Abstract
Background:
Elevated lipoprotein(a) [Lp(a)] is a common hyperlipidaemic condition with strong genetic predisposition and is independently associated with ischaemic heart disease (IHD). A Mendelian randomisation study has suggested that elevated Lp(a) is likely to confer similar causal risks as heterozygous familial hypercholesterolemia for premature IHD. We aimed to characterise the clinical profiles of admitted patients with IHD with at least one Lp(a) measurement. We also investigated whether elevated Lp(a) concentration was associated with premature onset of IHD.
Methods:
This is a descriptive, non-interventional, retrospective study with data from a single tertiary hospital IHD Lp(a) cohort in Singapore, which consecutively recruited 521 patients with IHD admitted to the hospital.
Results:
A total of 82.2% were men, 46.6% had newly diagnosed IHD and 10% had premature IHD. The median Lp(a) levels was 35.2 nmol/L. 70.8% of patients had normal Lp(a) concentrations (<70 nmol/L), 13.4% of people with Lp(a) ⩾ 70 to <120 nmol/L and 15.7% of patients with Lp(a) ⩾ 120 nmol/L. Lp(a) distribution was positively skewed to the right for all ethnicities. Patients of Indian ethnicity and of female gender had higher levels of Lp(a) compared with other ethnicities and gender, respectively. Multivariable regression analysis identified Lp(a) ⩾ 155 mmol/L to be associated with development of premature IHD (OR = 2.90, 95% CI: 1.26–6.67, p = 0.012).
Conclusion:
There exist differences in Lp(a) distribution across ethnicities and gender. The subgroup analysis suggests that Lp(a) ⩾ 155 mmol/L was associated with premature onset of IHD.
Keywords
Introduction
Lipoprotein(a) [Lp(a)] is an independent causal risk factor for ischaemic heart disease (IHD), calcific aortic valve stenosis, ischaemic stroke and peripheral artery disease, as shown in various epidemiologic and Mendelian randomisation studies.1,2 A meta-analysis of statin outcome trials reported that for individuals with Lp(a) levels more than 50 mg/dL (approximately 120 nmol/L), the elevated baseline Lp(a) and on-statin Lp(a) levels were both independently and linearly related to risk of cardiovascular disease with an increased risk of 31% and 43%, respectively. 3 Lp(a) is an atherogenic particle comprising of an apolipoprotein(a) [apo(a)] component bound to apolipoprotein B100 [apo B100] via one disulfide bridge.1,2 The Lp(a)-associated risk of atherosclerotic cardiovascular disease (ASCVD) depends on the degree of elevation of Lp(a); extremely elevated Lp(a) levels (>115 mg/dL or ⩾290 nmol/L) were associated with a 2.5-fold increased risk for myocardial infarction in a large prospective Danish general population study. 4 An approximate one-fifth of the world population has elevated blood Lp(a) concentrations that increase the risks of ASCVD and cardiovascular events. 5 The distribution of Lp(a) levels and apo(a) isoform size is known to vary considerably between different ethnicities.1,6
Among patients with IHD, patients with premature-onset IHD are at highest cardiovascular risk with higher rates of recurrent myocardial infarction, increased morbidity, shortened life expectancy and increased healthcare economic burden. 7 In the United States, 20% of all heart disease-related deaths in from 1999 to 2018 were in individuals less than 65 years old. 8 Moreover, in a US registry of 2097 individuals with myocardial infarction before or at 50 years old, 20% had myocardial infarction less than 40 years old. 9 Thus, addressing risk factors in individuals in the young adulthood is critically important to reduce premature onset of ASCVD and associated complications, including elevated Lp(a).7,10 Elevated Lp(a) is an important risk factor for premature IHD that is underdiagnosed.11,12 A Mendelian randomisation study has suggested that elevated serum Lp(a) levels above 430 nmol/L confer a similar causal risk for premature IHD as with heterozygous familial hypercholesterolemia (FH). 13 Hence, European Society of Cardiology and European Atherosclerosis Society (EAS) Guidelines (2019) followed by multiple other guidelines have since recommended that all adults should have their Lp(a) measured at least once in their lifetime.2,5,14 There are still knowledge gaps in the field of Lp(a) including the need to clarify at what threshold Lp(a) confers to additional risk to premature onset of IHD especially in Asian people.
To understand the clinical significance of elevated Lp(a) in Asian patients to inform effective implementation strategies related to Lp(a) management, it is important to study the differences in Lp(a) distribution among patients of different ethnicities, clinical subtypes of IHD and demographics. In this study, we investigated the distribution of serum Lp(a) in a multi-ethnic cohort of patients with IHD in Singapore and its association with other demographic and clinical characteristics. We also investigated the association of elevated Lp(a) with premature onset of IHD.
Methods
This is a descriptive, non-interventional, retrospective cohort study utilising data obtained from an investigator-led study, the Changi General Hospital IHD Lp(a) database, which recruited patients admitted to the cardiology wards from June to December 2020 with IHD. 11 Patient recruitment was based on electronic medical records with a diagnosis of IHD or if there was presence of coronary artery disease or ischaemic cardiomyopathy or coronary artery disease requiring coronary stent intervention or coronary artery bypass graft. Information on the age of diagnosis of IHD and presence of comorbidities at the time of initial diagnosis of IHD was obtained from history taking and their electronic medical records. 11 We defined premature IHD diagnosed <45 years for male, and <50 years for women.15,16 Family history of IHD refers to family history of IHD of any age.17,18
The demographic, clinical and lipid profiles of patients based on four pre-specified non-mutually exclusive subtypes of IHD were described. Myocardial infarction refers to the subtype of patients with a history of myocardial infarction or at time of recruitment. Unstable angina refers to the subtype of patients without history of myocardial infarction, but who had unstable angina. Post-revascularisation refers to the subtype of patients without history of myocardial infarction but had percutaneous revascularisation before. Stable angina subtype refers to the subtype of patients with history of stable angina or minor coronary disease and did not have history of acute myocardial infarction, percutaneous revascularisation or unstable angina before. Serum Lp(a) concentrations were measured in the hospital laboratory using particle-enhanced turbidimetric immunoassay with Tina-quant Lp(a) Gen.2 (Latex) Roche, with an inter-assay coefficient of variation of ⩽2.2%. The diagnosis of hyperlipidaemia were taken from electronic medical records as either hyperlipidaemia, dyslipidaemia and/or hypercholesterolaemia. Based on our previous report, we defined the condition of elevated Lp(a) or ‘hyper-Lp(a)’ as Lp(a) ⩾ 120 nmol/L, while the ‘grey zone’ was ⩾70 to <120 nmol/L. 19 Notably, various guidelines have adopted different thresholds for Lp(a), 5 with most guidelines adopting the EAS’s threshold using Lp(a) ⩾ 125 nmol/L as elevated Lp(a) and defining a grey zone’ of cardiovascular risk between ⩾75 and <125 nmol/L. 2 Thus, we also briefly describe the results according to EAS threshold classification in the results section.
Statistical analyses were performed using STATA version 16.1 StataCorp LLC. Continuous variables were expressed as medians with interquartile ranges (IQRs), as well as mean and standard deviation, median with IQRs (minimum and maximum). Categorial variables were expressed in percentages and number of observations (n). Chi-square test for categorical variables, t test for comparison of means and Mann–Whitney U test for comparison of medians (for continuous variables) were used to investigate the associations of demographic and clinical characteristics between the groups. As the distributions of Lp(a) are significantly skewed, Lp(a) were loge-transformed to normalise their distributions for linear regression analysis. In assessing the relationship between Lp(a) and onset of premature IHD, the multivariate regression model used was adjusted for gender, ethnicity, diabetes mellitus, hypertension, hyperlipidaemia and smoking status at the time of IHD diagnosis. Of note, in this subanalysis, comorbidities at the time of IHD diagnosis (instead of at time of study recruitment) were taken into consideration as comorbidities of some patients only occurred after IHD diagnosis and, hence, not attributable to the onset of IHD in some. The correlation between the Lp(a) and low-density lipoprotein-cholesterol (LDL-C) levels was analysed using Spearman’s rank correlations.
Results
Baseline characteristics
The demographics, clinical and lipid profiles of patients are detailed in Table 1. Among the 521 study participants, 82.2% were of male gender. The median age at study enrolment was 63.4 years (IQR: 56.0–71.0) and the median age of onset of IHD was 59.2 years (IQR: 51.3–66.8). A total of 46.6% of the study participants were newly diagnosed with IHD. Our multi-ethnic cohort included patients of Chinese (49.3%), Malay (31.3%), Indian (12.7%) and other ethnicities (6.7%). Common cardiovascular comorbidities present at point of enrolment to study included hyperlipidaemia (91%), hypertension (78.9%) and diabetes mellitus (48.5%). Of the patients, 73.1% had myocardial infarction, 7.6% had unstable angina without myocardial infarction, 64.5% underwent percutaneous revascularisation before and 14.2% did not have history of acute myocardial infarction, prior percutaneous revascularisation or unstable angina. This cohort did not have patients who had a baseline diagnosis of FH from the electronic database or during patient interview at study recruitment, but this is likely due to underdiagnosis as suggested by the elevated total cholesterol or LDL-C present in a few patients; Table 1 shows that the maximum total cholesterol was 11.83 mmol/L, and LDL-C was 9.86 mmol/L.
Baseline demographics and clinical characteristics of patients based on the clinical subtypes (not mutually exclusive) of ischaemic heart disease.

Comorbidities listed in this table are at time of study enrolment. For continuous variables, mean (standard deviation), median and range (minimum to maximum) are displayed.
CABG, coronary artery bypass grafting; COPD, chronic obstructive pulmonary disease; FH, familial hypercholesterolaemia; IQR, interquartile range; Lp(a), lipoprotein(a); SD, standard deviation.
Lp(a) distribution
The median Lp(a) level was 35.2 nmol/L in our study group (Tables 1 and 2; Figure 1). A total of 70.8% of patients had normal Lp(a) concentrations (<70 nmol/L), and 72.9% with Lp(a) < 75 nmol/L. There were 15.7% of patients with Lp(a) ⩾ 120 nmol/L and 14.6% with Lp(a) ⩾ 125 nmol/L. There were 13.4% of people with Lp(a) concentrations within the ‘grey zone’ of ⩾70 to <120 nmol/L, or 12.5% with Lp(a) ⩾ 75 to <125 nmol/L. There were 3.5% patients with Lp(a) between 150 and 170 nmol/L and 7.9% with Lp(a) ⩾ 170 nmol/L. At the 90th and 95th percentiles, Lp(a) concentrations were 155 and 195 nmol/L, respectively. The Lp(a) distribution was positively skewed to the right for all ethnicities (Figure 1). Across ethnicities, patients of Indian ethnicity had the highest median Lp(a) levels (Table 2; Figure 2). Using regression analysis with loge-transformed Lp(a) as dependent variable, when compared with Chinese ethnicity, the Indian ethnicity had higher Lp(a) (p = 0.031, β = 0.33, 95% CI: 0.03–0.62), and this remained positively associated when adjusted for gender (p = 0.029, β = 0.33, 95% CI: 0.03–0.62). Females also had significantly higher median Lp(a) levels than males (44.2 mmol/L vs 34.2 mmol/L, p = 0.022, β = 0.29, 95% CI: 0.04–0.53) on univariable regression analysis, shown in Table 2 and Figure 1.
Lp(a) distribution by ethnicity and gender.

IHD, ischaemic heart disease; IQR, interquartile range; Lp(a), lipoprotein(a).

Distribution of Lp(a) in nmol/L stratified by (a) ethnicity and (b) gender.

Violin plots of log-transformed Lp(a) levels across the three major ethnicities in patients with and without premature onset of IHD.
Medications
At study recruitment, 67.3% of patients were on statin (Table 3). There were no patients on agents of PSCK9 inhibition which includes evolocumab, alirocumab and inclisiran (Table 3). Only 7.7% of patients were on ezetimibe at the point of recruitment while 6.0% of patients were on fibrates. None of the patients were on niacin, hormone replacement therapy or fibrinolytic therapy. 46% of patients on statin had elevated LDL-C ⩾ 2.1 mmol/L, whereas 40% of patients on ezetimibe had LDL-C ⩾ 2.1 mmol/L. There was a smaller proportion of patients with statin that had elevated LDL-C ⩾ 2.1 mmol/L when compared with patients with LDL-C < 2.1 mmol/L (p < 0.001).
Medications of patients at the point of recruitment to study for all patients and by clinical subtypes (not mutually exclusive) of ischaemic heart disease.

ACEIs or ARB refers to angiotensin converting enzyme inhibitors and angiotensin receptor blockers. PCSK9i refers to agents that inhibit the proprotein convertase subtilisin/kexin type 9.
Correlation between LDL-C and Lp(a)
By Spearman’s rank correlation, the Spearman’s rho correlation between LDL-C levels with Lp(a) levels was 0.1768, p < 0.001. On stratification by LDL-C levels, the correlation between LDL-C and Lp(a) were 0.1–0.25 (LDL-C < 1.8 mmol/L, Spearman’s rho = 0.216, p = 0.0121; LDL-C < 2.1 mmol/L, Spearman’s rho = 0.256, p = 0.0002; LDL-C < 2.6 mmol/L, Spearman’s rho = 0.1577, p = 0.0074; LDL-C ⩾ 2.6 mmol/L, Spearman’s rho = 0.1280, p = 0.0119).
Lp(a) and Premature IHD
Ten percent of the study participants had a premature onset of IHD (Table 4, Figure 2). There was a higher proportion of patients with Lp(a) levels ⩾155 nmol/L in the premature IHD group when compared with non-premature IHD group (19.2%; vs 9.8%, p = 0.037). On univariate analysis, Lp(a) ⩾ 155 nmol/L (90th percentile), was associated with significantly increased risks of premature IHD (OR = 2.19, 95% CI: 1.03–4.65, p = 0.042). This relationship was also preserved on multivariable analysis (OR = 2.90, 95% CI: 1.26–6.67, p = 0.012) on adjustment for gender, ethnicity and comorbidities at time of diagnosis of IHD (diabetes mellitus, hypertension, hyperlipidaemia, smoking). Among the 52 patients with premature IHD, 10 patients (19.2%) had elevated Lp(a) levels of ⩾155 nmol/L; Among these 10 patients, all had hypertension, 6 of them had elevated LDL-C > 2.6 mmol/L, 5 patients had diabetes and 5 patients were present or past cigarette smokers.
Clinical characteristics of the study participants based on the age at onset of ischaemic heart disease (non-premature vs premature IHD).
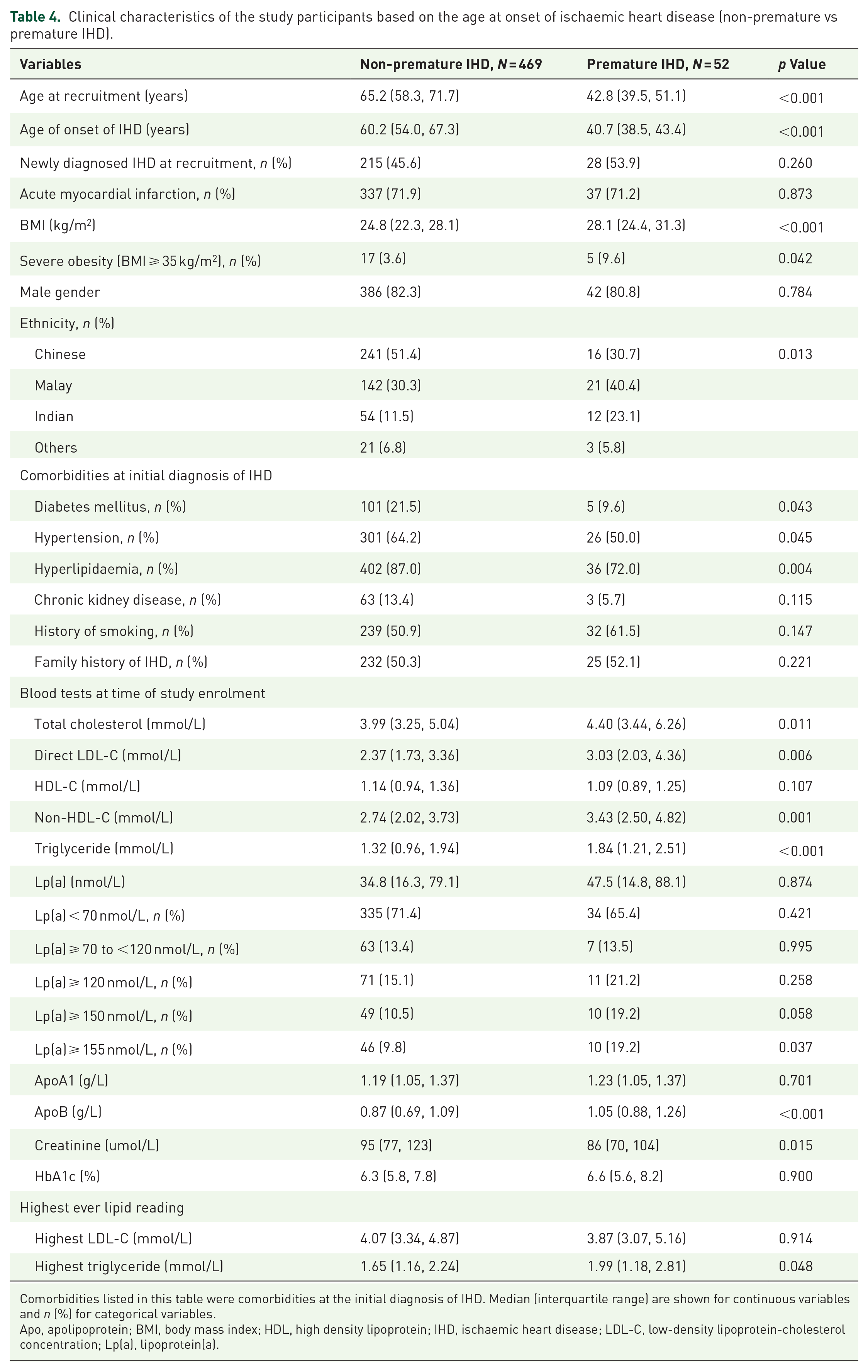
Comorbidities listed in this table were comorbidities at the initial diagnosis of IHD. Median (interquartile range) are shown for continuous variables and n (%) for categorical variables.
Apo, apolipoprotein; BMI, body mass index; HDL, high density lipoprotein; IHD, ischaemic heart disease; LDL-C, low-density lipoprotein-cholesterol concentration; Lp(a), lipoprotein(a).
Patients with premature IHD when compared with those without premature onset of IHD also had higher median BMI (28.1 kg/m2 vs 24.8 kg/m2, p < 0.001) and BMI ⩾ 35 kg/m2 (9.6% vs 3.6%, p = 0.042). The premature IHD group had lower cardiovascular comorbidities of hyperlipidaemia (72.0% vs 87.0%, p = 0.004), hypertension (50.0% vs 64.2%, p = 0.045) and diabetes mellitus (9.6% vs 21.5%, p = 0.043) at the time of diagnosis of IHD. However, premature IHD group compared with non-premature IHD group, had a higher median total cholesterol (4.40 mmol/L vs 3.99 mmol/L, p = 0.011), LDL-C (3.03 mmol/L vs 2.37 mmol/L, p = 0.006), triglyceride (1.84 mmol/L vs 1.32 mmol/L, p < 0.001) and apoB levels (1.05 g/L vs 0.87 g/L, p < 0.001), Table 4.
Discussion
In this cross-sectional study, we reported the differences which exist in Lp(a) distribution in our multi-ethnic Asian cohort by ethnicity and gender, with individuals of Indian ethnicity having the highest Lp(a) level followed by Malays and Chinese. Similar to published medical literature, women had higher median Lp(a) levels than men. 2 In our study, 15.7% of patients with IHD admitted to hospital had elevated Lp(a) ⩾ 120 nmol/L. Extremely elevated Lp(a) ⩾ 170 nmol/L affected 7.9% of the study cohort, and this group of patients are potential candidates for future Lp(a) lowering therapies pending the outcome of the ongoing phase II and III trials RNA-directed therapeutics targeting Lp(a). 1 In our cohort, there were 12%–14% within the grey zone. A recent retrospective study of 609 individuals showed that re-testing Lp(a) of individuals in the grey zone territory (30–50 mg/dL or approximately 75–125 nmol/L) did not help to reclassify patients already in high-risk category. 20 However, re-testing Lp(a) in individuals re-categorised 33% of individuals from intermediate category to high risk and re-categorised 20% individuals to low-risk categories. 20 While our cohort were already at very high cardiovascular risk category, a repeat Lp(a) testing few months later for patients with Lp(a) within grey zones is beneficial, as Lp(a) may be falsely decreased or increased during acute events. 21
Multiple studies have consistently shown that the prevalence of Lp(a) is greatly affected by ethnicity. In the Interheart study which enrolled participants from seven different ethnicities, the Lp(a) concentrations were lowest in Chinese people, and highest in Africans. 22 In the study of UK Biobank, the Lp(a) levels were highest in Black, followed in descending order by South Asian, White and lowest in Chinese. 23 Similar to our recently published study of 2025 individuals undergoing coronary angiogram in Singapore, our current study comprising hospitalised patients with IHD reported that patients of Indian ethnicity had higher Lp(a) levels compared with patients of Chinese and Malay ethnicities. 19 These differences in Lp(a) levels were thought to be related to the variation in apo(a) isoform sizes or Kringle-IV repeat polymorphism and genetic variants within the LPA locus and APOE genotypes.22,24 The ethnic difference may also explains the observation that the 90th percentile in this Asian study correlated to 155 nmol/L, and 95th percentile correlated to 195 nmol/L, but these percentiles corresponded to higher Lp(a) levels in other cohorts. In the UK Biobank study, the 90th and 95th percentiles in Whites were 168 and 221 nmol/L, respectively, whereas in Blacks, the 90th percentile was 212 nmol/L and 95th percentile was 263 nmol/L.2,23
Genetic studies suggest that elevated Lp(a) may be one of the most potent genetic risk factor of ASCVD and calcific aortic valve stenosis. 25 Lp(a) exhibits atherogenic, inflammatory and procalcific properties, partly because of the oxidised phospholipids carried by Lp(a).1,2 Similar to oxidised LDL-C, Lp(a) has inflammatory properties promoting the chemotaxis of monocytes and inducing the expression of vascular adhesion molecules, and promotes atherosclerotic plaque formation. 26 However, unlike LDL-C concentrations which can be modified by lifestyle factors including diet, Lp(a) concentrations are predominantly genetically determined with a small variable effect from diet and currently available lipid-lowering medications.1,27 Statins may be associated with a 10%–20% increase of Lp(a) levels in susceptible people as shown in one meta-analysis 28 ; however, this observation was not present in another meta-analysis. 29
We also found that elevated Lp(a) above 155 mmol/L was associated with the premature onset of IHD, although this was only a subgroup analysis and of small sample size; hence, further larger studies are warranted to confirm our findings. Interestingly, this concentration was similar to the threshold to define elevated Lp(a) used in a study of UK Biobank which showed that Lp(a) ⩾ 150 nmol/L was associated with hazard ratio of 1.50 and 1.16 in the contexts of primary and secondary prevention. 23 We noticed that all patients with premature onset of IHD as well as elevated Lp(a) levels of ⩾ 155 nmol/L had another cardiovascular risk factor such as hypertension, diabetes mellitus, hyperlipidaemia or smoking. Our observation suggests that even moderately elevated Lp(a) concentrations above 155 nmol in Southeast Asians was associated with premature onset of IHD particularly when other cardiovascular risk factors are present, contributing to an elevated overall cardiovascular risk for an individual.
Elevated Lp(a) is clearly underdiagnosed in Singapore. 11 Prior to the onset of this study, Lp(a) was not routinely measured even in high-risk cardiovascular patients with strong family history of premature IHD. For decades worldwide and especially regionally, Lp(a) was the ‘forgotten’ biomarker due to lack of awareness of this condition among doctors and other healthcare professionals. This is despite elevated Lp(a) being identified as an early risk factor for ASCVD, with a population cohort study demonstrating that youths with elevated Lp(a) having 2.5 times greater risk of developing adult ASCVD. 30 On a similar note, the Lp(a) Heritage study reported that only 13% of patients with established ASCVD ever had prior Lp(a) measured. 31 Through our study, we hope to highlight the need for regular Lp(a) measurement in all patients at high risk of IHD. FH is strongly associated with premature IHD, and has a high prevalence of 1 in 250 people in the general population. 24 The prevalence of elevated Lp(a) of world’s general population is high at 15%–20%.6,24,32 Individuals with both FH and Lp(a) has the highest risk of ASCVD event and mortality than presence of either alone. 24 Therefore, Lp(a) should be given at least equal importance in detection strategies in Asia.
The limitation of our study is that data were cross-sectional from a single centre. However, being one of the largest local hospitals and the only public hospital serving Singapore’s Eastern region, accounting for approximately 20% of the country’s total population, our data are likely representative of the regional population of patients with IHD which has a much higher prevalence of cigarette smoking, poor diet and obesity compared with the general population in Singapore. 11
The strengths of our study included a broad analysis of risk factors associated with IHD in a Southeast Asian cohort. Moreover, given that Lp(a) levels are dependent on isoform size, our study had the advantage of uniformly measuring Lp(a) using an isoform-insensitive assay in molar concentrations. 1 In conclusion, our study reported that there Indian ethnicity had the highest median Lp(a) concentration among the other major ethnicities, while female patients had higher median Lp(a) than male patients. Our sub-analysis study suggested that elevated Lp(a) at threshold ⩾155 nmol/L was associated with premature onset of IHD in our study participants.
