Abstract
Background:
Chronic obstructive pulmonary disease (COPD) is a leading global health burden, with high prevalence of comorbidities (e.g., hypertension and diabetes) that further increase healthcare utilization and mortality. Integrated care is proposed as a potential management strategy for COPD patients with comorbidities, but its overall effects remain unclear due to inconsistent evidence from prior studies.
Objectives:
To systematically evaluate the effects of integrated care on key health outcomes in patients with COPD and at least one comorbidity.
Design:
Systematic review and meta-analysis.
Data sources and methods:
Databases including MEDLINE, EMBASE, CENTRAL, CINAHL, and ClinicalTrial.gov were searched. Eligible studies were randomized controlled trials (RCTs) evaluating integrated care in patients with COPD and comorbidities. Two independent reviewers conducted study screening, data extraction, and quality assessment. Effects of integrated care were assessed using a random-effects model.
Results:
Seven RCTs from high-income countries were included. Common integrated care components were health education, self-management support, and (in two studies) telemonitoring. Meta-analysis showed that integrated care significantly reduced the number of COPD exacerbations and all-cause hospitalizations. No significant effects were observed for all-cause emergency visits or CAT scores.
Conclusion:
Integrated care effectively reduces COPD exacerbations and all-cause hospitalizations in patients with COPD and comorbidities, supporting its clinical value. However, high heterogeneity across studies, limited generalizability to non-high-income countries (e.g., China), and lack of impact on patient-reported outcomes (CAT scores) highlight the need for further localized research.
Trial registration:
Registered with PROSPERO, Registration ID: CRD420251170533.
Plain language summary
Chronic obstructive pulmonary disease (COPD), a serious lung condition affecting millions worldwide, often occurs alongside other health problems (e.g., heart disease, diabetes). These additional conditions can worsen outcomes, increasing hospital stays and even death. While “integrated care” (combining treatments, education, and long-term support) is often recommended for such patients, its benefits need clearer evidence. This study reviewed seven high-quality clinical trials from wealthier countries to evaluate how integrated care affects COPD patients with other health issues. Most programs included education, self-management training, and remote health monitoring. Key findings: Integrated care reduced sudden worsening of COPD symptoms (“exacerbations”). It also lowered the chance of being hospitalized for any health reason. However, it did not significantly reduce emergency room visits or improve patients’ self-reported symptom scores (CAT scores). These results suggest that integrated care can help stabilize COPD patients and reduce hospital stays, making it a valuable approach for healthcare systems. However, more research is needed to adapt these programs for low-income regions and to improve their impact on quality of life.
Summary box
It is estimated that 44.7% of COPD patients over 40 years old are suffering from at least one other disease, and 51% of the excessive medical burden for COPD patients could be attributed to comorbidities. A favorable way to improve the management of comorbid patients is integrated care. Previous studies, however, have not analyzed the effects of integrated care on patients with COPD and other comorbidities. This study fills this gap and shows that integrated care significantly reduced the number of exacerbations and all-cause hospitalization rates, underscoring the value of integrated care in managing comorbid COPD patients.
Introduction
Chronic obstructive pulmonary disease (COPD) is one of the leading health issues worldwide for the elderly. It affected over 200 million people and claimed over 3 million lives in 2019, ranking among the sixth leading causes of death in the global burden of disease.1,2 COPD affects about 3.2% of China’s total population and causes over 1 million deaths, making it the third leading disease burden. 2 The actual disease burden, however, might be much higher due to the low detection rate in China. 3 A population study in China revealed that the actual prevalence is up to 8.6% in the population over 20 years old, namely, about 100 million people are affected. 3
Due to common risk factors and pathogenic mechanisms, most COPD patients have multiple comorbidities, including hypertension, heart failure, osteoporosis, and diabetes, which not only accelerate the progression of COPD but also are themselves affected by COPD exacerbations.4–7 In China, it is estimated that 44.7% of COPD patients over 40 years old are suffering from at least one other disease.6,8–10 In addition, previous studies showed that 51% of the excessive medical burden for COPD patients could be attributed to comorbidities, and most patients died of comorbidities. 11 Given that COPD itself is a disease with multiple disease components and most patients have at least one comorbidity, treatment of patients requires the combination of different elements, which is integrated care.
Integrated care cannot be narrowly defined, but should be seen as an overarching term for a broad and multicomponent set of ideas and principles that seeks to better coordinate care around people’s needs. 12 According to different forms and purposes, integrated care can be further divided into five types: horizontal integration, vertical integration, sectoral integration, people-centered integration, and whole system integration. 12 In this study, integrated care is defined as people-center integration, which means integrated care between providers and patients and other service users to engage and empower people through health education, shared decision-making, supported self-management, and community engagement, and which specifically includes four core elements: health education on the association mechanism between COPD and comorbidities; personalized self-management support (e.g., medication reminders, diet, and exercise plans); multidisciplinary collaboration (interdepartmental consultations involving respiratory medicine, cardiology, etc.); dynamic monitoring (remote or onsite follow-up). This definition ensures consistency in screening eligible interventions.
The effects of integrated care have not been evaluated with any high-quality systematic evidence. Previous studies have adopted different data and elements of integrated care, showing varied results.13–19 This study uses systematic evaluation methods to analyze the effects of integrated care on patients with COPD and multiple comorbidities, to understand the strength of integrated care, and improve intervention implementations.
Methods
This systematic review and meta-analysis follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses, 20 with the complete checklist provided in Supplemental Material 1.
Data sources and search
This systematic review was conducted on the databases from MEDLINE, EMBASE, CENTRAL, CINAHL, and ClinicalTrial.gov. The search strategy can be found in Supplemental Material 1. The searching period extended from the database interception to December 2023. Eligibility criteria were the availability of full texts in any language. Details of the search process and compliance with PRISMA 2020 are shown in Supplemental Material 1.
Inclusion criteria
Inclusion criteria were: studies evaluating short or long-term effects of integrated care on health outcomes in patients with COPD and at least one other chronic disease. Studies had to include one of these outcomes: exacerbation-related outcomes (number of exacerbations, emergency caused by exacerbations, hospitalization caused by exacerbations, et al.), all-cause hospitalization, all-cause emergency, quality of life, exercise capacity, pulmonary function test, COPD symptoms, and mental health. The intervention must be integrated, covering at least two core elements of integrated care (health education on the association mechanism between COPD and comorbidities; personalized self-management support; multidisciplinary collaboration; dynamic monitoring). Only randomized controlled trials (RCTs) were included in this review. Sample size was not an exclusion criterion.
Exclusion criteria
Exclusion criteria were: Non-RCTs (e.g., cohort studies, case-control studies); Studies only including patients with pure COPD (without comorbidities); Interventions that only replicate standard management content (e.g., no additional self-management support); Studies without complete outcome data (e.g., only reporting p-values without effect sizes or not reporting baseline data).
Study selection and data collection process
Title and abstract were first screened by two investigators (XL, YQ) independently to identify potentially eligible articles. Manuscripts that met the inclusion criteria after screening were read in full text to determine their inclusion. Disagreements were resolved by consensus or discussion with a third investigator.
Each study was critically and independently appraised by two investigators (XL, YQ) for the following components: clinical trial sites, sample size, mean age and gender structure of participants, stage of COPD, types of comorbidity, inclusion and exclusion criteria, intervention (duration, frequency, and used elements), summary of results, strengths and limits of the study.
The risk of bias of each study was evaluated by two investigators under the following domains: random sequence generation, allocation concealment, blinding for participants or personnel, blinding for outcome assessors, incomplete outcome data, selective reporting, and other bias. The bias domain will be rated as “low risk,” “high risk,” and “unclear risk” if the researcher did not mention relevant information.
Statistical analysis
The outcomes were extracted from the studies identified above. To analyze the mean differences, the random-effect analysis model for continuous variable were used.
We analyzed the studies according to initial level, on the basis of the existing literature and recommendations. Data are presented as means, with their 95% confidence intervals. Heterogeneity was assessed by the I2 statistic for each comparison. An I2 > 40% was considered to reject the homogeneity of the comparison (and to accept the heterogeneity hypothesis). In the cases of heterogeneity, a random-effect model was used. For all the comparisons, a p value <0.05 was considered statistically significant.
All the analyses were conducted by Stata SE 16.
Results
Search results
The study selection flowchart is demonstrated in Figure 1. A total of 7214 references were identified, of which 664 were duplicates. After checking the type of studies, 468 references were excluded. By reading the titles and abstracts of 6081 references, 5858 did not meet the inclusion criteria. Reading the full text of 223 manuscripts, 215 were excluded for the following reasons: (i) participants were not comorbidity patients with COPD and other diseases; (ii) the intervention was not integrated care according to the definition mentioned in introduction; (iii) the study was not based on a RCT design; (iv) no access to full text; (v) the outcomes were not health related. Finally, 7 studies met the criteria for qualitative analysis.13–19 Different studies were considered for meta-analysis of specific outcomes.

Study selection.
Characteristics of studies included
A detailed summary of the characteristics of studies included can be found in Supplemental Material 2. Titles, study countries, sample sizes, types of comorbidities, interventions, and main results are provided.
Studies were all conducted in high-income countries, like Italy, Australia, the Netherlands, England, Russia, and Spain.
In the references included, the sample size ranged from 50 to 470 participants. The mean ages of participants were 59 and 48.3 for the two studies in Russia, respectively, while the mean age of studies conducted in other countries was 70. The gender distribution was balanced in all included studies. Inclusion criteria were similar across the studies: participants must be diagnosed with COPD and at least one other disease simultaneously. Most studies restricted the comorbidity types to cardiovascular diseases and metabolic diseases. In general, the populations included in the studies were homogeneous.
The interventions varied widely in different studies, but they at least combined two or more elements of COPD management, such as education, follow-up, and supported self-management to empower patients. Studies measured outcomes including all-cause hospitalization and emergency, the number of exacerbations, hospitalization days, and emergency caused by exacerbations, 6 min walking test (6MWT), life quality, COPD-related symptoms, blood pressure, and blood glucose. Follow-up length varied among studies, ranging from 4 months to 12 months.
Interventions
Studies included indicated a large variety of interventions regarding whether telemonitoring was adopted, and elements used to construct the integrated care model (as we limited the setting where studies were conducted, all studies were finished in community hospitals or at patients’ homes). The characteristics of interventions are shown as follows.
Specifically, two studies were based on telemonitoring techniques to help monitor patients’ lung function and other vital signs, patients’ medication adherence, daily diet, and scientific training. The monitor adjusted patients’ interventions like the frequency and intensity of exercise according to patients’ capacity. When any exceptional situation occurred, urgent assistance would be provided. For instance, when abnormalities in patients’ lung function or vital signs were detected, they would be automatically reported to the monitor, and further treatment would be decided. The other five studies did not adopt telemonitoring techniques, and they simply provided integrated care onsite.
The elements used to construct the integrated care model varied among these seven studies. All studies provided health education for participants, including improved knowledge about COPD, smoking cessation, healthy diet, and better lifestyle. Four studies helped participants make and adjust personalized plans for diet and exercise. Five studies offered suggestions on standardized medication for patients. Only one study provided priority medical services for participants if needed.
Outcomes
The outcomes analyzed by the selected studies varied widely and focused on the clinical status and life quality of comorbidity patients: exacerbation-related outcomes (the number of exacerbations, hospitalization, and emergency caused by exacerbations), hospitalization and emergency caused by all causes, lung function, mortality, 6 min walking test, EQ-5D, COPD assessment test (CAT), and depression. A quantitative analysis (meta-analysis) was performed on four specific outcomes: the number of exacerbations, all-cause hospitalization, all-cause emergency, and CAT. Figures 2–5 present the forest plots for each outcome, with detailed interpretations of effect sizes, 95% confidence intervals (CIs), and heterogeneity statistics provided below.
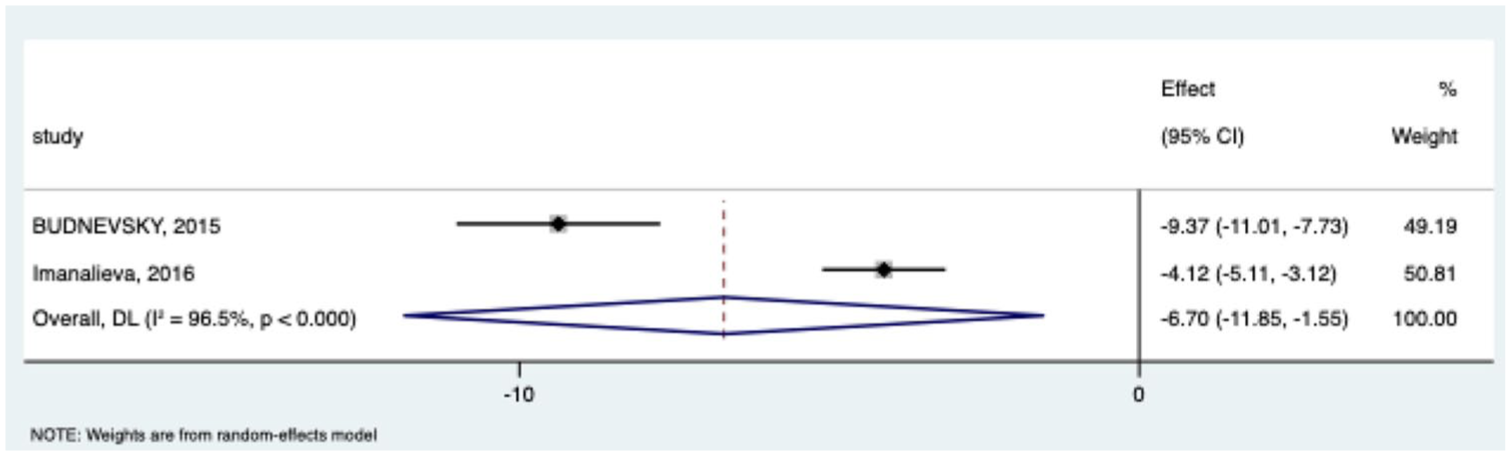
The number of exacerbations.

All-cause hospitalization.

All-cause emergency.

COPD assessment test.
The number of exacerbations
Two studies (n = 120 patients: Budnevskiy et al., Imanalieva et al.) reported the number of COPD exacerbations during follow-up. As shown in Figure 2, integrated care was associated with a significant reduction in exacerbations compared to usual care (weighted mean difference (WMD) = −6.70; 95% CI: −11.85 to −1.55). High heterogeneity was observed (I² = 96.5%; p < 0.001).
All-cause hospitalization
Three studies (n = 390 patients: Budnevskiy et al., Imanalieva et al., Rose et al.) contributed data on all-cause hospitalization. As shown in Figure 3, integrated care significantly reduced all-cause hospitalization compared to usual care (WMD = −3.12; 95% CI: −6.10 to −0.14). Heterogeneity was high (I² = 98.5%; p < 0.001), likely due to variations in follow-up duration and comorbidity severity.
All-cause emergency
Two studies (n = 340 patients: Budnevskiy et al., Rose et al.) reported all-cause emergency visits. As shown in Figure 4, integrated care did not significantly reduce all-cause emergency visits compared to usual care (WMD = −3.08; 95% CI: −8.87 to 2.72). Extreme heterogeneity was observed (I2 = 99.0%; p < 0.001), driven by conflicting results between studies: Budnevskiy et al. reported a large reduction (WMD = −6.06), while Rose et al. showed a negligible effect (WMD = −0.15).
COPD assessment test
Three studies (n = 459 patients: Budnevskiy et al., Walker et al., Gottlieb et al.) provided data on CAT scores. As shown in Figure 5, integrated care did not significantly improve CAT scores compared to usual care (WMD = −0.54; 95% CI: −1.45 to 0.36). High heterogeneity was present (I2 = 95.2%; p < 0.001), largely due to Budnevskiy et al. reporting a significant reduction (WMD = −2.65) while the other two studies showed minimal or no effect (WMD = −0.13 to 0.22).
Risk of bias in selected studies
The risk of bias in the selected studies should be considered when interpreting the results. While all included studies were RCTs, variations in study design, patient populations, and intervention specifics could introduce bias. The heterogeneity observed in the studies, particularly in the number of exacerbations, highlights the need for standardized approaches to measuring outcomes and reporting results. The results for the risk of bias of each study can be found in Supplemental Material 2.
Discussion
Association with existing literature
This study’s finding that integrated care reduces hospitalization rates is consistent with Dennett et al., who conducted a mixed-methods review on COPD patients with comorbidities and reported that adapted interventions improve healthcare utilization. 21 However, our study further narrowed the intervention from adapted management to integrated care and found that patients with comorbidities benefited more significantly.
Compared with Poot et al., 22 who only included patients with pure COPD and found no significant effect of integrated care on exacerbations, our study confirms that even patients with comorbidities can benefit from integrated care, filling the gap in evidence for this population.
Sources of heterogeneity
High heterogeneity primarily stems from three key factors, which we analyzed to contextualize result variability:
Intervention intensity and delivery
Variations in the mode of care delivery and frequency drove outcome differences. For example, Budnevskiy et al. implemented weekly in-person follow-ups plus daily physical training, resulting in a 1.8-fold reduction in COPD exacerbations—whereas Imanalieva et al. used monthly telephone check-ins, leading to a smaller (but still significant) 0.56 reduction in exacerbations. Telemonitoring studies also varied: Bernocchi et al. combined real-time cardiorespiratory monitoring with weekly nurse/physiotherapist calls, yielding a 60-m improvement in 6-min walk test (6MWT) scores, while Walker et al. relied solely on lung mechanics monitoring via forced oscillation technique (FOT) and reported no significant change in quality of life (EQ-5D). This suggests that multimodal support (combining remote monitoring with personalized clinical follow-up) may be more impactful than single-component telemonitoring.
Comorbidity composition
Patient populations differed in comorbidity profiles, reflecting regional health trends. Studies in Russia had a higher prevalence of metabolic syndrome (100% of participants in Budnevskiy et al. 19 ), leading to measurable improvements in metabolic outcomes (e.g., 4.37 mmHg reduction in systolic blood pressure). In contrast, Western European studies focused on cardiovascular comorbidities (e.g., 75%–76% prevalence of hypertension in Rose et al.), with outcomes centered on reducing cardiac-related hospitalizations. These differences highlight that integrated care effectiveness is modulated by comorbidity type, emphasizing the need for tailored models.
Outcome measurement timing
Inconsistent timing of outcome assessments contributed to heterogeneity in patient-reported outcomes. For example, CAT scores measured at 6 months (two studies) showed a non-significant trend toward improvement, while the only study measuring CAT at 12 months (Budnevskiy et al.) reported a larger, clinically meaningful reduction (WMD: −2.65; 95% CI: −3.29 to −2.00). This suggests that patient-reported symptoms may improve over longer follow-up, but short-term assessments (4–6 months) may underestimate benefits.
Study limitations
Geographical and generalizability constraints
All seven included studies were conducted in high-income countries (HICs: Canada, Russia, Denmark, Italy, etc.), which limits generalizability to middle- and low-income countries (MLICs)—notably China, where COPD affects over 100 million people. 3 China’s unique healthcare context includes higher hypertension prevalence (17.8% in patients with COPD), 23 extremely low use of telemedicine in rural China, 24 and a greater burden of undiagnosed comorbidities. 8 These factors may reduce the feasibility of HIC-derived integrated care models (e.g., telemonitoring) in China and other low- and middle-income countries, requiring localized adaptations (e.g., community-based in-person follow-up).
Short follow-up durations
Five studies had follow-up periods of 4–9 months, with only two extending to 12 months. This is insufficient to capture the long-term clinical course of COPD, which typically follows annual or biennial exacerbation patterns. Longer follow-up (e.g., 2 years) is needed to evaluate whether integrated care sustains reductions in exacerbations and hospitalizations, or modifies long-term outcomes, such as disease progression or mortality.
Incomplete outcome data
Key outcomes such as mortality and quality of life (EQ-5D) were excluded from meta-analysis due to insufficient data. For example, while Rose et al. reported a 44% lower mortality risk with integrated care, other studies did not measure mortality—precluding a pooled effect estimate. Similarly, EQ-5D scores were reported in only a few studies, limiting conclusions about patient-centered quality of life improvements.
Heterogeneity in “integrated care” definition
Despite our a priori definition (four core elements), studies varied in component inclusion: three studies omitted multidisciplinary collaboration, and two lacked structured self-management support. This variability complicates comparisons across interventions and highlights the need for standardized reporting of integrated care components in future RCTs.
Conclusion
This systematic review and meta-analysis of seven RCTs provides robust evidence that integrated care (combining health education, personalized self-management support, multidisciplinary collaboration, and dynamic monitoring) reduces two critical outcomes in patients with COPD and comorbidities: integrated care significantly lowers the number of exacerbations, a key driver of disease progression and healthcare costs; pooled results show a meaningful reduction in hospitalizations, alleviating the substantial burden of inpatient care for this population.
Notably, integrated care did not significantly reduce all-cause emergency visits or improve COPD Assessment Test (CAT) scores. These null findings likely reflect the complexity of emergency triggers (e.g., acute comorbidity flares) and the need for more granular, patient-specific symptom management to improve self-reported outcomes.
Given the high prevalence of comorbidities in COPD patients globally, our results support the adoption of integrated care as a standard component of COPD management in high-income settings. However, critical gaps remain: localization for MLICs: future research should adapt integrated care to low-resource contexts (e.g., China) by prioritizing community-based care over telemonitoring and addressing region-specific comorbidity patterns (e.g., hypertension); long-term effectiveness: studies with 2+ year follow-up are needed to assess whether integrated care modifies mortality and disease progression; component optimization: RCTs comparing individual integrated care elements (e.g., telemonitoring vs in-person follow-up) will clarify which components drive the greatest benefit.
In summary, integrated care is a clinically valuable strategy for reducing exacerbations and hospitalizations in COPD patients with comorbidities. With further refinement for diverse healthcare contexts and longer-term evaluation, it has the potential to improve global COPD management and reduce healthcare disparities.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251414315 – Supplemental material for Effects of integrated care on patients with COPD and comorbidities: a systematic review
Supplemental material, sj-docx-1-tar-10.1177_17534666251414315 for Effects of integrated care on patients with COPD and comorbidities: a systematic review by Xiaolong Guan, Yiqi Xia, Jinxu Ai, Zhenyu Shi, Xiaoping Wei and Ping He in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251414315 – Supplemental material for Effects of integrated care on patients with COPD and comorbidities: a systematic review
Supplemental material, sj-docx-2-tar-10.1177_17534666251414315 for Effects of integrated care on patients with COPD and comorbidities: a systematic review by Xiaolong Guan, Yiqi Xia, Jinxu Ai, Zhenyu Shi, Xiaoping Wei and Ping He in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-3-tar-10.1177_17534666251414315 – Supplemental material for Effects of integrated care on patients with COPD and comorbidities: a systematic review
Supplemental material, sj-pdf-3-tar-10.1177_17534666251414315 for Effects of integrated care on patients with COPD and comorbidities: a systematic review by Xiaolong Guan, Yiqi Xia, Jinxu Ai, Zhenyu Shi, Xiaoping Wei and Ping He in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We would like to thank the China Center for Health Development Studies, Peking University, for providing support in database retrieval; the authors of all included studies for providing original data; and the editorial team and reviewers of Therapeutic Advances in Respiratory Disease for their valuable comments on the manuscript.
Authors’ note
The work is done by the China Center for Health Development Studies, Peking University.
Declarations
Supplemental material
Supplemental material for this article is available online.
Artificial intelligence disclosure
No generative artificial intelligence (e.g., ChatGPT) was used in the creation of text, references, or images for this study. All content was original to the authors, and data analysis was performed manually using Stata SE 16.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
