Abstract
Background:
Since their emergence, drug-coated balloons (DCBs) have been used widely to treat in-stent lesions with coronary artery disease (CAD). However, despite their superior efficacy to balloon angioplasty, how DCBs affect neointimal characteristics is poorly understood.
Objectives:
We aimed to assess the neointimal characteristic changes following DCB treatment.
Methods:
Using optical frequency domain imaging (OFDI), we serially observed the in-stent lesion site just after and 1 year after DCB angioplasty in 12 lesions of 11 patients with repeated revascularization. Neoatherosclerosis was defined as lipid-laden neointima with or without calcification in the stented lesion. Progression or regression of neoatherosclerosis, newly formed neointimal calcification, newly formed uncovered strut and newly formed evagination were assessed. Tiny tissue protrusion was also recorded as mushroom-like protrusion.
Results:
Underlying stents were first-generation (n = 5) or newer (n = 7) drug-eluting stents (DESs) with implantation durations ranging from 1 to 15 years (median 8 years). Surprisingly, two-thirds of the lesions (67%, 8 of 12) showed progression of neoatherosclerosis, while a quarter of lesions (25%, 3 of 12) showed regression of neoatherosclerosis. The maximal lipid arc increased from 122° to 174°. Newly formed neointimal calcification was observed in 2 of 12 lesions (16%). Newly formed uncovered struts (33%; 4 of 12) and newly formed evaginations (33%; 4 of 12) were not rare. Mushroom-like protrusion was found in a quarter of lesions (25%; 3 of 12).
Conclusion:
Our study demonstrated that a considerable number of lesions showed varied neointimal characteristic changes in a small number of patients. Further studies in a larger population are needed to understand the clinical impact of these findings.
Introduction
Drug-coated balloons (DCBs) are a standard treatment for in-stent lesions with coronary artery disease (CAD). For managing in-stent restenosis, DCBs have demonstrated superior efficacy to balloon angioplasty or repeat implantation of bare metal stents (BMSs) or first-generation drug-eluting stents (DESs), according to a network meta-analysis. 1 Current guidelines thus recommend DCBs for in-stent lesions as a Class 1 therapeutic with an evidence level of “A.” 2
Paclitaxel is the most commonly used drug for DCBs, having also been used in first-generation DESs. 3 Ever since the introduction of first-generation DESs, substantial drawbacks have been reported, such as delayed arterial healing, local hypersensitivity reaction and neoatherosclerosis, all of which led to a steady increase in major adverse cardiovascular events over time. 4 Such drawbacks are not often reported with DCBs. However, newly formed lipid-laden neointima following DCB treatment has been reported by Alfonso et al., 5 whereas newly formed neointimal calcification following DCB treatment was reported by Mori et al. 6 Furthermore, the development of late stent malapposition after DCB treatment was reported by Yumoto et al. 7 However, while these finding may have some clinical significance, they were all limited to case reports.
The present study therefore was aimed to assess the neointimal characteristic changes following DCB treatment in multiple cases.
Methods
Study population
This study was performed as a retrospective observational study and complied with the guidelines of the Declaration of Helsinki.
We retrospectively reviewed patients who underwent percutaneous coronary intervention (PCI) for their in-stent lesion with DCBs from May 2018 to June 2019 in our facility. Regardless of the symptoms, 12 lesions from 11 patients assessed with optical frequency domain imaging (OFDI) images just after the DCB angioplasty and 1 year after DCB angioplasty were compared and assessed for neointimal characteristic changes.
DCB angioplasty procedure
The lesions were basically pre-dilated with a semi-compliant balloon or scoring balloon, followed by DCB angioplasty with a paclitaxel-coated balloon (SeQuent Please® B.Braun; Melsungen AG, Vascular Systems, Berlin, Germany). The balloon profile and inflation pressure (7–14 atm) and duration (30–40 s) were left to each operator’s discretion. OFDI run was performed just after DCB angioplasty.
OFDI and analyses
The FastView™ coronary catheter with the LUNAWAVE™ system (TERUMO Corp., Tokyo, Japan) was advanced distally to the target lesion over a 0.014-inch conventional angioplasty guidewire. Red blood cells were removed by automatically infusing contrast through a 6- or 7-Fr guiding catheter at a rate of 3–5 mL/s for 2–3 s. OFDI images were acquired through auto-pullback during blood removal and then stored digitally for an offline analysis.
All OFDI images at just after DCB angioplasty and at 1-year follow up were analyzed by two experienced investigators (Y.T. and H.M.) using a dedicated offline viewer system (TERUMO Corp., Tokyo, Japan). The side-by-side comparison system enabled us to perform a close observation of neointimal morphology, and target lesions were matched based on the distance from landmarks, such as branches, calcifications, and stents. Neoatherosclerosis was defined as lipid-laden neointima and/or macrophage accumulation with/without calcification showing a circumference of ⩾30° and a length ⩾0.3 mm. 8 The lipid arc was measured on the frame with the largest lipid by visual screening. Macrophage accumulation was semi-quantitatively graded as follows: grade 0, no macrophages; grade 1, localized macrophage accumulation; grade 2, clustered accumulation in ⩾1 quadrant but <3 quadrants; and grade 4, clustered accumulation in ⩾3 quadrants. 9 An uncovered strut was defined as a strut that showed no or <10 μm tissue coverage on the strut. The strut was considered malapposed if the distance between the blooming artifact and luminal border exceeded 150 μm. Evaginations were defined as outward bulges in the luminal contour between struts. 10 Mushroom-like protrusion was defined as tiny tissue protrusions, usually with a smooth surface.
To focus on the changes in neointimal characteristics between just after DCB angioplasty and 1-year follow up, several assessments were performed. Progression or regression of neoatherosclerosis was defined when an increase or decrease, respectively, was observed by a circumference of ⩾30° and a length ⩾0.3 mm. Newly formed uncovered struts and newly formed evaginations on side-by-side comparison were also recorded. Mushroom-like protrusions occasionally observed on follow-up were also assessed.
Statistical analyses
Categorical variables are expressed as numbers and percentages. Continuous variables are expressed as the mean ± standard deviation or median with interquartile range, as appropriate. Comparisons between baseline and follow-up were performed with Wilcoxon’s signed-rank test or the McNemar test, as appropriate. The JMP software program, version 16.0 (SAS, Cary, NC, USA), was used for the analyses. A p-value < 0.05 was considered statistically significant.
Results
The patients and lesion characteristics at baseline are shown in Table 1. The patients were mostly male (82%) and a mean 73 ± 9 years old. Prevalences of traditional coronary risk factors, including hypertension, diabetes, dyslipidemia, and smoking, were considerably high in this population. In addition, the prevalences of chronic kidney disease and hemodialysis were also relatively high. Two-thirds of patients (64%) had previous myocardial infarction while the median number of previous PCI procedures (not including last DCB angioplasty) was three. Lipid and diabetes were well controlled but not optimal. Most patients presented with chronic coronary syndrome, while a few presented with acute coronary syndrome.
Baseline characteristics.

ACS, acute coronary syndrome; CCS, chronic coronary syndrome; DCBs, drug-coated balloons; DES, drug-eluting stent; HDL, high-density lipoprotein; LAD, left anterior descending artery; LCX, left circumflex artery; LDL, low-density lipoprotein; PCI, percutaneous coronary intervention; RCA, right coronary artery; TLR, target lesion revascularization.
The majority of lesions showed restenosis, while three showed very late stent thrombosis. The type of underlying stent was a DES in all cases, with implantation durations ranging from 378 to 5602 days. There were six first-generation stents (four Cypher®, one TAXUS® and one NOBORI®) and four second-generation stents (one Resolute Onyx®, two Xience® and one PROMUS Element™) and two third-generation stents (both Synergy®). Underlying stent overlap was observed in 67% of lesions. The median time from index to follow-up was 328 days. Target lesion revascularization at follow-up was needed in 42% of lesions.
Qualitative and quantitative evaluations derived from OFDI images at each time point are shown in Table 2. The minimum lumen area was significantly reduced after the follow-up period compared to that at baseline (3.3 mm2 at baseline versus 3.2 mm2 at follow-up; p = 0.04). Similarly, the maximum neointimal thickness showed an increasing trend (0.7–0.8 mm, p = 0.17). The maximum lipid arc numerically increased (122°–174°, p = 0.30). In this population, the prevalence of lipid-laden neointima and macrophages was substantially high at both the index and follow-up examinations (83–100%, p = 1.00). Although the macrophage grade, neointimal calcification, uncovered struts, any malapposition and any evagination numerically increased, no significant differences were observed.
OFDI data.
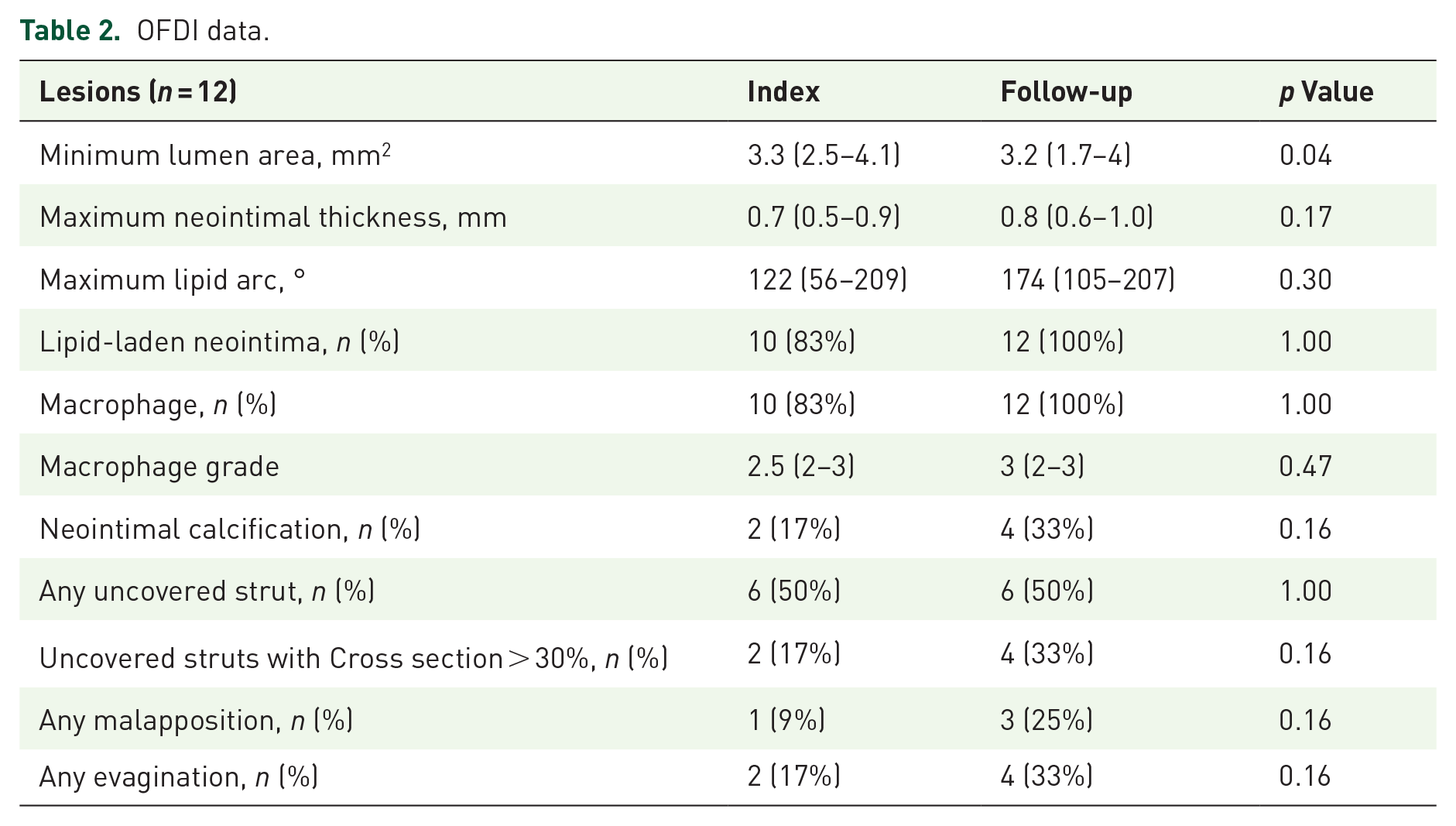
Table 3 shows the change in neointimal characteristics between index and follow-up. Representative images for progression or regression of neoatherosclerosis were shown in Figures 1 and 2. Two-thirds of the lesions showed progression of neoatherosclerosis (67%; 8 of 12), while a quarter of lesions showed regression (25%; 3 of 12). One case showed progression of neoatherosclerosis at one segment and regression at another segment. Newly formed neointimal calcification was observed in 2 of 12 lesions (16%) (Figure 3). Newly formed uncovered struts were observed in 4 of 12 lesions (33%), and newly formed evaginations were also observed in 4 of 12 lesions (33%) (Figure 4). Mushroom-like protrusions that hadn’t been observed at the index procedure were found in a quarter of lesions (25%; 4 of 12) (Figure 2). Rates of newly formed neointimal calcification and newly formed evagination tended to be greater in first-generation DESs while rates of regression of neoatherosclerosis tended to be greater in newer-generation stent. Target lesion revascularizations (TLRs) thereafter were observed in five lesions in which 80% (4 of 5) lesions showed proregression of neoatherosclerosis. Whereas TLRs thereafter were not observed in lesions with regression of neoatherosclerosis (0 of 3). Among those without TLR, progression of neoatherosclerosis was observed in 71% (5 of 7).
Neointimal characteristics changes.

Ca, calcification; DES, drug-eluting stent; TLR, target lesion revascularization; VLST, very late stent thrombosis.

Progression of neoatherosclerosis (lesion 5 in Table 3). (a) and (b) Show angiography at baseline and follow-up with corresponding OFDI images (c) and (d). The yellow line and arrows indicate newly formed lipid-laden neointima. The minimum thickness of the fibrous cap was 50 μm, suggesting a thin cap.
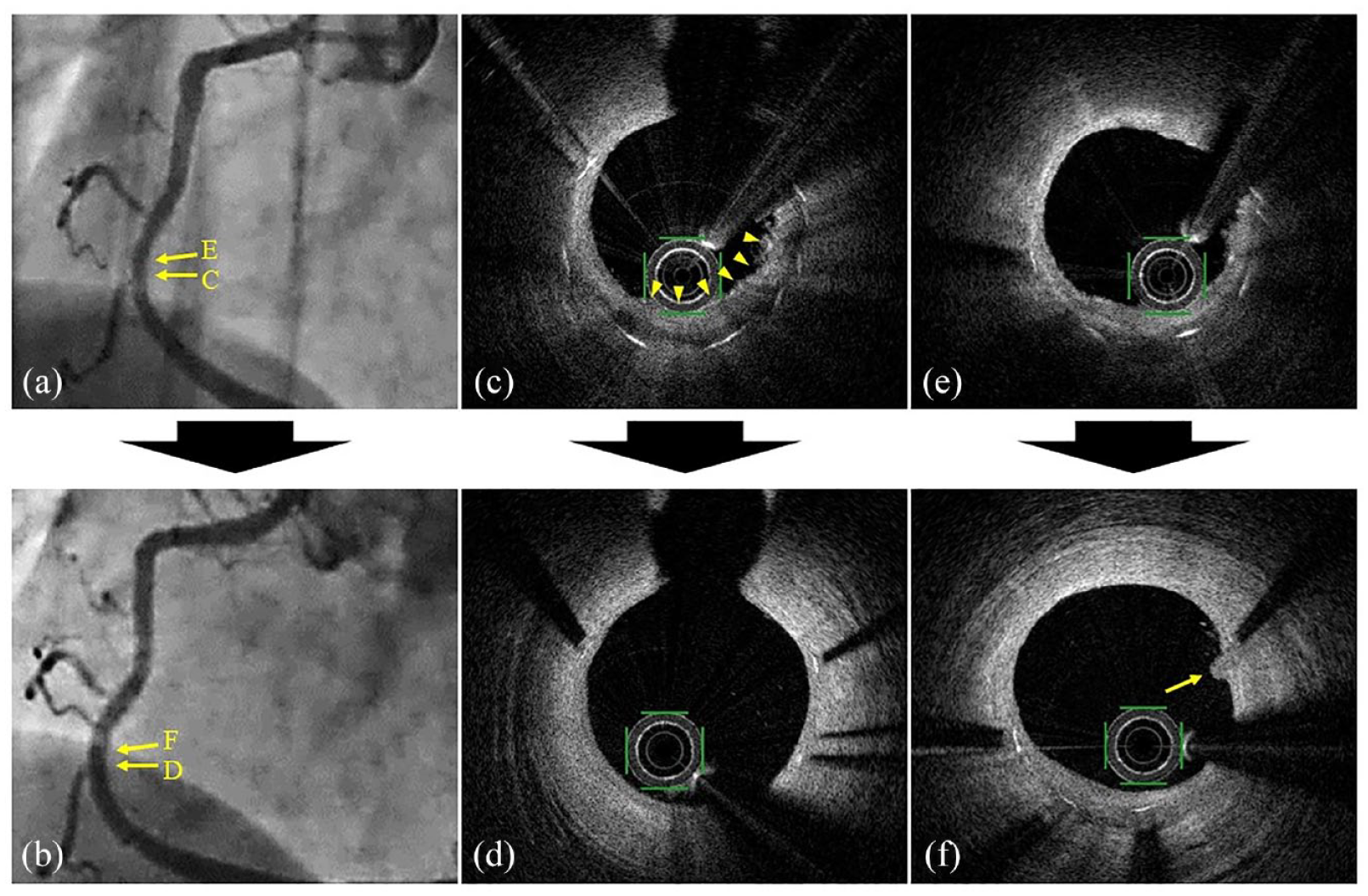
Regression of neoatherosclerosis (lesion 11 in Table 3). (a) and (b) Show angiography at baseline and follow-up with corresponding OFDI images (c) to (f). The yellow arrowheads in (c) indicate foamy macrophages at the index evaluation, which were diminished in (d). The yellow arrow in (f) indicates a mushroom-like protrusion not observed in (e).

Newly formed neointimal calcification (lesion 3 in Table 3). (a) and (b) Show angiography at baseline and follow-up with corresponding OFDI images (c) and (d). The lipid pool in (c) turned into neointimal calcification at follow-up (yellow arrows in d).

Newly formed evaginations and uncovered struts (lesion 9 in Table 3). (a) and (b) Show angiography at baseline and follow-up with corresponding OFDI images (c) to (f). The yellow arrowheads in (d) indicate newly formed evaginations not observed at baseline (c). The yellow arrows in (f) indicate newly formed uncovered struts not observed at baseline (f).
Discussion
As the main findings of this study, after DCBs treatment for in-stent lesions, a substantial proportion of lesions showed progression of neoatherosclerosis, while a few showed regressions. Newly formed evaginations and uncovered struts were not rare. Mushroom-like small protrusions were occasionally observed.
The mechanism underlying neoatherosclerosis has yet to be fully elucidated, although its prevalence is known to be significantly greater with DESs than with BMSs. 8 Even with newer-generation DESs, 50% of lesions showed neoatherosclerosis, while 20% of lesions with BMSs showed it, according to a human autopsy study (stent implantation duration: 1–5 years). 11 In a pre-clinical study, the endothelial barrier function as assessed by vascular endothelium cadherin (VE-cadherin) was significantly impaired with newer-generation DESs than with BMSs (22–39% versus 95%, respectively; p < 0.01). 12 Another in vitro study showed that a paclitaxel analog led to adhesion delay and shape change of human umbilical vein endothelial cells through the VE-cadherin-mediated integrin b1/FAK (Focal adhesion kinase)/ROCK (RhoA/Rho-associated coild coil containing kinase) signaling pathway. 13 It is thus unsurprising that paclitaxel-coated DCBs altered the neointimal endothelial function following balloon injury, leading to the progression of neoatherosclerosis.
However, why neoatherosclerosis regressed in a few lesions may be more difficult to interpret. An optical coherence tomography (OCT) study observed neointima following DES implantation at 270 and 551 days, and found that some heterogeneous neointima at the first follow-up became homogeneous neointima at the second follow-up (20%; 4 of 20). 14 Similarly, from another OCT study assessing 96 cases of in-stent restenosis treated by DCB, some of the heterogeneous neointima became homogeneous neointima (30%). 15 Accordingly, as shown in our study, neointimal characteristic changes may not be rare and in fact extremely variable, ranging from regression to progression.
Delayed arterial healing and hypersensitivity reaction were not uncommon in first-generation DES but became significantly rare in newer-generation DESs.11,16–18 Delayed arterial healing appears as uncovered struts on OCT/OFDI, while hypersensitivity reaction can lead to aneurysmal formation, appearing as peri-strut staining on angiography or evagination on OCT/OFDI.10,19 Both delayed arterial healing and hypersensitivity reaction are considered to be due to drug and/or polymer coatings on DESs.11,16–19 Similarly, drugs released from DCBs, drugs from previous DES implantation and polymers from previous DES implantation can be considerable contributing factors to the new formation of evaginations or uncovered struts.
In our study, all lesions were treated by DCBs without any additional DESs. Thus, drug release from DESs was expected to be minimal in these lesions, given the lengthy duration of previous DES implantation (8 years). Drug release from DCBs was likely minimal as well, since the tissue drug concentrations become almost zero after 2 months, according to pre-clinical studies assessing the pharmacokinetics of DCBs.20,21 Notably, all lesions with newly formed evaginations or uncovered struts had underlying first-generation DESs. Because drugs are basically combined with durable polymers in these early-generation devices, very low levels of drug might have some effect on neointima.11,22 Indeed, uncovered struts were reported in 7.6% of first-generation DES cases versus 0.8% of BMS cases, even 10 years after implantation. 23 Sites of overlapping first-generation DESs were reported to show significantly delayed healing and persistent inflammation compared to those with BMSs. 24 Taken together, these findings suggest that newly formed evaginations and uncovered struts can be triggered by DCBs placed for in-stent lesions of DESs.
Persistent fibrin deposition used to be a major pathological feature of paclitaxel-eluting stents. 17 Fibrin deposition is also observed on the surface of neointima treated by paclitaxel-coated balloons in chronic phase. 21 This fibrin deposition on the surface of neointima is often tiny and mimics mushroom-like protrusion on OFDI. When paclitaxel-eluting stents and paclitaxel-coated balloons are used in combination, fibrin deposition is reported to significantly increase compared to the individual use of either device. 25 Thus, we speculate that the mushroom-like protrusions likely represented the healed phase of fibrin. Some might worry about thrombotic events following the use of paclitaxel-including devices. However, these mushroom-like protrusions were all small and thus unlikely to have any marked influence on the clinical outcome. In some clinical studies, DCBs have been used to treat thrombotic culprit lesions of acute myocardial infarction, demonstrating a noninferior clinical outcome to DESs. 26 Therefore, fibrin deposition may occur and persist but is unlikely to have a clinical impact.
Different trends between first-generation DESs and newer-generation DESs were observed in our small number of lesions. Newly formed evagination was more frequently observed in first-generation DESs than in newer-generation DESs. Evagination following stent implantation has been reported to be much more frequent in first-generation DESs than in newer-generation DESs, possibly due in part to higher drug doses or less biocompatible polymers in first-generation DESs, which may have some impact even after a decade following stent implantation.10,22 Newly formed neointimal calcification which is a more advanced form of neoatherosclerosis, was also more frequently observed in first-generation DESs than in newer-generation DESs. 8 Neointimal calcification can depend on multiple factors, such as duration of underlying DES implantation, age and traditional risk factors. 8 Thus, it is hard to interpret too much from our data, but it is notable that newly formed calcification can occur 1 year following DCB treatment. In contrast, regression of neoatherosclerosis was more frequently observed in newer-generation DESs than in the first-generation DESs, possibly due to differences in the nature of newer-generation DESs, as they were made to attain better neointima.12,22
Overall, our data indicates that there is an inclination of neoatherosclerosis progression approximately 1 year after DCB therapy, the clinical significance of this – especially in a longer term – is yet to be understood and shouldn’t be taken as a notion to discourage the use of DCB as a therapeutic option to treat In-Stent lesions. While it is assumed that lesion with less neoatherosclerosis is applicable for DCB therapy, the most suitable lesion for its use is still unclear.
Several limitations associated with the present study warrant mention. First, this was a small, nonrandomized, observational, single-center study. Second, although OFDI findings were observed by multiple experienced physicians, interpretation is still subjective. Third, possible selection bias exists for the study population because these patients had a history of multiple revascularizations with a greater prevalence of traditional coronary risk factor than normal patients. In addition, the present study included only patients who underwent both index OFDI and follow-up OFDI. Patients who did not undergo OFDI would have exerted a different vascular response from others. Thus, it is not appropriate to generalize the phenomenon observed in this study. Nevertheless, this kind of systemic assessment has never been performed, so we are confident in the value of our study.
Conclusion
Neointimal characteristic changes, including progression/regression of neoatherosclerosis, newly formed evaginations, newly formed uncovered struts and mushroom-like protrusion, were not rare in DCB-treated in-stent lesions of CAD in lesions with repeated revascularization. Further studies in a larger population with a longer duration are needed to confirm the clinical impact of these findings.
Supplemental Material
sj-docx-1-tak-10.1177_17539447231199660 – Supplemental material for Neointimal characteristic changes following drug-coated balloons in lesions with repeated revascularization
Supplemental material, sj-docx-1-tak-10.1177_17539447231199660 for Neointimal characteristic changes following drug-coated balloons in lesions with repeated revascularization by Yosuke Takei, Hiroyoshi Mori, Takahiro Tezuka, Ayumi Omura, Daisuke Wada, Hiromoto Sone, Kazuma Tashiro, Masahiro Sasai, Tokutada Sato and Hiroshi Suzuki in Therapeutic Advances in Cardiovascular Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
