Abstract
Extracorporeal membrane oxygenation (ECMO) assists blood circulation and gas exchange via a heart–lung machine. ECMO is used mainly in intensive care units as bridging therapy until heart and respiratory failure can be addressed or until transplantation can be performed. ECMO is sometimes used during surgery under general anaesthesia, depending on the patient’s underlying diseases and the nature of the operation. If the oxygen supply and carbon dioxide removal capacity are limited, venovenous (VV)-ECMO can be helpful. Here, we describe the use of VV-ECMO for surgical resection of an endotracheal mass through rigid bronchoscopy in a patient who developed decompensating dyspnoea due to central airway obstruction (CAO).
Keywords
Introduction
Central airway obstruction (CAO) refers to obstruction of the trachea and main bronchi causing dyspnoea, suffocation and complications, such as atelectasis and pneumonia. 1 Patients with severe respiratory distress due to CAO require immediate treatment. Generally, surgical treatment of endotracheal lesions involves bronchoscopy; however, general anaesthesia for these patients is challenging. Because CAO makes conventional endotracheal intubation and mechanical ventilation difficult, alternatives should be considered. Additionally, the anaesthesiologist must supply adequate oxygen and remove carbon dioxide during surgery. During endotracheal procedures, temporary extubation and apnoea may occur. Therefore, it is necessary that the anaesthesiologists understand these procedures fully and communicate sufficiently with the surgeon.
When adequate ventilation during anaesthesia is impossible with conventional methods, extracorporeal membrane oxygenation (ECMO) is a useful alternative. ECMO has been used successfully in patients under anaesthesia requiring airway surgery and in those with tracheal obstruction.2–4 We successfully induced general anaesthesia, using venovenous (VV)-ECMO as alternative ventilation support, in a patient who underwent surgical resection of an endotracheal mass that caused CAO.
Case Report
A 70-year-old man (height, 170.7 cm; weight, 74.8 kg; American Society of Anesthesiologists physical status, IV) was admitted with a severe cough and dyspnoea. He had a history of diabetes mellitus and had received palliative chemotherapy for non-small cell lung cancer, stage 4 (Figure 1). The initial arterial blood gas analysis (ABGA) showed an arterial pH of 7.43, partial pressure of arterial carbon dioxide (PaCO2) of 5.1 kPa, partial pressure of arterial oxygen (PaO2) of 12.4 kPa and bicarbonate (HCO3) level of 25.2 mmol/L. Flexible bronchoscopy and chest radiography revealed a mass blocking most of the tracheal lumen (Figure 2). Despite high-flow oxygen therapy using an AIRVO™ device (Fisher & Paykel Healthcare, Auckland, New Zealand), delivered via a nasal cannula at a rate of 50 L/min, with concurrent treatment with an antitussive expectorant, steroids and a bronchodilator, his dyspnoea quickly worsened. Nine days after admission, ABGA showed hypercapnoea and hypoxaemia (Table 1). The patient was then transferred to the intensive care unit (ICU) and scheduled for surgical treatment for the dyspnoea after conservative treatment failed. The otorhinolaryngologist decided to remove the endotracheal mass using rigid bronchoscopy under general anaesthesia. However, considering the location and size of the tracheal mass, there was a risk of insufficient ventilation causing hypoxaemia during anaesthesia induction. In similar cases, because conventional endotracheal intubation could interfere with the procedure, anaesthesiologists and surgeons should consider these issues. Accordingly, we decided to use VV-ECMO before general anaesthesia following a multidisciplinary consultation involving anaesthesiologists, pulmonologists, otorhinolaryngologists and cardiothoracic surgeons. A cardiothoracic surgeon cannulated each of the patient’s femoral veins using a 21-Fr and 23-Fr catheter, respectively, and initiated VV-ECMO in the ICU. The main parameters of the VV-ECMO were set as follows: blood flow velocity: 4.61 L/min, speed: 1885 rpm and gas flow: 4.5 L/min. Thirty minutes after starting ECMO, the patient’s pulse oxygen saturation was 96%, and the arterial oxygen saturation was 99%. After confirming adequately maintained oxygenation, the patient was transferred to the operating room.
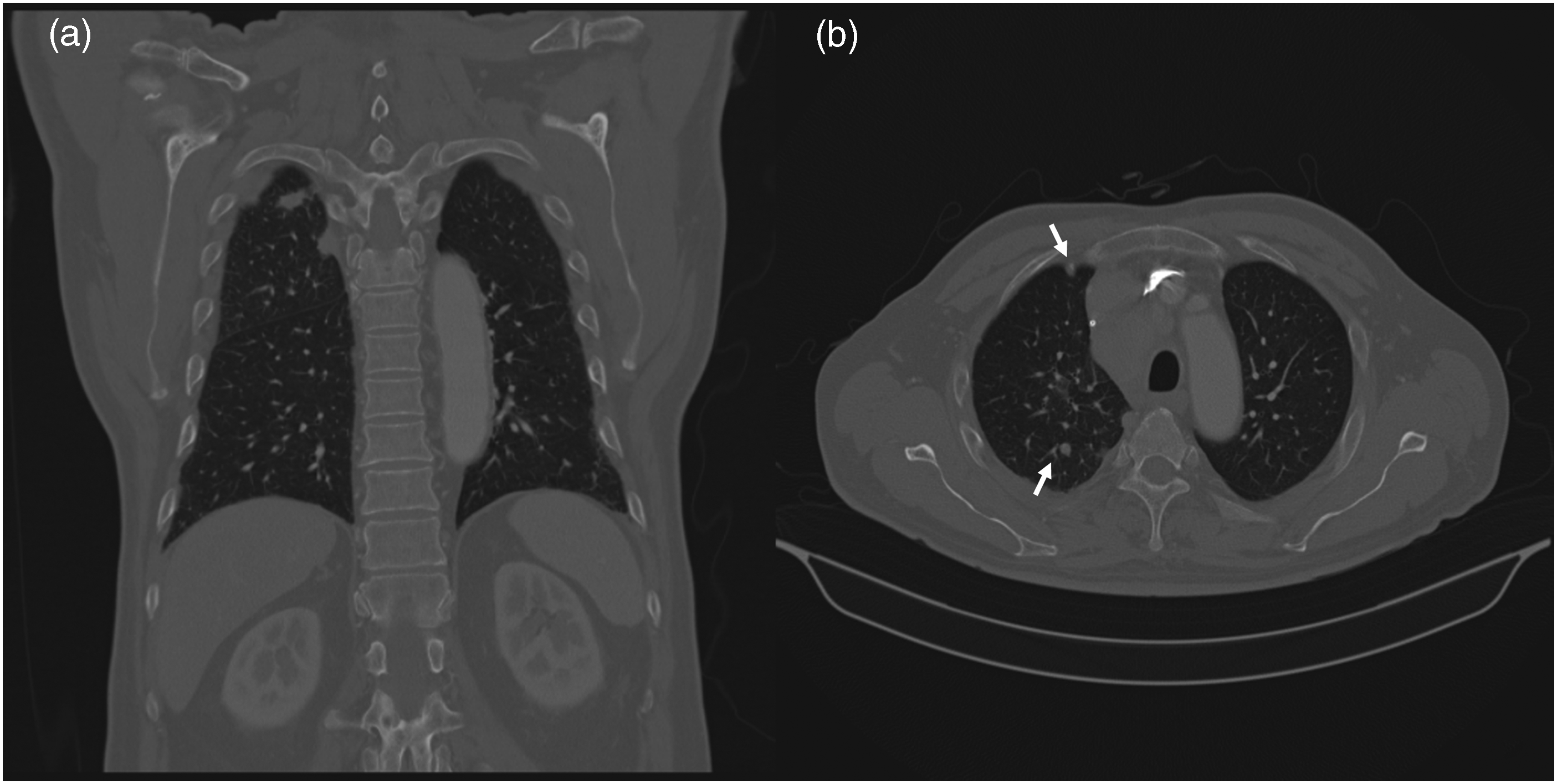
Computed tomography images 2 months prior to surgery (a) A 1.6-cm lung cancer mass (white arrow) is visible in the right apical area and (b) multiple nodules (white arrows) are also visible.

Preoperative examination findings (a) chest radiograph showing an endotracheal mass (arrows) above the carina and (b) flexible bronchoscopy revealed a mass obstructing most of the tracheal lumen.
Perioperative arterial blood gas analysis results.

pH, arterial pH; PaCO2, partial pressure of arterial carbon dioxide; PaO2, partial pressure of arterial oxygen; SaO2, arterial oxygen saturation; FiO2, fraction of inspired oxygen; Admission, the day of admission; 9th day, 9 days after admission; After ECMO, 30 minutes after initiating VV-ECMO; PostOP: 1 day after the operation; VV-ECMO, venovenous extracorporeal membrane oxygenation.
On arrival in the operating room, non-invasive blood pressure monitoring, three-lead electrocardiography, pulse oximetry and bispectral index monitoring were initiated. Then, the arterial and central venous pressures were monitored. The initial blood pressure was 150/92 mmHg, while the heart rate was 113 beats/min and oxygen saturation was 88%. While on VV-ECMO and while receiving supplemental high-flow oxygen at 5 L/min, general anaesthesia was induced with 2% propofol, remifentanil and rocuronium. The propofol and remifentanil were administered via an infusion pump (Orchestra® Workstation; Fresenius Kabi, Bad Homburg, Germany), using target-controlled infusion. Then, endotracheal intubation was attempted using a McGrath MAC (Medtronic, Dublin, Ireland) video laryngoscope and 5.0-mm inner diameter tube. However, the tube did not pass beyond 23 cm because of the endotracheal mass. After tracheal intubation, mechanical ventilation was initiated (pressure control mode; tidal volume, 500 mL; respiratory rate, 12/min; fraction of inspired oxygen (FiO2), 0.4). However, at 130 to 150 mL, the tidal volume was significantly lower than the pre-set value. During anaesthesia induction, the patient’s pulse oxygen saturation was 88% despite continuous ECMO at 5 L/min. We suspected low-flow ECMO, and the cardiothoracic surgeon adjusted the inflow cannula under ultrasound guidance. Thereafter, the pulse oxygen saturation recovered gradually to 95% and was maintained stably without mechanical ventilation soon after. Anaesthesia was maintained with continuous intravenous infusion of 2% propofol and remifentanil (target concentration infusion). After anaesthetic induction, the endotracheal tube was removed to insert a rigid bronchoscope. The mass was seen through the rigid bronchoscope and was removed using forceps (Figure 3). After confirming that the mass had been removed without immediate complications, the patient was reintubated using an 8.0-mm inner diameter endotracheal tube. Total anaesthesia time was 93 minutes, and the operating time was 32 minutes. The patient was transferred to the ICU postoperatively, weaned from ECMO 2 days later and extubated 7 days postoperatively. The excised endotracheal mass was diagnosed pathologically as malignant adenocarcinoma.

Intraoperative findings (a) rigid bronchoscopy revealed an endotracheal mass; (b) the carina was visible after mass removal and (c) the excised mass.
This report conformed with the CARE guidelines. 5
Discussion
We successfully used ECMO with general anaesthesia for the surgical treatment of severe respiratory distress caused by CAO. In this patient, most of the tracheal lumen was obstructed, so we anticipated that sufficient ventilation with intubation and mechanical ventilation would be impossible. We started ECMO before general anaesthesia, and the operation was completed safely while supplying sufficient oxygen to the patient.
Although there are various causes of CAO, the main cause is lung malignancy. Airway obstruction develops in 20% to 30% of patients with lung cancer. 6 CAO is also caused by metastasis of thyroid, breast and colon cancer. 7 Non-malignant diseases that can cause endotracheal lesions are lymphadenopathy, sarcoidosis, iatrogenic tracheal stenosis after intubation or tracheostomy and infectious diseases. 1 Safe and effective treatments for CAO are airway dilatation, surgical resection (core-out), laser therapy and stent insertion via bronchoscopy. 8 In our patient, the mass that caused CAO was surgically removed using rigid bronchoscopy, which is usually performed under general anaesthesia. For surgery in cases with endotracheal lesions, intravenous anaesthetics with sufficient neuromuscular blockade are helpful. 9 During the surgical treatment of endotracheal lesions, anaesthesiologists must consider unconventional ventilation strategies because an endotracheal tube interferes with surgical access. For a short operation, the patient can tolerate apnoea with temporary extubation or maintain spontaneous breathing under moderate sedation. 10 As an example, jet ventilation, which supplies high-pressure oxygen through a nozzle, can be used effectively because it can supply oxygen to the patient without endotracheal intubation. 11 However, despite these ventilation strategies, if the remaining lumen is too narrow owing to significant CAO, the oxygen supply may be insufficient. In such situations, ECMO can assist patient ventilation.
While previous guidelines for difficult airway management did not mention ECMO,12,13 recent guidelines issued by the American Society of Anesthesiologists suggested ECMO as an alternative invasive intervention. 14 Although the use of ECMO for airway management is uncommon, it should be considered in situations where the benefits outweigh the potential harm. In a case report published in 2020, ECMO was used successfully for the surgical removal of airway lesions to provide adequate oxygenation. 15 When conventional ventilators cannot ensure adequate oxygenation and removal of carbon dioxide, anaesthesiologists should consider ECMO because of its usefulness and safety. Another important consideration when using ECMO is the need to monitor the equipment continuously to ensure that it is functioning properly. Hypoxaemia and impaired oxygen delivery can occur in patients receiving VV-ECMO, which can be lethal in decompensating patients. 16 In our case, temporary hypoxaemia (<90% pulse oxygen saturation) may have been induced by unintentional repositioning of the inflow cannula while transferring the patient from the ICU to the operating room. Nevertheless, practitioners with sufficient knowledge of the ECMO equipment and the patient’s cardiopulmonary function were on standby, and the cause of the problem was immediately identified and resolved. Therefore, it is necessary to not only monitor the equipment function carefully, but also to ensure that human and material resources are on hand to respond immediately should a problem arise. In this case, the medical staff who participated in the operation decided to transfer the patient from the intensive care unit to the operating room after initiating ECMO in the ICU. However, there was a risk that unexpected cannula malpositioning or mechanical malfunction in the medical equipment could occur while the patient was being transported. Considering these risks, it could be safer to initiate ECMO after the patient arrives in the operating room instead of the intensive care unit.
In conclusion, ECMO is a useful alternative for ventilation support in patients with severe CAO. When using ECMO, it is necessary to monitor the oxygen supply and carbon dioxide removal carefully, and personnel and equipment must be available immediately in the case of a mechanical malfunction.
Footnotes
Author contributions
ML designed the report and wrote the manuscript; HCJ and YS reviewed the literature and interpreted the data; JJ and WSC were the anaesthesiologists and reviewed the manuscript; YHC collected the data and BSK revised the manuscript and supervised the work. All authors read and approved the final manuscript.
Data availability statement
The data in this study are available from the medical records of Soonchunhyang University Bucheon Hospital. The data are also available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement and informed consent
Written informed consent was obtained from the patient for the publication of this report and any accompanying images. This study was approved by the Soonchunhyang University Bucheon Hospital Institutional Review Board (approval no. SCHBC 2021-12-013).
Funding
The authors disclosed receipt (pending publication) of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Soonchunhyang University Research Fund.
