Abstract
Background:
Different accessible and low-cost biomarkers have been investigated to stratify patients with acute heart failure (AHF). One of them is the red cell distribution width (RDW), which proved to be a greater prognostic marker of mortality than other conventional markers.
Objective:
Therefore, the objective of our study is to determine whether the anemic status modifies the magnitude of association between high RDW and 1-year mortality in patients with AHF.
Design:
Observational, analytical, retrospective cohort study.
Methods:
We included participants ⩾18 years old hospitalized with a diagnosis of AHF. As an association measure, a crude and adjusted generalized linear model of the Poisson family calculated the risk ratio (RR) with a 95% confidence interval (95% CI).
Results:
In all, 709 participants with an average age of 73.48 years were included. At 1 year, the risk of dying was three times higher with high RDW (RR = 3.05, 95% CI: 1.39–6.66; p < 0.01). In anemic participants, the risk of dying at 1 year is two times greater with high RDW (RR = 2.18, 95% CI: 0.91–5.22; p = 0.07), while in non-anemic participants, the risk of dying increased almost seven times (RR = 6.95, 95% CI: 1.65–29.23; p < 0.01).
Conclusion:
High RDW is a risk factor for mortality at 1 year in patients with AHF. A greater magnitude of association was found in non-anemic patients.
Plain language summary
This study explores whether having anemia (a condition where the body doesn’t have enough red blood cells) changes how the size of red blood cells, known as RDW, can help predict the risk of death within a year in people with heart failure. We found that patients with larger red blood cells were more likely to pass away within a year, and this risk was even higher for those who didn’t have anemia. These insights could help doctors better understand and care for people living with heart failure.
Background
Acute heart failure (AHF) presents with sudden symptoms (dyspnea, edema, fatigue) and signs (pulmonary crackles, jugular venous distension) due to volume overload, often requiring emergency care. 1 Globally, AHF affects an estimated 26 million people. 2 In Latin America and the Caribbean, data are limited, largely due to inconsistent definitions and diagnostic methods. 3 In Peru, there is no current record of the prevalence and incidence of this condition. Only one study in 2017 reported that advanced age and comorbidities were risk factors for death in patients with AHF. 4
While AHF mortality has declined—attributed to novel therapies and devices—its prevalence and healthcare burden have grown. 5 AHF increases hospitalizations and healthcare costs, impacting families and communities. 2 In Spain, in-hospital mortality ranges from 7% to 11% within 60–90 days post-discharge. 6 In Peru, a study at a tertiary hospital reported 7.2% in-hospital mortality in AHF patients, though broader national data are lacking. 4
The American College of Cardiology/American Heart Association and the European Society of Cardiology (ESC) recognize baseline brain natriuretic peptide (BNP) and cardiac troponin levels as prognostic markers at hospital admission and discharge in patients with AHF. 7 Emerging serum biomarkers, such as adiponectin, sST2, galectin-3, osteoprotegerin, and Heart-type Fatty Acid–Binding Protein (H-FABP), are under investigation for their roles in AHF pathophysiology and their potential utility in diagnosis, prognosis, and risk stratification. 8
In new biomarkers research, red blood cell distribution width (RDW), a measure of erythrocyte volume variability, has been associated with morbidity and mortality in various conditions, including cardiovascular and infectious diseases.9–12 Its levels may be influenced by factors such as erythropoietin (EPO) activity, chronic inflammation, and iron deficiency.13,14
Anemia and RDW are closely linked to cardiovascular diseases. Reduced erythrocyte phagocytosis allows greater variability in red blood cell size (anisocytosis), increasing RDW levels. Chronic inflammation can suppress EPO production, further worsening anisocytosis and elevating RDW. 10 This may contribute to heart failure progression by impairing erythrocyte deformability and oxygen delivery to the myocardium and peripheral tissues. 13 RDW has also been identified as an independent predictor of medium-term mortality, even after adjusting for hemoglobin levels, comorbidities, and other biomarkers. 15
Despite growing evidence, few studies in Latin America have explored the role of anemia in modulating RDW levels among patients with AHF. This study aims to evaluate whether anemia modifies the association between RDW and 1-year mortality in patients with AHF.
Materials and methods
Study design and population
An observational, analytical, retrospective cohort study was conducted covering December 2013 to March 2017. In addition, we performed a secondary analysis using the database of the study Pariona et al. “Clinical-epidemiological characteristics of AHF in a tertiary level hospital in Lima, Peru” 4 attended in the cardiology service of the Edgardo Rebagliati Martins Hospital. We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cohort studies, and the completed checklist is provided in the Supplementary Material.
The study population was defined according to Pariona et al.’s inclusion criteria: 4 adults (≥18 years) hospitalized for acute heart failure (AHF) in the adult emergency department of the Edgardo Rebagliati Martins Hospital. Hospitalized patients had the diagnosis of AHF, as defined by the ESC, regardless of the etiology or cause of decompensation. Diagnosis began with an initial clinical evaluation and auxiliary examinations such as chest X-ray, electrocardiogram, echocardiogram, and laboratory tests (natriuretic peptides). In addition, only patients with RDW dosage and complete confounding variables were included. Patients with a history of congenital heart disease, chronic inflammatory disease, cancer, and active gastrointestinal bleeding were excluded from data collection.
Sample power and sampling
We carried out a statistical power calculation with participants who had the complete data of our variables of interest. For this, we used the study by Melchio et al., 15 where it was reported that the prevalence of high RDW was 48.7%, while mortality in those with high RDW was 56.7% and 32% in those with low RDW. With these parameters, a confidence level of 95% and the 709 available participants, the statistical power of 99% was calculated. The study by Pariona et al. used non-probabilistic sampling for convenience.
Study variables
The main outcome variable in our research is all-cause mortality, assessed 1 year after discharge due to AHF. The outcome was categorized as “alive” or “dead” at the end of the follow-up period. The main independent variable was RDW, categorized using a cut-off point of 15% to define high RDW levels, and <15% as normal. This threshold was selected based on the study by Nakashima et al., which reported its prognostic value for in-hospital mortality in patients with AHF. 16 Moreover, other studies have used similar RDW cut-offs, such as 14.5%,17,18 14.8%, 15 and 16.7%, 19 showing associations with mortality and readmissions. Given this consistency across populations and study designs, we selected 15% to facilitate comparability with prior evidence. Anemia was stratified according to the WHO definition: hemoglobin <12 g/dL in females and <13 g/dL in males. Confounding variables included sex, age (years), comorbidities (hypertension, coronary artery disease, diabetes mellitus, dyslipidemia, arrhythmia, renal failure), and pro natriuretic peptide type B (proBNP; pg/mL).
Data collection procedure
The database from the study of Pariona et al. was used. This study collected data from medical records between December 2013 and March 2017. In addition, the data were recorded in an electronic file specially designed for this work. Laboratory and RDW values were recorded upon admission to hospitalization. Complete blood counts, including RDW measurements, were performed using the Sysmex XN-1000™ automated hematology analyzer (Sysmex Corporation, Kobe, Japan). This high-performance analyzer utilizes fluorescence flow cytometry for leukocyte, erythrocyte, and reticulocyte analysis; hydrodynamic-focused impedance for red blood cell and platelet counting; and a non-cyanide SLS (sodium lauryl sulfate) method for hemoglobin measurement. All data were entered in the Excel program version 2013, where quality control of the atypical data was made, and those participants who did not meet the selection criteria and did not have complete data before the statistical analysis were excluded from the study.
Statistical analysis
Statistical analyses were performed with the STATA version 14.0 program (StataCorp LP, College Station, TX, USA). If numerical variables had a symmetric distribution, they were presented as a mean with standard deviation, if they met a biased distribution as a median with an interquartile range. The categorical variables were presented as frequencies and percentages. Bivariate associations were evaluated with the t-Student parametric test for symmetric distribution variables and the chi-square for categorical variables. We applied a crude and adjusted generalized linear model of the Poisson family with robust standard errors to measure the strength of association between mortality and RDW. The risk ratio (RR) was established as the measure of association with their respective 95% confidence interval (95% CI). The variables were entered into the adjusted model following an epidemiological criterion, and the collinearity relationships were evaluated with the inflation factor of variance. In addition, patients were stratified into two “anemic” and “non-anemic” groups to assess whether there was a change in the effect of the association between RDW and mortality in these groups. We considered effect modification from a 30% increase or decrease in the adjusted RRs of the anemic and non-anemic participants compared to the entire sample.
Ethical aspects
The present study was presented to the ethics committee of the Universidad Peruana de Ciencias Aplicadas FCS-CEI/365-06-2. Because a secondary database analysis was used, no informed consent was required. The database was analyzed without identifiers to preserve the participants’ anonymity. The patients’ information was stored in encrypted form on two computers, only accessible by the researchers.
Results
The database recorded 1075 patients, of which 366 patients were excluded because they did not have the required variables (Figure 1). The mean age of the participants was 73.5 years (12.0), and 55.3% of the participants were male. The most frequent comorbidities were arterial hypertension (59.0%), anemia (57.3%), and arrhythmia (49.4%). Laboratory data showed a mean hemoglobin of 11.8 g/dL (2.1), RDW of 15.2% (1.8), and a median of 4.0 × 103 pg/mL of proBNP (Table 1).
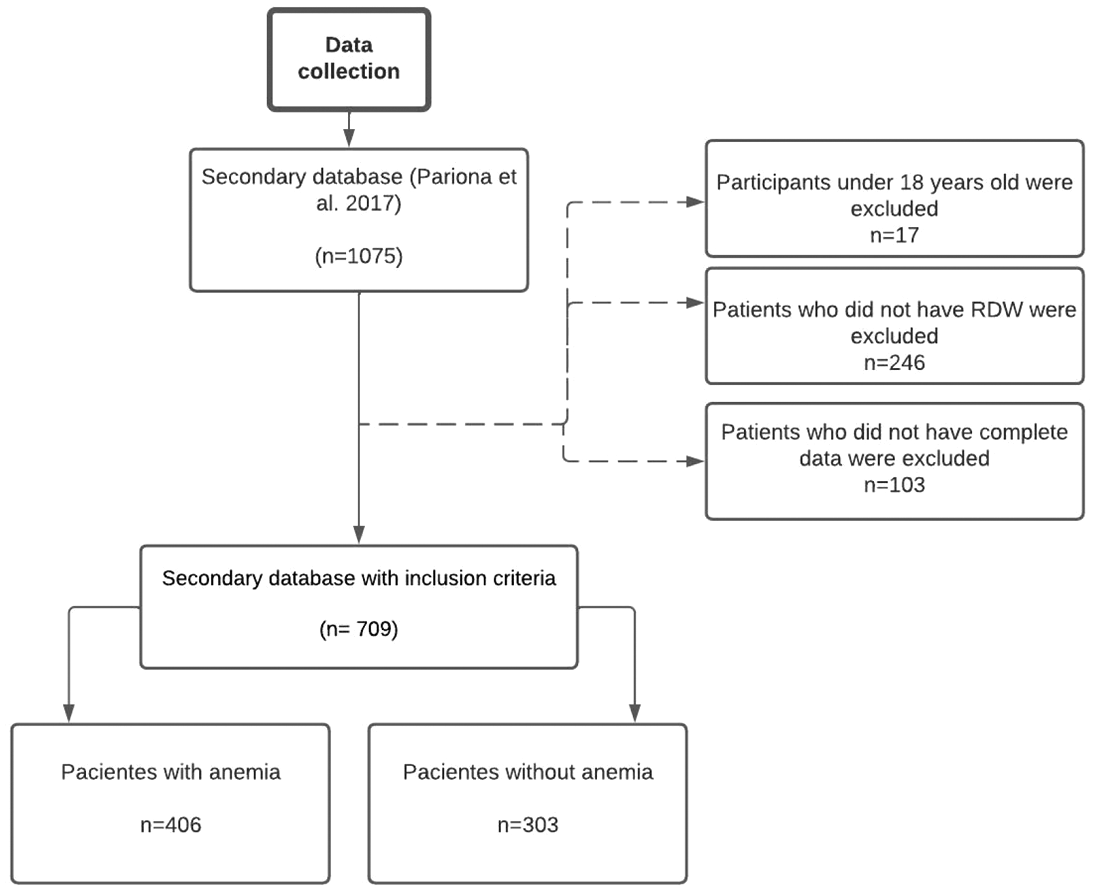
Flowchart for selecting study participants.
Baseline patient characteristics according to RDW status.
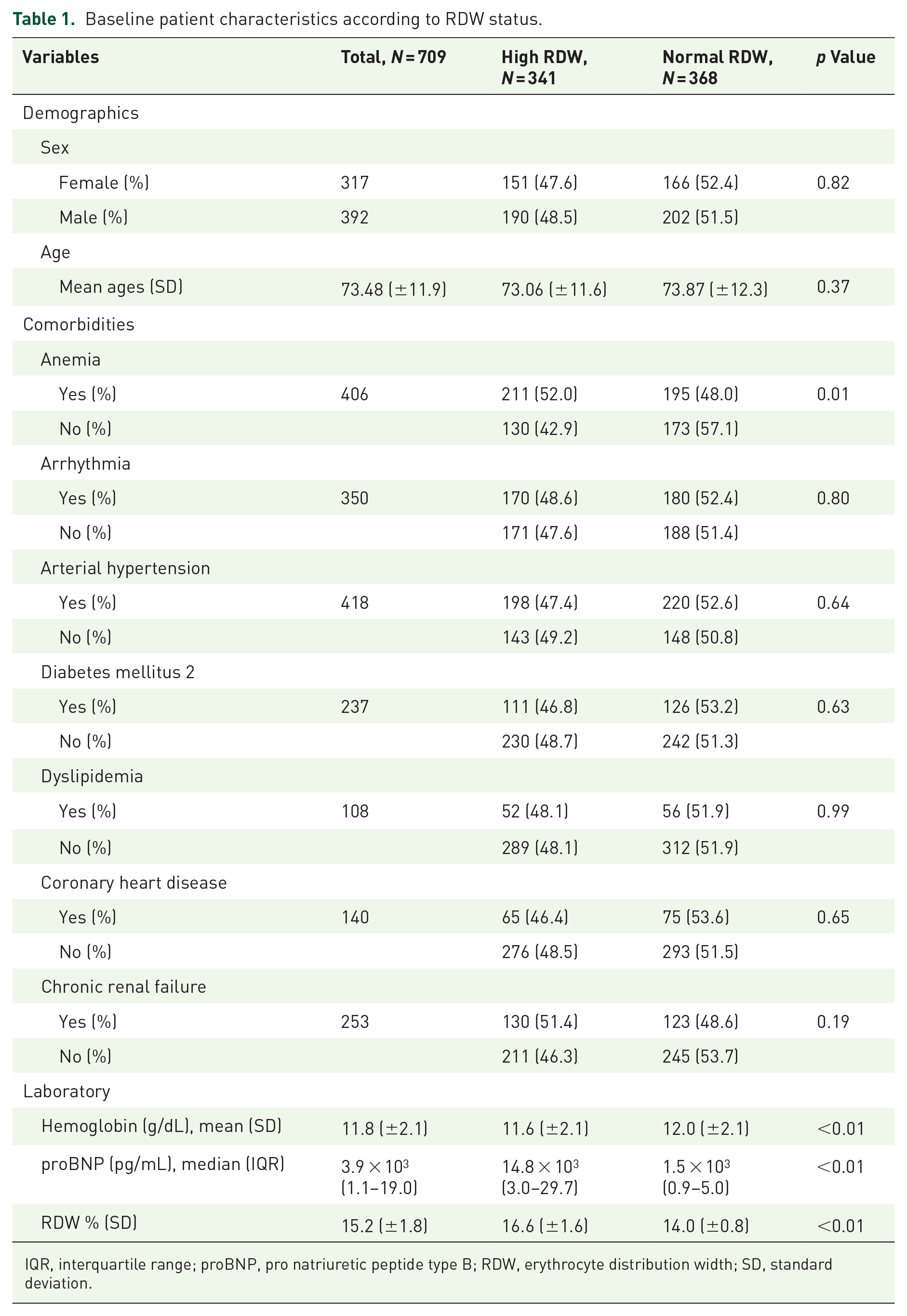
IQR, interquartile range; proBNP, pro natriuretic peptide type B; RDW, erythrocyte distribution width; SD, standard deviation.
The prevalence of high RDW in the participants was 48.1%. The high RDW was 52.0% in the anemic sample, while in the non-anemic sample, it was 42.9% (p = 0.01). Hemoglobin levels were lower in those with high RDW, 11.6 g/dL (2.2), compared to 12.0 g/dL (2.1; p < 0.01). Median proBNP values were highest in the group of participants with high RDW (p < 0.01). There was no association of high RDW with the rest of the study variables (p > 0.05; Table 1).
The mortality during follow-up was 8.9% (n = 63). There was a statistically significant association between anemia and mortality, 10.8% versus 6.3% in non-anemic (p = 0.03). In addition, we found that 16% of participants with high RDW died compared to only 2.3% of those with normal RDW (p < 0.01). Similarly, there were lower hemoglobin levels in the group of dead participants, 11.0 g/dL (2.4) versus 11.9 g/dL (2.0). On the other hand, the median serum proBNP was higher in the dead than in the survivors (p < 0.01; Table 2).
Bivariate analysis according to 1-year mortality.

IQR, interquartile range; proBNP, pro natriuretic peptide type B; RDW, erythrocyte distribution width; SD, standard deviation.
In the crude Poisson regression analysis, a sevenfold risk of dying 1 year after discharge from hospitalization for AHF was found in those participants with high RDW (RR = 7.41, 95% CI: 3.58–15.35). In the adjusted analysis, the triple risk of dying was evidenced in those participants with high RDW (RR = 3.05, 95% CI: 1.39–6.66; Table 3).
Poisson regression models for the total sample and stratified by anemia status.

Adjusted for age, sex, anemia, arrhythmia, arterial hypertension, diabetes mellitus 2, dyslipidemia, coronary heart disease, chronic renal failure, and pro natriuretic peptide type B.
Adjusted for age, sex, arrhythmia, arterial hypertension, diabetes mellitus 2, dyslipidemia, coronary heart disease, chronic renal failure, and pro natriuretic peptide type B.
CI, confidence interval; RDW, erythrocyte distribution width; RR, risk ratio.
In the crude Poisson regression analysis only with the anemic participants of the study, we found more than five times the risk of dying with high RDW (RR = 5.85, 95% CI: 2.52–13.55) at 1 year after discharge from hospitalization for AHF. In the adjusted analysis, there was a double risk of dying with high RDW (RR = 2.18, 95% CI: 0.91–5.22) but without statistically significant differences (Table 3).
In the crude Poisson regression analysis only with non-anemic study participants, more than 11 times the risk of dying was found in participants with high RDW (RR = 11.31, 95% CI: 2.65–48.2). In the adjusted analysis, the variable high RDW maintained statistical significance with mortality 1 year after discharge due to hospitalization for AHF (RR = 6.95, 95% CI: 1.65–29.23; Table 3).
Discussion
Our study found that high RDW is a risk factor for 1-year all-cause mortality after being discharged from the hospital for AHF. Furthermore, by stratifying the sample, the non-anemic population presented a greater strength of association for the risk for 1-year mortality after being discharged from hospitalization for AHF than the anemic population.
Our study reports that high RDW levels were associated with increased mortality. This result has already been demonstrated in different studies related to worse prognosis, mortality, and hospital recurrence in patients with AHF and other cardiovascular diseases.15–22 Similar results were found in different studies that concluded that anemia is not an independent factor of adverse outcomes.15,20,21 Furthermore, correction of hemoglobin levels and treatment of anemia have not demonstrated beneficial clinical results. 23 Another study found that high RDW is a strong predictor of the transition from non-anemia to anemia. 24 Both results could be explained by pathophysiological mechanisms of cardiovascular diseases, which involve the release of inflammatory cytokines, microvascular disorders, oxidative stress, and nutritional deficiencies, among others, that alter the shape of erythrocytes by inhibiting the synthesis of EPO and hemoglobin. All these phenomena would cause an increase in RDW.25–27
The association between high RDW and mortality may be more strongly influenced by inflammation than by anemia status itself. It is possible that patients with established anemia are better adapted to chronic inflammation, whereas those transitioning to anemia may be more vulnerable to its deleterious effects, contributing to all-cause mortality 1 year after hospitalization for AHF. Both anemia and inflammation are linked to bone marrow dysfunction and increased production of reactive oxygen species, promoting anisocytosis and adverse cardiac remodeling. 28
One proposed mechanism involves the overexpression of hepcidin, a key regulator of iron metabolism. Inflammatory stimuli such as interleukin-6 induce hepatic hepcidin production, which reduces intestinal iron absorption and iron release from reticuloendothelial stores by downregulating ferroportin expression—a process known as “reticuloendothelial blockade.”15,29,30 This mechanism appears to be more prominent in the acute stages of heart failure, as described by Jankowska et al. 31
Furthermore, recent evidence shows that iron deficiency in non-anemic patients is associated with a higher mortality risk than in anemic patients without iron deficiency, suggesting that iron status itself may independently impact prognosis.31–33 Both absolute and functional iron deficiency impair erythropoiesis and oxidative metabolism, reduce cellular energy, and compromise immune function. Over time, these alterations can affect myocardial structure and function by lowering oxygen reserves in myoglobin and decreasing tissue oxidative capacity, thereby impairing left ventricular performance.33,34 Unfortunately, our study did not include serum markers of iron status, such as total iron, ferritin, or transferrin, which limits further exploration of these pathways. In addition, future studies should also explore sex-specific differences in the prognostic role of RDW, particularly by stratifying analyses among anemic and non-anemic patients. This could provide deeper insights into RDW’s clinical relevance across diverse patient profiles.
Regarding the relevance of our findings, RDW is a low-cost marker that can be easily obtained from any routine automated blood count. Unlike biomarkers such as NT-proBNP, which are widely validated and strongly recommended for the diagnosis, prognosis, and management of AHF,35,36 RDW has the advantage of being universally available and more affordable, making it especially valuable in settings with limited resources. Moreover, RDW is not only limited to patients with anemia; it has also been shown to be an independent predictor of poor prognosis in patients with AHF. For example, Xanthopoulos et al. conducted a cohort study to test a score for early risk stratification in AHF, including RDW values at hospital admission, history of arterial hypertension, and acute myocardial infarction. They concluded that RDW is an excellent prognostic marker independent of other variables that can predict mortality and re-hospitalization at 1 year. 37 While NT-proBNP remains a cornerstone biomarker, future studies should assess whether the combination of RDW and NT-proBNP could improve risk stratification models, particularly where access to NT-proBNP testing is restricted.
Potential limitations of the present investigation need to be mentioned. Since the study is retrospective, there could be errors in the medical records. However, quality controls were carried out to ensure no implausible data. Furthermore, since the study was secondary data analysis, only one hemoglobin value was available at the beginning of hospitalization, and there is no information on measurements or variations of hemoglobin throughout the time of hospitalization or during the year of follow-up of the participants. Neither was it possible to obtain the etiology of AHF in the participants, although there is a description of their main comorbidities at hospital admission. Also, Peru has a fractional health system, and this study was carried out in a tertiary-level health center; probably, the data cannot be extrapolated to other health centers that do not have the same complexity level (bias Berkson). In addition, the investigation could only calculate the cumulative incidence since neither the exact date nor the main cause of death could be obtained. Finally, important clinical variables such as New York Heart Association (NYHA) functional class, specific heart failure treatments (e.g., beta-blockers, Angiotensin-Converting Enzyme (ACE) inhibitors/Angiotensin II Receptor Blocker (ARBs), mineralocorticoid receptor antagonists, Sodium-Glucose Co-Transporter 2 (SGLT2) inhibitors), ejection fraction, blood pressure, soluble sST2 protein, body mass index, and harmful habits (e.g., smoking, alcohol consumption) were not available in the dataset. These factors may have significantly influenced prognosis and should be considered in future studies.
Conclusion
We found that 6 out of 10 participants with anemia had high RDW. One year after hospital discharge for AHF, mortality was consistent with other studies. An association was found between high RDW and 1-year all-cause mortality in patients with AHF, both anemic and non-anemic. However, we found a more significant association between high RDW and mortality in non-anemic patients in the stratified analysis by anemia status. RDW is a practical, easily accessible, and low-cost marker that, together with anemia, could stratify patients at low or high risk.
Supplemental Material
sj-doc-1-tak-10.1177_17539447251366798 – Supplemental material for Impact of anemia on the association between red cell distribution width and 1-year mortality in acute heart failure patients
Supplemental material, sj-doc-1-tak-10.1177_17539447251366798 for Impact of anemia on the association between red cell distribution width and 1-year mortality in acute heart failure patients by Sherelym Alessandra Maita-Arauco, Sthephanie María Quispe-Vasquez, Vicente Aleixandre Benites-Zapata and Pedro Antonio Segura-Saldaña in Therapeutic Advances in Cardiovascular Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
