Abstract
Background:
Evidence regarding the relationship between dietary calcium intake and severe abdominal aortic calcification (AAC) is limited. Therefore, this study aimed to investigate the association between dietary calcium intake and severe AAC in American adults based on data from the National Health and Nutrition Examination Survey (NHANES).
Methods:
The present cross-sectional study utilized data from the NHANES 2013–2014, a population-based dataset. Dietary calcium intake was assessed using two 24-h dietary recall interviews. Quantification of the AAC scores was accomplished utilizing the Kauppila score system, whereby severe AAC was defined as having an AAC score greater than 6. We used multivariable logistic regression models, a restricted cubic spline analysis, and a two-piecewise linear regression model to show the effect of calcium intake on severe AAC.
Results:
Out of the 2640 individuals examined, 10.9% had severe AAC. Following the adjustment for confounding variables, an independent association was discovered between an augmented intake of dietary calcium and the incidence of severe AAC. When comparing individuals in the second quartile (Q2) of dietary calcium intake with those in the lowest quartile (Q1), a decrease in the occurrence of severe AAC was observed (odds ratio: 0.66; 95% confidence interval: 0.44–0.99). Furthermore, the relationship between dietary calcium intake and severe AAC demonstrated an L-shaped pattern, with an inflection point observed at 907.259 mg/day. Subgroup analyses revealed no significant interaction effects.
Conclusion:
The study revealed that the relationship between dietary calcium intake and severe AAC in American adults is L-shaped, with an inflection point of 907.259 mg/day. Further research is required to confirm this association.
Keywords
Introduction
Cardiovascular calcification is a prevalent etiology of cardiovascular morbidity and mortality, exhibiting arterial and valve calcification as defining features.1,2 Vascular calcification possesses life-threatening implications, exemplified by calcified abdominal aorta, calcified uremic arterioles, and calcified valvular disease. 3 Furthermore, vascular calcification serves as a risk factor for ischemic vascular disease, and mitigation of calcification has the potential to decrease patient mortality. 4 Nonetheless, the complete pathogenesis of vascular calcification remains inadequately elucidated, and a specific preferred treatment approach is yet to be established. 5 Consequently, comprehending the factors influencing vascular calcification and implementing preventive measures becomes imperative. Numerous studies have indicated a close association between vascular calcification and factors such as hemoglobin A1c, blood uric acid, diet, and hypertension.6–9 Nevertheless, a comprehensive analysis investigating the impact of diet intake, particularly dietary calcium intake, on the development of severe abdominal aortic calcification (AAC) is presently lacking.
Calcium (Ca), an essential metal element abundant in the human body, primarily constitutes bones. 10 It is predominantly acquired through dietary intake, with sources such as dairy products, soy, grains, seafood, and fortified foods being significant contributors. 11 Dietary calcium typically exists in a bound form that is enzymatically broken down into calcium ions by gastric acid. 12 Calcium ions play crucial roles in various physiological processes, including muscle contraction, coagulation promotion, neural signal transmission, and bone formation. Previous research has primarily examined the relationship between calcium intake and cardiovascular disease (CVD). For instance, a South Korean study identified a correlation between dietary calcium intake and CVD, potentially influenced by obesity. 13 A meta-analysis involving postmenopausal women suggested a 15% increased risk of CVD with calcium supplements in healthy postmenopausal women. 14 Furthermore, a nonlinear dose–response meta-analysis demonstrated an inverse linear correlation between dietary calcium intake and hypertension risk. 15 Although previous studies have emphasized the overall risk of CVD, there is limited research specifically focusing on vascular calcification. Studies have indicated that dietary patterns enhancing calcium intake can effectively reduce blood pressure in patients with normal hypertension, improve the survival of vascular smooth muscle, and impede calcification.16,17 Conversely, some studies have suggested that elevated levels of calcium and phosphorus act synergistically as essential stimuli for vascular calcification in chronic kidney disease. 18
Therefore, the relationship between calcium levels and vascular calcification is not entirely clear, and further investigation is necessary. We hypothesized that dietary calcium intake is associated with the prevalence of severe AAC. To examine this association, we conducted an observational study using data from the 2013 to 2014 National Health and Nutrition Examination Survey (NHANES), which monitors the nutritional status of civilians in the United States.
Materials and methods
Data sources and study population
The NHANES, a nationally representative cross-sectional study, employs a stratified multistage probability and oversampling design to ensure an accurate representation of the noninstitutionalized civilian population of the United States. 19 Biennial data releases from the study consist of participants who collectively represent approximately 50,000 US citizens. The administration of standardized interviews and examinations is carried out by trained staff, including physicians, dentists, health technologists, interviewers, and laboratory technicians. 20 Comprehensive information regarding the NHANES study design, recruitment, procedures, and demographic characteristics can be found on the CDC website (https://www.cdc.gov/nchs/nhanes/index.htm). 21 In summary, the NHANES study employed a four-stage design with oversampling of specific subgroups to enhance precision, and an advanced computer system was used for data collection and processing. The survey findings play a crucial role in determining disease prevalence and associated risk factors.
In this study, we used population-based data from NHANES to investigate the association between dietary calcium intake and severe AAC. During the 2013–2014 cycle, a total of 10,175 participants were initially included in the study. Exclusions were made for individuals under 40 years old (n = 6360), those with missing data about AAC (n = 675), and finally those with missing dietary calcium intake data (n = 500). Ultimately, a total of 2640 participants met the eligibility criteria and were included in the study. The selection process and search strategy are illustrated in detail in Figure 1, which provides a flowchart of the study. NHANES received approval from the NCHS Research Ethics Review Board, and written informed consent was provided by all participants. 22 All procedures adhered to the guidelines of the Declaration of Helsinki and Strengthening the Reporting of Observational Studies in Epidemiology (STROBE).
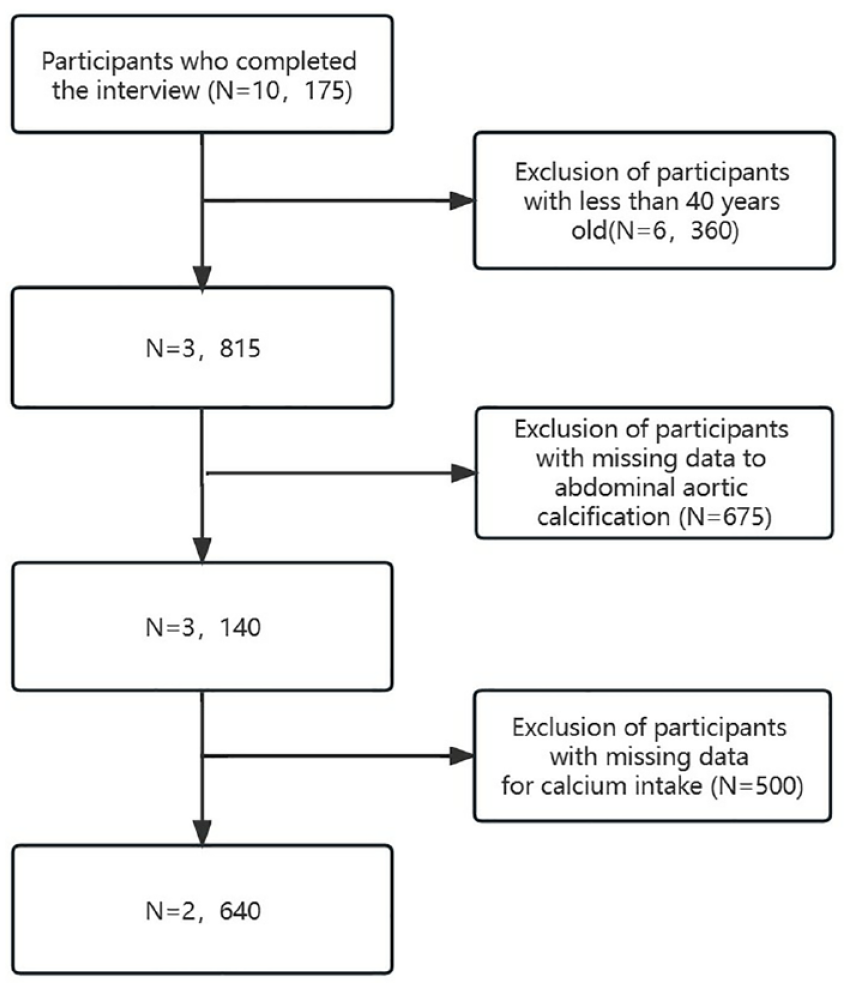
Flowchart of patient selection.
Dietary calcium intake
Dietary intake interviews were conducted at the NHANES mobile examination clinics through in-person sessions. To gather dietary intake data, participants of NHANES underwent two 24-h dietary recalls.23,24 The initial recall interview occurred face-to-face at the Mobile Examination Center, administered by trained interviewers. The second interview took place via telephone or mail within a period of 3–10 days. The dietary assessments were based on the average of these two recalls. 25 The analysis of calcium intake involved examining it as categorical variables (Q1–Q4). Calcium intake quartiles (Q1–Q4) were determined by dividing the calcium intake distribution into four parts, reflecting low to high intake levels. The range of quartiles (Q1–Q4) are as follows: Q1 (⩽570.5 mg/day), Q2 (570.5–806.2 mg/day), Q3 (806.2–1096.1 mg/day), and Q4 (⩾1096.1 mg/day).
Abdominal aorta calcification
As previously documented,26–28 the assessment of AAC was conducted with precision by employing dual-energy X-ray absorptiometry to perform a lateral scan of the lumbar spine, specifically targeting vertebrae L1–L4. To quantify the extent of AAC, the Kauppila score system was utilized.26–29 Additional comprehensive information can be accessed on the official website: https://wwwn.cdc.gov/Nchs/Nhanes/2013-2014/DXXAAC_H.htm. The AAC score ranges from 0 to 24, and a severity threshold of greater than 6 (>6) has been established based on prior investigations.8,26,30–32
Covariates
To address potential confounding variables, we incorporated various covariates in our analysis, including sex (male or female), race (Mexican Americans, other Hispanics, non-Hispanic whites, non-Hispanic blacks, non-Hispanic Asians, or other groups), education level (below high school, high school, or above high school), body mass index (BMI; <25, 25–30, or ⩾30 kg/m2), heart failure (yes or no), CHD (yes or no), angina (yes or no), stroke (yes or no), smoking (yes or no), hypertension (yes or no), alcohol (yes or no), diabetes (yes or no), kidney stone (yes or no), albumin, total cholesterol, 25OHD2 + 25OHD3, phosphorus, aspartate transaminase (AST), serum creatinine, serum uric acid, and serum calcium. Hypertension was defined as the use of antihypertensive drugs, a diagnosis of hypertension, or three consecutive systolic or diastolic blood pressure readings ⩾140 or ⩾ 90 mmHg. 33 Diabetes was defined as taking hypoglycemic agents or having been diagnosed with diabetes, hemoglobin A1c ⩾6.5%, and fasting blood glucose ⩾126 mg/dL. As a routine liver function test index, AST reflects the damage to the liver. To examine the relationship between dietary calcium intake and severe AAC, we performed stratified multivariate regression analysis. 34
Statistical analysis
The chi-square test and one-way analysis of variance were used to compare the characteristics of the study participants at baseline. Continuous variables were reported as mean and standard deviation or median and interquartile range, and categorical variables were expressed as population proportions and percentages. To assess statistical differences among dietary calcium intake for baseline characteristics analysis, we employed one-way ANOVA and chi-square tests. We utilized multiple imputations to fill in missing baseline data. Values based on the dietary calcium intake of our population were used in this study. Participants were divided into four groups according to dietary calcium intake in mg/day [Q1 (⩽570.5 mg/day), Q2 (570.5–806.2 mg/day), Q3 (806.2–1096.1 mg/day), and Q4 (⩾1096.1 mg/day)].
We used binary logistic regression models [odds ratio (OR) and 95% confidence interval (CI)] to evaluate the impact of dietary calcium intake on severe AAC, adjusting for major covariates. These included sex (male or female), race (Mexican Americans, other Hispanics, non-Hispanic whites, non-Hispanic blacks, non-Hispanic Asians, or other groups), an education level (below high school, high school, or above high school), BMI (<25, 25–30, or ⩾30 kg/m2), heart failure (yes or no), coronary heart disease (CHD) (yes or no), angina (yes or no), stroke (yes or no), smoking (yes or no), hypertension (yes or no), alcohol (yes or no), diabetes (yes or no), kidney stone (yes or no), albumin, total cholesterol, 25OHD2 + 25OHD3, phosphorus, AST, serum creatinine, serum uric acid, and serum calcium. We used restricted cubic spline analysis and smooth curve fitting to handle the nonlinear relationship between dietary calcium intake and severe AAC. If the nonlinear relation existed, a piecewise regression analysis based on the logistic regression models was performed to determine the inflection point.
Finally, to conduct subgroup analyses, we employed hierarchical binary logistic regression models, transforming continuous variables into categorical variables based on clinical cutoffs or quantiles, and performing interaction tests. We conducted effect adjustment tests on subgroup measures, followed by likelihood ratio tests. We utilized R-4.1.0 (R Foundation for Statistical Computing, Vienna, Austria) and FREE Statistics (version 1.7) to perform data analysis. We deemed p-values less than 0.05 statistically significant using a two-tailed test. We adhered to the STROBE reporting guidelines.
Results
Baseline characteristics of participants
Table 1 presents the baseline characteristics of the study participants, stratified by their calcium intake levels. A total of 2640 individuals (1243 males and 1397 females) with a mean age of 58.8 ± 11.9 years were included in our analysis. Among them, 288 individuals were diagnosed with severe AAC. Participants were categorized into four quartiles based on their daily calcium intake: quartile 1 (⩽570.5 mg/day), quartile 2 (570.5–806.2 mg/day), quartile 3 (806.2–1096.1 mg/day), and quartile 4 (⩾1096.1 mg/day). In both male and female participants, individuals with high calcium intake were characterized by being younger, non-Hispanic white, with less hypertension and diabetes, more likely to consume alcohol, and exhibited elevated levels of AST and phosphorus in comparison to those in the low calcium intake group.
Characteristics of the study population, National Health and Nutrition Examination Survey (NHANES) 2013–2014 (N = 2640).

The unit of calcium is mg/day.
p Values of multiple comparisons were corrected by the false discovery rate method.
Q1–Q4: quartile according to calcium absorption.
AAC, abdominal aortic calcification; AST, aspartate transaminase; no severe AAC, AAC score ⩽6; severe AAC, AAC score >6; %, weighted proportion.
Association between dietary calcium intake and severe AAC
To further elucidate the relationship between calcium intake and the occurrence of severe AAC, multivariate logistic regression models were employed, and the calcium intake was considered both as continuous and as categorical variables (Table 2). In model 1, when calcium intake was analyzed as a continuous variable, per 100 mg/day increase, the OR (95% CI) for severe AAC was [0.96 (0.94–0.99), p = 0.014]. An unadjusted analysis revealed a decreased incidence of severe AAC in the second quartile (Q2) compared to the lowest quartile (Q1) (OR = 0.7, 95% CI: 0.5–0.99, p = 0.041). Model 2, which accounted for demographic variables (sex, age, race, education poverty–income ratio), exhibited a similar pattern with a decreased incidence of severe AAC in Q2 compared to Q1 (OR = 0.68, 95% CI: 0.47–0.98, p = 0.04). Model 3, adjusting for demographic variables, body mass index, and basic diseases (heart failure, coronary heart disease, angina, stroke, kidney stone, alcohol consumption, hypertension), continued to show a decreased incidence of severe AAC in Q2 compared to Q1 (OR = 0.66, 95% CI: 0.45–0.98, p = 0.041). Similarly, in model 4, after considering demographic variables, body mass index, basic diseases, and lifestyle factors (smoking, alcohol consumption), Q2 exhibited a decreased incidence of severe AAC compared to Q1 (OR = 0.67, 95% CI: 0.45–0.99, p = 0.047). Notably, when contrasting the lowest quartile (Q1) to the higher quartiles (Q2, Q3, and Q4), the ORs for AAC, yielding values of 0.67 (95% CI: 0.45–0.99, p = 0.047), 0.86 (95% CI: 0.57–1.29, p = 0.462), and 0.86 (95% CI: 0.57–1.3, p = 0.476), respectively, after adjusting for all potential confounders. Moreover, a sensitivity analysis was performed after excluding completely missing patients, and we found similar results to our main analysis (Supplemental Table 4). We further analyzed the correlation between dietary calcium intake, AAC score, and AAC risk, and similar trends can be observed. Due to the length limitation of the article, we present it in Supplemental Tables 1 and 2. The differences described were not statistically significant after weighting the analyses with the survey weights (Supplemental Table 3).
Multivariable logistic regression to assess the association of calcium intake with severe AAC.

Calcium intake per 100 mg is a continuous variable. Model 1: No adjustment; Model 2: Adjusted for demographic variables (sex, age, race, poverty–income ratio, education); Model 3: Adjusted for demographic variables, body mass index, basic diseases (heart failure, CHD, angina, stroke, kidney stone, diabetes, hypertension); Model 4: Adjusted for demographic variables, body mass index, basic diseases, lifestyle (smoking, alcohol); Model 5: Adjusted for demographic variables, body mass index, basic diseases, lifestyle and biochemical index (albumin, total cholesterol, 25OHD2 + 25OHD3, phosphorus, AST, serum creatinine, serum uric acid, and serum calcium).
AAC, abdominal aortic calcification; CI, confidence interval; no severe AAC, AAC score ⩽6; OR, odds ratio; Ref, reference; severe AAC, AAC score >6; %, weighted proportion.
Dose–response relationships
Dietary calcium intake was considered a continuous variable in restricted cubic spline analysis. Utilizing multivariable-adjusted restricted cubic spline analysis, a distinct ‘L-shaped’ association between dietary calcium intake and severe AAC was observed (P for non-linearity = 0.048) (see Figure 2). By integrating the graphical interpretation with clinical significance, the optimal inflection point for dietary calcium intake was determined to be 907.259 mg/day. In instances where dietary calcium intake was less than 907.259 mg/day, each 1 mg/day increase in dietary calcium intake correlated with a 0.1% reduction in the incidence of severe AAC (OR = 0.999, 95% CI: 0.998–1, p-value = 0.0444). However, this association was not evident when dietary calcium intake was equal to or exceeded 907.259 mg/day, as demonstrated in Table 3.
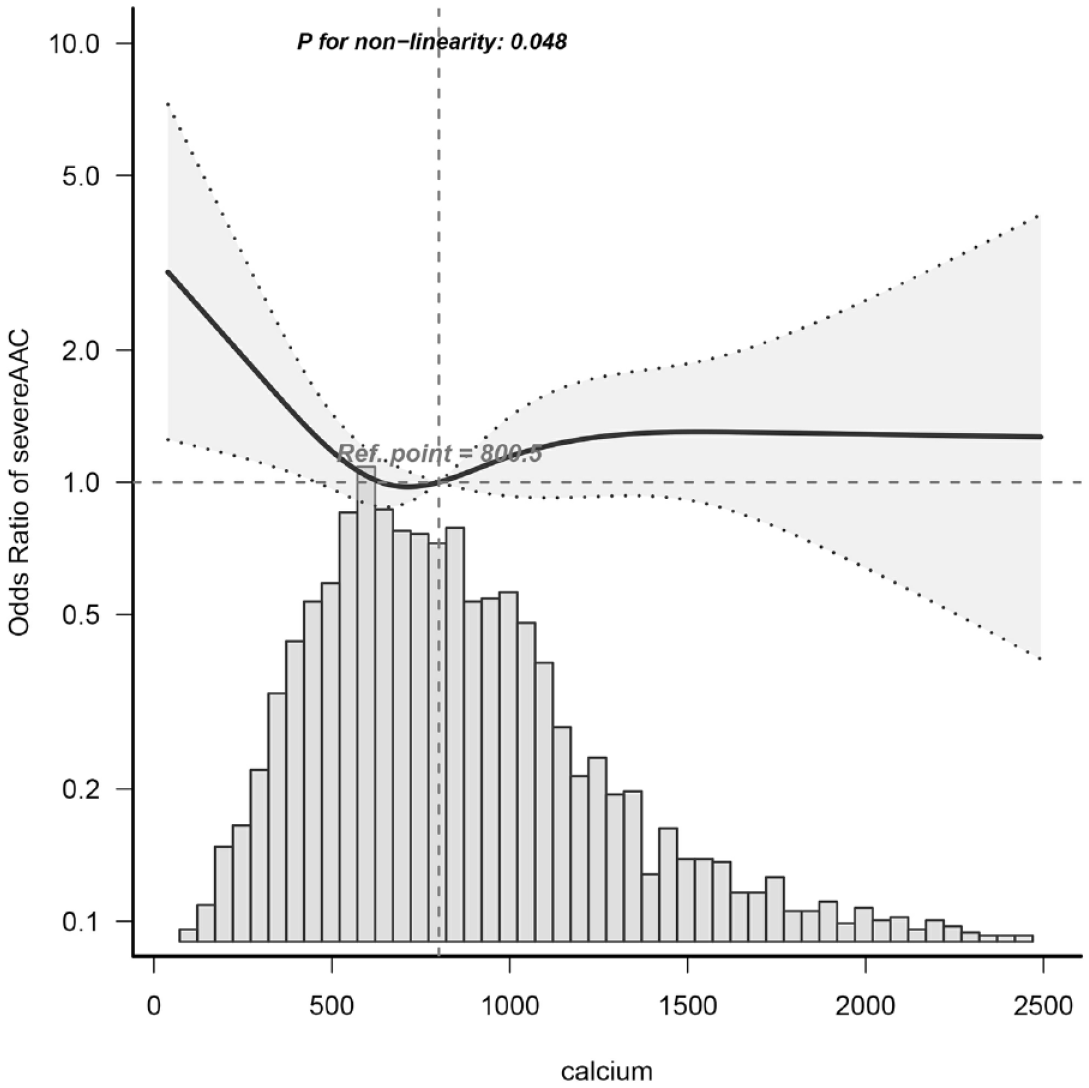
Dose–response relationship between dietary calcium intake and severe AAC odds ratio.
Threshold effect analysis of the relationship between dietary calcium intake and severe AAC.

Adjusted for sex, age, race, PIR, education level, body mass index, heart failure, coronary heart disease, angina, stroke, smoking, hypertension, alcohol, diabetes, kidney stones, albumin, total cholesterol, 25OHD2 + 25OHD3, phosphorus, AST, serum creatinine, serum uric acid, and serum calcium.
AAC, abdominal aortic calcification; CI, confidence interval; no severe AAC: AAC score ⩽6; OR, odds ratio; Ref, reference; severe AAC: AAC score >6; %, weighted proportion.
Subgroup analysis
To further investigate the influence of other risk factors on the correlation of dietary calcium intake and severe AAC, subgroup analyses were carried on according to the following stratification variables: sex, age, education, PIR, and BMI. The results of subgroup analyses and interactions were summarized in the Supplemental Figure. Subgroup analysis results were in concordance with those of the multivariable logistic regression analysis. Considering multiple testing, a p value of less than 0.05 for the interaction of family poverty-to-income ratio may not be statistically significant.
Discussion
In a large-scale cross-sectional study with 2640 participants, we investigated the impact of dietary calcium intake on severe AAC in American adults. Our findings suggest a nonlinear L-shaped trend between dietary calcium intake and severe AAC, with a turning point of 907.259 mg/day. There is an independent correlation between increased dietary calcium intake and severe AAC. Our sensitivity and stratification analyses confirmed the robustness of this relationship.
Previous research has found conflicting results regarding the relationship between dietary calcium intake and CVD mortality. While one study showed a U-shaped trend where a lower risk of CVD death was associated with lower dietary calcium intake at intakes below 800 mg/day, 35 another experimental study found no significant effect of a calcium-rich diet on coronary calcium deposition in pigs with metabolic syndrome. 36 Moreover, high calcium intake has been linked to a higher risk of CVD death in males but not in females, and increasing calcium and phosphorus levels have been associated with vascular calcification in CKD patients.18,37 A randomized controlled trial in postmenopausal women also showed an increasing trend in the incidence of cardiovascular events with calcium supplementation. 38 Therefore, the specific dosage of dietary calcium intake and the health status of the study population may influence the association between dietary calcium intake and CVD or severe AAC. This study is the first to explore the association between dietary calcium intake and severe AAC, which is of significant importance for further understanding the specific relationship between the two.
Another notable contribution of this paper is the identification of a relationship between dietary calcium intake and severe AAC, characterized by an L-shaped curve, indicating a nonlinear association and a potential threshold effect. In addition, subgroup and sensitivity analyses have demonstrated the overall stability of the results. Specifically, for populations with dietary calcium intake below 907.259 mg/day, there is a decreasing incidence of severe AAC with an increasing intake of dietary calcium. However, for populations with dietary calcium intake above 907.259 mg/day, we did not find an association between dietary calcium intake and severe AAC. Restricted cubic spline of calcium intake suggested the protective effect of calcium intake may not be linear and there appears to be a saturation threshold effect. Perhaps due to the presence of a certain proportion of calcium balance in the body, moderate intake of calcium can effectively supplement it, while excessive intake of calcium cannot be directly applied by the body, resulting in a certain saturation threshold effect. Good dietary sources of calcium include dairy products, soy products, grains, seafood, and fortified foods.39–41 Studies have shown that the optimal calcium intake for women aged 25–50 years is 1000 mg/day, while pregnant or lactating women require a daily dose of 1200–1500 mg, and postmenopausal women require a daily dose of 1500 mg. For men aged 25–65, a recommended daily intake of 1000 mg/day is suggested, and for all women and men aged 65 and above, a recommended daily intake of 1500 mg/day is suggested. 42 Our findings indicate that dietary calcium intake is associated with an incidence of severe AAC, which can inform future research on CVDs.
Further exploration may be warranted to investigate the negative correlation observed between dietary calcium intake and AAC in this study. The lack of statistical significance in the disparity among various quartiles of dietary calcium intake and severe AAC at baseline could potentially stem from an inadequate sample size. Notably, a decline in severe AAC incidence was solely evident in the second quartile, exhibiting an OR close to 1.00. Moreover, weighted regression analysis revealed a lack of statistically significant association between dietary calcium intake and severe AAC. These findings may imply limited applicability of the study results solely to the specific study population, necessitating further research for comprehensive understanding and generalizability.
The relationship between dietary calcium intake and severe AAC exhibits a biologically plausible mechanism. Calcium levels are crucial for proper adhesion of endothelial cells lining the blood vessels, and alterations in blood calcium levels resulting from dietary calcium intake can induce significant changes in cell morphology. These changes may trigger epithelial–mesenchymal transition (EndMT) and differentiation into diverse cell types, including osteoblasts, chondrocytes, and adipocytes.43–47 In addition, insulin-like growth factor 1 (IGF1) signaling contributes to the regulation, proliferation, and differentiation of calcified vascular cells. 48 Calcium and osteoprotegerin (OPG) can modulate the expression of the IGF1 receptor (IGF1R), which inhibits calcium-induced calcification.49,50 Excessive calcium concentration augments OPG expression, consequently obstructing the protective IGF1R mechanism. Furthermore, elevated calcium intake can elevate OPG expression, further impeding the protective IGF1R mechanism. 51 Finally, insufficient calcium intake can contribute to CVD through pathways affecting blood pressure, insulin secretion, and sensitivity. However, additional research is imperative to comprehensively grasp the potential mechanisms underlying the association between dietary calcium intake and severe AAC.52–54
Our study has several limitations. First, due to the cross-sectional nature of the study, we were unable to obtain a causal relationship between dietary calcium intake and severe AAC. However, our findings create a foundation for further human studies to reveal the relationship between dietary calcium intake and arterial calcification. Our future research will conduct clinical trials to test our conclusions. Moreover, the reliance on self-reported data from interviews and 24-h dietary assessments in this study introduces the potential for recall and reporting bias, despite the validation of these assessment tools in other research. 55 Finally, data for the study were derived from NHANES, which focused on populations in the United States, and the dietary guidelines were designed for Americans. All participants in the study were 40 years and older due to the limitations of AAC measurement. Since the occurrence of vascular calcification is age-related 56 and the middle-aged, and elderly population is more prone to vascular calcification, our study focused on the general adult population aged 40 years and older, which is of greater significance for the prevention of vascular calcification. However, whether the association is applicable in other countries or among young subjects requires further research.
Conclusion
In summary, our study suggests that there is an independent association between dietary calcium intake and severe AAC. The dose–response analysis revealed an L-shaped relationship between dietary calcium intake and severe AAC. Further studies are needed to determine the relationship between dietary calcium intake and severe AAC, as well as to explore the underlying mechanisms.
Supplemental Material
sj-docx-1-tak-10.1177_17539447241232774 – Supplemental material for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey
Supplemental material, sj-docx-1-tak-10.1177_17539447241232774 for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey by Kai Zhang, Fangming Gu, Yu Han, Tianyi Cai, Zhaoxuan Gu, Jianguo Chen, Bowen Chen, Min Gao, Zhengyan Hou, Xiaoqi Yu, JiaYu Zhao, Yafang Gao, Jinyu Xie, Rui Hu, Tianzhou Liu and Bo Li in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-docx-2-tak-10.1177_17539447241232774 – Supplemental material for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey
Supplemental material, sj-docx-2-tak-10.1177_17539447241232774 for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey by Kai Zhang, Fangming Gu, Yu Han, Tianyi Cai, Zhaoxuan Gu, Jianguo Chen, Bowen Chen, Min Gao, Zhengyan Hou, Xiaoqi Yu, JiaYu Zhao, Yafang Gao, Jinyu Xie, Rui Hu, Tianzhou Liu and Bo Li in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-docx-3-tak-10.1177_17539447241232774 – Supplemental material for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey
Supplemental material, sj-docx-3-tak-10.1177_17539447241232774 for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey by Kai Zhang, Fangming Gu, Yu Han, Tianyi Cai, Zhaoxuan Gu, Jianguo Chen, Bowen Chen, Min Gao, Zhengyan Hou, Xiaoqi Yu, JiaYu Zhao, Yafang Gao, Jinyu Xie, Rui Hu, Tianzhou Liu and Bo Li in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-docx-4-tak-10.1177_17539447241232774 – Supplemental material for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey
Supplemental material, sj-docx-4-tak-10.1177_17539447241232774 for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey by Kai Zhang, Fangming Gu, Yu Han, Tianyi Cai, Zhaoxuan Gu, Jianguo Chen, Bowen Chen, Min Gao, Zhengyan Hou, Xiaoqi Yu, JiaYu Zhao, Yafang Gao, Jinyu Xie, Rui Hu, Tianzhou Liu and Bo Li in Therapeutic Advances in Cardiovascular Disease
Supplemental Material
sj-docx-5-tak-10.1177_17539447241232774 – Supplemental material for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey
Supplemental material, sj-docx-5-tak-10.1177_17539447241232774 for Association between dietary calcium intake and severe abdominal aorta calcification among American adults: a cross-sectional analysis of the National Health and Nutrition Examination Survey by Kai Zhang, Fangming Gu, Yu Han, Tianyi Cai, Zhaoxuan Gu, Jianguo Chen, Bowen Chen, Min Gao, Zhengyan Hou, Xiaoqi Yu, JiaYu Zhao, Yafang Gao, Jinyu Xie, Rui Hu, Tianzhou Liu and Bo Li in Therapeutic Advances in Cardiovascular Disease
Footnotes
Acknowledgements
The authors want to express their sincere gratitude to all participants and the schools for their participating in the study. In addition, we gratefully thank Dr. Jie Liu of Department of Vascular and Endovascular Surgery, Chinese PLA General Hospital for his contribution to the statistical support, study design consultations, and comments regarding the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
