Abstract
Objective
Sleep disorder is prevalent in modern society and has been linked to multiple adverse health outcomes; however, evidence regarding its association with gallstone disease remains limited. This study aimed to investigate the association between sleep disorder and gallstones among adults in the United States.
Methods
We conducted a cross-sectional analysis of data from 7438 adults in the United States aged ≥20 years who participated in the National Health and Nutrition Examination Survey (2017–2020). The relationship between sleep disorder and gallstones was assessed using multivariable logistic regression models, with sensitivity analyses performed using multiple imputation, weighted analysis, and propensity score matching.
Results
Among all participants, 780 (10.5%) reported gallstones. Individuals with sleep disorder had significantly higher odds of gallstones (odds ratio = 1.49, 95% confidence interval = 1.26–1.76,
Conclusion
Our findings suggest that sleep disorder is independently associated with gallstones. These results highlight the importance of identifying and managing sleep problems in individuals at risk of gallstone disease; however, longitudinal studies are needed to clarify causal pathways.
Keywords
Introduction
Gallstones represent a common gastrointestinal disease with a high global incidence; it can lead to complications such as biliary colic, cholecystitis, and pancreatitis in severe cases. 1 The formation of gallstones is influenced by various factors, including genetic factors, dietary habits, body mass index (BMI), metabolic syndrome, and hormone levels. Despite established risk factors, many potential factors need further research. 2
Sleep disorder, characterized by difficulties in falling asleep and maintaining sleep as well as early awakening, is an increasingly common health issue in modern society. 3 Long-term sleep disorder not only affects the quality of life but is also associated with various health problems, such as obesity, diabetes, hypertension, and cardiovascular diseases. 4 The high prevalence of sleep disorder and its widespread health impacts make it a public health concern. 5 Recent studies have indicated that late bedtimes and short sleep duration elevate the risk of gallstone disease, potentially through circadian disruption and metabolic dysregulation. 6 In parallel, computational analyses have revealed important risk factors for insomnia, underscoring a multifactorial interplay between sleep disturbances and metabolic outcomes. 7
However, direct evidence on the relationship between sleep disorder and gallstones remains limited, especially in large, nationally representative cohorts. Most existing studies have focused on the associations between sleep and metabolic or cardiovascular diseases, with insufficient evidence on whether sleep disorder increases the risk of gallstone disease. 8 Therefore, this study aimed to explore the relationship between sleep disorders and gallstone disease among adults in the US using data from the National Health and Nutrition Examination Survey (NHANES, 2017–2020) and determine whether this association varies across different subgroups based on sex, age, and health status. This study sought to bridge this research gap and provide scientific evidence for the management of sleep disorders and prevention of gallstone disease.
Methods
This retrospective cross-sectional study used data from the NHANES (2017–2020) conducted by the Centers for Disease Control and Prevention (CDC) to assess the health and nutritional status of noninstitutionalized US residents. Participants were selected through a multistage, stratified probability sampling design. The NHANES collects health-related data across various parameters, including demographic characteristics, physical examination results, laboratory tests, and health-related questionnaire responses. For the present analysis, we included individuals aged ≥20 years who had completed the survey and had available data on sleep disorders, gallstone disease, and all required covariates; participants with missing data for any of these variables were excluded. This study was conducted in accordance with the principles of the Declaration of Helsinki (1975), as revised in 2024, and the NHANES protocols were approved by the National Center for Health Statistics (NCHS) Research Ethics Review Board (Protocol #2018-01). All participants had provided written informed consent for study participation. Our secondary analysis adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cross-sectional studies and did not require further institutional review board approval. 9 These data are publicly available on the NHANES website.
Definition of gallstone disease
Gallstone disease cases were identified using questionnaires during the NHANES (2017–2020) survey. Participants were asked by trained interviewers, “Have you ever had gallstones?” This screening method has been used in previous studies. 10
Definition of sleep disorders
The presence of sleep disorder was determined by asking, “Have you ever told a doctor or other health professional that you have sleep disorder?” The possible answers were “Yes,” “No,” “Refused,” and “Don’t know.” Participants who answered “Refused” or “Don’t know” were coded as missing and excluded from the analysis. 11
Definition of covariates
Potential covariates based on previous studies included age, sex, race, educational level, marital status, BMI, cholesterol, diabetes, hypertension, smoking status, drinking status, long-term sedentary behavior (LTSB), and sleep duration. Race data were classified as non-Hispanic White, non-Hispanic Black, Mexican American, or other races. Participants were categorized into two groups based on their marital status: (a) living alone (never married, separated, divorced, or widowed) and (b) living with a partner (married and living with a partner). Educational level was divided into three levels based on the years of education received: <9 years, 9–12 years, and >12 years. Diabetes was defined as a medical diagnosis of diabetes by a doctor. 12 Hypertension was defined as a medical diagnosis of hypertension by a doctor. 13 Smoking status was categorized as smoking (having smoked 100 cigarettes in a lifetime) and nonsmoking (not having smoked 100 cigarettes in a lifetime). 14 Drinking status was categorized as drinking (at least once a month) and nondrinking (less than once a month). 10 Sedentary behavior was defined as sedentary time ≥600 min per day, classified as LTSB. 15 Sleep duration was calculated as the time between usual sleep time and usual wake time. According to the consensus of the American Academy of Sleep Medicine (AASM), Sleep Research Society (SRS), and National Sleep Foundation, 16 short sleep duration was defined as ≤6 h of sleep per night, and long sleep duration was defined as ≥9 h of sleep per night, as applied in previous studies. 11
Statistical analyses
Participants’ characteristics were summarized as mean (SD) values for continuous variables and frequencies (percentages) for categorical variables. Continuous data were compared using two-sample (unpaired) t-tests, and categorical data were compared using chi-square tests. To assess the association between sleep disorder and gallstones, univariable and multivariable logistic regression models were used. Model 1 was adjusted for age and sex. Model 2 was additionally adjusted for BMI and hypertension; these variables were included because they are well-established risk factors for gallstone disease, showed strong associations with the outcome in our univariable analyses, and satisfied the change-in-estimate criterion (≥10%) for identifying key confounders. Model 3 was further adjusted for race, educational level, marital status, cholesterol, diabetes, smoking status, drinking status, LTSB, and sleep duration as part of a fully adjusted model. Interactions between subgroups were tested using likelihood ratio tests. A
Additionally, several sensitivity analyses were conducted to evaluate the robustness of our results. First, missing covariate values were imputed using multiple imputation by chained equations (MICE), generating five imputed datasets based on the variables included in the final statistical model; the results across these datasets were then combined according to Rubin’s rules. 17 Second, when using the NHANES dataset, it is recommended to use sampling weights and design variables in all analyses to avoid biased estimates and overestimation of significance levels. Therefore, according to the NHANES analysis guidelines, a complex sampling design and sampling weights were used in the analyses. Third, to make our results more robust, propensity score matching (PSM) was performed. A 1:1 nearest neighbor matching algorithm with a caliper width of 0.2 was used. The variables selected to generate propensity scores were age, sex, race, educational level, marital status, BMI, cholesterol, diabetes, hypertension, smoking status, drinking status, LSTB, and sleep duration. The degree of PSM was checked using standardized mean differences, with <0.1 considered an acceptable threshold. Statistical analyses were performed using R software (version 4.2.1, R Foundation for Statistical Computing, Vienna, Austria), R survey package (version 4.1-1), and Free Statistics software (version 1.7.1, Beijing FreeClinical Medical Technology Co., Ltd.)
Results
Study population
Of the 15,560 participants who completed the interviews, 6328 were excluded because they were aged <20 years. Thus, 9232 participants aged ≥20 years were included. Further data screening excluded 1794 participants for the following reasons: (a) 22 had missing gallstone disease data; (b) 8 had missing sleep disorder data; and (c) 1764 had missing data for other covariates. Ultimately, 7438 participants were included in the final analyses (Figure 1).

Study flow chart.
Baseline characteristics
In total, 7438 adult participants were enrolled, including 6658 without gallstones (no gallstone group) and 780 with gallstones (gallstone group). The average age of the overall study population was 50.6 years (SD = 17.5 years), and 51.9% were women. Table 1 shows the demographic characteristics of the two groups. Compared with the no gallstone group, the average age was higher (58.0 vs. 49.7 years,
Baseline characteristics of participants in the NHANES (2017–2020).

BMI: body mass index; LTSB: long-term sedentary behavior; NHANES: National Health and Nutrition Examination Survey.
Relationship between sleep disorder and gallstones
Univariable analysis (Supplementary Table 1) showed that participants with sleep disorder had 2.08 times higher odds of having gallstones than those without sleep disorder (95% confidence interval (CI) = 1.79–2.42,
Multivariable logistic regression results are shown in Table 2. In the unadjusted model, sleep disorder was positively associated with the odds of having gallstones (odds ratio (OR) = 2.08, 95% CI = 1.79–2.42). After sequential adjustment in Models 1–3, the association was attenuated but remained statistically significant (Model 1: OR = 1.79, 95% CI = 1.53–2.09; Model 2: OR = 1.56, 95% CI = 1.33–1.83; and Model 3: OR = 1.49, 95% CI = 1.26–1.76).
Association between sleep disorder and gallstones.
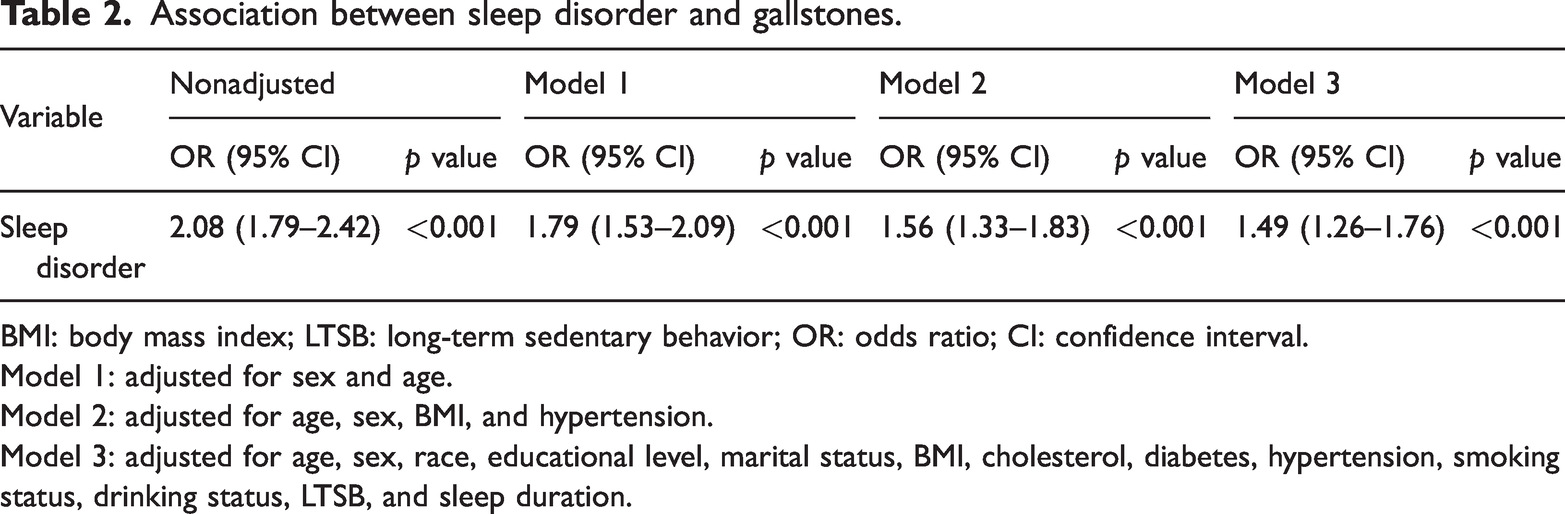
BMI: body mass index; LTSB: long-term sedentary behavior; OR: odds ratio; CI: confidence interval.
Model 1: adjusted for sex and age.
Model 2: adjusted for age, sex, BMI, and hypertension.
Model 3: adjusted for age, sex, race, educational level, marital status, BMI, cholesterol, diabetes, hypertension, smoking status, drinking status, LTSB, and sleep duration.
Subgroup analyses
Subgroup analyses were conducted to examine whether the association between sleep disorder and gallstones differed across population subgroups (Figure 2). Across sex, obesity, diabetes, hypertension, and sleep duration categories, the associations were generally consistent. However, a significant interaction was observed for age (
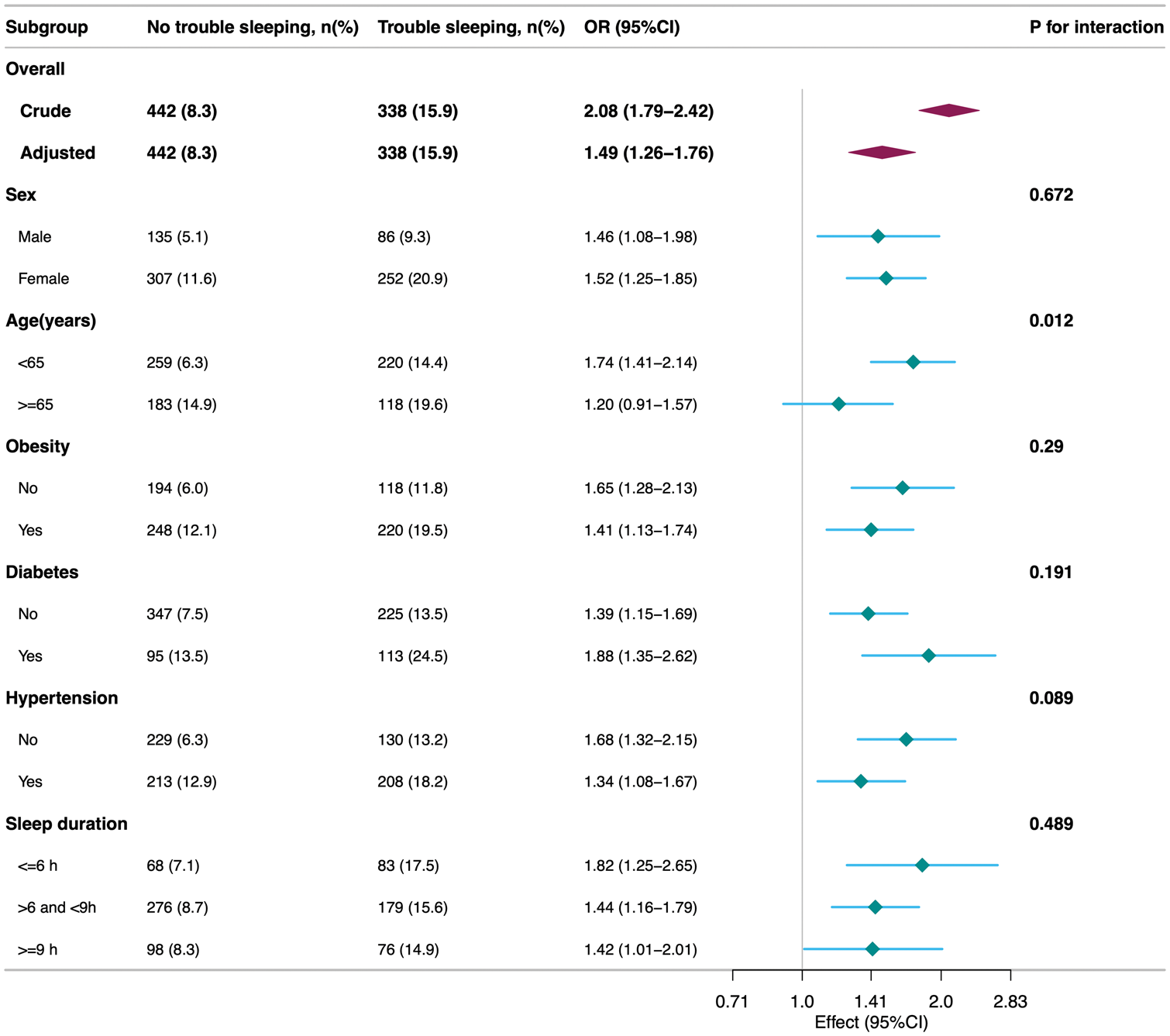
Subgroup analyses for the association of sleep disorder with gallstones.
Sensitivity analyses
To validate the robustness of the results, multiple imputation, weighted analysis, and PSM analyses were conducted (Supplementary Table 2). Among the 9232 participants, 30 were excluded because of missing data on gallstones and sleep disorder; multiple imputation was performed for the remaining 9202 participants with missing covariates. Participants with sleep disorder had 1.44 times higher odds of having gallstones than those without sleep disorder (95% CI = 1.24–1.67,
Discussion
This cross-sectional study, based on the NHANES (2017–2020) data, examined the association between sleep disorder and gallstone disease among adult Americans. The results showed that participants with sleep disorder had higher odds of having gallstones (OR = 1.49, 95% CI = 1.26–1.76). The study also validated the robustness of the results through multiple sensitivity analyses, such as multiple imputation, weighted analysis, and PSM.
The findings of this study are consistent with those reported by Chen et al., according to which, obstructive sleep apnea is significantly associated with an increased risk of gallstones. They reported a higher incidence of gallstones in patients with obstructive sleep apnea, suggesting that sleep disorder is associated with a higher likelihood of gallstones. 18 Tian et al. analyzed data from the UK Biobank and the FinnGen Consortium using Mendelian randomization and found that napping is causally associated with an increased risk of gallstones, possibly because napping inevitably reduces nighttime sleep quality. 19 However, other studies have failed to determine a clear association between sleep disorder and the risk of gallstones. 20 Inconsistencies in study results may be due to various factors, including sample size, study design, and differences in the definition and measurement of sleep disorder. This study utilized a large national dataset and adjusted for multiple confounding factors, providing more reliable evidence supporting a positive association between sleep disorder and the prevalence of gallstones. Although our principal analyses used standard logistic regression models, we also compared unweighted and weighted approaches to account for the NHANES’ complex sampling design. Both methods produced qualitatively similar point estimates for the association between sleep disorder and gallstones. However, the weighted analysis yielded slightly wider confidence intervals, reflecting the impact of stratified sampling and oversampling of certain demographic groups. These minor discrepancies underscore the importance of applying survey weights when using nationally representative data to minimize potential biases and better approximate true population-level effects.
In the sex subgroup, both men (OR = 1.46; 95% CI = 1.08–1.98) and women (OR = 1.52; 95% CI = 1.25–1.85) with sleep disorder exhibited higher odds of gallstones, although the risk was slightly higher in women. This finding is consistent with that reported by Srivastava et al., wherein there were biological differences in many metabolic and hormonal factors related to gallbladder diseases in females, possibly explaining the role of sex in gallstone susceptibility. 21 Another study has also indicated that female patients with sleep disorder had 1.62 times higher odds of having gallstones than those without sleep disorder. 22 Younger participants (<65 years) with sleep disorder exhibited higher odds of having gallstones (OR = 1.74; 95% CI = 1.41–2.14), while the risk was lower in the older group (>65 years), with significant interaction observed in the age subgroup. This is consistent with Su et al.’s observation that age is a risk factor for gallbladder diseases, and younger individuals may be more susceptible to gallstone formation due to lifestyle and physiological factors. 23 Obese, diabetic, and hypertensive participants with sleep disorder had significantly higher odds of gallstones. These three conditions are often associated with metabolic syndrome, which may be related to gallstone formation via its influence on the chemical composition of bile and gallbladder dynamics. This is consistent with Svistunov et al.’s study that emphasized the connection between these metabolic diseases and gallbladder diseases. 24 The sleep duration subgroup analyses showed significant differences in the impact of sleep disorder combined with different sleep durations on the risk of gallstones. Specifically, the group with sleep disorder and short sleep duration (≤6 h) had the highest risk of gallstones (OR = 1.82, 95% CI = 1.25–2.65), indicating that insufficient sleep was strongly associated with higher odds of gallstones in those with sleep disorder. The group with sleep disorder and normal sleep duration (>6 h and <9 h) had a relatively lower risk of gallstones, OR = 1.44 (95% CI = 1.16–1.79), suggesting that maintaining normal sleep duration partially mitigates the impact of sleep disorder on gallstone prevalence. The group with sleep disorder and long sleep duration (≥9 h) had the lowest risk of gallstones (OR = 1.42, 95% CI = 1.01–2.01). Future research should further explore the association of sleep duration with gallstone risk and evaluate the potential benefits of improving sleep quality in relation to gallstone outcomes.
Numerous studies have linked sleep disruptions to other metabolic and gastrointestinal (GI) disorders, such as nonalcoholic fatty liver disease and irritable bowel syndrome, in addition to gallstones.25,26 These conditions share overlapping pathophysiological features, particularly chronic inflammation, insulin resistance, and altered gut microbiota, which may be further exacerbated by disturbed sleep. Although our cross-sectional design precludes firm causal inferences, prospective or interventional studies could aid in clarifying the causal pathways underlying these associations. Further evidence linking sleep disorder to gallstone disease comes from an experimental mouse study in which feeding occurred during the animals’ usual rest phase, thereby disturbing normal circadian rhythms. 27 This shift resulted in altered expression of hepatic genes involved in bile acid and cholesterol metabolisms, imbalanced gut microbiota, and a marked increase in cholesterol gallstones. These findings suggest that circadian misalignment contributes to conditions that favor gallstone formation by disrupting hepatic lipid metabolism and gut microbial communities, echoing our cross-sectional observations in human populations.
The biological mechanisms underlying the relationship between sleep disorder and gallstone formation are not fully elucidated but can be speculated as follows:
Although this study provides important insights into the relationship between sleep disorder and gallstones among US adults, certain limitations should be acknowledged. First, the cross-sectional design precludes causal inference. We cannot conclude whether sleep disorder precedes the development of gallstones, whether gallstones adversely affect sleep, or whether both conditions arise from shared underlying factors. Second, information on gallstones and sleep disorder was based on self-reported questionnaires, which may have introduced recall bias and misclassification. Self-reported sleep problems may not capture the full spectrum or severity of sleep disorders, and self-reported gallstones may not always be confirmed using imaging or clinical records. Third, despite adjustment for a broad set of sociodemographic, lifestyle, and clinical variables, residual confounding cannot be ruled out. In particular, we did not include detailed data on dietary patterns, objectively measured physical activity, use of hormone therapy or lipid-lowering drugs, and some comorbidities, all of which could influence both sleep and gallstone formation. Future studies incorporating more granular assessments of diet and physical activity as well as comprehensive medication and comorbidity data are needed to better address residual confounding and clarify the temporal relationship between sleep disorder and gallstone disease.
Conclusion
Individuals with sleep disorder had higher odds of gallstones in this nationally representative sample. This cross-sectional association does not establish causality but suggests that sleep problems are linked to gallstone disease or reflect higher underlying risk of gallstones. Future longitudinal and interventional studies with objective sleep measures are needed to clarify causal pathways and assess whether improving sleep can reduce the burden of gallstone disease.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261418359 - Supplemental material for Association of sleep disorder with increased risk of gallstone disease in adults in the United States: A cross-sectional study of the National Health and Nutrition Examination Survey (2017–2020)
Supplemental material, sj-pdf-1-imr-10.1177_03000605261418359 for Association of sleep disorder with increased risk of gallstone disease in adults in the United States: A cross-sectional study of the National Health and Nutrition Examination Survey (2017–2020) by Yaqiong Zhuang, Yisen Huang, Yingxuan Huang, Chanchan Lin, Boming Xu, Yilin Zeng, Peizhong Chen, Yubin Wang and Xiaoqiang Liu in Journal of International Medical Research
Footnotes
Acknowledgments
The author expresses gratitude to all the National Health and Nutrition Examination Survey (NHANES) participants for their valuable contributions to this study.
Author contributions
Conceptualization: Xiaoqing Liu and Yaqiong Zhuang. Data curation: Yisen Huang, Yingxuan Huang, and Chanchan Lin. Formal analysis: Boming Xu. Investigation: Yinglin Zeng. Methodology: Peizhong Chen. Resources: Xiaoqiang Liu. Software: Yaqiong Zhuang. Supervision: Yubin Wang. Writing—original draft: Yaqiong Zhuang. Writing—review and editing: Xiaoqiang Liu.
Data availability statement
Declaration of conflicting interests
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Ethics approval and consent to participate
Ethics approval was obtained from the National Center for Health Statistics (NCHS) Ethics Review Committee, and participants provided written informed consent. All procedures were performed in accordance with the Declaration of Helsinki (1975, as revised in 2024) and relevant NCHS guidelines and regulations. The secondary analyses did not require additional institutional review board approval.
Funding
This study was supported by the Fujian Medical University (grant No. 2022QH1268). The funder played no role in the study design, analysis, decision to publish, or manuscript preparation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
