Abstract
Despite recent advances in treatments and knowledge of biomarkers, patients with metastatic lung cancer have a 5-year survival rate of 5%. Rearranged during transfection (RET) fusions occur in 1% to 2% of lung cancer patients. Pralsetinib has been used to treat non-small cell lung cancer with a single RET fusion; however, there have been no reports regarding its use in patients with multiple RET fusions. Genetic mutations in tumor tissues were tested using Amplification Refractory Mutation System-PCR and next-generation sequencing (NGS). Pleural fluids obtained from a male patient with non-small cell lung cancer were also used to detect genetic aberrations by NGS. Pleural fluid-based NGS revealed three RET rearrangements: CCDC6-RET (C2:R12), RET-NRG3 (R11:N3), and CCDC6-RET (C1:R12). All three rearrangements were targeted by pralsetinib, a RET fusion inhibitor. Pralsetinib drastically improved the patient’s condition within 4 days, and a partial response was achieved 1 week after pralsetinib was administered. We report for the first time the important clinical observation of a patient with multiple RET fusions who was effectively treated with pralsetinib.
Keywords
Introduction
Lung cancer patients have greatly benefited from newly identified biomarkers and advances in clinical therapeutic methods. Nonetheless, the 5-year survival rate for patients with metastatic disease remains less than 5%. 1 Rearranged during transfection (RET) is a proto-oncogene that belongs to the receptor tyrosine kinase (RTK) family. RET rearrangements have been shown to promote cell survival, migration, and growth.2,3 RET fusions were first discovered in non-small cell lung cancer (NSCLC) in 2012 and show an incidence rate of approximately 1% to 2%. 4 RET fusions are primarily observed in non-smokers or light-smokers with lung adenocarcinoma (LUAD). 5 RET has been found to fuse with a variety of heterologous upstream partner genes in NSCLC, with KIF5B-RET accounting for approximately 54% of the cases, followed by CCDC6-RET (17%) and NCOA4-RET (3%), 4 while the rest of the cases are rare RET fusions. 6
Although patients with RET rearrangements are primarily treated with chemotherapy and non-specific site-targeted therapies, the Food and Drug Administration has approved the use of selpercatinib and pralsetinib (BLU-667), which selectively target RET fusions, in patients. However, there have been no reports regarding their use in patients with multiple RET fusions. This case report describes the treatment of a patient with three RET rearrangements.
Case presentation
A male patient in his early 40s was first diagnosed with lung cancer via a chest positron emission tomography/computed tomography scan, and multiple metastasis were found in both lungs (Figure 1). He underwent lymph node puncture at Shanghai Pulmonary Hospital in April 2019, and the disease was pathologically confirmed as LUAD. Amplification Refractory Mutation System-PCR (ARMS-PCR) analysis revealed no genetic aberrations. Pemetrexed combined with platinum complexes was administered for six cycles in April 2019 as first-line treatment. Pemetrexed was administered as a single agent thereafter. Re-examination through CT analysis conducted in December 2019 showed disease progression. Second-line treatment with docetaxel plus Sintilimab (a PD-1 inhibitor, Innovent Biologics Co., Ltd., Suzhou, China) was adopted to treat the patient for seven cycles, which ended in May 2020. In June 2020, a blood liquid biopsy sample was analyzed using a next generation sequencing (NGS)-based gene panel, which identified an IGR (downstream CCSER2)-RET fusion; however, this rearrangement was not the classical breakpoint that could justify the use of a RET fusion-targeted therapy. In September 2020, another NGS-based gene panel was used to screen for genetic aberrations. No classical RET fusion breakpoints were detected, although an intergenic-RET (intergenic: R12) and a RET-NRG3 (R11:N3) fusion were identified. Owing to the failure to detect classic breakpoints, RET fusion-targeted therapy was not used to treat the patient. Nab-paclitaxel and cisplatin plus bevacizumab was adopted from November 2020 as third-line treatment.
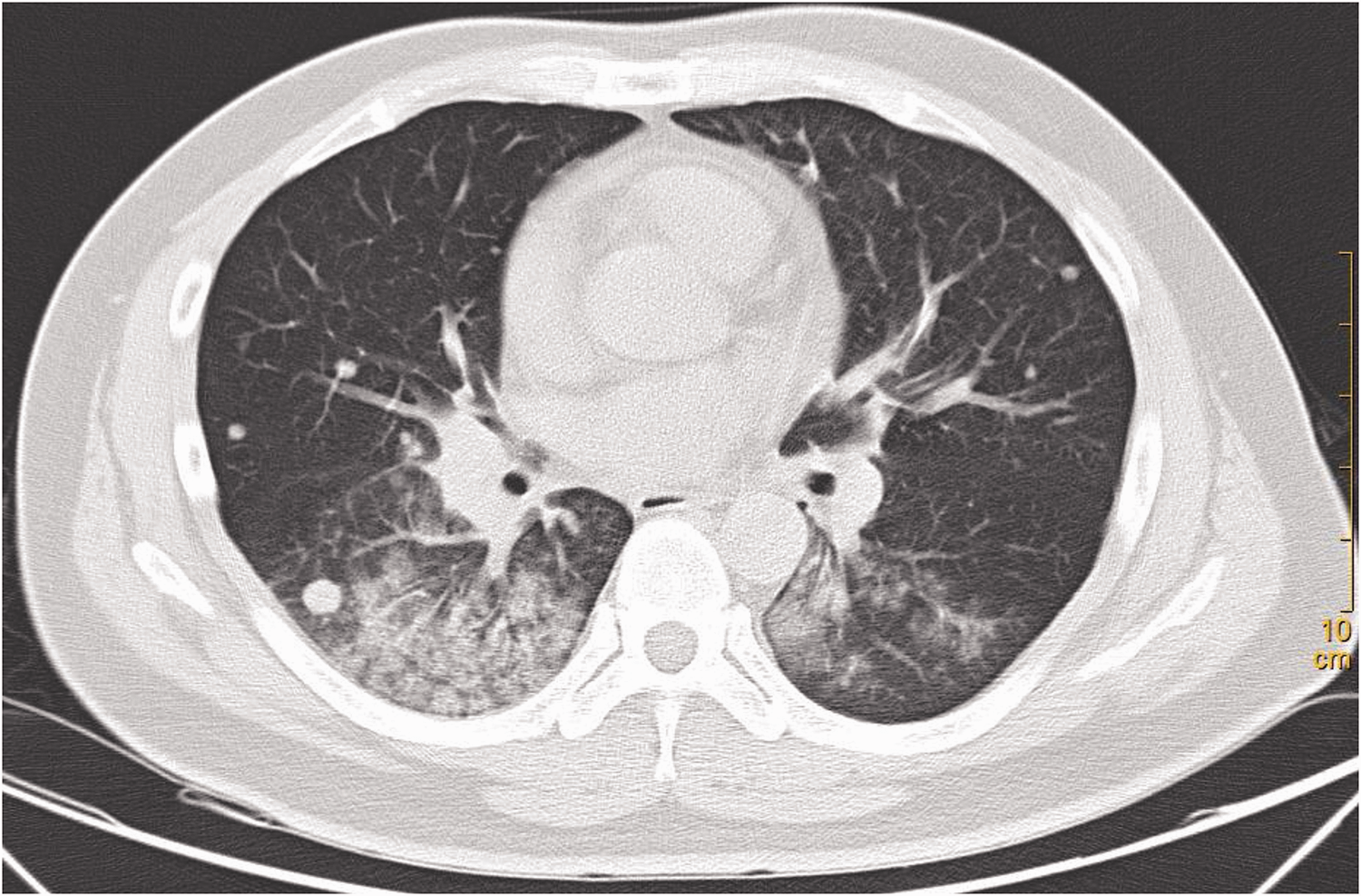
Chest computed tomography image revealing multiple metastases in the left and right lungs when the patient was initially diagnosed.
The patient was treated with radiotherapy in March 2021. Camrelizumab (a PD-1 inhibitor) was administered in April 2021 after disease progression was detected again. In June 2021, CT examination revealed that the patient was exhibiting progressive disease. The patient’s functionality was evaluated according to the Eastern Cooperative Oncology Group system 7 as performance status (PS) level 3. Pleural fluid was analyzed using an eleven-gene NGS panel for molecular diagnostics, and three RET rearrangements were identified: CCDC6-RET (C1:R12) (2.55%), CCDC6-RET (C2:R12) (3.89%), and RET-NRG3 (R11:N3) (0.83%). Of these, the CCDC6-RET (C1:R12) rearrangement is a classic breakpoint targeted by pralsetinib (BLU-667). The patient was administered pralsetinib (400 mg once daily, orally) from June 2021. After 4 days, the physical condition of the patient had substantially improved, and the PS score after 1 week was 1. In July 2021, a follow-up CT showed that a partial response (PR) had been achieved (Figure 2). The patient’s disease has remained stable to date. The timeline of treatments and diagnostics is outlined in Figure 3. Ethics approval was obtained from the Ethics Committee of Jiangyin People’s Hospital. Written informed consent was obtained from the patient to publish the case report for scientific study, and CT images were used for publication only. Patient consent for treatment was obtained. All patient details were de-identified, and the reporting of this study conforms to CARE guidelines. 8

Comparison of computed tomography images of the patient before and after pralsetinib treatment. The orange circle shows that tumor size shrank drastically.
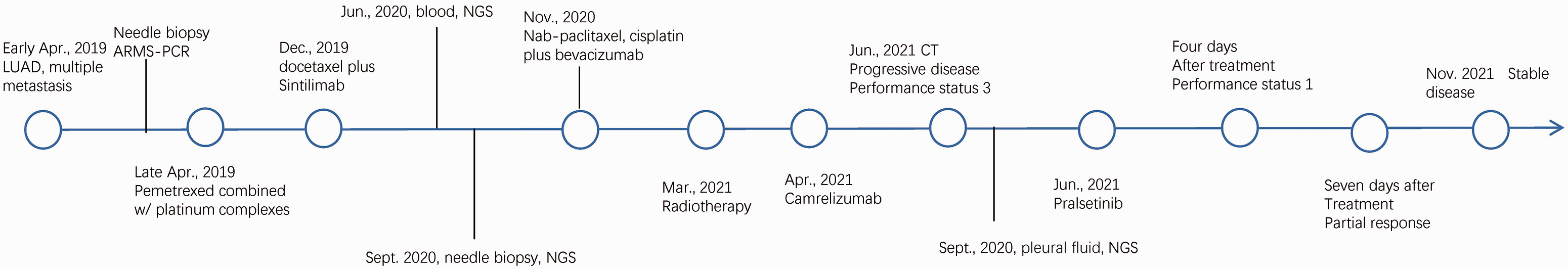
Timeline of clinical treatments. LUAD: lung adenocarcinoma; ARMS-PCR: Amplification Refractory Mutation System-PCR; PD: progressive disease; PS: performance status; PR: partial response; SD: stable disease.
Discussion
Several targeted drugs for rare mutations such as RET fusions were approved in 2020, providing a wider range of therapeutic options for clinicians. We report for the first time that pralsetinib can be used to treat LUAD patients with multiple RET fusions.
RET fusions have been widely reported in NSCLC patients.4,9 The most frequently observed RET fusions involve partner genes such as KIF5B, CCDC6, and NCOA4, which are intrachromosomal rearrangements. The specific fusion partner and location of the RET breakpoints dictate the properties of the newly formed oncoproteins. Different fusion partner genes are believed to confer distinctive properties, such as a highly expressed, activated RET oncoprotein 10 or the formation of multikinase signaling hubs, 11 with potential to influence drug sensitivity of the newly formed fusion protein.
While chemotherapy remains a treatment choice for driver-mutation-positive metastatic LUAD, targeted therapies such as tyrosine kinase inhibitors are increasingly being used. NSCLC patients with oncogenic driver mutations generally show a quick initial response to targeted therapies, yet tumor recurrence is common due to acquired resistance. 12 Treatment for cancers harboring a RET fusion has evolved from traditional chemotherapy to non-selective RTK inhibitors, selective RTK inhibitors, and immunotherapy. Although immune checkpoint inhibitors have become a standard-of-care for treating metastatic LUAD, most NSCLC patients with RET fusions have low PD-L1 expression and tumor mutation burden, and therefore show inferior responses to immune checkpoint inhibitors.13–15 Differential efficacy has also been observed between different RET fusion partner genes, 15 making the precise selection of RTK inhibitors clinically important. Pralsetinib (BLU-667) was approved by the FDA in September 2020 after clinical trials showed that it demonstrates significant clinical activity against RET fusion genes. 16
Herein, we report for the first time the treatment of a LUAD patient harboring multiple RET fusions with pralsetinib. The achievement of PR in our case indicates that pralsetinib is effective against multiple RET fusions. Unfortunately, due to the limited tumor tissue available, we were unable to verify the expression of the RET fusion genes by fluorescence in situ hybridization or immunohistochemistry.
Our case has an obvious limitation, i.e., its limited generalizability. However, our report does support the use of pralsetinib for treating patients with multiple RET-rearranged NSCLC and provides clinically meaningful information.
Supplemental Material
sj-jpg-1-imr-10.1177_03000605221105368 - Supplemental material for Pralsetinib treatment for multiple RET fusions in lung adenocarcinoma: a case report
Supplemental material, sj-jpg-1-imr-10.1177_03000605221105368 for Pralsetinib treatment for multiple RET fusions in lung adenocarcinoma: a case report by Xiangming Cao, Xiongwei Liu, Simin Wang, Zhen Liu, Xin Ren, Debin Sun and Lichun Deng in Journal of International Medical Research
Footnotes
Author contributions
LD and XC conceived and supervised the manuscript. XC, XL, SW, ZL, and XR treated the patient and collected the clinical data. DS analyzed the data. XC drafted the manuscript. All authors reviewed the manuscript and agreed to submit for publication.
Data availability statement
Data are available upon request.
Declaration of conflicting interests
Debin Sun works for Genecast Biotechnology Co., Ltd. All authors declare that they have no conflicting interests.
Funding
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
