Abstract
The incidence and mortality of coronary heart disease are increasing annually, and the disease is now one of the leading causes of death in China. Percutaneous coronary intervention has become the preferred approach for treating coronary artery disease. The use of a drug-coated balloon is a new treatment strategy for coronary artery disease that has been shown to be safe and effective in intravascular restenosis, bifurcation disease, and small-vessel disease, and this approach has been extended to other indications, such as large-vessel coronary artery disease. However, some experts believe that the intima muscularis fibrosa of large vessels is thick. After dilation of large vessels with drug-coated balloons, elastic contraction may occur. Additionally, the use of drug-coated balloons cannot prevent the occurrence of vessel dissection; therefore, their use for treating large-vessel coronary disease remains controversial. This review has aimed to discuss the findings of clinical trials demonstrating the efficacy and safety of drug-coated balloons for the treatment of coronary artery de novo large-vessel lesions.
Keywords
Introduction
The multi-step transformation of China’s economy, society, natural environment, lifestyle, and healthcare system has had a significant effect on the burden of cardiovascular disease and major cardiovascular risk factors in China. Currently, coronary heart disease-related morbidity and mortality have been increasing, and the disease has become one of the leading causes of death in China, with an expected increase of >50% in cardiovascular disease events per year between 2010 and 2030. 1 Studies have shown that cardiovascular disease is beginning to affect younger people. 2 Moreover, the number of people disabled by coronary heart disease is increasing, causing a serious medical, social, and economic burden. 3 The first-line treatment for coronary artery disease (CAD) is percutaneous coronary intervention, 4 which has progressed from plain old balloon angioplasty, bare-metal stents (BMSs), and first- and second-generation drug-eluting stents (DESs). 5 However, plain old balloon angioplasty is associated with acute complications, such as acute-vessel closure and early elastic retraction, and late complications, such as neointimal proliferation.6,7 BMSs also have disadvantages, such as difficult stent delivery and high restenosis rates (20%–30%).5,8 These problems have led to the development of DESs, and many clinical trials have demonstrated the safety and efficacy of second-generation DESs. 9 However, DESs are still associated with restenosis, late in-stent thrombosis, and neoatherosclerosis.10–12
The drug-coated balloon (DCB) is a new therapeutic strategy for CAD that can deliver drugs rapidly and uniformly to the vessel wall through a lipophilic matrix in a single-balloon inflation without a permanent implant. 13 With the advent of DCBs, various complications caused by DES can be resolved, and stent restenosis has become a class-IA indication for DCBs. National and international expert guidelines have indicated that DCBs can be used in small-vessel, bifurcated, and other complex coronary lesions. Many long-term randomized controlled trials have shown that the major adverse cardiovascular events (MACEs) of DCBs are similar to those of DESs. 14 However, large-vessel disease is more common in CAD, 2 and proximal large-vessel disease is more common than distal small-vessel disease in autopsy and angiographic studies. 15 Several clinical trials have demonstrated the safety and efficacy of DCBs for treating coronary artery de novo large-vessel lesions. In particular, DCBs avoid foreign-body implantation, reduce the endothelial inflammatory response, decrease the risk of thrombosis of DCBs, shorten dual antiplatelet therapy duration, and make pharmacological balloon angioplasty a better option for treating large-vessel coronary lesions. This review presents and discusses the DCB technique and its clinical trials in coronary artery de novo large-vessel lesions.
Methods
We performed a detailed search of PubMed, Web of Science, and other databases to select the most recent and comprehensive data up to 10 December 2022. The following keywords were used: (“paclitaxel-coated balloon” OR “paclitaxel-eluting balloon” OR “drug-eluting balloon” OR “drug-coated balloon” OR “DCB” OR “drug-eluting stent” OR “DES”) AND (“coronary atherosclerotic disease” OR “coronary atherosclerotic disease” OR “coronary macrovascular disease”).
Large-vessel CAD
The reference vessel diameter is defined as the average of the proximal and distal diameters of the target-lesion segment, and there is no universally acceptable definition to differentiate coronary vessel diameter. In this review, we chose 2.75 mm as a cutoff and defined vessels with a diameter ≥2.75 mm as large coronary vessels.16,17
DCB technology
DCB uses single-balloon dilatation, which is designed to provide rapid and uniform drug delivery through a lipophilic matrix. The DCB drug type, effective excipients, delivery drug loss, pharmacokinetic rate, optimal drug loading, release kinetic profile, specific balloon selections, and lesion preparation all affect the drug-delivery efficiency. 18
Drugs used in DCBs
Paclitaxel was the first drug used in a DCB.14,19–21 The drug reduces inflammation and plaque progression. 22 Some meta-analyses have suggested that the use of paclitaxel-coated balloons is associated with increased mortality.23,24 This association may be related to the lack of patient-level data, lack of a quantitative effect size, crossover and selection bias in follow-up, lack of plausible pathophysiological explanations, and the fact that high-dose paclitaxel is safe in other medical indications. 25 Meta-analyses by Ko et al. 26 and Gray et al. 27 on paclitaxel-coated balloon safety in peripheral arteries as well as several large meta-analyses on the use of paclitaxel-coated balloons in coronary arteries28,29 have demonstrated that intervention with paclitaxel-coated balloons is safe.
Sirolimus is another antiproliferative drug currently used in DCBs. Although sirolimus is considerably less lipophilic than paclitaxel, it reduces long-term vasodilation better than paclitaxel. 30 In the Nanoluté trial 31 and SIRolimus–PAClitaxel study, 32 the efficacy of sirolimus was not significantly different from that of paclitaxel-coated balloons. Therefore, sirolimus-coated balloons can be considered safe for use in CAD (Table 1).
Currently approved drug-coated balloons.
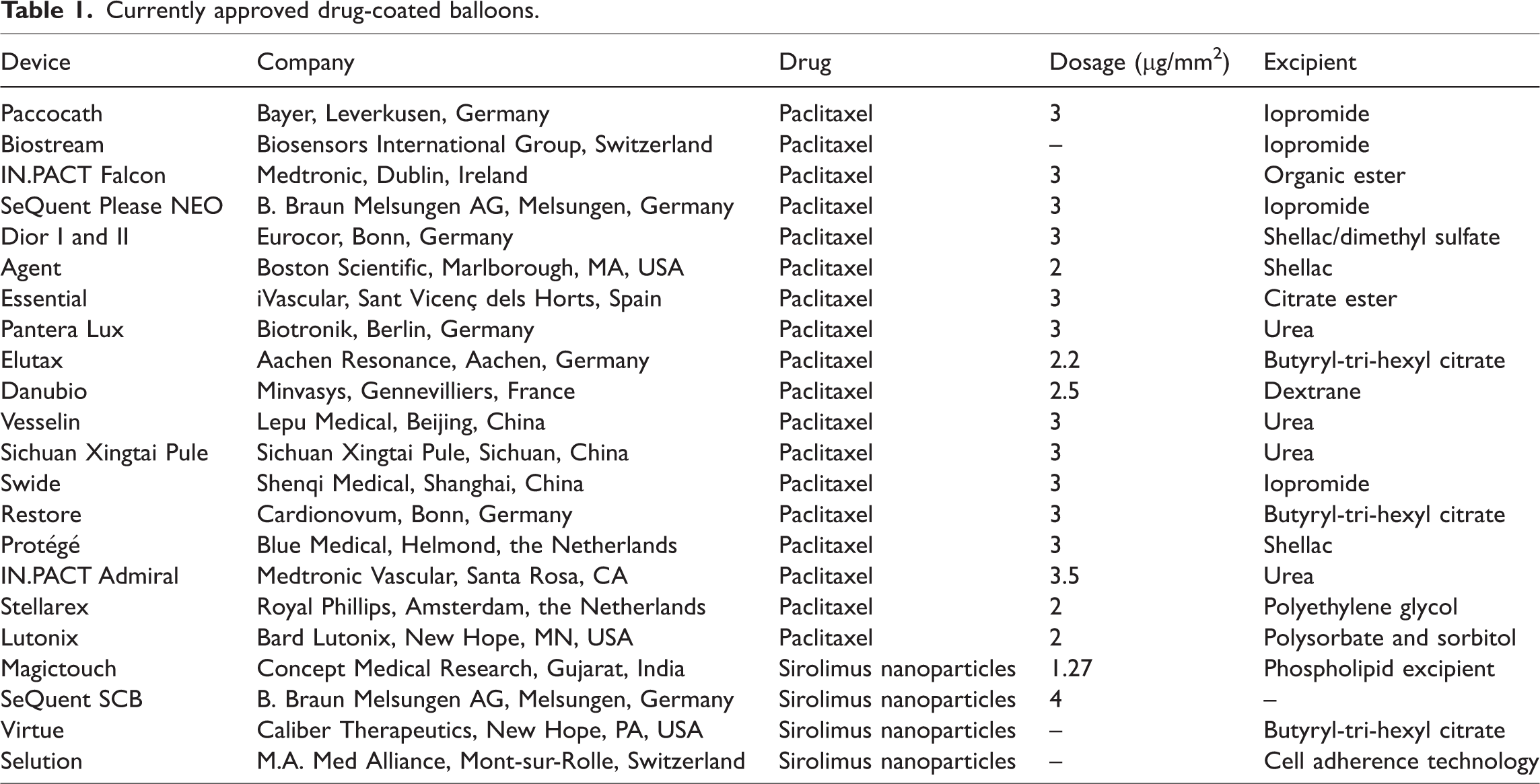
Currently, betamethasone dipropionate, bortezomib, and green tea extract are also used in DCBs, but recent studies have shown that these drugs are less effective for paclitaxel-coated balloons in clinical treatment. 33
Excipients
Currently, investigational adjuvants, such as iopromide and urea (Table 1), have been used in DCBs. Resveratrol, 34 keratin, 35 and magnesium stearate 36 have also been shown to have an adjuvant effect on DCB interventions in animal studies. However, further studies are needed to determine if they can be used clinically.
Drug loss during delivery
During DCB preparation, insertion, and inflation, antiproliferative drug particles are lost from the intended coating, and drug loss during DCB preparation and insertion affects delivery efficiency. Faenger et al. 37 compared drug loss in seven different DCBs during treatment and showed that the drug loss decreased by 0.4%–12% after removal of the protective shield, whereas none of the DCBs showed significant drug loss when pushed into the vessel sheath. Furthermore, drug loss during preparation differed by the type of DCB; the lower the loss, the more efficient the DCB delivery. To avoid excessive drug loss from DCBs during delivery, care is needed to not touch or wet the DCB material, to not pre-inflate the DCBs, and to use each DCB only once.
Preoperative preparation
Balloon selection and lesion preparation are critical to DCB delivery and treatment. Clinical trials in coronary neovascular lesions have also shown that good lesion preparation is an important factor in the successful use of DCBs. For example, different balloons may be used for the pre-dilatation of different lesions: 1. For uncomplicated lesions, a semi-compliant or noncompliant balloon with a 1:1 balloon-to-artery ratio is recommended for lesion preparation. 2. If balloon delivery is difficult, inadequate vessel filling or possible undersizing are anticipated, a smaller balloon may be used initially, and vessel size can be reassessed after administering vasodilators. 3. High-pressure noncompliant or cutting balloons may be used if the lesion shows calcification. After lesion preparation, the following conditions should be met before DCB deployment: (a) a fully-inflated balloon sized to the vessel; (b) residual stenosis ≤30%; (c) thrombolysis in myocardial infarction (MI) grade 3; and (d) absence of grade C or greater occlusion. If these requirements are not met, a DES may be considered.13,38
DCBs in large-vessel disease clinical trials
Efficacy of DCB-only application in coronary artery de novo large-vessel lesions
In a single-center retrospective study, 39 102 patients (vessel diameter >2.8 mm) were treated with DCBs (SeQuent Please or SeQuent Please NEO; B. Braun Melsungen AG, Melsungen, Germany). Seventeen (16.3%) patients developed vessel dissection after pre-expansion, of whom eight underwent repeat stenting due to severe dissection or target-lesion hematoma. The incidences of MACEs (n = 4, 3.9%) and target-lesion revascularization (TLR) (n = 4, 3.9%) at 12 months were low, with a late lumen loss (LLL) of 0.01 ± 0.52 mm. There were no deaths or MIs at follow-up. In a single-center prospective study, 40 120 patients with coronary artery de novo lesions with a vessel diameter ≥3.0 mm were treated with DCB (SeQuent Please). The incidence of vessel occlusion after pre-expansion was 35% (n = 42), of which 23 (19.1%) and 2 (1.6%) were types C and D, respectively, and two patients with type D occlusion underwent corrective stent implantation. The incidence of target-lesion failure was 3.4%, with no patients experiencing death or target-vessel thrombosis. Another multi-center prospective study 41 included 119 patients (vessel diameter ≥2.75 mm) treated with DCB alone (SeQuent Please or Swide; Shenqi Medical, Shanghai, China) with a type C vessel dissection during treatment but without remedial stenting, and the TLR rate was 3.4% (n = 4). In a prospective study by Lu et al., 42 92 patients (vessel diameter ≥2.7 mm) were enrolled for DCB-only treatment (SeQuent Please) and 6 required adjunctive DES (sirolimus-eluting stent) implantation. At the 9-month follow-up, the TLR and overall MACE rates were 4.3% and 4.3%, respectively. The LLL was −0.02 ± 0.49 (Table 2).
DCB-only application in coronary artery de novo large-vessel lesions.

MACE: major adverse cardiac event, MI: myocardial infarction; TLF: target-lesion failure; TLR: target-lesion revascularization.
These studies showed a low incidence of adverse cardiovascular events in coronary artery de novo large-vessel lesions treated with DCBs alone, demonstrating the safety and efficacy of DCBs in the treatment of coronary artery neovascular lesions. Although the use of DCBs can lead to the development of vessel dissection, these studies have shown that the vast majority of cases of vessel dissection require only clinical observation and do not require stenting. However, these were single-arm observational studies, not controlled trials; therefore, randomized controlled trials are needed to prove the value of DCBs in coronary artery neovascular lesions.
Comparison of DCB application in large and small vessels
In a retrospective study, 43 527 patients with CAD were divided according to vessel diameter into a large-vessel diameter (n = 200) group with vessel diameters ≥2.8 mm and a small-vessel diameter (n = 327) group with vessel diameters <2.8 mm. Both groups were treated with DCBs (SeQuent Please). In all lesions treated with DCBs, there were 196 (32.9%) lesions with vessel dissection. There were fewer cases of large-vessel disease than small-vessel disease (63 vs. 133, p = 0.04) and more cases of type-B small-vessel disease than large-vessel disease (16 vs. 24, p = 0.007). However, the numbers of retreatment stents implanted were similar between the groups (1 vs. 1, p = 0.715). During follow-up, one patient in the small-vessel disease group had an MI. There was no MI or target-vessel thrombosis in the large-vessel disease group and no significant difference in MACE (0% vs. 1.4%, p > 0.05) and TLR (0% vs. 1.1%, p > 0.05) rates between the groups. In a prospective study, 44 215 patients with CAD were enrolled for paclitaxel-coated balloons (SeQuent Please or SeQuent Please NEO) intervention according to lesion vessel diameter, divided into the large-vessel disease group (≥2.8 mm, n = 85) and small-vessel disease group (<2.8 mm, n = 130). During paclitaxel-coated balloon treatment, one patient each in the large-vessel disease and small-vessel disease groups underwent stenting (1.1% vs. 0.7%, p > 0.05). At post-procedural follow-up, one patient in the small-vessel disease group and none in the large-vessel disease group had a MACE (0 vs. 2.3%, p > 0.05), and the target-lesion failure rates (0% vs. 1.5%) and LLL (−0.16 ± 0.48 vs. −0.19 ± 0.41, p = 0.81) were similar between the groups. Rosenberg et al. 17 placed 234 patients from the International Multi-center DCB Registry data into the large-vessel disease group (≥2.75, n = 117) and small-vessel disease group (<2.75, n = 117), and both groups underwent DCB intervention (SeQuent Please); a total of 19 patients required repeat stenting (7.6% vs. 7.1%, p = 0.880). During follow-up, there was one definite vascular thrombosis in the large-vessel disease group and none in the small-vessel disease group (p = 0.300), and the MACE (6.1% vs. 5.1%, p = 0.903), TLR (1.0% vs. 3.8%, p = 0.200), and death (3.0% vs. 0%, p = 0.071) rates were similar between the large-vessel and small-vessel disease groups at 9 months (Table 3).
Comparison of DCB application in large and small vessels.

DAPT: dual antiplatelet therapy; DCB: drug-coated balloon; LVD: large-vessel disease; MACE: major adverse cardiac event; SVD: small-vessel disease; TLF: target-lesion failure; TLR: target-lesion revascularization.
Recent expert consensus has shown that DCBs can be used in small-vessel lesions. These clinical trials, all of which included large numbers of patients, have shown that DCBs used in large-vessel lesions have similar efficacy and safety to those used in small vessels. Vessel occlusion is inevitable with DCB deployment, but these studies showed that only a few type C and D vessel occlusions required corrective stent implantation, demonstrating that DCBs are safe and effective in large-vessel disease. However, the follow-up time of the abovementioned study was short, and it was not a randomized controlled trial, so trials with long-term follow-ups are needed to compare the role of DCBs in large-vessel and small-vessel disease lesions.
Comparison of the efficacy of DCB and DES in de novo large-vessel lesions
The randomized PEP CAD NSTEMI 45 trial enrolled 210 patients (vessel diameter: 2.5–3.5 mm) treated with DCB (n = 104, SeQuent Please or SeQuent Please NEO) or stenting (n = 106, DES: 44%, BMS: 56%). Nine cases required repeat stenting during DCB treatment (one with BMS, eight with DES). At the 9-month follow-up, there was no significant difference in the target-lesion failure (4.1% vs. 6.3%, p = 0.75) and MACE (5.9% vs. 14.4%, p = 0.056) rates between DCB and stenting. In a simple, single-center randomized controlled trial, 46 60 patients received either an appropriately sized DCB (n = 30; Lacrosse non-slip element balloon) or an appropriately sized second-generation DES (n = 30; everolimus-eluting stent). DES stenting was required in three of the DCB patients. There were no MACEs after either post-procedure treatment. At the 8-month follow-up, the TLR rates (0.0% vs. 6.1%, p = 0.193) and LLL (0.25 ± 0.25 vs. 0.37 ± 0.40, p = 0.185) were not significant in either group. In the prospective, single-center REVELATION randomized controlled trial, 47 120 patients (vessel diameter ≥2.75 mm) were randomized to treatment with DCB (n = 60, Pantera Lux; Biotronik, Berlin, Germany) or DES (n = 60, Orsiro; Biotronik or Xience; Abbott Vascular Devices, Santa Clara, CA, USA). One patient required emergency stenting during DCB treatment. During follow-up, there were no deaths or MACEs in the patients treated with DCB or DES. The TLR rates (DCB 3% vs. 2%, p > 0.05) and LLL (–0.40 to 0.20 vs. –0.16 to 0.10, p = 0.51) were similar. The safety of DCB and of DES in de novo large-vessel lesions were compared in a prospective cohort study 48 of 1237 patients treated with a DCB (n = 544) or a DES (n = 693). The majority of these patients had large-vessel lesions: 73% in the DCB group vs. 86% in the DES group had vessel diameters ≥3 mm. At a mean follow-up of 3.5 years, mortality (p = 0.24), acute MI, and stroke were similar between DCB and DES. Gobić et al. 49 compared the difference between DCB-only treatment and DES treatment in primary ST-elevation MI in a randomized controlled trial that included 78 patients (vessel diameter: 2.5–4.0 mm) treated with DCB (n = 41, SeQuent Please) or DES (n = 37, Biomime; Meril Life Sciences, Vapi, India) and 3 (7.3%) treated with DES due to DCB treatment with persistent stenosis (n = 2) or C–F type vessel occlusion (n = 1). Two patients in the DCB group experienced post-procedure bleeding with black stool during the post-procedure hospital stay, but no bleeding was reported in the DES group (5.3% vs. 0%, p = 0.49). DCB and DES treatments were similar in the Canadian Cardiovascular Society angina classifications (p = 0.39) and MACE rates (5.3% vs. 5.4%, p = 0.98) at 1 month and Canadian Cardiovascular Society angina classification (p = 0.68) and MACE rates (0% vs. 5.4%, p = 0.26) at 6 months, with no significant difference. There was also a trend toward more MACEs in the DES group than in the DCB group at 6 months (relative risk, 5.13; 95% CI: 0.25–103.42, p = 0.29). Another single-center randomized controlled trial 50 enrolled 84 patients (vessel diameter: 2.5–4 mm) randomized to DCB (n = 42, Yinyi Biotech Bingo, Liaoning, China) or DES-eluting stent (n = 42). Four patients who developed severe vessel dissection during DCB treatment were treated with salvage DES implantation. At the 1-year follow-up, two patients in the DCB-treated group died of systemic cardiovascular disease, and one patient was treated with DES for TLR. Three patients in the stent group received DCB for TLR, and two patients died of MI. MACE rates were similar between the DCB and stent groups at 1 year (11% vs. 12%, p > 0.05). There was no significant difference in the Canadian Cardiovascular Society angina classifications between groups at 1, 6, and 12 months. There was no significant difference in LLL (0.13 ± 0.36 vs. −0.11 ± 0.45, p > 0.05) between the two groups. A single-center study 51 included 40 patients treated with paclitaxel-coated balloons (Eurocor GmbH, Bonn, Germany) who were compared with patients who underwent BMS (n = 51) and DES (n = 49) treatment. Four patients had salvage stents for types C and D artery dissection. At the 6-month follow-up, the LLL of the paclitaxel-coated balloon group was 0.51 ± 60.59 mm, which was not significantly different from that for BMS alone (0.74 ± 60.57 mm, p = 0.64) and DES alone (0.21 ± 60.32 mm, p < 0.01), indicating a high restenosis rate with paclitaxel-coated balloon treatment. The incidence of MACEs was 17.5% for paclitaxel-coated balloons, 23.5% for BMS (p = 0.20) and 4.1% for DES alone (p = 0.90). These findings indicate that the incidence of late cardiovascular adverse events with paclitaxel-coated balloons was similar to that of the other three treatments. A retrospective study 52 compared 49 patients treated with DCB (SeQuent Please) with patients who underwent DES treatment (n = 49). During treatment, two (2%) cases were treated with stenting for acute-vessel occlusion. In this study, the follow-up times (DCB: 12 months vs. DES: 16 months, p < 0.01) were significantly different between the two groups, but Cox regression analysis showed that only DCB treatment was not an independent risk factor for adverse events (p = 0.43), and the follow-up MACE (6% vs. 6%, p = 1.0) and TLR (2% vs. 4%, p = 0.56) outcomes were not significantly different. To compare the efficacies of DCB and DES in the treatment of coronary artery de novo large-vessel lesions, a single-center cohort study 53 enrolled 148 patients, of which 47 received DCB (80% SeQuent Please NEO, 20% SeQuent Please) and 107 received DES treatment. During treatment, coronary dissection occurred in some patients in the DCB group; there were only two cases of DES implantation due to coronary dissection. Follow-up at 33 months showed that the all-cause mortality (DCB group: 20%, DES group: 15%, p = 0.129), cardiovascular mortality (DCB group: 4.9%, DES group: 6.5%, p = 0.210), and target-vessel reconstruction (DCB group: 7.3%, DES group: 8.3%, p = 0.86) rates were not significantly different, indicating that the safety of DCB angioplasty alone was similar to that of second-generation DES in the treatment of coronary artery de novo large-vessel lesions (Table 4).
Comparison of DCB and DES in coronary artery in situ macrovascular studies.

BMS: bare-metal stent; DCB: drug-coated balloon; DES: drug-eluting stent; LLL: late lumen loss; MACE: major adverse cardiac event; RCT: randomized controlled trial; TLF: target-lesion failure; TLR: target-lesion revascularization.
DESs are currently the mainstay of treatment for CAD and have demonstrated safety and efficacy in coronary interventions. The abovementioned trials demonstrated that the incidences of MACEs were similar for DCB and DES in the treatment of coronary artery de novo large-vessel lesions and that the use of DCB in the treatment of no foreign-body implantation truly achieved “intervention without implantation.” However, these studies included a small number of participants with a short follow-up, so large randomized controlled trials are still needed to prove the efficacy of both in coronary macrovascular disease. There is currently an ongoing randomized controlled trial on the long-term efficacy of DCB and DES in the treatment of neonatal CAD (NCT05101005). This trial is expected to further demonstrate the efficacy and safety of the DCB.
Application of DCBs in patients with large vascular disease complicated with diabetes
Diabetes mellitus is an independent risk factor for cardiovascular disease, and 25% of patients treated with percutaneous coronary intervention have comorbid diabetes mellitus. Compared with DES, DCB delivers antiproliferative drugs uniformly to the vessel wall and has superior efficacy to DES in patients with CAD and comorbid diabetes mellitus.54,55 In a prospective, multi-center, observational study 56 with 1156 patients (vessel diameter: 2–4 mm) treated with DCB that compared the use of DCB in patients with diabetes (n = 578) in nondiabetic patients (n = 578), differences in the target-lesion failure (2.77% vs. 5.36%, p = 0.025) and TLR (1.90% vs. 4.15%, p = 0.026) rates were observed in the non-diabetic patients after 1 year of follow-up, with a lower annual incidence. There were no significant differences in MACE (odds ratio (OR): 1.580, p = 0.100), cardiac deaths (OR: 1.608, p = 0.403), MI (OR: 4.042, p = 0.057), and any revascularization (OR: 1.534, p = 0.058). Although the patients with diabetes had higher rates of target-lesion failure and TLR after DCB angioplasty, there were no significant increases in the MACE risk, cardiac deaths, MI, or revascularization, and the above observational studies demonstrated that the efficacy of DCB was not lower in patients with diabetes mellitus than in those without. However, clinical evidence on the efficacy of DCB for coronary macroangiopathy in patients with diabetes is still lacking.
DCB in patients with high bleeding risk
In patients with atrial fibrillation and elderly patients, the incidence of bleeding complications in the first year after percutaneous coronary intervention ranges from 25%–40%, whereas post-percutaneous coronary intervention bleeding increases patient mortality and predisposes to other complications. 57 The use of DCBs in patients at high risk of bleeding reduces the duration of dual antiplatelet therapy use. 58 After 1 month of dual antiplatelet therapy, the incidence of MACE was significantly lower with DCB than with concomitant DES. 59 Ni et al. 60 included 76 patients with a high risk of bleeding and primary CAD, for whom the vessel diameter was ≥2.75 mm in 49 (64.5%) and ≥1 mm in 41 (51.2%). All were treated with paclitaxel-coated balloons (SeQuent Please). At 12 months, two (2.6%) patients died of MI and one (1.3%) developed TLR. A multi-center, single-blind, randomized controlled trial 61 enrolled 163 patients with increased bleeding who received a DCB (n = 81) or BMS (n = 82), of whom 84% had a vessel diameter ≥3.0 mm. At 9 months post-intervention, TLR (0% vs. 6%, p < 0.01) and MACE (2.5% vs. 13.4%, p = 0.01) rates were significantly lower with DCB than with BMS. These findings demonstrate that DCBs are superior to BMSs in efficacy and safety in patients with a high risk of bleeding. A randomized controlled trial 62 enrolled 40 patients at high risk of bleeding randomized to treatment with paclitaxel-coated balloons (n = 20) or BMSs (n = 20). There was no significant difference in postoperative fractional-flow reserve between paclitaxel-coated balloons and BMSs (0.87 ± 0. 06 vs. 0.89 ± 0.06, p = 0.254). At 9 months, the LLL was significantly lower with paclitaxel-coated balloons than with BMSs (0.2 ± 0.3 vs. 1.2 ± 0.8 mm, p < 0.001), and restenosis occurred only with BMSs (0% vs. 25.0%, p = 0.049). Therefore, in high-risk patients with CAD, DCB is significantly better than BMS. The use of DCBs in patients at high risk of bleeding can significantly reduce damage to the coronary arteries and significantly shorten dual antiplatelet therapy,58,63 thereby reducing the risk of bleeding.
Conclusion
DCB is a true intervention that requires no implantation. Compared with DES, DCB requires no foreign-body implantation, which reduces the intimal inflammatory response and thrombosis risk. The latest expert consensus indicates that DCB can be used for in-stent restenosis and small-vessel lesions, and the current clinical data prove that DCB is not inferior to DES in coronary macrovascular lesions, indicating that DCBs are safe and effective for large-vessel coronary disease. The use of DCB may reduce the duration of dual antiplatelet therapy, which is appropriate for patients with a high risk of bleeding associated with coronary macrovascular disease. Further, a DCB can reduce the inflammatory response of patients, and the coronary artery damage in patients is less than that of DES, which is suitable for patients with diabetes combined with CAD. In DCB treatment, treatment preparation and inflation will cause drug loss from the DCB, thus affecting its efficacy. Compared with DES, a more meticulous operation is required to retain the drugs in the DCB as much as possible to ensure that an adequate drug concentration is delivered to the lesion. At present, there is still a lack of clinical evidence on the role of DCB in coronary macrovascular disease, especially in complex cases with a high bleeding risk and diabetes, so further long-term, large-scale randomized trials are needed to prove the efficacy of DCB in coronary macrovascular disease.
Footnotes
Author contributions
All authors contributed to the study conception and design. Yong Cao and Cheng Shen had the idea for the article. Material preparation, data collection, and analysis were performed by Yong Cao, Cheng Shen, Lizhi Lin, Xiaorong Yin, Meng Chen, and Lu Cui. The first draft of the manuscript was written by Lizhi Lin, and all authors commented on previous versions of the manuscript. All authors have read and approved the final manuscript.
Data availability statement
Data availability is not applicable to this article as no new data were created or analyzed in this study.
Declaration of conflicting interests
The authors declare no conflicts of interest.
Funding
This study was supported by the Key R&D Plan Projects in Jining City (2022YXNS022 to Yong Cao and 2024YXNS068 to Cheng Shen) and Specialised clinical research programme for teams of attending physicians (ZZTD-2022-002 to Yong Cao).
