Abstract
Background:
Radial endobronchial ultrasound (R-EBUS), virtual bronchoscopic navigation (VBN), and electromagnetic navigation bronchoscopy (ENB) are widely used bronchoscopic techniques for diagnosing peripheral pulmonary lesions (PPLs). However, the applications of their combinations remain unclear.
Objectives:
This study aimed to investigate the diagnostic performance and safety of R-EBUS versus its combination with ENB or VBN and lesion characteristics.
Design:
This study is a retrospective, single-center cohort study.
Methods:
Patients who underwent R-EBUS without and with ENB or VBN (R-EBUS+ENB, R-EBUS+VBN) for peripheral pulmonary. Diagnostic yield, sensitivity, specificity, and complications were compared using inverse probability of treatment weighting (IPTW) for baseline difference adjustment.
Results:
R-EBUS, R-EBUS+ENB, and R-EBUS+VBN groups had diagnostic yields of 74.6%, 78.2%, and 73.0%, respectively (no significant differences after IPTW adjustment). Multimodal approaches significantly improved diagnostic yield in patients with emphysematous lungs (R-EBUS vs R-EBUS+ENB: odds ratio (OR): 3.51; 95% confidence interval (CI): 1.38–8.95; p = 0.009; R-EBUS vs R-EBUS+VBN: OR: 3.14; 95% CI: 1.05–9.35; p = 0.04). R-EBUS+ENB demonstrated superior diagnostic performance in lesions ⩽20 mm (OR: 3.58; 95% CI: 1.28–9.98; p = 0.015), lesions with positive bronchial signs (OR: 1.98; 95% CI: 1.07–3.67; p = 0.029), and solid lesions with combined positive bronchial signs (OR: 2.67; 95% CI: 1.18–6.07; p = 0.019). Mild bleeding was more frequent in the R-EBUS+ENB group than in the R-EBUS group (OR: 3.21; 95% CI: 1.13–9.13; p = 0.029); severe complications did not significantly differ among groups.
Conclusion:
Comparable diagnostic performances were observed among R-EBUS, R-EBUS+ENB, and R-EBUS+VBN groups. Multimodal approaches significantly enhanced diagnostic accuracy in subtypes with lesions of small size, positive bronchial signs, or emphysematous lungs. These findings highlight the importance of tailored multimodal strategies to improve diagnostic yield and procedural safety in PPL evaluation.
Plain language summary
Why was the study done? Lung nodules are small spots in the lungs that may be signs of cancer or other conditions. Diagnosing them early and accurately is important for proper treatment. Doctors often use bronchoscopy—a procedure that uses a thin tube to collect tissue samples from the lungs. While several advanced techniques are available, it is unclear whether combining them improves diagnostic accuracy and safety. This study compared three common methods—radial endobronchial ultrasound (R-EBUS), R-EBUS with electromagnetic navigation bronchoscopy (ENB), and R-EBUS with virtual bronchoscopic navigation (VBN)—to see which worked best. What did the researchers do? The team reviewed data from 441 patients who underwent bronchoscopy between 2016 and 2024. Patients were grouped based on the method used: R-EBUS alone, R-EBUS+ENB, or R-EBUS+VBN. The researchers compared how well each method diagnosed lung nodules and examined any complications, such as bleeding or collapsed lung. What did the researchers find? All three methods had similar overall accuracy, with success rates between 73.0% and 78.2%. However, combined approaches offered better results in certain cases. Patients with emphysema had improved diagnostic outcomes when ENB or VBN was added to R-EBUS. The R-EBUS+ENB combination also performed better for small nodules (⩽20 mm) and those near the airways. Mild bleeding was slightly more common with ENB, but serious complications were rare across all groups. What do the findings mean? R-EBUS alone is effective for most patients, but combining it with ENB or VBN may improve diagnosis in specific situations—such as for patients with emphysema or small, hard-to-reach nodules. These results can help doctors choose the best diagnostic approach based on individual characteristics of the patient and lung lesion.
Keywords
Introduction
Lung cancer remains the most common type of cancer worldwide and the leading cause of cancer-related deaths. 1 As lung cancer advances to later clinical stages, treatment options have become increasingly limited. Accordingly, a timely and accurate diagnosis is crucial. The widespread use of low-dose computed tomography (CT) screening has contributed to reducing lung cancer mortality and increasing the detection of peripheral lung lesions (PPLs).2,3 However, obtaining histological diagnoses of these PPLs remains challenging. Various modalities, including transthoracic needle aspiration biopsy (TTNA) and bronchoscopy, are used to diagnose PPLs. 4 TTNA demonstrates excellent sensitivity for these lesions; however, it has significant limitations, particularly causing a higher risk of pneumothorax of 15%–25% and hemoptysis of 1%–5%.5 –7 In contrast, conventional bronchoscopy is a less invasive method that accesses the nodule through the natural bronchial lumen, reducing complications. Nonetheless, the yield of conventional transbronchial biopsies is suboptimal.8,9
In recent years, several advanced bronchoscopic procedures, such as radial endobronchial ultrasound (R-EBUS), virtual bronchoscopy navigation (VBN), and electromagnetic navigational bronchoscopy (ENB), have been developed to improve the biopsy of PPLs. 10 R-EBUS has become a widely used technique recommended by the American College of Chest Physicians (ACCP) and the European Society for Medical Oncology (ESMO) for diagnosing PPLs.11,12 Although R-EBUS can confirm biopsy sites, it does not provide navigational guidance for bronchoscopy. Therefore, using R-EBUS alone may leave the lesion inaccessible in 8%–20.8% of cases.13,14 Therefore, other advanced guidance techniques, including VBN and ENB, have been introduced for PPL diagnosis. ENB is an image-guided bronchoscopic technique based on electromagnetic navigation. By leveraging information from CT scans and electromagnetic sensors, ENB reconstructs the tracheobronchial tree and visualizes the airway, leading to the target lesion. VBN generates virtual bronchoscopic images of the bronchial pathway to guide the bronchoscope toward peripherally located lesions. 10 Studies suggest that multimodal approaches combining R-EBUS with VBN or ENB may outperform R-EBUS alone in diagnosing PPL, as these techniques address R-EBUS’s navigational limitations.15,16 Nevertheless, a recent meta-analysis of 46 prospective trials showed that the diagnostic rate of 75.6% for R-EBUS combined with VBN or ENB was not significantly different from the 71.9% diagnostic rate for R-EBUS alone. 17 However, VBN and ENB are associated with significantly higher costs than R-EBUS. 18 Therefore, it is crucial to identify lesion characteristics that could help the combination of R-EBUS and ENB/VBN achieve an accurate diagnosis.
In recent years, newer technologies, such as robot-assisted bronchoscopy and other advanced navigation platforms, have emerged, offering improved precision and reach for PPLs. However, their broader adoption remains constrained by requirements for dedicated equipment, operator training, and procedural integration. Although these technologies were introduced at our center, they were not routinely implemented during the study period. This study aimed to evaluate the diagnostic performance and safety of R-EBUS versus its combination with ENB or VBN, which continue to represent widely used and clinically relevant strategies in current practice. We employed inverse probability of treatment weighting (IPTW) based on lesion characteristics across the three patient groups to minimize selection bias and control for preoperative confounders.
Methods
Study design and participants
This retrospective, single-center study included 441 patients with suspected malignant lung lesions who required pathological evaluation. Between February 2016 and June 2024, these patients underwent R-EBUS, R-EBUS combined with ENB, or R-EBUS combined with VBN at the West China Hospital of Sichuan University, a university-affiliated tertiary care center. Patients with suspected malignant pulmonary nodules who required a bronchoscopic biopsy were included in this study. The exclusion criteria were as follows: (1) bronchoscopy for nondiagnostic purposes, (2) unsuccessful bronchoscopic approach to the lung nodule, and (3) incomplete or missing information on medical history or follow-up. This study was approved by the Institutional Review Board of West China Hospital, Sichuan University (Approval No. 2023 [2315]), and the requirement for informed consent was waived due to its retrospective design. The reporting of this study conformed to the STARD 2015 guidelines for studies on diagnostic accuracy. 19 A complete STARD checklist is provided in Supplemental Material 2.
Bronchoscopic procedures
All patients underwent preprocedural chest CT. Before each procedure, the bronchoscopist determined whether to perform a biopsy using R-EBUS alone or in combination with ENB or VBN. All procedures were performed in the inpatient and outpatient settings under moderate sedation by one of four experienced bronchoscopists, each with relevant professional qualifications and more than 3 years of independent procedural experience. Intravenous midazolam and fentanyl were administered by a trained nurse based on the clinical judgment of the attending pulmonologist. When R-EBUS was combined with ENB, the SuperDimension Navigation System (SD300LG; Medtronic, Minneapolis, MN, USA) was used for navigation, followed by radial probe confirmation of the lesion location. When R-EBUS is combined with VBN, the probe is inserted into the endoscopic channel and guided by a virtual bronchoscopic image to verify the position of the lesion and locate the targeted nodule. For R-EBUS procedures, a 1.4-mm UM-S20-17S probe (Olympus Corporation, Tokyo, Japan) was used as the ultrasound probe. The choice of the flexible bronchoscope (BF-1TQ290; Olympus, Tokyo, Japan) depended on the size and location of the lesion. The use of additional techniques, such as rapid on-site evaluation, was determined by an attending pulmonologist.
Lesion characteristics and endpoints
Clinical data were collected and analyzed from medical records, including demographic characteristics, radiological findings, surgical reports, pathological results, final diagnosis of biopsied lesions, and surgery-related complications. The reviewed clinical data included age, sex, and history of smoking. Chest CT scans obtained within 1 month before bronchoscopy were evaluated to determine the location and characteristics of the lungs. Lesion size was measured as the largest diameter on the axial CT section. Lesion locations were classified according to distance from the hilum into two groups: the inner group (lesions in the inner or middle third of the ellipse) and the outer group (lesions in the outer third of the ellipse). 20 Lesion structures were categorized as solid or subsolid (partially solid or pure ground glass). The background lung parenchyma was described as normal lung, emphysema, or interstitial lung abnormalities (ILA) based on imaging findings. Emphysema was defined as the presence of low-attenuation areas lacking visible lung walls,21,22 and ILA was identified by reticular or reticular-nodular opacities, honeycombing, and fibrosis. 23 Bronchial signs on CT were graded as follows: grade 0 (no bronchial signs), grade 1 (airways immediately adjacent to the lesion), and grade 2 (airways directly aligned with the lesion). 24
The primary endpoint was the overall diagnostic yield, defined as the proportion of cases in which the bronchoscopic diagnostic results matched the final diagnosis established during the index procedure or follow-up. This approach was guided by the recent ATS/Chest Delphi consensus research statement, 25 although we also incorporated follow-up results to confirm benign diagnoses in cases with initial nonspecific pathology, consistent with previous studies. 26 Specifically, a procedure was considered diagnostic if it resulted in either a specific malignant or benign diagnosis during bronchoscopy or a benign outcome confirmed by clinical and radiologic follow-up. Cases with nonspecific inflammation or nondiagnostic pathology were not considered diagnostic unless follow-up confirmed a benign disease. Diagnostic yield was calculated as the number of diagnostic cases divided by the number of patients who underwent an attempted biopsy. A lesion was considered malignant if histological or cytological specimens showed tumor cells. A lesion was considered benign if pathological evaluation showed specific benign features and/or positive microbiological findings. For lesions without a definitive pathological diagnosis, clinical and imaging follow-ups were conducted for at least 6 months to determine the lesion status. False-negative cases were defined as follows: (1) malignancy confirmed by repeat biopsy or surgery, (2) lesion growth observed on follow-up imaging, (3) initiation of treatment for presumed lung cancer without pathological confirmation, or (4) malignancy diagnosed at a different site during subsequent clinical evaluation. True-negative cases were defined as follows: (1) subsequent diagnostic testing confirming a nonmalignant condition, (2) spontaneous regression without lung cancer treatment, or (3) absence of lesion progression during follow-up imaging. The secondary endpoints included sensitivity, specificity for malignancy, and procedure-related complication rates. Safety events, including bleeding and pneumothorax, were graded according to the Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. 27 Severe bleeding was defined as bleeding requiring invasive intervention, such as blood transfusion. Pneumothorax was confirmed by chest radiography performed immediately after the procedure and 24 h later.
Statistical analysis
Baseline patient and lesion characteristics were described as median (interquartile range) or mean (±standard deviation) for continuous variables and as frequency (percentage) for categorical variables. Patients were categorized into the R-EBUS, R-EBUS+ENB, and R-EBUS+VBN groups for analysis. Given the observational design of this study, many baseline characteristics may have differed among the three groups. To address potential confounding factors, we used IPTW, a method widely applied in clinical retrospective studies.28 –30 Propensity scores were estimated using a multivariable logistic regression model with baseline covariates as predictors and each group assignment as the outcome variable. The IPTW was implemented using the R package ipw (version 1.0-11; CRAN, Vienna, Austria). For IPTW, patient weights were calculated as 1/(probability of receiving R-EBUS alone) for patients receiving only R-EBUS and 1/(1 − probability of receiving R-EBUS alone) for patients not receiving R-EBUS alone. Stabilized weights were obtained by multiplying the raw weights by the marginal probability of receiving each treatment group. In addition, the weights below the 1st percentile or above the 99th percentile were trimmed to reduce the effects of extreme values. Covariate balance was assessed using absolute standardized differences (ASDs), with values ⩽0.1 indicating adequate balance.31,32 Outcomes were deemed comparable if no between-group differences were observed for all covariates. The main outcomes analyzed were the diagnostic performance and safety of R-EBUS, R-EBUS+ENB, and R-EBUS+VBN. In addition, we performed subgroup analyses to compare the effectiveness of these three biopsy navigation methods for lung lesions with different characteristics. All results are reported as point estimates with 95% confidence intervals (CIs), and p-values of <0.05 were considered statistically significant. More details are provided in the Supplemental Methods. Statistical analyses were performed using the R software (version 4.3.2; R Foundation for Statistical Computing, Vienna, Austria). All statistical procedures were reviewed and confirmed by a member of the research team with formal biostatistics training.
Results
Patient characteristics before and after IPTW
During the study, 492 patients underwent R-EBUS alone or in combination with ENB/VBN. After applying the exclusion criteria, 441 eligible participants remained and were divided into three groups according to the biopsy navigation method: 173 in the R-EBUS alone group, 173 in the R-EBUS+ENB group, and 89 in the R-EBUS+VBN group. The baseline clinical and radiological characteristics of all patients and the differences among the three groups after IPTW adjustment are presented in Table 1. Following IPTW adjustment, all ASDs for baseline covariates fell to ⩽0.1, confirming the successful elimination of between-group differences (Table 1). The distribution of the procedures performed during the study is shown in Supplemental Figure 4. Procedural volumes gradually increased over time, reflecting evolving clinical practice patterns.
Preprocedural demographic and radiologic characteristics among the R-EBUS group, the R-EBUS+ENB group, and the R-EBUS+VBN group before and after IPTW adjustment.
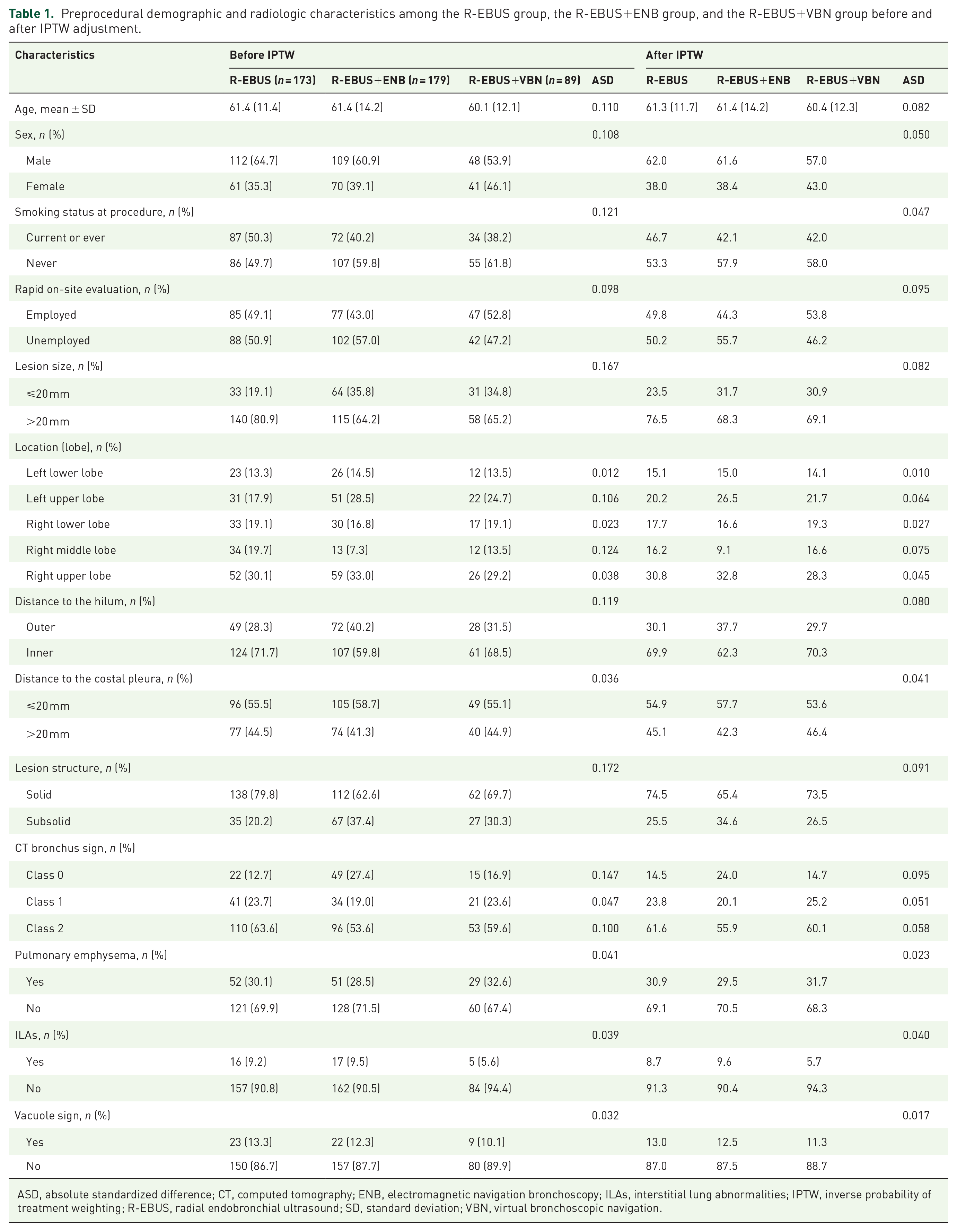
ASD, absolute standardized difference; CT, computed tomography; ENB, electromagnetic navigation bronchoscopy; ILAs, interstitial lung abnormalities; IPTW, inverse probability of treatment weighting; R-EBUS, radial endobronchial ultrasound; SD, standard deviation; VBN, virtual bronchoscopic navigation.
Diagnostic yield and safety profiles
The diagnostic results are shown in Figure 1. Among the 441 patients, 175 malignant tumors were identified using the index procedure. After follow-up, of the 266 cases initially classified as nonmalignant, 159 (36.1%) were confirmed as true negatives, and 107 (24.3%) were deemed false negatives. The overall diagnostic yields were 74.6% (129/173), 78.2% (140/179), and 73.0% (65/89) for the R-EBUS-alone, R-EBUS-ENB, and R-EBUS-VBN groups, respectively. After IPTW adjustment, no significant differences were observed in overall diagnostic rates between R-EBUS alone and the multimodal groups (IPTW R-EBUS-alone vs R-EBUS+ENB: odds ratio (OR): 1.58; 95% CI: 0.92–2.69; p = 0.095; R-EBUS-alone vs R-EBUS+VBN: OR: 0.92; 95% CI: 0.50–1.69; p = 0.795). Similarly, no significant differences were observed in the sensitivity and specificity of diagnosing malignant lesions between the combined and R-EBUS-alone groups (Tables 2 and 3; Supplemental Figures 1 and 2).

Details of bronchoscopic findings and final diagnoses.
Primary and secondary outcomes in the R-EBUS and R-EBUS combined with ENB groups before and after the IPTW adjustment.

Referred to the R-EBUS group.
95% CI, 95% confidence interval; CTCAE, Common Terminology Criteria for Adverse Events; ENB, electromagnetic navigation bronchoscopy; IPTW, inverse probability of treatment weighting; OR, odds ratio; R-EBUS, radial endobronchial ultrasound.
Primary and secondary outcomes in the R-EBUS and R-EBUS combined with VBN groups before and after the IPTW adjustment.
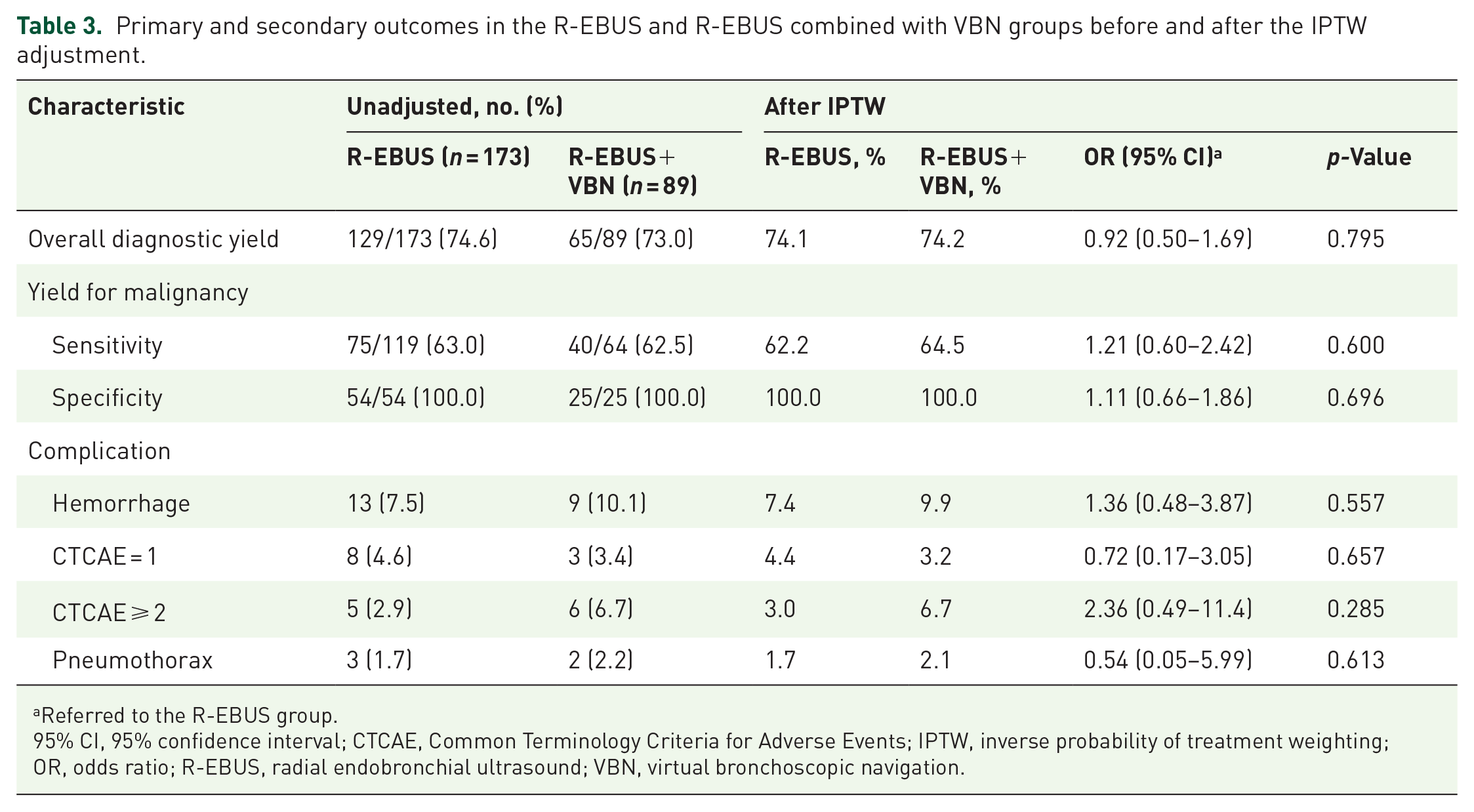
Referred to the R-EBUS group.
95% CI, 95% confidence interval; CTCAE, Common Terminology Criteria for Adverse Events; IPTW, inverse probability of treatment weighting; OR, odds ratio; R-EBUS, radial endobronchial ultrasound; VBN, virtual bronchoscopic navigation.
Based on the IPTW analysis, the R-EBUS+ENB group showed a higher risk of mild bleeding than the R-EBUS-alone group (IPTW OR: 3.21; 95% CI: 1.13–9.13; p = 0.029) but did not exhibit an increased risk of moderate or severe bleeding (IPTW OR: 1.95; 95% CI: 0.66–5.76; p = 0.226). No significant differences were observed in the bleeding risk between the R-EBUS+VBN group and R-EBUS-alone group and in the risk of pneumothorax between the combined ENB/VBN group and the R-EBUS-alone group (IPTW R-EBUS-alone vs R-EBUS+ENB: OR: 1.50; 95% CI: 0.44–5.06; p = 0.515; R-EBUS-alone vs R-EBUS+VBN: OR: 0.54; 95% CI: 0.05–5.99; p = 0.613; Tables 2 and 3; Supplemental Figures 1 and 2).
Subgroup analysis
Subgroup analysis showed that multimodal approaches achieved a higher diagnostic yield in emphysematous lungs than R-EBUS alone (IPTW R-EBUS-alone vs R-EBUS+ENB: OR: 3.51; 95% CI: 1.38–8.95; p = 0.009; R-EBUS-alone vs R-EBUS+VBN: OR: 3.14; 95% CI: 1.05–9.35; p = 0.04; Figure 2; Supplemental Figure 3). R-EBUS+ENB was more effective in diagnosing lesions of ⩽20 mm in size (IPTW OR: 3.58; 95% CI: 1.28–9.98; p = 0.015), those with positive bronchial signs (IPTW OR: 1.98; 95% CI: 1.07–3.67; p = 0.029), and solid lesions combined with positive bronchial signs (IPTW OR: 2.67; 95% CI: 1.18–6.07; p = 0.019), resulting in a significantly higher diagnostic yield than the R-EBUS-alone group (Figure 2). However, these findings should be interpreted with caution due to the small sample sizes of some subgroups.
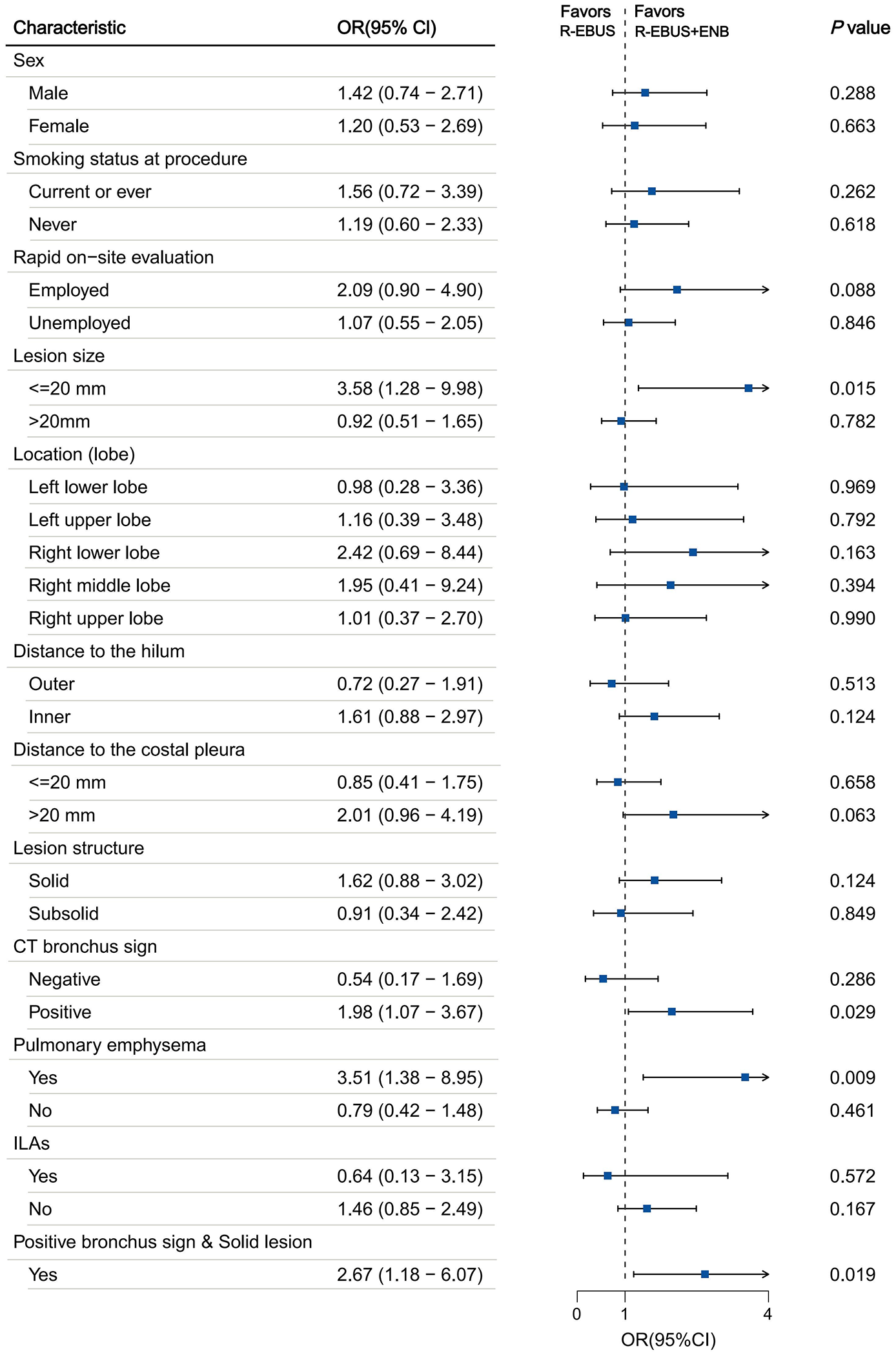
Subgroup analysis comparing the diagnostic performance of the R-EBUS group versus the R-EBUS combined with ENB group. Odds ratios and 95% confidence intervals were referenced to R-EBUS.
Discussion
This study comprehensively evaluated the diagnostic performance and safety of R-EBUS and its combination with ENB or VBN for PPL diagnosis. Although no significant differences in the overall diagnostic rates were observed between the groups after IPTW adjustment, our results suggest that multimodal approaches combining R-EBUS with VBN or ENB may enhance diagnostic efficiency, particularly with emphysema in the background lung. In addition, the combination of R-EBUS and ENB demonstrated superior diagnostic performance in specific subgroups, including those with ⩽20 mm lesions, lesions with positive bronchial signs, and solid lesions exhibiting combined bronchial signs. These findings highlight the potential value of tailored multimodal strategies in addressing challenging PPL cases.
Over the past decade, advanced bronchoscopic techniques have revolutionized the diagnostic landscape of PPL. R-EBUS, as a cornerstone tool, provides real-time confirmation of the lesion location during biopsy. However, its inherent limitation, the lack of intrinsic navigational capability, can reduce its efficacy for identifying peripheral or small-sized lesions. Navigation-based technologies, such as ENB and VBN, have been developed to address this limitation, enabling precise lesion localization via preprocedural CT imaging pathways.33,34 Both observational studies and recent randomized trials have demonstrated that combining R-EBUS with a navigation system, such as ENB or VBN, can enhance diagnostic accuracy.15,35,36 Despite these advancements, combined approaches are associated with higher costs, longer procedural times, and additional training requirements. 18 In our center, implementing ENB or VBN requires additional capital equipment, dedicated navigation systems, and specialized staff coordination, contributing to greater procedural complexity and institutional resource demands relative to R-EBUS alone. Furthermore, meta-analyses and prospective studies have yielded inconsistent results, with some failing to find significant superiority of multimodal approaches over R-EBUS alone.17,37,38 These discrepancies underscore the need for further research to identify specific scenarios or lesion characteristics that most likely benefit from adding navigational modalities, such as VBN or ENB. Given these discrepancies, our study aimed to determine whether combining R-EBUS with VBN or ENB provides distinct advantages for specific lesion characteristics while maintaining a favorable safety profile.
Our study found no significant differences in the overall diagnostic rates, malignant lesion sensitivity, or specificity among the three groups. However, the tendency to prioritize multimodal approaches for technically challenging cases likely influenced these findings. Surgeons often select ENB or VBN for more complex lesions, potentially underestimating the broader utility of these approaches in routine practice. Meanwhile, the R-EBUS+ENB group showed a higher incidence of mild bleeding complications than other groups, which could be attributed to the additional tissue manipulation required during multimodal procedures.
Our subgroup analysis revealed that multimodal approaches achieved a higher diagnostic rate for lesions in the emphysema-dominated lungs than a monomodal approach. Emphysematous lungs are characterized by a distorted anatomy and complicated bronchoscopic navigation. In such cases, the enhanced mapping provided by ENB and VBN likely overcomes these structural challenges, allowing more precise lesion targeting. Moreover, R-EBUS-ENB demonstrated superior diagnostic performance in identifying smaller lesions and those with positive bronchial signs. The enhanced localization capabilities of ENB electromagnetic-guided navigation allow for accurate access to peripheral or less visible lesions, improving diagnostic accuracy. Similarly, lesions with positive bronchial signs that align with identifiable airway structures benefit from the precision of advanced navigational modalities, further supporting the advantages of a multimodal approach under these circumstances. Similar findings have been observed in recent studies evaluating combined navigational strategies. 39 Although we also examined other lesion characteristics, including density and anatomical location, no significant differences in diagnostic performance were observed between R-EBUS alone and multimodal strategies. This may be partly due to the relatively small sample size, which might limit the ability to detect true differences. Further investigations with larger cohorts are warranted to clarify whether these lesion features influence the effectiveness of multimodal approaches.
In addition to the techniques evaluated in this study, new bronchoscopy technologies have emerged in recent years. Developing newer technologies, such as robot-assisted bronchoscopy and other advanced navigation platforms, offers greater precision in targeting peripheral lesions. These technologies have been introduced at our institution in recent years and are currently being explored for selected clinical applications. Because these technologies were not routinely implemented during the study, they were excluded from the analysis. We plan to conduct future prospective studies to evaluate the diagnostic performance, safety, and cost-effectiveness of these emerging technologies compared to conventional strategies. Our findings apply to institutions where traditional navigation methods remain the primary diagnostic approaches.
This study had limitations. First, as a single-center retrospective analysis, even with IPTW adjustment to control for observed confounders, the potential for selection bias, such as physician preferences, limits the generalizability of the study’s findings to other clinical settings. In addition, excluding cases where bronchoscopic access to the lesion was unsuccessful may have overestimated the diagnostic yield, introducing another form of selection bias that should be considered when interpreting our results. Second, the small sample sizes of certain subgroups may have reduced the statistical power of these analyses, warranting caution when interpreting these results. Third, operator experience, a critical determinant of bronchoscopic success, was not explicitly considered, potentially introducing variability in the outcomes. In addition, because procedure times were not uniformly documented in this retrospective dataset, we were unable to conduct a meaningful comparison of the time requirements across different biopsy strategies. Finally, as the study spanned several years, technological advances and evolving clinical practices might have influenced procedural strategies and outcomes. Future research should prioritize prospective multicenter studies with standardized protocols and larger cohorts to validate these findings and refine the indications for multimodal bronchoscopic strategies while assessing cost-effectiveness and integrating emerging technologies, such as robotic-assisted bronchoscopy, into diagnostic algorithms for PPLs.
Conclusion
This study indicates that R-EBUS alone may be sufficient for relatively simple lesions, providing comparable diagnostic performances to combined modalities while avoiding the additional costs and complexity associated with ENB or VBN. In contrast, multimodal approaches could offer superior diagnostic yields in more complex scenarios, such as smaller lesions, emphysematous lungs, and lesions exhibiting positive bronchial signs. These findings highlight the importance of adopting lesion-specific diagnostic strategies to enhance the effectiveness and efficiency of bronchoscopy. Future studies should further evaluate the cost-effectiveness of these technologies and help define more precise indications for their use to optimize clinical and economic outcomes.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666251355130 – Supplemental material for Comparative diagnostic performance and safety of radial endobronchial ultrasound versus its combination with electromagnetic or virtual bronchoscopic navigation for peripheral pulmonary lesions: a retrospective study
Supplemental material, sj-pdf-1-tar-10.1177_17534666251355130 for Comparative diagnostic performance and safety of radial endobronchial ultrasound versus its combination with electromagnetic or virtual bronchoscopic navigation for peripheral pulmonary lesions: a retrospective study by Qi Qi, Wanqin Fang, Linhui Yang, Yi Liu, Rui Xu and Dan Liu in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-pdf-2-tar-10.1177_17534666251355130 – Supplemental material for Comparative diagnostic performance and safety of radial endobronchial ultrasound versus its combination with electromagnetic or virtual bronchoscopic navigation for peripheral pulmonary lesions: a retrospective study
Supplemental material, sj-pdf-2-tar-10.1177_17534666251355130 for Comparative diagnostic performance and safety of radial endobronchial ultrasound versus its combination with electromagnetic or virtual bronchoscopic navigation for peripheral pulmonary lesions: a retrospective study by Qi Qi, Wanqin Fang, Linhui Yang, Yi Liu, Rui Xu and Dan Liu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
