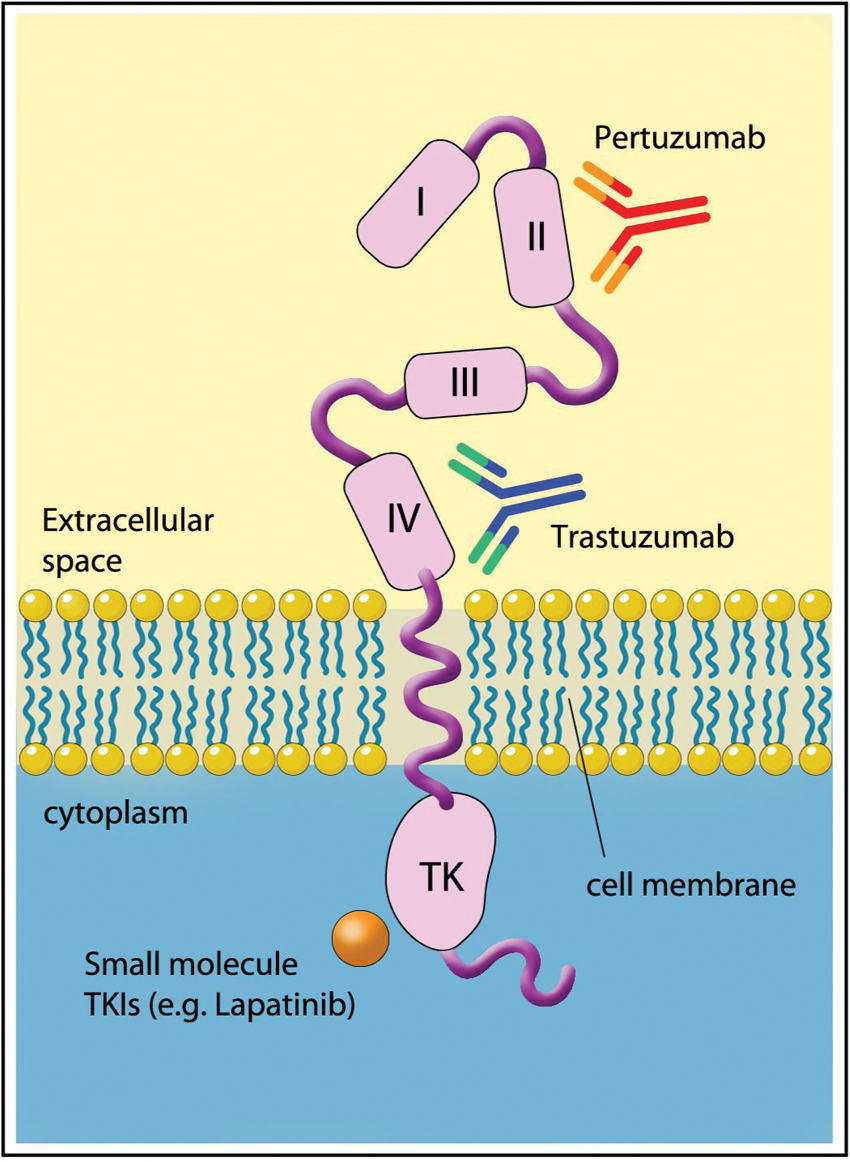
The HER-2 receptor and binding sites for trastuzumab, pertuzumab and small molecule inhibitors. (Illustration courtesy of Alessandro Baliani, Copyright © 2012.)
Further preclinical studies of trastuzumab provided the rationale for the clinical evaluation of trastuzumab in combination with chemotherapy for HER-2-positive gastric cancer; with enhanced antitumour effect observed with capecitabine, cisplatin, paclitaxel, docetaxel and irinotecan [Fujimoto-Ouchi et al. 2007], and a synergistic effect reported with cisplatin in HER-2 positive gastric cancer cell lines [Kim et al. 2008b].
Clinical evaluation of trastuzumab in gastroesophageal cancer
A single phase II study evaluating trastuzumab in advanced gastric cancer has been recently published, 4 years after the results were initially presented. A total of 228 patients with locally advanced unresectable or metastatic adenocarcinoma of the stomach or OGJ were screened to select 22 patients with HER-2-positive cancers for treatment with trastuzumab (8 mg/kg loading dose then 6 mg/kg q 21 days) plus cisplatin monotherapy (75 mg/m2 q 21 days). The response rate (32%) and median PFS (5.1 months) [Gravalos et al. 2011] do not compare favourably with those reported with combination chemotherapy, probably reflecting the suboptimal chemotherapy backbone selected. For the phase III ToGA trial, a chemotherapy doublet comprising cisplatin (80 mg/m2 day 1) with either infused 5-fluororacil (5-FU; 800 mg/m2 days 1–5) or capecitabine (2000 mg/m2/day days 1–14) was chosen for combination with trastuzumab (8 mg/kg loading dose then 6 mg/kg) every 21 days. A total of 3665 patients were centrally screened for HER-2 overexpression by IHC or amplification by fluorescent in situ hybridization (FISH; HER2:CEP17 ratio ≥2), of whom 810 were HER-2 positive, defined as IHC3+, or FISH+ with any IHC result. Of those, 584 patients were randomized to the chemotherapy doublet, with or without trastuzumab in this open-label phase III study designed to detect an increase in overall survival from 10 to 13 months with trastuzumab (hazard ratio [HR] 0.77) with 80% power and a two-sided log rank test α = 0.05. The statistical calculations had been amended due to a lower than expected event rate during the study; the anticipated poor prognosis of HER-2-positive patients and projected median survival of just 7 months with chemotherapy alone (and expected increase to 10 months with the addition of trastuzumab, HR 0.70) was incorrect. In the intent-to-treat population, at the final analysis, median survival was 11.1 months with the chemotherapy doublet, significantly increased to 13.8 months with the addition of trastuzumab (HR 0.74, 95% confidence interval [CI] 0.60–0.91, p = 0.0046). Median PFS (6.7 compared with 5.5 months, HR 0.71, 95% CI 0.59–0.85, p = 0.0002) and radiological response rate (47% compared with 35%, odds ratio 1.70, 95% CI 1.22–2.38, p = 0.0017) were similarly improved with the antibody. Patients with the strongest expression of HER-2 gained the greatest benefit in a preplanned subgroup analysis, with an unprecedented median survival of 17.9 months in patients with IHC3+ and FISH-positive tumours who received trastuzumab, compared with 12.3 months with chemotherapy. Furthermore, in a post hoc exploratory analysis, the subgroup of 446 patients with IHC2+/FISH+ or IHC3+ cancers had a 4.2 month prolongation of median overall survival with the addition of trastuzumab to chemotherapy (16.0 versus 11.8 months), which has formed the basis of the EMEA approval of trastuzumab in gastric cancer. Efficacy outcomes for the ITT population and these molecular defined subgroups who benefitted most are recorded in Table 3.
Efficacy outcomes in the ToGA trial.
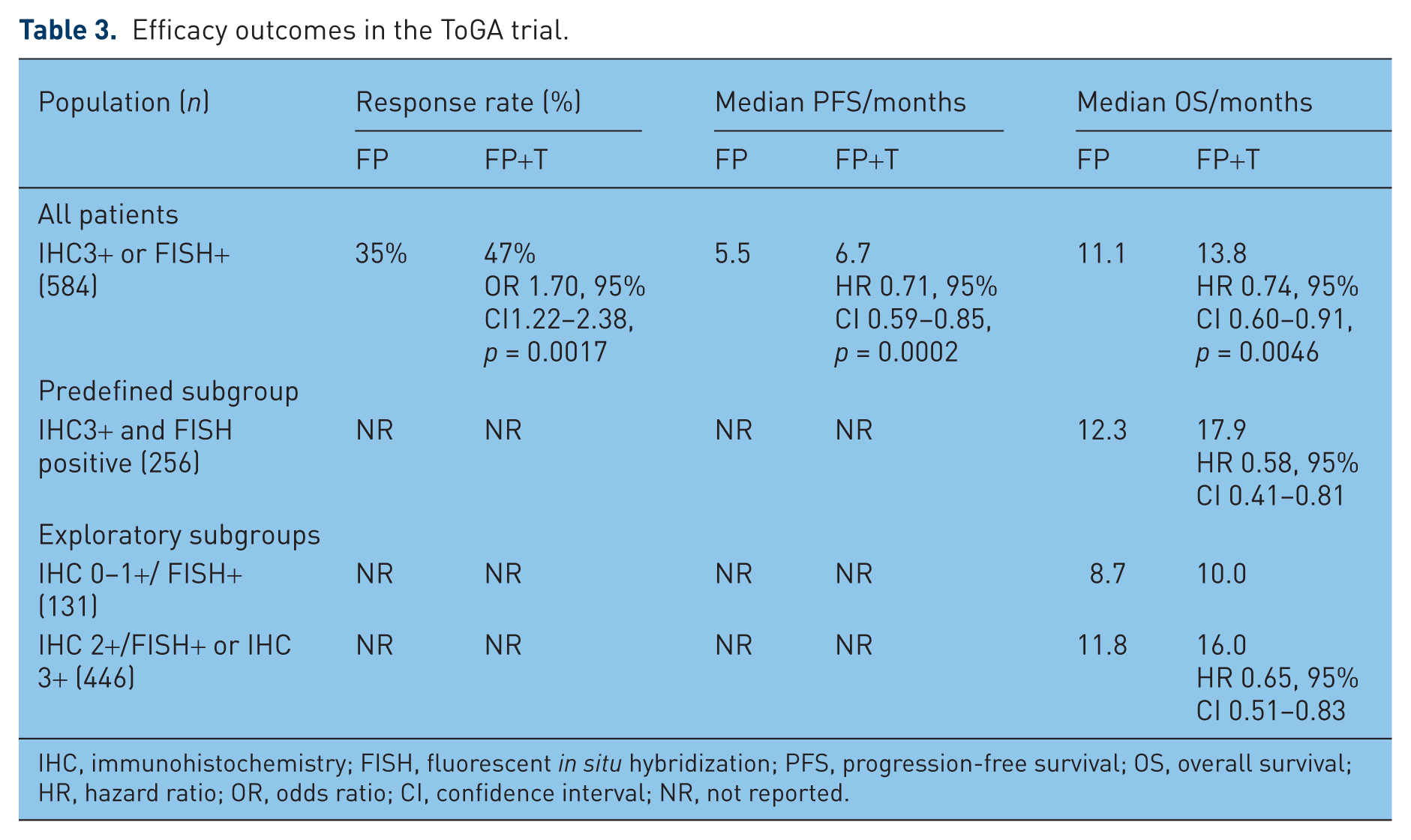
IHC, immunohistochemistry; FISH, fluorescent in situ hybridization; PFS, progression-free survival; OS, overall survival; HR, hazard ratio; OR, odds ratio; CI, confidence interval; NR, not reported.
Whilst some clinicians would not consider a platinum–fluoropyrimidine doublet the optimal chemotherapy regimen, it is well tolerated and remains an internationally accepted first-line treatment option. Furthermore, additive cardiac toxicity might have been expected had the investigators selected an epirubicin-based triplet based upon previous studies of trastuzumab with doxorubicin in breast cancer [Seidman et al. 2002], although more recent studies have safely combined trastuzumab with moderate cumulative doses (360 mg/m2) of the less cardiotoxic epirubicin [Untch et al. 2010]. With the doublet regimen selected for the ToGA trial, trastuzumab added little toxicity, with no increase in chemotherapy related grade 3–4 toxicities (68% both arms) or cardiac events (6% both arms), although a numerical increase in treatment related deaths was observed (3% compared with 1%). The only increase in cardiotoxicity reported with trastuzumab was a modest increase in asymptomatic left ventricular dysfunction defined as a drop of left ventricular ejection fraction (LVEF) of ≥10% to below 50% (5% compared with 1% with chemotherapy alone) detected on 12-weekly cardiac assessments. Whilst this is of little relevance to patients with advanced disease whose median survival is so limited, it may be more important in the curative-intent setting where, as we have seen in studies of early breast cancer [Slamon et al. 2011], cardiac failure could occur in a small proportion of long-term survivors. Current studies of trastuzumab in gastroesophageal cancer are summarized in Table 4. Notably at present, none evaluate trastuzumab with an anthracycline-based triplet, although two phase II studies are combining the antibody with a novel triplet chemotherapy backbone comprising docetaxel, oxaliplatin plus a fluoropyrimidine, one with the addition of the anti-VEGF-A monoclonal antibody, bevacizumab.
Selected current phase II and III studies evaluating trastuzumab in gastroesophageal cancer.

DFS, disease-free survival; PFS, progression-free survival; pCR, pathological complete remission; OS, overall survival; HR, hazard ratio; OR, odds ratio; CI, confidence interval; OGJ, oesophagogastric junction; ORR, overall response rate; CAPOX, capecitabine + oxaliplatin; ECF, epirubicin, cisplatin + infused 5-fluorouracil; ECX, epirubicin, cisplatin + capecitabine.
Other agents targeting HER-2
Trastuzumab emtansine (T-DM1)
Trastuzumab emtansine (T-DM1) is a novel antibody–drug conjugate linking trastuzumab to the cytotoxic agent, DM1, a derivative of maytansine, a potent microtubule inhibitor developed in the early 1970s [Kupchan et al. 1972]. As a systemic therapy, severe, mainly gastrointestinal toxicity was dose-limiting and efficacy was minimal at tolerable doses [Issell and Crooke, 1978]. Therefore, attention turned to strategies to deliver the cytotoxicity directly into tumour cells, via conjugation with antibodies. Initial conjugates utilizing disulphide bonds, which were anticipated to undergo endosomal reduction thereby releasing the active drug within the target tumour cell [Chari et al. 1992], were limited by insufficient cleavage of the disulphide linker with consequent poor drug delivery [Austin et al. 2005]. These have been superseded by a thioether linkage, which despite being nonreducible, show improved efficacy and reduced toxicity [Lewis Phillips et al. 2008]. T-DM1 is well tolerated and has encouraging monotherapy activity in patients with advanced breast cancer, who are resistant to trastuzumab. The most common toxicities are hypokalaemia, thrombocytopenia, transaminitis and fatigue [Burris et al. 2011; Krop et al. 2010a, 2010b]. Two phase III studies are currently underway in the first- [ClinicalTrials.gov identifier: NCT01120184] and second-line treatment [ClinicalTrials.gov identifier: NCT00829166] of advanced breast cancer. With promising preclinical activity reported in HER-2-positive gastric cancer cell lines [Barok et al. 2011], we are hopeful that evaluation in patients HER-2-positive advanced gastroesophageal cancer will soon follow.
Pertuzumab
Pertuzumab is a monoclonal antibody targeting HER-2 in domain II, the extracellular subdomain required to dimerize with other HER family receptors, preventing formation of the highly mitogenic HER-2/HER-3 dimer. Like trastuzumab, the antibody is not effective in patients without HER-2 amplification [Gianni et al. 2012]. However, in HER-2 positive disease, promising results from a phase II study combining pertuzumab with trastuzumab in patients with previously trastuzumab-treated advanced breast cancer [Baselga et al. 2010] have translated into a 6-month improvement in PFS from the addition of pertuzumab to first-line docetaxel and trastuzumab in the recent phase III CLEOPATRA study in advanced breast cancer [Baselga et al. 2012]. Computer models and molecular dynamic simulations undertaken to investigate the apparent synergy with trastuzumab have reported that the HER-2 receptor is altered by trastuzumab binding, increasing the plasticity of the receptor and enhancing association with pertuzumab. Similarly, pertuzumab may enhance trastuzumab binding [Fuentes et al. 2011]. The landmark CLEOPATRA study could be considered as significant an advance as the initial phase III study of trastuzumab in advanced breast cancer, with a comparable magnitude of benefit reported with each additional antibody over standard therapy: the HR for the addition of trastuzumab to chemotherapy for time to disease progression was 0.51 (95% CI 0.41–0.63) [Slamon et al. 2001]. Similarly, the HR for the addition of pertuzumab to docetaxel plus trastuzumab for PFS was 0.62 (95% CI 0.51–0.75) [Baselga et al. 2012]. A trend towards improved survival was noted at this preplanned interim analysis 0.64 (95% CI 0.47–0.88; p = 0.005) which did not meet the study’s early stopping rule, allowing completion of planned accrual. The second antibody adds moderate toxicity, with higher rates of diarrhoea, rash and febrile neutropenia with the triplet regimen, but no increase in left ventricular dysfunction was evident [Baselga et al. 2012]. As a result of these data, an adjuvant study adding pertuzumab to trastuzumab has opened to accrual in early breast cancer (APHINITY). Evaluation in advanced gastric cancer is also planned, with a phase II study of cisplatin, capecitabine and trastuzumab, randomizing patients to two different pertuzumab dose schedules, expected to open in 2012 [ClinicalTrials.gov identifier: NCT01461057].
Lapatinib
Lapatinib is an orally active small molecule inhibitor of HER-2 and EGFR, which is licensed for the treatment of HER-2-positive advanced breast cancer. Preclinical efficacy in HER-2 amplified gastric cancer cell lines has been reported, with synergy observed with trastuzumab [Wainberg et al. 2010], oxaliplatin [Kim et al. 2008a] and the oral fluoropyrimidine S-1 [Tanizaki et al. 2010]. Of interest, paclitaxel and cisplatin demonstrated a sequence-dependent synergistic effect, where the cytotoxic agent was optimally delivered before lapatinib [Kim et al. 2008a]. Lapatinib also appears to reverse irinotecan resistance in vitro [Yashiro et al. 2011], providing rationale for its use in the second- or third-line treatment of advanced gastroesophageal cancer.
As monotherapy, lapatinib has been evaluated in a phase II study of previously untreated patients with advanced gastric cancer. Perhaps unsurprisingly, the drug had limited efficacy in the unselected cohort (confirmed partial responses reported in 4 of 47 patients, median survival 4.8 months), and a significantly longer median survival was noted in patients with HER-2 gene expression above the median value (6.8 compared with 3.0 months in patients with expression less than median, p = 0.0031) [Iqbal et al. 2011]. However, even a median survival of 6.8 months in the high HER-2 expressers compares poorly to first-line studies of combination chemotherapy [Cunningham et al. 2008]. A small phase II study of lapatinib monotherapy in 25 patients with previously treated EGFR or HER-2-positive advanced gastric cancer (measured by FISH or IHC) reported no objective responses, and remains unpublished [Hecht et al. 2008]. Owing to the limited efficacy as monotherapy, combination of lapatinib with chemotherapy is now being investigated: a placebo-controlled randomised phase III study (LOGiC) will determine whether the addition of lapatinib to capecitabine and oxaliplatin improves survival in previously untreated patients with HER-2-positive advanced gastric or OGJ cancer. Early blinded safety results suggested no unmanageable toxicity, with grade 3 diarrhoea in 2 of 21 evaluable patients (9.5%) [Hecht et al. 2009], despite potential concerns regarding overlapping gastrointestinal toxicities of the three agents. A phase III study in the second-line setting (TYTAN) has also reported blinded safety data for the combination of paclitaxel with or without lapatinib, with grade 3 diarrhoea similarly reported in 10% of patients.[Satoh T, 2010] The clinical applicability of the TYTAN study results will largely depend upon the efficacy in the subgroup of patients who received first-line trastuzumab, which became standard therapy during the study accrual. An EORTC randomized phase II study is evaluating the combination of lapatinib with a combination of epirubicin, cisplatin and either 5-FU or capecitabine (ECF or ECX) in patients with HER-2 positive advanced gastric cancer, which will also be investigated in the operable disease setting in a planned amendment to the phase III MRC ST03 trial. These studies in progress are summarized in Table 5.
Current phase II and III studies evaluating other HER-2 targeted agents in gastroesophageal cancer.

Planned amendment to the current study of ECX ± bevacizumab, subject to completion of pilot safety study.
DFS, disease-free survival; PFS, progression-free survival; pCR, pathological complete remission; OS, overall survival; HR, hazard ratio; OR, odds ratio; CI, confidence interval; OGJ, oesophagogastric junction; ORR, overall response rate; 5-FU, 5-fluorouracil; CAPOX, capecitabine + oxaliplatin; ECF, epirubicin, cisplatin + infused 5-fluorouracil; ECX, epirubicin, cisplatin + capecitabine.
Other small molecule inhibitors of HER-2
Dacomitinib (PF00299804) is a ‘pan-HER’ small molecule inhibitor, with antitumour activity reported in HER-2-positive gastric cancer cell lines and xenografts, and synergy observed with several commonly used cytotoxics (5-FU, cisplatin, docetaxel and paclitaxel), targeted agents (trastuzumab and investigational inhibitors of IGF1R, ERK1/2 and PI3K/mTOR) [Nam et al. 2011a]. A phase II study will determine its monotherapy activity in patients with previously treated HER-2-positive advanced gastric or OGJ cancers.
A phase II study of Afatinib (BIBW2992), an irreversible inhibitor of EGFR and HER-2 has recently been initiated in trastuzumab-refractory advanced oesophagogastric cancer [ClinicalTrials.gov identifier: NCT01522768]. HM781-36B is an irreversible pan-HER inhibitor, which also demonstrates potent preclinical activity in HER-2-positive gastric cancer cell lines and xenografts. Of interest, although the targeted agent was inactive as monotherapy in HER-2-negative cell lines, synergy with chemotherapy was reported in both HER-2 amplified and nonamplified models [Nam et al. 2011b]. The drug is currently undergoing phase I evaluation in solid tumours [ClinicalTrials.gov identifiers: NCT01455584 and NCT01455571].
Other pan-HER inhibitors, including AZD8931 and BMS-599626, are undergoing early phase clinical testing, but currently lack published data in gastroesophageal cancers, with no trials in this disease registered as yet.
Resistance to trastuzumab
Whilst some patients with HER-2-positive advanced gastric cancer exhibit primary resistance to trastuzumab, all acquire resistance after a relatively short treatment duration (median PFS 6.7 months) [Bang et al. 2010]. At present, we are not aware of any published studies which have evaluated mechanisms of trastuzumab resistance in gastroesophageal cancers.
In the decade since the integration of trastuzumab into the routine management of breast cancer, much research has focused on the mechanisms of primary and acquired resistance, the latter of which develops in the majority of patients. Whilst resistance remains poorly elucidated on an individual patient basis, important mechanisms in breast cancer have been established. These can be broadly divided into aberrations in the downstream signalling PI3K/AKT/mTOR pathway, shedding of the extracellular domain of the receptor with formation of the constitutively activated p95 HER-2, and redirection of signalling via other growth factor receptors.
PTEN (phosphatase and tensin homologue protein), is the protein product of the PTEN tumour suppressor gene which antagonizes the function of PI3K, thereby inhibiting signal transduction through the PI3K/AKT pathway. Loss of expression occurs in approximately one third of breast cancers [Perez-Tenorio et al. 2007] and renders the tumour at least partially resistant to trastuzumab [Nagata et al. 2004]. This resistance can potentially be overcome by PI3K inhibitors, which abrogate the constitutive signalling through this pathway in preclinical studies [Nagata et al. 2004]. Similarly, combination of trastuzumab with AKT or mTOR inhibitors in preclinical studies reverses PTEN loss-induced trastuzumab resistance [Lu et al. 2007]. PTEN loss has also been reported to predict benefit from lapatinib [Dave et al. 2011].
Activating mutations in the gene encoding the p110 catalytic subunit α of PI3K (PIK3CA) also switch on signalling through this important pathway, negating the upstream inhibitory effect of trastuzumab [Berns et al. 2007] and additionally conferring resistance to lapatinib [Wang et al. 2011]. Such mutations are relatively common in breast cancer, reported in approximately 25% of patients and associated with HER-2 overexpression [Saal et al. 2005]. Again, PI3K, AKT and mTOR inhibitors are under evaluation in advanced breast cancer in combination with trastuzumab to circumvent this resistance mechanism. A phase I study in patients with HER-2-positive advanced breast cancer previously treated with trastuzumab reported a 44% response rate to the mTOR inhibitor, everolimus (RAD001) combined with paclitaxel and trastuzumab [Andre et al. 2010], despite the majority of patients having progressed on or within 3 months of receiving the antibody for advanced disease or within 12 months of adjuvant trastuzumab. A 15% response rate was reported in a phase II study of the chemotherapy-free combination of trastuzumab and everolimus in patients with a more stringent definition of trastuzumab resistance, where all patients had progressed on trastuzumab-based therapy [Morrow et al. 2011]. Of interest, neither loss of PTEN nor PIK3CA mutations was predictive for benefit from everolimus.
P95 HER-2, a truncated form of the receptor formed by cleavage of the extracellular domain, lacks a binding site for trastuzumab, bestowing resistance to the antibody. Preclinical studies have confirmed that breast cancer cell lines expressing p95 HER-2 are resistant to trastuzumab, but remain sensitive to the small molecule inhibitor, lapatinib. Correlation with archival tumour from 47 patients with advanced breast cancer treated with trastuzumab, with or without chemotherapy, revealed expression of p95 in 9 out of 46 patients, 8 of whom had not responded to trastuzumab [Scaltriti et al. 2007]. The combination of trastuzumab and lapatinib has been evaluated in a phase III study of 296 patients with trastuzumab-resistant advanced breast cancer, in whom the combination was superior to lapatinib alone (HR for progression 0.73, 95% CI 0.57–0.93, p = 0.008). Unfortunately, correlation with p95 expression was not performed [Blackwell et al. 2010]. This combination is particularly attractive for patients with trastuzumab-resistant gastric cancer, potentially providing a chemotherapy-free treatment option after first-line chemotherapy plus trastuzumab. Inhibitors of the chaperone protein, HSP90, have also been reported to abrogate signalling through p95 HER-2 in vivo [Chandarlapaty et al. 2010]. A study evaluating an HSP90 inhibitor in combination with trastuzumab in the second-line treatment of HER-2-positive advanced gastric cancer [ClinicalTrials.gov identifier: NCT01402401] is underway.
Increased signalling through other receptor tyrosine kinases including EGFR [Diermeier et al. 2005], MET [Shattuck et al. 2008] and the insulin-like growth factor 1 receptor (IGF1R) [Lu et al. 2001] has been described in HER-2-resistant cells. Inhibition of IGF1R improves trastuzumab sensitivity in preclinical studies [Browne et al. 2011], providing rationale for a dual HER-2/IGF1R targeted strategy to prevent or at least delay acquired resistance via this mechanism.
Cyclin E overexpression has also been recently correlated with reduced trastuzumab sensitivity in HER-2-positive cell lines [Scaltriti et al. 2011], but we are not aware of any clinical confirmation of this putative mechanism.
A recently described further mechanism of resistance, is hyperactivation of the nonreceptor tyrosine kinase, SRC, which may modulate trastuzumab resistance induced by the other mechanisms described above, potentially providing a more broadly applicable therapeutic target. The investigators demonstrated that inhibition of SRC by the small molecule saracatinib in combination with trastuzumab, inhibited AKT activation, a surrogate marker of HER-2 signalling, in both PTEN-deficient and EGFR-overexpressing HER-2-positive cells [Zhang et al. 2011].
Correlative translational studies are vital to identify whether these, or other, novel mechanisms of trastuzumab resistance are also relevant to gastric and OGJ cancers, to allow rationally designed clinical trials for patients with primary or acquired trastuzumab-resistant disease.
Future directions
Two phase II and one phase III study of trastuzumab have already been initiated in early oesophagogastric cancer, to determine whether the benefit observed in the advanced disease setting will translate into a meaningful increase in survival in patients with operable disease. At present, possibly due to concerns over cardiotoxicity, none will evaluate the antibody with epirubicin-based chemotherapy in advanced or early disease. Similarly, no study of trastuzumab beyond first disease progression has yet been initiated to the best of the authors’ knowledge. This common practice in advanced breast cancer is supported by retrospective studies and small prospective studies [von Minckwitz et al. 2009], but should now be robustly tested in advanced oesophagogastric cancer, evaluating continuation of trastuzumab with second-line irinotecan or docetaxel chemotherapy. The patent life of intravenous trastuzumab will inevitably limit investment in large studies in gastroesophageal cancer, and whether the new subcutaneous formulation of the drug can be substituted currently remains unproven.
Attention has naturally turned to the trastuzumab–pertuzumab dual antibody blockade, which will soon be tested in a phase II study in advanced gastric and OGJ cancers, which it is hoped will lead to phase III evaluation. However, the cost–efficacy analysis of this combination will be critical to determine its applicability and will inevitably limit its utility in countries where health resources are finite. The combination of T-DM1 and pertuzumab may be even more effective and potentially provide a systemic chemotherapy-free strategy, which would be popular with both patients and physicians if efficacious. Certainly, accrual to two phase III studies evaluating this combination in advanced breast cancer has been extremely rapid due to widespread expectation of improved outcomes. It is our hope that the evaluation of T-DM1 in advanced gastroesophageal cancer will follow swiftly, avoiding the almost decade-long delay experienced with trastuzumab.
Correlative translational studies are now integral to prospective clinical trials of targeted agents, therefore the identification of mechanisms of resistance to HER-2-targeted agents should be expedient, and novel agents to circumvent these mechanisms swiftly tested in molecularly selected patients.
Whilst only 15–25% of patients with gastroesophageal cancer are HER-2 positive, the integration of trastuzumab into the routine treatment of this molecularly selected group represents an important advance towards personalized medicine for these patients, which can perhaps be replicated once further subgroups of patients with druggable molecular aberrations can be identified.
Footnotes
Acknowledgements
The authors acknowledge NHS funding to the NIHR Biomedical Research Centre.
Funding
The authors received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors for this work.
Conflict of interest statement
Dr Okines previously received an honorarium from Roche and travel support to attend international scientific meetings from Roche, Bayer and Amgen. Professor Cunningham has received research funding from Roche, Amgen and Merck and have attended advisory boards for Roche and Amgen and acted as an expert witness for Amgen (all uncompensated).
