Abstract
Atherosclerotic cardiovascular disease is a major cause of morbidity and mortality in the Western world. Despite tremendous strides in understandings its pathogenesis, it still remains a challenge because of gaps in our understanding of its initiation, progression and complications leading to the clinical syndromes of angina, acute coronary syndrome, cerebrovascular disease and peripheral vascular disease. Recent studies have provided impetus on the shift from models of atherosclerosis based on cellular interactions to models where the important role of extracellular matrix is recognized. Proteoglycans, especially those belonging to the small leucine-rich proteoglycan family of which decorin is a representative example, have come under close scrutiny for their role in atherogenesis. There is evidence from
Introduction
Atherosclerotic cardiovascular disease is the most common cause of morbidity and mortality in the Western world [Roger et al. 2011]. Atherosclerosis is also the underlying pathology in a vast majority of cases of peripheral vascular disease and aortic aneurysms. Despite tremendous strides in understanding atherogenesis, precise steps involved in its initiation, progression and clinical outcomes are not fully understood. Commonly accepted theories of atherogenesis incorporate endothelial dysfunction, abnormal lipid retention and extrusion, oxidative stress and inflammation. All of these phenomena have caveats which do not explain all aspects of this complex malady. Further, these theories of atherogenesis are predominantly based either on the premise of active cellular interactions in the complex plaque milieu or passive role for extracellular matrix (ECM) components in lipid retention.
The ECM provides the immediate environment for different cell types in the vessel wall. By virtue of their close proximity and complex structure, different components of ECM play a critical role in endothelial, inflammatory and vascular smooth muscle cell (VSMC) migration, proliferation and apoptosis.
Extracellular matrix and proteoglycans
The ECM is composed of collagen fiber meshwork, dominated by type I and III fibers. Individual collagen molecules are linked to cell membrane via proteins, such as fibronectin, osteopontin and laminin. This layout of collagen and interconnecting proteins provides the structural framework for tissue organization. Another component of the ECM is proteoglycans which are complex molecules and were initially thought to be biologically inert and provide only space-filling function. Off late, their role in physiologic and pathophysiologic processes in the vessel wall is being increasingly recognized. In fact, the theory of lipid retention in the subendothelial space is dependent on interaction of lipids with the glycosaminoglycan side-chains of proteoglycans [Williams and Tabas, 1995]. The proteoglycan superfamily contains more than 30 well-characterized individual molecules [Iozzo, 1998]. As shown in Figure 1, a typical proteoglycan is composed of a protein molecule with domain structure to which a varying number of glycosaminoglycan chains of variable length are attached [Iozzo, 1998].

Decorin, a small leucine rich proteoglycan, has a 4-domain structure. Domain I is constituted by a signal peptide and propeptide chain. This is followed by domain II which is the cysteine-rich region and domain III which has leucine rich repeats and is primarily involved in binding and interacting with other protein molecules. Domain IV represents the carboxyl terminal of the protein and does not have well defined function.
Proteoglycans have been identified in all three layers of the vessel wall [Merrilees et al. 2001]. Different proteoglycans have predominance in different layers of the vessel wall. Apart from their role in lipid retention which is mediated via negatively charged glycosaminoglycan chains interacting with both native and oxidized-low density lipoproteins (ox-LDL), proteoglycans have been identified to play a significant role in in-stent restenosis, smooth muscle cell (SMC) proliferation and endothelial function. Among proteoglycans, members of the small leucine-rich proteoglycan (SLRP) family seem to play an especially important role in vascular biology. Decorin (OMIM ID 125255, DECORIN) is a type of SLRP whose role in the pathogenesis of both native and graft vessel atherosclerosis is being increasingly recognized [Yamakawa et al. 2000]. In this context, we review the role of decorin in the pathogenesis of atherosclerosis.
Structure of decorin
Decorin is a member of the class 1 SLRP family (Table 1) with a relative molecular mass (
Classification of proteoglycans.

proline/arginine-rich end leucine-rich repeat protein.
Decorin has glycosaminoglycan chains attached to the core protein which can be of either chondroitin or dermatan sulfate type. Each molecule of decorin can have either one or two chains of either of these glycosaminoglycans [Zamfir et al. 2011].
Decorin gene maps to the long arm of chromosome 12, between q21 and q23. It has a complex promoter region flanking exon1b. The promoter region has been shown to have tumor necrosis factor-alpha (TNF-α), activating protein-1 and transforming growth factor-beta (TGF-β) responsive elements. These relationships highlight the complex mechanisms involved in the regulation of transcriptional activity of decorin gene [Iozzo, 1998].
Distribution of decorin in normal and atherosclerotic arteries
Decorin is present in ECM in abundance and its role in relation to collagen fibrillogenesis has been well described [Reed and Iozzo, 2002]. Given the recent information on its role in modulating cellular function, it is believed that decorin may have a role in homeostasis beyond that of maintaining structural integrity of ECM [Seidler and Dreier, 2008; Iozzo and Schaefer, 2010].
In the arterial wall while versican and biglycan both types of chondroitin sulfate proteoglycan (CSPG) play a crucial role in lipid retention; decorin which is another type of CSPG, seems to have a smaller role in this process [Williams, 2001].
Merrilees and colleagues analyzed the distribution of decorin and vesican/biglycan in different blood vessels [Merrilees et al. 2001]; the normal human coronary, internal thoracic and radial arteries and saphenous veins. They observed that decorin is more prominent in the intima of internal thoracic and radial arteries as compared to that of the coronary artery and saphenous vein grafts. In contrast, versican and biglycan showed a strong staining in the intima of the coronary artery and saphenous veins as compared with light staining in radial and internal thoracic arteries. There was no significant difference in staining pattern of these three proteoglycans in the adventitia or media with usually strong staining for all three in adventitia and light staining in the media. Further, these authors observed in organ culture experiments a preferential expression of versican and biglycan in the intima of coronary artery and saphenous vein graft as compared with decorin in internal thoracic artery and fairly even and mild expression in case of radial artery [Merrilees et al. 2001]. This study provides a strong evidence for dichotomy regarding the predominance of proteoglycans in the intima, depending on the type of the blood vessel. It is of note that in studies of different types of blood vessels in humans there is a gradation in the risk of atherogenesis with the highest propensity in saphenous vein grafts and the least in the internal thoracic artery. This risk seems to correlate well with the pattern of proteoglycan distribution in the intima of vessel walls.
Radhakrishnamurthy and colleagues examined proteoglycan distribution in normal and atherosclerotic coronary arteries and identified low levels of decorin in the intima of normal coronary arteries [Radhakrishnamurthy et al. 1998], consistent with the findings of Merrilees and colleagues [Merrilees et al. 2001]. Interestingly, in this study decorin was found to be expressed in significant quantities in association with a fibrous cap of atherosclerotic lesions. This colocalization of decorin with fibrous cap region is consistent with the known collagen stabilizing function of this proteoglycan. Colocalization of decorin with fibrous regions was also observed in directional atherectomy samples obtained from both native atherosclerotic segments and in segments with restenosis following angioplasty [Riessen et al. 1994]. In addition to the fibrous areas of the plaque, decorin appears to colocalize with areas rich in profibrotic cytokines, TGF-β and platelet derived growth factor (PDGF), and macrophages, as suggested by Evanko and colleagues in a study of a nonhuman primate model of diet-induced atherosclerosis [Evanko et al. 1998]. The proximity of macrophages, profibrotic cytokines and decorin to fibrous areas of plaque suggests a close interplay between these cellular and ECM components. Similar observation of decorin colocalization with fibrous caps of the atherosclerotic lesions was made by Strom and colleagues in the ApoE/LDLr knockout mice model of accelerated atherosclerosis [Strom et al. 2004]. We have observed abundant expression of decorin in relation to a fibrous cap in atherosclerotic lesions of LDLr null mice fed a high-cholesterol diet (unpublished data). It is of note that these arteries express large amounts of collagen throughout the intima and media.
Kolodgie and colleagues analyzed proteoglycan distribution in coronary atherosclerotic lesions in patients with sudden coronary death [Kolodgie et al. 2002]. Lesions examined in this series were plaques that had ruptured, plaques with erosion, or plaques with atherostenosis involving more than 75% luminal narrowing. There was minimal expression of decorin in all these lesions; in contrast, there was abundance of versican and hyaluronan in lesions with plaque rupture or erosion. Based on this study, the absence of decorin in plaques may reflect a ‘vulnerable’ phenotype or advanced atherosclerotic lesion with a higher tendency for precipitation of acute coronary syndromes. Decorin has also been shown to result in a significant inhibition of neointimal hyperplasia, a precursor of advanced atherosclerosis in an
Decorin and in-stent restenosis
In-stent restenosis represents a frequent and a major problem in the clinical practice of interventional cardiology. Despite the availability of drug-eluting stents, the incidence of clinically significant in-stent restenosis is ~12% at 1 year [Cutlip et al. 2002]. Mechanistically in-stent restenosis represents tissue response to foreign body with proliferation of SMCs and accumulation of ECM and macrophages.
In a balloon angioplasty model, decorin overexpression was associated with the inhibition of PDGF receptor activation and significant reduction in collagen synthesis leading to decreased intimal hyperplasia [Nili et al. 2003]. In a study of human atherectomy specimens showing advanced in-stent restenosis, biglycan was the predominant proteoglycan with minimal expression of decorin which was colocalized within areas of collagen deposition [Glover et al. 2002]. This is similar to the findings in atherectomy specimens from native coronary arteries.
Decorin also seems to be associated with long-term modulation of intimal hyperplasia at the stent site. Farb and colleagues observed that as the time from percutaneous coronary intervention increased, there was an increase in decorin deposition associated with a change from type III to type I collagen, and a decrease in versican accumulation [Farb et al. 2004]. This remodeling was evident even in coronary arteries that underwent stenting more than 18 months ago. This finding suggests that in-stent restenosis may run a more indolent course than initially thought and that decorin may play an active role in modulating this process.
Decorin and atherogenesis
Decorin affects many aspects of atherogenesis. Here, we briefly discuss the steps in atherogenesis where decorin has been shown to have an important role. The biological effects of decorin on various aspects of atherosclerosis are summarized in Table 2.
Functions of decorin in relation to atherosclerosis.

TGF-β, transforming growth factor beta; PDGF, platelet derived growth factor.
Endothelial cell function
Normally functioning endothelium represents the first line of defense against inflammatory cell deposition. Upon repeated exposure to hemodynamic, physical, or chemical stress, endothelium becomes activated with expression of receptors for ox-LDL and decrease in nitric oxide availability/activity, and a stage for initiation of atherosclerosis sets in. Decorin seems to have functional dichotomy in relation to endothelial cell function and survival. Some investigators have observed potent anti-angiogenic effects of decorin in tumors [Grant et al. 2002], while others have shown it to participate in the maintenance of endothelial function; actually decorin may have a role in promoting angiogenesis in noncancerous states [Jarvelainen et al. 1992; Schonherr et al. 1999]. Decorin also seems to play a role in angiogenesis associated with inflammation [Nelimarkka et al. 2001].
Endothelial activation often results in apoptosis, a key event in early atherosclerosis. Endothelial apoptosis generally parallels endothelial dysfunction in the progression of atherosclerosis. In fact, endothelial apoptosis may lead to increased SMC proliferation via a paracrine loop [Raymond et al. 2004]. Decorin has been shown to inhibit endothelial apoptosis via both Akt-dependent and Akt-independent pathways [Schonherr et al. 2001, 2002]. Decorin-mediated Akt stimulation is related to phosphatidyl-inositol-3,4,5-triphosphate dependent kinase-1 activation, followed by increase in intracellular p21 and cyclin A. Stimulation of Akt activity also leads to inactivation of pro- apoptotic Bad. Decorin also induces an increase in p27, a cell cycle regulator, via Akt-independent pathway. These proposed mechanisms of action of decorin on endothelial cell biology and the effects on other cell types in the vessel wall involved in atherosclerosis are summarized in Figure 2.
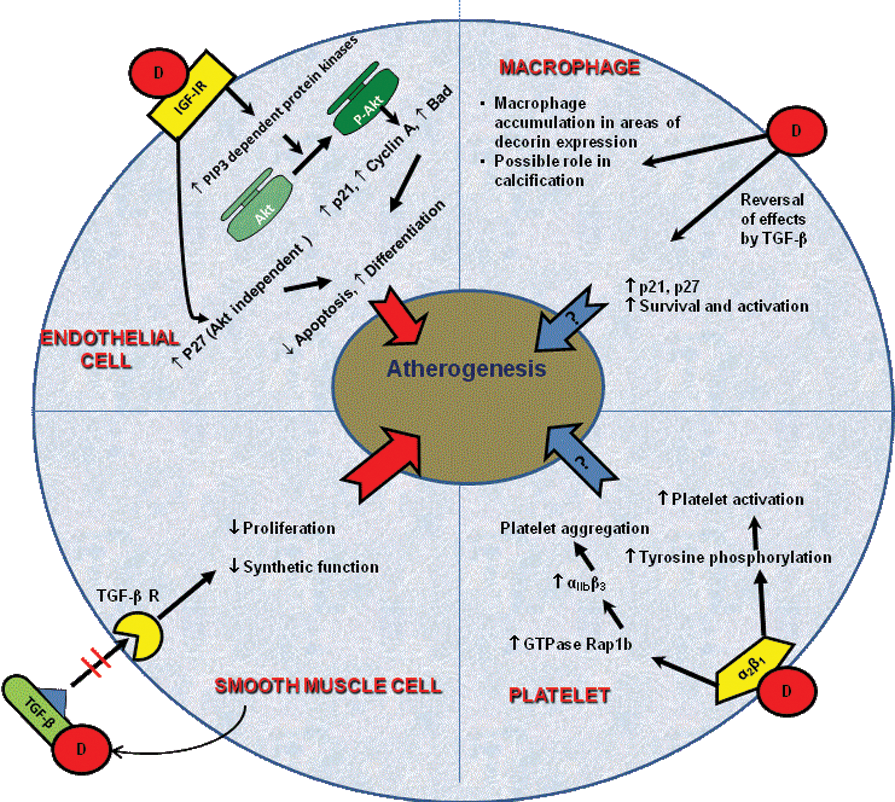
Decorin interacts with multiple cellular elements involved in atherogenesis. Abbreviations (in alphabetical order): Akt= protein kinase B; Bad= Bcl-2- associated death promoter; D= Decorin; IGF-1R= insulin like growth factor-1 receptor; M-CSF= macrophage colony stimulating factor; p21 and p27= cyclin dependent kinase inhibitor; SR-A = Scavenger receptor A; SMC= smooth muscle cells; TGF-β= transforming growth factor beta; TGF-β R= Transforming growth factor beta receptor; αIIbβ3 and α2β1 = surface integrins on platelets. (see text for details)
In contrast to the tumor cell lines where decorin binds to epidermal growth factor receptor (EGFR) and inhibits tyrosine kinase activity [Comalada et al. 2003] or causes depletion of these surface receptors by internalization [Zhu et al. 2005], it has been known for a while that endothelial cells interact with decorin in EGFR-independent fashion [Schonherr et al. 2002]. Recent work has demonstrated the putative receptor for decorin on endothelial cells to be insulin-like growth factor receptor-1 [Schonherr et al. 2005]. Decorin seems to regulate endothelial cell behavior by binding to this receptor. Decorin also affects endothelial cell motility by interaction with α2β1 integrin on collagen matrix [Fiedler et al. 2008].
Vascular smooth muscle cells and macrophages
SMC migration, proliferation and synthetic function play key roles in the evolution of atherosclerosis and in-stent restenosis lesions. This is true both for native arterial and grafted venous conduits. SMCs contribute significantly to intimal hyperplasia, a precursor of advanced atherosclerotic lesions. Fischer and colleagues demonstrated that the effect of decorin on SMCs is mediated via inhibition of TGF-β activity and leading to decreased proliferation and less synthetic activity of these cells [Fischer et al. 2001]. There is conflicting data regarding the role of decorin in inhibiting PDGF-related SMC proliferation [Nili et al. 2003; Fischer et al. 2001]. Further, arterial SMCs express more decorin as compared with venous SMCs [Franch et al. 2006]. This difference may be linked to increased propensity of venous SMCs for proliferation and synthetic (collagen and collagenases) function [Wong et al. 2005].
Decorin synthesis by SMCs is regulated at multiple levels. There is evidence to suggest role for farnesoid X receptor (FXR), a nuclear receptor, in this regulation. Activation of FXR increases decorin synthesis at the transcriptional level [He et al. 2008]. Interestingly activation of FXR is also associated with inhibition of SMC migration and proliferation. Decorin synthesis is also upregulated in SMC by exposure to adiponectin [Yamakawa et al. 2000]. Since adiponectin has some anti-atherogenic activity, it is plausible that the stimulation of decorin synthesis is one mechanism by which adiponectin exerts its anti-atherogenic effect.
Mechanical strain has been shown to directly affect the proteoglycan composition of vessel wall with resultant decrease in synthesis of decorin while versican and biglycan are synthesized in larger quantities by SMCs [Lee et al. 2001]. Disease states such as hypertension are associated with increased mechanical strain, suggesting that decreased decorin synthesis may be yet another unrecognized step linking hypertension to atherogenesis.
Decorin also seems to have a role in vascular calcification as it localizes to the areas of calcification and increases calcium deposition in cultured bovine arterial SMCs [Fischer et al. 2004]. The effect of decorin on the mineralization ability of SMCs in response to oxidative stress, as induced in the presence of ox-LDL, has been shown to be related to glycosaminoglycan chains attached to decorin core protein [Yan et al. 2011]. Further, macrophages colocalize within the areas of decorin expression [Evanko et al. 1998] and calcium deposition in the plaque milieu [Jeziorska et al. 1998]. The presence of macrophages seems to be closely linked to vascular calcification as interleukin-1 produced from these cells leads to increased decorin secretion by SMCs [Edwards et al. 1994].
Decorin colocalizes with macrophages in both atherosclerotic plaques and lesions of in-stent restenosis [Yamakawa et al. 2000; Evanko et al. 1998]. Decorin expression is associated with increased survival of macrophages mediated via induction of p21 and p27 and decreased proliferation related to inhibition of macrophage colony stimulating factor [Xaus et al. 2001]. Decorin has also been shown to be associated with macrophage activation, which is related to blockade of the inhibitory effects of TGF-β on these cells [Comalada et al. 2003].
Decorin and platelets
In addition to a critical role in the formation of occlusive thrombi in plaques with erosion or rupture, activation of platelets is thought to be related to progression of atherosclerosis by variety of mechanisms [Weber, 2005]. Guidetti and colleagues in
Decorin and scavenger receptors
Scavenger receptors play a critical role in the progression of atherosclerosis, especially given their role in the uptake of ox-LDL [Pluddemann et al. 2007]. Among the scavenger receptors class E molecule lectin like ox-LDL receptor-1 (LOX-1) seems to play an important role in atherogenesis as it is involved in multiple facets of atherosclerosis [Mehta et al. 2006]. However, little is known about the interaction between decorin and different types of scavenger receptors. In one study, scavenger receptor type A was shown to act as a cellular receptor for decorin, resulting in increased adhesion of macrophages to ECM [Santiago-Garcia et al. 2003]. How this finding and potential interaction of decorin with other scavenger receptors translates into affects on atherosclerosis is not known as yet.
Role of decorin as evident in animal models of atherosclerosis
Danielson and colleagues first demonstrated increased fragility and decreased tensile strength of skin in decorin null mice [Danielson et al. 1997]. Further studies in decorin null mice suggest its role in preventing tumor progression and limiting abnormal tissue fibrotic response [Ameye and Young, 2002]. There is as yet no information on the effect(s) of decorin deficiency in relation to changes in vessel wall and its effect on progression of atherogenesis, although work in a model of corneal injury in decorin null mice suggests a role for decorin in angiogenesis [Schonherr et al. 2004]. Al Haj Zen and colleagues addressed the issue of whether decorin is just a marker of extent of atherosclerosis or if it plays an active role in inhibition of atherogenesis [Al Haj Zen et al. 2006]. In their study, systemic decorin overexpression resulted in a significant decrease in atherosclerotic plaque formation in Apo-E null mice. This was associated with a decrease in macrophage and collagen accumulation and decrease in gelatinase activity. In addition, serum triglyceride levels were also decreased suggesting a possible role for systemic decorin expression in the modulation of lipid profile.
Perspective
Very few cases of decorin deficiency have been described in humans. In a case described by Gotte and Kresse, decorin deficiency was associated with features of accelerated ageing (progeria) [Gotte and Kresse, 2005]. Rare cases of Marfan’s [Pulkkinen et al. 1990] and Ehler Danlos [Wu et al. 2001] phenotype have also been described in association with decorin deficiency. These patients manifest increased skin elasticity and corneal opacities, consistent with the known effects of decorin on collagen fibrillogenesis. While these cases highlight importance of decorin in the organization of ECM, all these patients were too young to have developed manifestations of atherosclerosis, making it difficult to predict how decorin deficiency might alter the course of atherosclerosis. However, based on the data discussed here, decorin or decorin-based molecules may prove to be promising therapeutic agents for altering the course of atherosclerosis and preventing its complications.
Overall, we hypothesize that decorin overexpression may result in a plaque that is ‘more stable’ and less likely to undergo rupture and erosion. In fact, decorin may prove to be a starter given that related molecules of SLRP family, such as fibromodulin, have been shown to inhibit neo-intimal hyperplasia in saphenous venous grafts [Ranjzad et al. 2009]. Conceptually, local delivery of decorin at the site of coronary stenting (analogous to delivery of antimitotic drugs to prevent in-stent restenosis) may prove to be useful at it will not only decrease the risk of restenosis, but also decrease the risk of in-stent thrombosis by promoting endothelial healing.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
The authors declare no conflicts of interest in preparing this article.
