Abstract
Thoracic SMARCA4-deficient undifferentiated tumor (SMARCA4-UT), as a rare and highly malignant neoplasm associated with a high mortality risk, is easily confused with SMARCA4-deficient nonsmall-cell lung cancer (NSCLC). To date, no standard and effective protocol for thoracic SMARCA4-UT has been established. Immunotherapy has demonstrated efficacy in advanced NSCLC, achieving unprecedented survival benefits. However, immune-related adverse events (irAEs) remain a significant clinical challenge. Here, we reported the first case of thoracic SMARCA4-UT with immune-related cystitis and hypothyroidism, in which the patient benefited from first-line immune checkpoint inhibitor (ICI)-based combination therapy, achieving a remarkable overall survival of over 100 weeks. Furthermore, we performed a review and analysis of the diagnosis, differential diagnosis, immunotherapy, and prognosis of thoracic SMARCA4-UT, proposing that first-line therapy combining immunotherapy with platinum-based chemotherapy (induction and maintenance phases) with or without radiotherapy, may improve the prognosis of such patients. Additionally, we hypothesized a potential role of macrophages in the pathogenesis of immune-related cystitis for the first time and detailed the clinicopathological characteristics and evidence-based management of this irAE.
Keywords
Introduction
Thoracic SMARCA4-deficient undifferentiated tumor (SMARCA4-UT), as a newly recognized disease entity characterized as a highly aggressive neoplasm with inactivated mutations in
Due to the rarity of thoracic SMARCA4-UT, an optimal strategy concerning specific SMARCA4-UT treatments has not yet been identified. However, some studies have demonstrated that immunotherapy could effectively improve clinical outcomes in patients with thoracic SMARCA4-UT.2,4 However, immune-related adverse events (irAEs) have emerged as a major challenge with bladder and urinary tract irAEs remaining poorly characterized.
Herein, we reported the first case of thoracic SMARCA4-UT that developed immune-related cystitis (with potential macrophage involvement) and hypothyroidism during first-line therapy combining immunotherapy with platinum-based chemotherapy, achieving an exceptional overall survival (OS) of over 100 weeks. Additionally, we conducted a literature review to characterize the clinical features and management strategies of immune-related cystitis.
Case presentation
A 75-year-old man with a 3-year history of unmedicated Parkinson’s disease and a 40-pack-year smoking history (cessation 1 year prior) was admitted to our hospital with urinary frequency, urgency, and painful urination following 15 cycles of an ICI-based combination therapy in first-line for lung cancer.
Approximately 100 weeks prior to presentation, chest computed tomography (CT) performed during a routine health screening revealed a 51 mm × 49 mm mass in the right lung. Subsequently, a transbronchial lung biopsy was performed at another hospital. Histopathological examination showed undifferentiated spindle-shaped and round tumor cells with prominent nucleoli, focal rhabdoid features, accompanied by sheet necrosis (Figure 1(a)). Immunohistochemical staining demonstrated the loss of

Pathology of the patient. (a) H&E staining: undifferentiated spindle-shaped and round tumor cells with prominent nucleoli, focal rhabdoid features accompanied by sheet necrosis (× 200). (b, c) Expression of SMARCA4 and SMARCA2 in tumor cells was absent for immunohistochemical staining (× 400 and × 200, respectively) (Endothelial and inflammatory cells as internal positive controls). (d, e) Immunohistochemical staining showed diffuse expression for INI-1 and SOX2 in tumor cells, respectively (both × 200). (f) Immunohistochemical staining was negative for Pan-cytokeratin in tumor cells (× 200). (g) H&E staining: sheets of tumor cells with large nuclei, heteromorphism, considering metastatic carcinoma (poorly differentiated) (× 400). (h) H&E staining: infiltration of inflammatory cells in mucosa and interstitium (× 200). (i, j) Immunohistochemical staining was positive for CD68 and PD-L1, respectively (both × 200).

Imaging of the patient. (a, b) The longest diameter of the right lung mass with 18F-FDG highly concentrated was 60 mm with emphysema in both lungs on PET-CT before therapy. (c) The 18F-FDG was highly concentrated in lymph nodes in the mediastinum, right hilum, left submandibular, and right clavicle areas on PET-CT before therapy. (d) The longest diameter of the right lung mass was 30 mm on chest CT after chemotherapy combined with immunotherapy for two cycles. (e) The longest diameter of the right lung mass was 17 mm on chest CT after antitumor therapy for 13 cycles. (f) CT urography: no obvious thickening of bladder wall, no localized filling defect in bladder cavity.
The patient received two cycles of Pembrolizumab combined with nab-Paclitaxel and Carboplatin, achieving an immune partial response (iPR) (Figure 2(d)). Subsequently, 11 additional cycles of the immunotherapy-containing combination regimen were administered, maintaining the iPR status (Figures 2(e) and 3). However, immune-related hypothyroidism developed after 13 cycles, requiring initiation of thyroid hormone replacement therapy. The patient received one additional cycle of immunotherapy combined with chemotherapy. Nevertheless, evaluation at this time confirmed a new right cervical mass as metastatic disease, while the lung mass remained stable. The progression-free survival (PFS) was 55 weeks, significantly exceeding the reported mOS for thoracic SMARCA4-UT. The right cervical mass was surgically resected, followed by one cycle of Pembrolizumab combined with nab-Paclitaxel plus Carboplatin. Approximately 2 weeks later, the patient developed urinary frequency, urgency, painful urination, and nocturia. Ultrasonography demonstrated benign prostatic hyperplasia (BPH) with calcification, while urinalysis showed leukocyturia and proteinuria. Symptoms improved following treatment with Levofloxacin, Doxazosin, and Mirabegron. Subsequently, the patient received one cycle of Pembrolizumab combined with Vinorelbine and Carboplatin. Unfortunately, the urethral irritation symptoms worsened. Multiple urine cultures and fungal tests were negative. CT urography revealed no significant abnormalities (Figure 2(f)), and cystoscopy demonstrated diffuse congestion of the bladder mucosa with surface follicular changes. Histological and immunohistochemical analysis revealed chronic mucosal inflammation with interstitial inflammatory cell infiltration (Figure 1(h)), predominantly activated T lymphocytes (CD3+, CD4+, CD8+, programmed cell death protein 1 (PD-1)+, and intracellular antigen 1 (TIA-1)+) and macrophages (CD68+ and PD-L1+) (Figure 1(i) and (j)), accompanied by scattered CD20+ B lymphocytes and CD117+ mast cells. A diagnosis of immune-related cystitis was established during the 9th week following the onset of urinary symptoms. Oral prednisone (40 mg per day) was initiated, leading to rapid resolution of urinary irritative symptoms within 5 days, after which the prednisone dose was gradually tapered (Figure 3).

Clinical course of the patient.
Subsequently, the patient received chemotherapy with Vinorelbine and Carboplatin. Unfortunately, urinary frequency and urodynia recurred on two occasions, necessitating adjustments to the glucocorticoid therapy regimen (Figure 3). During this period, a neck mass recurred, and the corresponding management involved Gemcitabine monotherapy or combined modality therapy with neck radiotherapy plus Gemcitabine (Figure 3). Unfortunately, liver metastases were subsequently detected. The patient ultimately succumbed to the disease, with an OS exceeding 100 weeks (Figure 3), significantly longer than the reported mOS for thoracic SMARCA4-UT.
Method
The reporting of this study conforms to the CARE checklist. 5
Literature review method
To investigate current management strategies for thoracic SMARCA4-UT and immune-related cystitis, a comprehensive literature search was conducted in PubMed, with selection restricted to peer-reviewed publications containing English abstracts. For SMARCA4-UT, the search terms were “thoracic,” “SMARCA4-deficient undifferentiated tumor”, “SMARCA4-deficient sarcoma” and “SMARCA4-deficient sarcomatoid tumor.” The search string was: (((((((((SMARCA4-deficient undifferentiated tumor[MeSH Terms]) OR (SMARCA4-deficient sarcoma[MeSH Terms])) OR (SMARCA4-deficient sarcomatoid tumor[MeSH Terms])) OR (SMARCA4-deficient undifferentiated tumor[Title/Abstract])) OR (SMARCA4-deficient undifferentiated tumors[Title/Abstract])) OR (SMARCA4-deficient sarcoma[Title/Abstract])) OR (SMARCA4-deficient sarcomas[Title/Abstract])) OR (SMARCA4-deficient sarcomatoid tumor[Title/Abstract])) OR (SMARCA4-deficient sarcomatoid tumors[Title/Abstract])) AND (thoracic[Title/Abstract]). For immune-related cystitis, the search terms were “immune-related cystitis,” “immunotherapy,” “immune checkpoint inhibitor” and “cystitis.” The search string was: ((immune-related cystitis[MeSH Terms]) OR (Immune-related cystitis[Title/Abstract])) OR ((((Immune checkpoint inhibitors[Title/Abstract]) OR (Immune checkpoint inhibitor[Title/Abstract])) OR (Immunotherapy[Title/Abstract])) AND ((cystitis[Title/Abstract]))). Inclusion criteria were as follows: (1) type of study: no limits. (2) species: human. (3) article language: English. (4) text availability: full text. Retrieved: 24 November 2024.
Discussion
Diagnosis and differential diagnosis
Histopathological examination of the right lung mass revealed undifferentiated round and spindle-shaped tumor cells exhibiting prominent nucleoli, along with focal rhabdoid features and sheet necrosis. Immunohistochemical analysis demonstrated the loss of SMARCA4 and SMARCA2 expression, absence of CK-7 and Pan-cytokeratin staining, diffuse SOX2 expression, and retained INI-1 expression in tumor cells. SMARCA4-deficient NSCLC comprises approximately 10% of NSCLC cases. 6 In contrast, SMARCA4-deficient NSCLC typically exhibits better differentiation, with immunohistochemical staining positive for SMARCA2, Claudin-4 and epithelial markers, but negative for SOX2.2,7 The mOS was 20.7 months for SMARCA4-deficient NSCLC compared with 5.2 months for thoracic SMARCA4-UT. 7
Thoracic SMARCA4-UT, malignant rhabdoid tumor (MRT), and small-cell carcinoma of the ovary, hypercalcemic type (SCCOHT) share overlapping morphological features. Notably, differential diagnosis becomes crucial when SMARCA4 and SMARCA2 expression is absent. MRT primarily affects infants and young children and shows loss of INI-1 expression by immunohistochemistry, 8 whereas SCCOHT is of ovarian origin. 9
Immune-related adverse events
Hypothyroidism is one of the more common irAEs associated with ICIs. 10 Immune-related hypothyroidism typically necessitates lifelong thyroid hormone replacement therapy; corticosteroid therapy is generally not recommended, and ICI discontinuation is usually not required. 11
In the present case, urinary tract irritation symptoms failed to resolve completely with antibiotic therapy and management for BPH, and subsequently worsened following continued immunotherapy. Consequently, cystoscopy was performed to establish a diagnosis. Currently, there are no criteria for diagnosing immune-related cystitis. 12 The precise pathophysiology underlying immune-related adverse events remains incompletely understood. Proposed mechanisms contributing to irAE development include: T cell cross-reactivity against tumor and normal tissue13,14; expansion of T cell receptor diversity 13 ; increase in tissue-resident Th1, Th2, and Th17 cells, 14 memory T cells, 13 and B cells13,14; and a reduction in regulatory T cells (Tregs). 14 Bladder tissue infiltration by inflammatory cells including CD3+, CD8+, CD20+, PD-1+, and/or TIA-1+ lymphocytes, along with CD117+ mast cells has been observed in patients with immune-related cystitis.15–17 In our patient, immunohistochemical analysis revealed bladder tissue infiltration by CD3+, CD4+, CD8+, and PD-1+ T lymphocytes; CD20+ B lymphocytes; CD68+ and PD-L1+ macrophages; and CD117+ mast cells. Immune-related cystitis was confirmed based on these pathological findings and the rapid symptomatic relief following glucocorticoid administration. Immune-related myocarditis has been associated with the expansion of interferon-gamma (IFN-γ)-induced macrophages expressing CXCL9 (C-X-C motif chemokine ligand 9) and CXCL10 (C-X-C motif chemokine ligand 10). In murine models, IFN-γ blockade and macrophage depletion reduced the population of CXCL9+CXCL10+ macrophages, attenuated immune-related myocarditis. and prolonged survival. 18 In patients with ICI-induced Stevens–Johnson syndrome/toxic epidermal necrolysis (SJS/TEN), tumor necrosis factor (TNF)-induced overexpression of macrophage-derived CXCL10 recruited CXCR3 (C-X-C motif chemokine receptor 3)+ cytotoxic T lymphocytes. TNF blockade resulted in significantly rapid recovery and no recurrence with continued ICI therapy in patients with ICI-induced SJS/TEN. 19 Both macrophage depletion and CXCR3 blockade ameliorated the immune-related myocarditis phenotype in murine models of ICI-induced myocarditis. 20 Macrophages have been implicated in mediating pancreatic β-cells death in PD-1 inhibitor-induced diabetes. 21 To our knowledge, this is the first report implicating macrophage involvement in immune-related cystitis; however, the underlying mechanisms remain to be elucidated. Most notably, the IFN-γ and TNF signaling pathways represent potential therapeutic targets for immune-related cystitis.
In order to achieve symptom resolution and prevent irAE recurrence, an appropriate corticosteroid treatment course with a carefully planned tapering strategy is crucial. 11 However, immune-related cystitis is not well documented, and definitive recommendations regarding the initial corticosteroid dose and subsequent tapering strategies are lacking. In our case, the initial prednisone dose was 40 mg per day, which was tapered over 4 weeks. During the tapering process, symptoms recurred. As previously reported, 15 rapid steroid tapering may cause recurrence of immune-related cystitis. Following temporary intravenous methylprednisolone administration, the prednisone dose was tapered over 10 weeks. However, symptoms recurred 1 week after Prednisone discontinuation, suggesting possible steroid dependence. Therefore, the prednisone dose was increased to 30 mg per day and then tapered by 5 mg every 2 weeks to a maintenance dose of 15 mg per day, with no subsequent recurrence of immune-related cystitis. One case of steroid-resistant immune-related cystitis was successfully managed with Infliximab, 22 suggesting that Infliximab may be a therapeutic option for steroid-dependent immune-related cystitis.
With close monitoring for irAE recurrence, immunotherapy rechallenge may be considered once a grade 2 irAE has resolved to grade 1 or below. 11 The patient experienced two distinct irAEs, and the immune-related cystitis exhibited potential steroid dependence. Although initiating corticosteroid therapy after irAE onset does not appear to diminish ICI efficacy, 11 immunotherapy should not be resumed unless the corticosteroid dose is reduced to ⩽10 mg prednisone equivalent per day. 23 In our case, immunotherapy rechallenge was not pursued.
Among patients with advanced NSCLC, 24% develop a single irAE, and 9% experience multisystem irAEs; the majority of the latter group present with two concurrent irAEs. 10 Thyroiditis and dermatitis are the most frequent initial irAEs, while the most common multisystem irAE patterns were pneumonitis with thyroiditis, hepatitis with thyroiditis, dermatitis with pneumonitis, and dermatitis with thyroiditis. 10 Longer duration of ICI therapy and better Eastern Cooperative Oncology Group (ECOG) performance status were identified as independent risk factors for multisystem irAEs. 10
Therapy and prognosis
Currently, there is no established standard of treatment for SMARCA4-UT. For advanced NSCLC, ICI therapy has been shown to be effective regardless of PD-L1 expression in tumor cells 24 and has achieved unprecedented survival prolongation. 25 The combination therapy of Atezolizumab, Bevacizumab, Carboplatin, and Paclitaxel has demonstrated efficacy in thoracic SMARCA4-UT, with one patient (40% PD-L1 expression) achieving a PFS of at least 17 months. 4 Additionally, patients with thoracic SMARCA4-UT have benefited from chemotherapy in combination with ICI or ICI monotherapy. 2 with one patient (100% PD-L1 expression) achieving an OS of 26 months. 26
In our case, the stage IV patient with PD-L1 expression of < 1% received nab-Paclitaxel plus Carboplatin combined with Pembrolizumab, achieving an iPR after two cycles of treatment. However, lung mass slightly enlarged after 10 cycles of treatment. The efficacy of PD-1 inhibitors is enhanced by the release of tumor-associated antigens and inflammatory cytokines following radiotherapy, which activates dendritic cells, promotes antigen presentation, recruits CD8+ T cells, induces tumor cells killing, 27 and upregulates PD-L1 expression on tumor cells and PD-1 expression on tumor infiltrating lymphocytes. 27 A meta-analysis has demonstrated that radiotherapy in combination with ICIs can significantly prolong PFS and OS in patients with advanced NSCLC. 28 Patients with stage IVB thoracic SMARCA4-UT who underwent chemoradiation (nab-Paclitaxel and Carboplatin) in first line and ICI in second line or beyond demonstrated survival durations of at least 11 months and 22 months.29,30 Subsequently, our patient underwent immunotherapy with concurrent local thoracic radiotherapy, maintaining iPR status for additional three cycles. The abscopal effect induced by combined radiotherapy and immunotherapy has been regarded as the dawn of cancer therapy.28,31 However, a right cervical mass was confirmed as metastatic disease, with PFS reaching 55 weeks. Fortunately, the right lung lesion remained stable despite immunotherapy discontinuation for approximately 39 weeks and subsequent development of distant metastases. The survival time of our patient exceeded 100 weeks, which far surpassed the mOS of thoracic SMARCA4-UT. Li et al. 2 suggested chemotherapies (with or without radiotherapy) combined with Pembrolizumab in the first line may improve the prognosis in thoracic SMARCA4-UT patients with low PD-L1 expression. Furthermore, the patient developed two immune-related adverse events. Development of irAEs, especially multisystem involvement, has been demonstrated to be associated with improved survival in lung cancer treated with ICIs.10,32,33 Given the aggressive nature and poor prognosis of SMARCA4-UT, maintenance therapy with the initial combination regimen was selected over Pembrolizumab monotherapy after six cycles of systemic therapy, although Carboplatin was intermittently discontinued due to myelosuppression. In fact, the patient achieved iPR after two cycles of first-line therapy, which persisted for 55 weeks during which 14 cycles of treatment were administered. It appeared that, after immunotherapy in combination with chemotherapy (without Carboplatin) and the reduction in the dose of chemotherapy drugs due to myelosuppression, right lung lesion enlargement occurred, and then right cervical lymph nodes metastasis emerged. We infer that for highly malignancy, initial regime maintenance therapy, if tolerated, may achieve sustained response and prevent recurrence and metastases.
Literature review on immune-related cystitis
We reviewed the literature on immune-related cystitis15,16,22,34–43 and summarized its clinical features, pathological characteristics, and management strategies (Table 1 and Supplemental Table 1). Patients were predominantly male, with a median age of 57 years. Immune-related cystitis occurred as early as after the 2nd cycle of Nivolumab and as late as after the 77th cycle of Nivolumab, with a median onset at the 6th cycle (Supplemental Table 1). Most reported cases of immune-related cystitis occurred after the third to seventh cycle of immunotherapy.
12
ICIs inducing immune-related cystitis were mainly PD-1/PD-L1 blockers, including Nivolumab (
Clinical and pathological characteristics and treatment of immune-related cystitis.
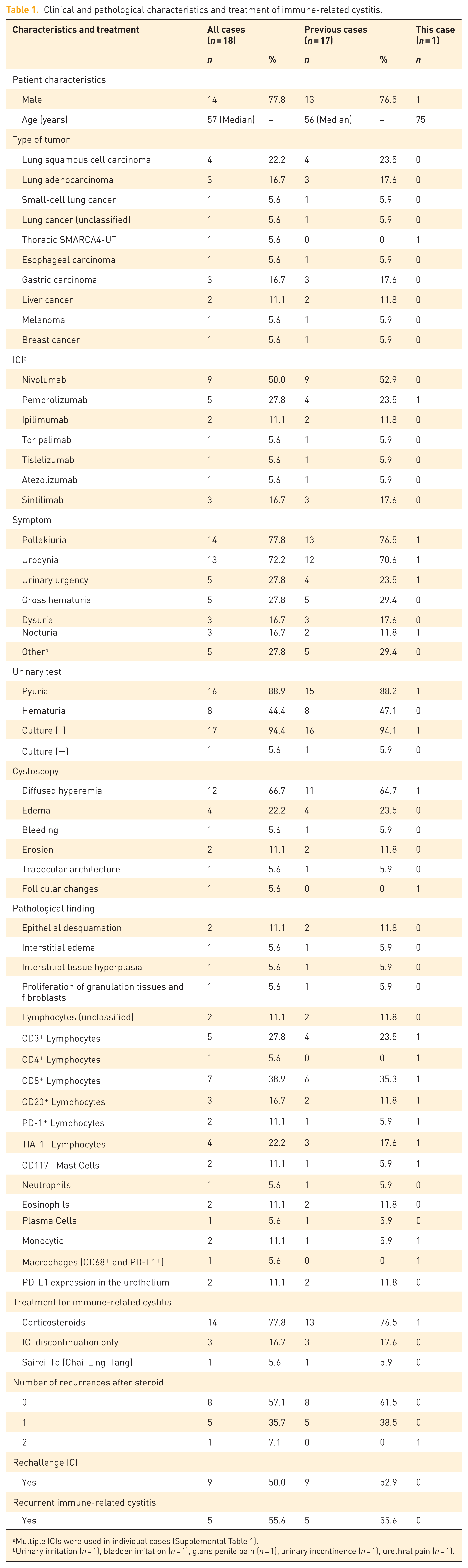
Multiple ICIs were used in individual cases (Supplemental Table 1).
Urinary irritation (
Urinary irritation symptoms were common and could be accompanied by dysuria, nocturia and even gross hematuria (Table 1). The diagnostic delay from symptom onset to confirmation ranged from 4 days to 6 months (Supplemental Table 1), reflecting insufficient disease awareness. When immunotherapy recipients presenting with pollakiuria, urodynia, dysuria or nocturia accompanied by sterile pyuria, hematuria or proteinuria are unresponsive to initial therapy (especially antibiotic therapy), immune-related cystitis should be suspected, and cystoscopy with biopsy should be performed. Cystoscopy commonly showed diffused hyperemia,15,16,34 edema,34,35 erosion,36,37 and even bleeding 22 of the bladder mucosa without signs of carcinoma infiltration15,17,34; the bladder mucosa was partially arranged in a trabecular pattern. 16 Bladder tissue in immune-related cystitis showed epithelial desquamation,16,36 interstitial edema, 36 and / or interstitial infiltrations of monocytes and eosinophils.37,43 Frequently, CD3+, CD8+, and/or TIA-1+ lymphocytes infiltrated the bladder tissue in patients with immune-related cystitis, while CD20+, PD-1+ lymphocytes, and CD117+ mast cells were also observed. In the urothelium, PD-L1 was positive expression (Table 1).
In a few cases, ICI discontinuation could relieve symptoms, but 77.8% of patients received glucocorticoid therapy (Table 1). The response rate to glucocorticoid therapy was 96.0% (24/25, based on the number of glucocorticoid treatment courses). Initial dose of Prednisone or Methylprednisolone ranged from 25 mg to 500 mg per day. Symptoms resolved within 1 week of glucocorticoid administration in most patients (with some requiring 2–3 weeks) and recurred in 42.9% of patients. Immune-related cystitis appeared to be less likely to relapse in patients who received glucocorticoid with a tapering period exceeding 6 weeks (Supplemental Table 1). Notably, the National Comprehensive Cancer Network (NCCN) and American Society of Clinical Oncology (ASCO) have not provided recommendations for glucocorticoid treatment strategies in immune-related cystitis. Initial glucocorticoid dose and tapering strategies for immune-related cystitis require further investigation. In addition, ICI therapy was resumed in 50% of patients, with immune-related cystitis recurring in 55.6% of these patients (Table 1).
Conclusion
We reported the first documented case of thoracic SMARCA4-UT that developed immune-related cystitis and hypothyroidism during combination therapy with first-line immunotherapy, with significant survival prolongation. We proposed that first-line therapy combining immunotherapy with platinum-based chemotherapy (induction and maintenance phases) with or without radiotherapy may improve the prognosis of such patients. Furthermore, we hypothesized a potential role of macrophages in the pathogenesis of immune-related cystitis for the first time. In the future, further investigation seems necessary for optimal corticosteroid dosing and tapering regimens.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251385689 – Supplemental material for Prolonged survival of thoracic SMARCA4-deficient undifferentiated tumor with immune-related cystitis: a case report and literature review
Supplemental material, sj-docx-1-tar-10.1177_17534666251385689 for Prolonged survival of thoracic SMARCA4-deficient undifferentiated tumor with immune-related cystitis: a case report and literature review by Xiang Li, Minshu Ao, Sen Tian, Yilin Chen, Hui Chen, Na Ta, Xiang Ni, Jingyu Zhang, Haidong Huang, Yao Fang, Lei Qu, Fang Yang, Zhenhong Hu, Yuchao Dong, Hui Shi and Chong Bai in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251385689 – Supplemental material for Prolonged survival of thoracic SMARCA4-deficient undifferentiated tumor with immune-related cystitis: a case report and literature review
Supplemental material, sj-docx-2-tar-10.1177_17534666251385689 for Prolonged survival of thoracic SMARCA4-deficient undifferentiated tumor with immune-related cystitis: a case report and literature review by Xiang Li, Minshu Ao, Sen Tian, Yilin Chen, Hui Chen, Na Ta, Xiang Ni, Jingyu Zhang, Haidong Huang, Yao Fang, Lei Qu, Fang Yang, Zhenhong Hu, Yuchao Dong, Hui Shi and Chong Bai in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
