Abstract
Background:
Radial endobronchial ultrasound (r-EBUS) is a minimally invasive procedure used to evaluate pulmonary lesions suspicious of cancer. Current information on the effect of combining different sampling tools used during r-EBUS or the addition of linear EBUS (EBUS-TBNA) on its diagnostic performance is limited.
Objectives:
To evaluate the effect on diagnostic performance of the systematic addition of different sampling tools and EBUS-TBNA during r-EBUS, as well as the rate of peri-procedural complications.
Design:
This was an observational, analytical cohort study designed to evaluate diagnostic accuracy.
Methods:
We calculated diagnostic accuracy statistics and used the Cochran–Armitage statistical test to assess the effect of combining techniques on diagnostic performance. Diagnostic success (DS) was defined as true positives and true negatives, while diagnostic failure was defined as false positives and false negatives.
Results:
A total of 309 patients were included, with a mean age of 67.9 years (standard deviation: 10.97); 50.8% (157/309) were male. The bronchial washing had a DS rate of 49%, while bronchial brushing showed a DS rate of 61%. The combination of bronchial washing and bronchial brushing improved the r-EBUS performance to 63%. Combining bronchial washing, bronchial brushing, and transbronchial biopsy increased the performance to 70%, and the addition of EBUS-TBNA raised the diagnostic performance to 80% (p < 0.001; Cochran–Armitage test). The overall complication rate was 6.4% (20/309), with pneumothorax occurring in 1.2% (4/309), bronchospasm in 3.8% (12/309), and bleeding in 1.2% (4/309).
Conclusion:
The use of multiple sampling tools significantly contributes to the DS of r-EBUS, particularly with the addition of EBUS-TBNA. This approach maintains a low complication rate.
Keywords
Introduction
Radial endobronchial ultrasound (r-EBUS) is a minimally invasive procedure used for the evaluation of pulmonary lesions suspected of malignancy.1 –3 The diagnosis of lung cancer is crucial for guiding treatment and improving prognosis.4,5 In elderly patients and those with comorbidities who present physiological limitations for undergoing invasive diagnostic methods, such as thoracoscopy, r-EBUS becomes a valuable diagnostic tool. Its good diagnostic capacity and low complication rate position it as a useful minimally invasive technique for these patients.5 –8
EBUS employs two techniques that can be used alone or in combination, depending on the anatomical characteristics of the disease to be diagnosed or staged.9,10 Linear EBUS (EBUS-TBNA) is primarily used for lesions or lymph nodes near the central airways, providing a one-plane image that is useful for guiding real-time biopsies and ultrasound-guided transbronchial needle aspiration (TBNA).5,9,10 r-EBUS is used for peripheral lesions located distal to the segmental bronchi, offering a 360-degree view of the lesion.11,12 Combining EBUS-TBNA with r-EBUS may enhance diagnostic accuracy for suspected malignant pulmonary lesions.5,11,13 However, the impact of this combined approach on diagnostic performance remains underexplored.3,9 –12
A systematic review of seven studies involving a total of 510 patients with pulmonary lesions reported the diagnostic performance of r-EBUS with TBNA as 75% (95% CI: 67%–84%) according to a random effects model. 14 The diagnostic performance of r-EBUS depends on factors such as lesion size, lesion etiology, anatomical location in the bronchial tree, lesions associated with bronchial signs on computed tomography (CT), using rapid on-site evaluation (ROSE), and operator expertise.15 –20 The main complications occurring in the peri-procedural period are pneumothorax and bleeding. However, these have been reported with a frequency of less than 5% in observational studies.5,6,8,15
Current information on the diagnostic capacity of combining r-EBUS with EBUS-TBNA and the various sampling techniques used during the procedure is limited.5 –10,15,18 –23 Therefore, this study was designed to evaluate the effect on diagnostic performance of the systematic addition of different sampling tools and EBUS-TBNA during r-EBUS, as well as the rate of peri-procedural complications.
Methods
We conducted an observational analytical cohort study with longitudinal follow-up using a retrospective institutional registry of patients who underwent EBUS in a tertiary care university medical center between 2017 and 2023.
Eligibility criteria
Patients over 18 years of age who underwent simultaneous r-EBUS and EBUS-TBNA were included. The procedure was indicated for patients with peripheral pulmonary lesions requiring mediastinal staging or pulmonary lesions in contact with the central airways. Patients whose histopathology samples were processed in institutions lacking certified cytopathologists trained in EBUS sample evaluation were excluded.
Procedures
The procedures were performed on an outpatient basis, assisted by two expert pulmonologists—selected from a team of four interventional pulmonologists in our service, all certified in EBUS with a minimum of 50 supervised procedures—and an anesthesiologist. Procedures were conducted under deep sedation with propofol and remifentanil using a laryngeal mask, with local lidocaine added to the airways. During the procedure, ROSE was performed for a preliminary evaluation of the obtained samples. A slide from the bronchial brushing and a touch imprint cytology from the first or second biopsy were prepared for the pathologist to quickly assess sample quality and detect malignant cells, aiding the diagnostic process. Histopathological analysis was performed by a pathologist with expertise in EBUS samples, specifically trained in respiratory pathology and with experience interpreting samples from over 50 patients who underwent EBUS. 24 The bronchoscope was introduced through the laryngeal mask in all cases using a bite block to protect the endoscope. All patients underwent four standardized sampling techniques: bronchial washing, bronchial brushing, transbronchial biopsy, and EBUS-TBNA. The sampling sequence was uniform across cases (Figure 1), beginning with bronchial brushing, followed by biopsy, bronchial washing, and finally EBUS-TBNA, which was performed to obtain samples from mediastinal lymph nodes and those pulmonary lesions in the proximity of the central airways or esophagus. TBNA of the pulmonary lesion was performed at the end of mediastinal staging, and these samples were in addition to those obtained via r-EBUS. Linear EBUS with TBNA was consistently performed after completion of the r-EBUS procedure.

Procedure flow.
Radial endobronchial ultrasound
A bronchoscope with an external diameter of 5.9 mm and a working channel of 2.8 mm was advanced as far as possible into the segmental bronchus closest to the target lesion after reviewing the pathways under the pulmonary window setting of a high-resolution chest CT (HRCT) scan. A bronchial navigation catheter with an external diameter of 2.68 mm and an internal diameter of 2.08 mm was inserted through the bronchoscope into the bronchus leading to the lesion identified by HRCT and then guided fluoroscopically to the lesion (navigation software was not used). Subsequently, a 20 MHz radial mechanical probe (Olympus UM-S20-17S) connected to EBUS (Olympus UM-S20-17S or UM-S20-20R) was introduced. Once the lesion was confirmed through r-EBUS imaging and X-ray fluoroscopy, the probe was removed while maintaining the catheter in place. Brushes and/or biopsy forceps were introduced through the catheter to obtain samples for histopathological, cytological, and microbiological studies. A minimum of five pathology samples with a diameter of about 2 mm were obtained by biopsy.
Endobronchial ultrasound-TBNA
Linear EBUS-TBNA was used for both diagnostic and staging purposes in our study. The procedure began with systematic mediastinal staging, following the standard N3 → N2 → N1 order according to tumor location. After completing the staging, TBNA samples were obtained from pulmonary lesions. During the staging of mediastinal lymph nodes, samples were taken from nodes with a short-axis diameter > 5 mm or a diameter of 3–5 mm with at least one sign of malignancy (heterogeneous density, central necrosis, rounded shape, edge enhancement, absence of the hilar structure). A scanning frequency of 7.5 MHz was used with 21G needles (Olympus NA-201SX-4021), applying a negative pressure of −15 cm H2O and inserting the needle 15–20 times within the lesion. Three to six passes were made per node, ensuring that at least two histological cores were obtained. Doppler ultrasound was utilized as necessary to identify and avoid puncturing blood vessels.
From each puncture, three types of samples were collected for analysis: a cell block using the inner stylet to release the core from the needle, which was immediately fixed in formalin; the contents of the needle were deposited on a glass cytological slide by injecting air with a syringe and then fixed; and a needle wash with saline was performed and deposited in a specimen jar for additional cytological analysis. Samples obtained from EBUS-TBNA were first deposited on histology slides for cell smears (cytology smears), followed by a cell block preserved in formalin, and finally a needle wash with normal saline for cytological analysis.
Variables
The variables considered included age, sex, comorbidities, Charlson index, forced vital capacity (FVC), forced expiratory volume in one second (FEV1), diffusion capacity of the lungs for carbon monoxide (DLCO), symptoms and indications related to EBUS, ASA score, size of the lesion (short-axis diameter of the lymph node or long-axis diameter of the pulmonary lesion), number of punctured stations, ultrasonographic characteristics of malignancy, histopathological results, performance of positron emission tomography/computed tomography (PET-CT), number of avid lymph nodes (standardized uptake value [SUV] > 2.5), and maximum SUV. Complications related to the procedure and the need for inpatient management or observation due to complications were quantified.
Diagnostic reference standard
A combined reference standard was used based on the results of histopathological and cytological findings from samples, biopsy/surgical resection findings, clinical and imaging follow-up, and postmortem analyses. True positives (TP), true negatives (TN), false positives (FP), and false negatives (FN) were defined according to the criteria described below.
TP (True Positive): Histopathology or cytology confirming a specific diagnosis (cancer or a benign condition) that was not contradicted by biopsy/surgical resection findings, clinical follow-up with diagnostic imaging for 12 months, or postmortem analysis. Diagnostic imaging findings consistent with the benign EBUS diagnosis were also considered for benign disease follow-up, with a minimum follow-up period of 12 months to confirm diagnostic stability.
TN (True Negative): For EBUS-TBNA, TN was defined as histopathology or cytology negative for cancer or other conditions, showing only lymph node tissue with a background of lymphocytes, without contradiction from biopsy/surgical resection, 12-month clinical follow-up with imaging, or postmortem analysis.
For r-EBUS used to sample pulmonary lesions, TNs were not assumed. All negative results were considered FNs, as the presence of normal tissue in a patient undergoing r-EBUS-guided biopsy of a pulmonary lesion suggests sampling from an incorrect site, thereby classifying the result as FN.
FP: Histopathology or cytology compatible with cancer, or a specific benign condition refuted by a different diagnosis from a biopsy/surgical resection, clinical follow-up with diagnostic imaging, or postmortem analysis.
FN: Nonconclusive histopathology or cytology (including reports of suspicion of malignancy) with biopsy/surgical resection, clinical follow-up with diagnostic imaging, or postmortem analysis compatible with cancer or a specific benign diagnosis. As explained earlier, all negative results in r-EBUS (without a specific benign or malignant diagnosis or with normal tissue) were considered FN. EBUS failures with an inability to perform the procedure or obtain samples were also considered FN.
In cases of silicosis diagnosis, a TP was considered if the patient had a history of exposure to causes of this condition (wood smoke, work in mining, masonry, or construction) or if the maximum SUV of the lesion on the PET-CT was <2.5 and the diagnosis of silicosis was not refuted by biopsy/surgical resection findings, clinical follow-up with diagnostic imaging, or postmortem analysis. In cases of silicosis that did not meet these conditions, the case was considered an FN.
Diagnostic success (DS) was defined as TP and TN, while diagnostic failure (DF) was defined as FP and FN. 25
Statistical analysis
Categorical variables were reported as absolute frequencies and percentages; continuous variables were reported as means ± standard deviation (SD) or medians and interquartile range (IQR: 25th–75th percentile) according to their distribution. Bivariate analyses for categorical variables were performed using the chi-square test (exact method), while for continuous variables, the Student’s t-test or Mann–Whitney U test was used, depending on their distribution. Normality was assessed using the Shapiro–Wilk test. Overall diagnostic accuracy was calculated using the equation (TP + TN)/(TP + FP + FN + TN).24,25
A logistic regression model was developed with independent variables considered relevant based on biological plausibility and previous publications associated with the outcome (p < 0.2 in bivariate analysis) to adjust for potential confounding factors. In the statistical analysis, a stepwise backward regression method was used for variable selection. It began with a saturated model that included all possible variables, eliminating the least significant variable at each step until all remaining variables were significant according to fit criteria such as the p-value.
Various performance metrics were determined, including sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), positive likelihood ratio (LR+), and negative likelihood ratio (LR-), as well as DS, diagnostic odds ratio (DOR), and number needed to diagnose (NND). The NND was interpreted as the average number of patients who needed to be evaluated for a correct diagnosis to be made in one of them.
The statistical significance of the trend in the use of combined techniques was evaluated using the Cochran–Armitage statistical test.24,25 All associations were considered statistically significant with a p-value < 0.05 (two-tailed). Statistical analyses were performed using Microsoft Excel 2017 (Microsoft Corporation, Redmond, WA, USA) and Stata version 17.0 (StataCorp LLC, College Station, TX, USA).
The reporting of this study conforms to the Standards for Reporting Diagnostic Accuracy Studies (STARD) 2015 guidelines for diagnostic accuracy studies 26 (Supplemental File).
Results
General population characteristics
A total of 309 patients were included (Figure 2), with a mean age of 67.9 years (SD: 10.97), and 50.8% (157/309) were male (Table 1). Systemic hypertension was present in 38.8% (120/309) of the patients, and the average Charlson index was 0.98 (SD: 1.13). Pulmonary function tests showed pre- and post-bronchodilator FVC values of 2.99 L (SD: 0.9) and 3.1 L (SD: 0.86), respectively, and an average DLCO of 79.43% of the predicted value (SD: 25.39). The most common symptoms leading to the detection of lesions requiring EBUS for diagnosis were cough (38.1%; 118/309) and dyspnea (22.9%; 71/309). Adenocarcinoma was diagnosed in 34.6% (107/309) of the patients, and 8.7% (27/309) had squamous cell carcinoma.
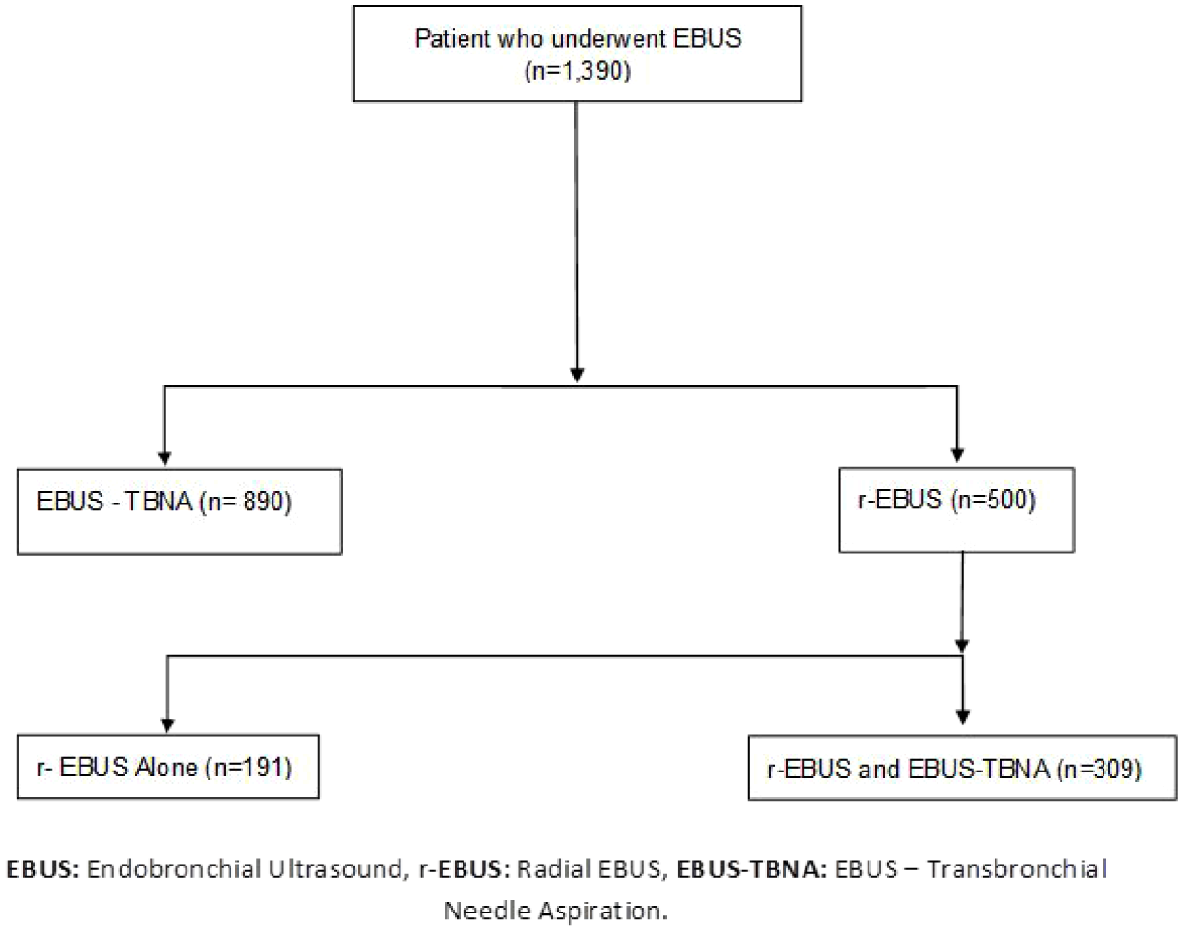
Patient flow diagram.
General characteristics of the population undergoing radial and linear EBUS.

COPD, chronic obstructive pulmonary disease; DLCO, diffusion capacity of the lungs for carbon monoxide; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; m, mean; n, number; r-EBUS, radial endobronchial ultrasound; SD, standard deviation.
Systematic additions of sampling tools
The bronchial washing had a yield of 49%, and bronchial brushing had a yield of 61%. The combination of bronchial washing and bronchial brushing improved the yield to 63%. The combination of bronchial washing, bronchial brushing, and transbronchial biopsy increased the yield to 70%, and the addition of EBUS-TBNA—providing cell smears, cell blocks, and needle wash samples—further increased the diagnostic yield to 80% (p < 0.001; Cochran–Armitage test; Figure 3). The combination of EBUS-TBNA and r-EBUS had a sensitivity of 72.5% and a specificity of 99.5%. The PPV was 99.7%, and the NPV was 59.9%. The LR+ was 131,950, and the LR- was 0.277. The DS rate was 80%, and the DOR was 477.182. The NND was 1.4 (Table 2).

Diagnostic performance of sampling tools used during Radial and Linear EBUS.
Diagnostic accuracy of radial and linear EBUS.

DOR, diagnostic odds ratio; LR+, positive likelihood ratio; LR−, negative likelihood ratio; NND, number needed to diagnose; NPV, negative predictive value; PPV, positive predictive value.
Bivariate analysis
In the DS group, a mean of 3.3 (SD: 1.44) mediastinal node stations were punctured, compared to 2.9 (SD: 1.41) in the DF group (p = 0.090) (Table 3). The mean diameter of the lesion in the DS group was 16.8 mm (SD: 8.87) compared to 13.6 mm (SD: 8.33) in the DF group (p = 0.001). In the group that underwent PET-CT, 65.9% (60/91) achieved DS, compared to 34.1% (31/91) with DF (p = 0.007). The average SUVmax of the pulmonary lesions in the DS group was 10.5 (SD: 7.05), compared to 8.5 in the DF group (SD: 5.49) (p = 0.019; Table 3).
Bivariate analysis of procedural characteristics and EBUS performance.

EBUS, endobronchial ultrasound (r-EBUS was combined with EBUS-TBNA); n, number; PET/CT, positron emission tomography; ROSE, rapid on-site evaluation; SD, standard deviation; SUVmax, maximum standardized uptake value.
Multivariate analysis
The multivariate logistic regression model adjusted for confounding factors found significant associations with diagnostic performance regarding lesion size (OR = 1.06; 95% CI: 1.01–1.14; p = 0.045) and a ROSE result suspicious for malignancy (OR = 5.78; 95% CI: 2.31–14.47; p < 0.001). The presence of chronic obstructive pulmonary disease (COPD) (OR = 0.32; 95% CI: 0.11–0.93; p = 0.037) and connective tissue disease (OR = 0.09; 95% CI: 0.01–0.95; p = 0.045) was significantly associated with DF (Table 4).
Multivariate analysis for EBUS performance.

COPD, chronic obstructive pulmonary disease; EBUS, endobronchial ultrasound (r-EBUS was combined with EBUS-TBNA); OR, odds ratio; ROSE, rapid on-site evaluation.
EBUS safety
The overall complication rate was 6.4% (20/309). The frequency of pneumothorax was 1.2% (4/309), bronchospasm was 3.8% (12/309), and bleeding occurred in 1.2% (4/309) of EBUS procedures.
Discussion
Our study analyses the impact of combining different sampling tools during r-EBUS, as well as factors related to DS and the frequency of complications associated with the procedure in a cohort of patients with pulmonary lesions suspicious of malignancy, similar to other large cohorts of patients undergoing r-EBUS.27,28 The sequential addition of bronchial washing, bronchial brushing, transbronchial biopsy, and EBUS-TBNA to r-EBUS showed a statistically significant trend in increasing DS. Variables associated with DS included lesion size and a ROSE result suspicious of malignancy. Our findings indicate that both EBUS-TBNA and r-EBUS are valuable interventional pulmonology techniques, demonstrating high diagnostic efficacy and a low risk of complications, whether used alone or in combination with various sampling tools. To our knowledge, the quantitative impact and clinical significance of incorporating additional sampling tools in patients with peripheral lung lesions requiring mediastinal staging, or in those with lesions near the central airways, have not been previously reported.4,5,10,12,29 –38 The simultaneous use of sampling tools during minimally invasive procedures in interventional pulmonology has enhanced DS.39 –42 The combination of multiple sampling techniques during r-EBUS showed a statistically significant trend in consecutive additions, making our results novel in the diagnosis and staging of pulmonary and mediastinal diseases. Therefore, the systematic incorporation of various sampling techniques by experienced pulmonologists improved DS among our cohort, even in the more distal locations of the lung parenchyma, where instrumental maneuvers were challenging and direct visualization of the target lesion was not possible.
In our cohort, all patients underwent r-EBUS as the primary diagnostic procedure for peripheral pulmonary lesions. EBUS-TBNA was used as a complementary technique in two specific scenarios: (1) for mediastinal staging when indicated, and (2) for obtaining TBNA samples from pulmonary lesions located in proximity to the central airways. This differs from the standard approach in which r-EBUS is typically used alone. The combined strategy employed in our study enabled a more comprehensive diagnostic assessment within a single procedure, improving DS while maintaining procedural safety.4,5,10,12,29 –38 This integrated approach influences procedural planning and tissue acquisition and offers practical advantages in clinical settings, particularly for therapeutic decision-making when both pulmonary lesions and mediastinal structures require evaluation. Future studies should focus on defining specific clinical scenarios where the combined approach provides the most significant benefit.
The frequency of ROSE use varies among published studies, which may contribute to the variability in reported DS.42 –48 ROSE likely improves DS by providing immediate information about sample quality.41,47,48 In our study, a ROSE result suspicious of malignancy was significantly associated with higher diagnostic performance. We found that our r-EBUS DS was good and comparable with that reported in the medical literature, and the addition of EBUS-TBNA allowed us to increase the DS to values above average.49-51 This may be related to the additional samples obtained by EBUS-TBNA, the use of a fluoroscopy guide for navigation, and the persistence in searching for the lesion when the ROSE indicated a nondiagnostic sample.
The addition of EBUS-TBNA to r-EBUS significantly improves DS because it adds a tool for fine-needle aspiration biopsy under real-time imaging of lesions in contact with the central airway.14,15,17,18,30,39 –41 This facilitates the diagnosis of pulmonary lesions adjacent to the central airway and of peripheral lesions with mediastinal involvement, where r-EBUS alone may not be sufficient. This synergy between both techniques allows for a more comprehensive and simultaneous evaluation, which is crucial for therapeutic planning and effective clinical management of patients with challenging thoracic tumors.39 –41
COPD and connective tissue disease are independently associated with DF due to airway structure alterations that may hinder the EBUS navigation catheter from adequately reaching peripheral nodules.52 –57 In COPD, the lungs undergo significant changes due to alveolar destruction and hyperinflation, which can displace pulmonary nodules from their normal location, making them more difficult to visualize and access with radial EBUS.52,53,58 In addition, changes due to parenchymal destruction, hyperinflation, and airway edema or collapse may hinder the ability to navigate the catheter through the bronchi accurately to reach peripheral pulmonary nodules.52,58 Conversely, in connective tissue diseases such as systemic sclerosis or rheumatoid arthritis, the thickening of pulmonary tissue or fibrosis can complicate the clear visualization of peripheral pulmonary nodules and make navigation through distorted bronchi difficult.54,55 Interstitial fibrosis and altered lung architecture can also interfere with the quality of images obtained via radial EBUS, making it more challenging to correctly localize nodules.55,56 The high prevalence of silicoanthracosis in our cohort is a noteworthy finding. Given its potential association with local pollution levels, we plan to conduct a dedicated study to investigate this relationship further. Additional analyses of silicoanthracosis will be included in future research.
The use of r-EBUS in the diagnosis of pulmonary and mediastinal tumors has become increasingly common due to its ability to access a wide range of anatomical locations and lymph node stations compared to more invasive techniques or those with a higher risk of adverse effects, such as surgical and percutaneous procedures.9,10,17 –19 r-EBUS and EBUS-TBNA offer less invasive procedures with good diagnostic accuracy and low complication rates. Our results showed a low frequency of complications such as pneumothorax, a high sensitivity, an elevated NPV, good DS, and low NND, reaffirming what has been described in the medical literature regarding EBUS as a safe, minimally invasive procedure for the diagnosis and staging of thoracic or extrathoracic neoplasms. We prioritized achieving a definitive diagnosis in a single procedure, which we believe is safer for the patient and likely associated with a better cost-utility balance. However, further studies are needed to explore this hypothesis.
Limitations
Among the limitations of the study is the inclusion of patients from a single hospital, which may limit external validity. However, the fact that the characteristics of our population are very similar to those in other series, along with the use of standard techniques, suggests that the impact of this limitation may not be significant. The observational nature of our study exposes it to the risk of confounding factors. Nonetheless, measures were taken to reduce bias risks, such as training personnel responsible for medical data collection, constructing the manuscript based on a checklist of items to include in cohort study reports (STROBE checklist), using standard techniques for the procedures, and adjusting for potential confounding variables in multivariate analysis. In addition, the sample size was not sufficient to perform adequately powered subgroup analyses to identify any group of patients in whom the addition of EBUS-TBNA might offer limited benefit, although such a scenario would be relevant only in cases where EBUS-TBNA is not indicated for mediastinal staging. We acknowledge this as an important area for future studies with larger sample sizes to enable adequately powered subgroup analyses.
Conclusion
The use of multiple sampling tools significantly contributes to the DS of r-EBUS, particularly with the addition of EBUS-TBNA. The variables independently associated with DS were lesion size and a ROSE result of suspected malignancy, while COPD and connective tissue disease were associated with DF. The frequency of complications, such as pneumothorax during procedures, was low.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251357699 – Supplemental material for Optimizing diagnostic yield in pulmonary lesions: impact of combined sampling tools and EBUS-TBNA during radial EBUS
Supplemental material, sj-docx-1-tar-10.1177_17534666251357699 for Optimizing diagnostic yield in pulmonary lesions: impact of combined sampling tools and EBUS-TBNA during radial EBUS by Eduardo Tuta-Quintero, Luis F. Giraldo-Cadavid, Catalina Sanmiguel-Reyes, Maria E. Navia, Ricardo Cardenas, Alirio Bastidas, Angelica Mora, Nelson Páez-Espinel, Lucía Viola, Miguel Suárez, Libardo Jiménez-Maldonado, Mauricio Durán, Jacqueline Mugnier and Javier Flandes in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

