Abstract
Background:
Pulmonary function tests offer crucial parameters for evaluating lung health and predicting clinical outcomes. Hyperlipidemia, a prevalent metabolic disorder, has been linked to declining pulmonary function. Statins are an essential therapy for lowering lipid levels in hyperlipidemia.
Objectives:
This study aims to investigate the therapeutic potential of statins in mitigating the decline in pulmonary function.
Design:
This is a retrospective cohort study.
Methods:
Out of 8286 patients who underwent spirometry testing from January 2018 to December 2020, 492 patients were included in the final analysis. The relationship between statin usage, dosage, along with other biometric indices and spirometry parameters were evaluated. Multivariate logistic regression analyses were employed to assess the association between statin use and the decline in pulmonary function.
Results:
In patients with persistent hyperlipidemia, the use of statins was associated with a higher predicted percentage of forced expiratory volume in 1 second (FEV1) compared to non-users (84.0% vs 78.0%, p = 0.015). Logistic regression models further revealed that statin use independently prevented FEV1 decline, irrespective of dosage (adjusted OR 0.036, 95% CI: 0.002–0.618 in lower statins dose group and adjusted OR 0.170, 95% CI: 0.019–1.552 in higher statins dose group).
Conclusion:
The findings suggested that statin usage, regardless of dosage, independently mitigated the decline in pulmonary function among patients with persistent hyperlipidemia. Early initiation of statin therapy may hold promise for individuals experiencing hyperlipidemia and declining pulmonary function.
Introduction
Pulmonary function tests are widely utilized clinical tools that measure parameters including lung volume, capacity, and flow rate. They have pivotal roles in assisting clinical decision-making, as well as the diagnosis of various pulmonary diseases. It mainly provides information on identifying obstructive or restrictive ventilatory defects. Common pulmonary diseases that could be assessed via pulmonary function tests include but are not limited to asthma, chronic obstructive pulmonary disease (COPD), and pulmonary fibrosis. 1 Previous literature exhibited that pulmonary function is a valuable predictor of all-cause mortality and cardiovascular mortality.2,3 Spirometry is the most common pulmonary function test. 4
Meanwhile, hyperlipidemia is a prevalent metabolic disorder that causes severe healthcare burden around the world. Hyperlipidemia is determined by low-density lipoprotein (LDL), total cholesterol, triglyceride (TG), and high-density lipoprotein (HDL). 5 Abnormal LDL levels are highly associated with the development of atherosclerotic cardiovascular disease and with all-cause/cardiovascular mortality.6,7 Thus, lipid-lowering therapy is the mainstay of reducing cardiovascular disease risks, with statins being the cornerstone among available therapeutic agents. They are inhibitors of the hydroxymethylglutaryl-CoA reductase enzyme, which leads to reduced cholesterol synthesis and promotes LDL clearance from circulation.8,9 Recently, increasing studies shifted focus to the additional therapeutic values of statins outside of lowering serum LDL, such as in depression or the field of cancer.10,11
Previous publications indicated that hyperlipidemia may be associated with worse pulmonary function in various lung diseases.12 –14 Due to hyperlipidemia’s relation to chronic systemic inflammation and dysregulation of various metabolic pathways, whether lowering blood cholesterol levels or anti-inflammatory intervention could lead to improved pulmonary outcomes became the topic of interest in recent studies. Thus, it came to researchers’ interest that statins have a beneficial effect on pulmonary function. Cell and animal models established that statins could alleviate the progression of airway diseases such as COPD via inhibiting inflammatory response, as well as modulating key elements of the profibrogenic mechanism responsible for lung fibrosis.15 –17 However, to date whether statin usage could lead to favorable pulmonary function outcomes in the real world remains questionable. Conflicting results were collected from previous clinical studies focusing on statin’s impact on smokers, as well as airway diseases including asthma and COPD. 18 Several studies reported beneficial effects19,20; however other studies also exhibited non-significant effects,21,22 or even worsened pulmonary function. 23 Moreover, studies with focused subgroup analysis, such as in populations with underlying hyperlipidemia, are also lacking.
Due to the aforementioned knowledge gap, we conducted a retrospective cohort study to investigate whether statins could enhance pulmonary function in patients with underlying hyperlipidemia and to ascertain any potential relationship between protective effects and drug doses.
Methods
Study design and population
In this retrospective cohort study, a total of 8286 patients who underwent pulmonary function tests from January 2018 to December 2020 were initially enrolled. Out of these, 756 participants were randomly selected for further analysis. Patients lacking LDL data or essential medical records (such as statin use records) were excluded due to the inability to comprehensively analyze the correlation between hyperlipidemia, statin use, and pulmonary function. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (STROBE checklist; Supplemental Material). 24
Spirometry
For data analysis, two flow-sensing spirometers (MS-IOS Jaeger, Würzburg, Germany, and Vmax 22 SensorMedics, Yorba Linda, Calif) connected to a computer were used. The peak expiratory flow (PEF) forced vital capacity (FVC) and forced expiratory volume in 1 second (FEV1) were recorded from the forced expiratory flow–volume curve. The predicted values for these indices were calculated as percentages. Airflow limitation was defined as FEV1/FVC <70%. 25
Data collection and measurements
Demographic parameters were attained through a thorough examination of patient medical records. Historical health data encompassing hypertension, diabetes, smoking, and alcohol consumption were collated for inter-group comparative analysis. Body mass index (BMI) was computed as weight in kilograms divided by the square of height in meters. Fatty liver was diagnosed using chest computed tomography (CT) scans performed within 1 year prior to enrollment. Moreover, a comprehensive array of biometric indices was performed, allowing us to assess inflammatory markers such as white blood cells (WBC), neutrophil-to-lymphocyte ratio (NLR), C-reactive protein (CRP), and metabolic profiles including triglycerides (TG), cholesterol (Chol), HDL, LDL, and fasting glucose. Hyperlipidemia was defined as LDL ⩾130 mg/dL, 26 while an LDL level < 130 is also the control target for most patients in Taiwan. 27 Clinical history including cardiovascular disease history and pulmonary disease history were included, along with enrolled patients’ medication history (long-acting bronchodilators, antihypertensive, antidiabetic agents) and clinical outcomes such as emergency room visits and hospitalizations.
Statins exposure evaluation
The usage of statins was defined according to the Anatomical Therapeutic Chemical classification. 28 Simvastatin, atorvastatin, fluvastatin, lovastatin, pitavastatin, rosuvastatin, and pravastatin were identified as the medications of interest. 9 The intensity of statins dosage was stratified via calculating cumulative defined daily dose (cDDD), 29 as it is the assumed sufficient maintenance dosage to reach the medication’s main indication in adult patients. We calculated the cDDD of statins for 1 year prior to enrollment.
Statistical analysis
The findings were presented as either the median with interquartile range or as proportions (percentages) as appropriate. Continuous variables were compared using the Mann–Whitney U test, while categorical variables were analyzed using Pearson’s chi-square test. Logistic regression was employed to calculate odds ratios (ORs) and corresponding 95% confidence intervals (CIs). Clinical variables exhibiting between-group differences with p-values <0.1 in the univariate model were included in the multivariate analysis. The threshold for statistical significance was set at a p-value <0.05. Post hoc power analysis was conducted to evaluate the effect of sample size. Statistical analyses were conducted using IBM SPSS Statistics version 25.0 (IBM Corp., Armonk, NY, USA).
Results
Patient characteristics
In all, 756 participants were randomly selected from the 8286 patients who underwent spirometry tests from January 2018 to December 2020 (Figure 1). In all, 248 patients were excluded due to lacking LDL data, and an additional 16 patients were excluded due to incomplete medical records of statin use. Out of the remaining 492 patients included for final analysis, 180 patients had documented usage of statins (36.5%). These patients characterized by statins usage were characterized by higher BMI (statins group vs without statins use group, 26.8 vs 25.5 L, p = 0.001), a higher portion of hypertension (63.7% vs 50.5%, p = 0.006), and diabetes (35.1% vs 23.4%, p = 0.008). When comparing their baseline lipid profile 1 year before and at enrollment, patients with statin usage had higher initial TG levels (121.0 vs 99.5 mg/dL, p = 0.003). At enrollment, patients with statin use had a higher percentage of persisted hyperlipidemia (19.3% vs 7.0%, p < 0.001), higher TG (115.0 vs 98.5 mg/dL, p = 0.001), and fasting glucose level (107.0 vs 102.0 mg/dL, p = 0.016), as demonstrated by Table 1. The statin group also demonstrated a higher proportion of acute coronary syndrome (ACS) (27.8% vs 8.0%, p < 0.001) and ischemic stroke (10.0% vs 5.1%, p = 0.040), along with a higher percentage of anti-hypertensive (29.4% vs 15.4%, p < 0.001) and anti-diabetic (15.0% vs 6.7%, p = 0.003) medication history. However, there were no significant differences in cardiac and pulmonary disease history and clinical outcomes between groups. Despite pulmonary function not showing a statistically significant difference since many factors are associated with hyperlipidemia, the patient population with persistent hyperlipidemia was singled out for further analysis (Table 2).

Study flow chart.
Baseline characteristics of patients categorized according to statin use.
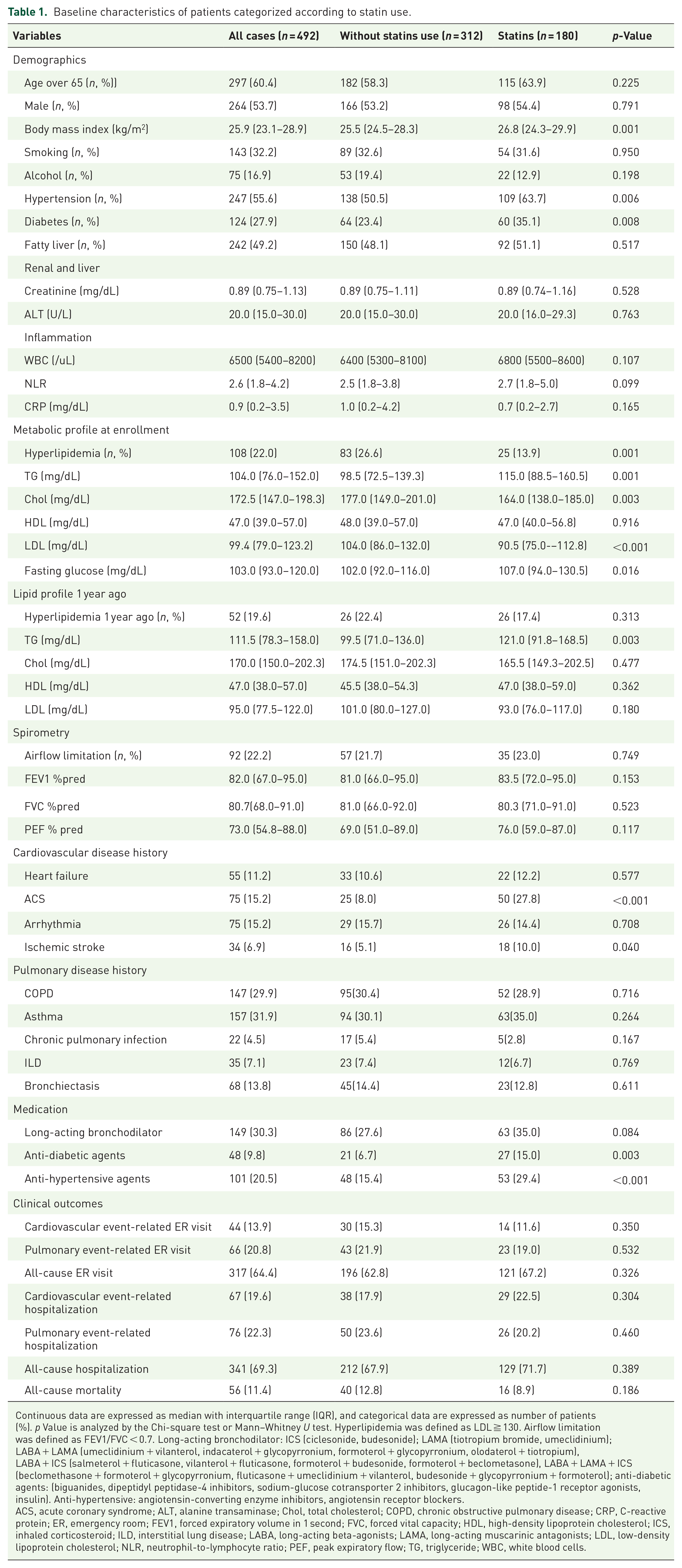
Continuous data are expressed as median with interquartile range (IQR), and categorical data are expressed as number of patients (%). p Value is analyzed by the Chi-square test or Mann–Whitney U test. Hyperlipidemia was defined as LDL ≧ 130. Airflow limitation was defined as FEV1/FVC < 0.7. Long-acting bronchodilator: ICS (ciclesonide, budesonide); LAMA (tiotropium bromide, umeclidinium); LABA + LAMA (umeclidinium + vilanterol, indacaterol + glycopyrronium, formoterol + glycopyrronium, olodaterol + tiotropium), LABA + ICS (salmeterol + fluticasone, vilanterol + fluticasone, formoterol + budesonide, formoterol + beclometasone), LABA + LAMA + ICS (beclomethasone + formoterol + glycopyrronium, fluticasone + umeclidinium + vilanterol, budesonide + glycopyrronium + formoterol); anti-diabetic agents: (biguanides, dipeptidyl peptidase-4 inhibitors, sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide-1 receptor agonists, insulin). Anti-hypertensive: angiotensin-converting enzyme inhibitors, angiotensin receptor blockers.
ACS, acute coronary syndrome; ALT, alanine transaminase; Chol, total cholesterol; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; ER, emergency room; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; HDL, high-density lipoprotein cholesterol; ICS, inhaled corticosteroid; ILD, interstitial lung disease; LABA, long-acting beta-agonists; LAMA, long-acting muscarinic antagonists; LDL, low-density lipoprotein cholesterol; NLR, neutrophil-to-lymphocyte ratio; PEF, peak expiratory flow; TG, triglyceride; WBC, white blood cells.
Baseline characteristics of patients with or without persisted hyperlipidemia.
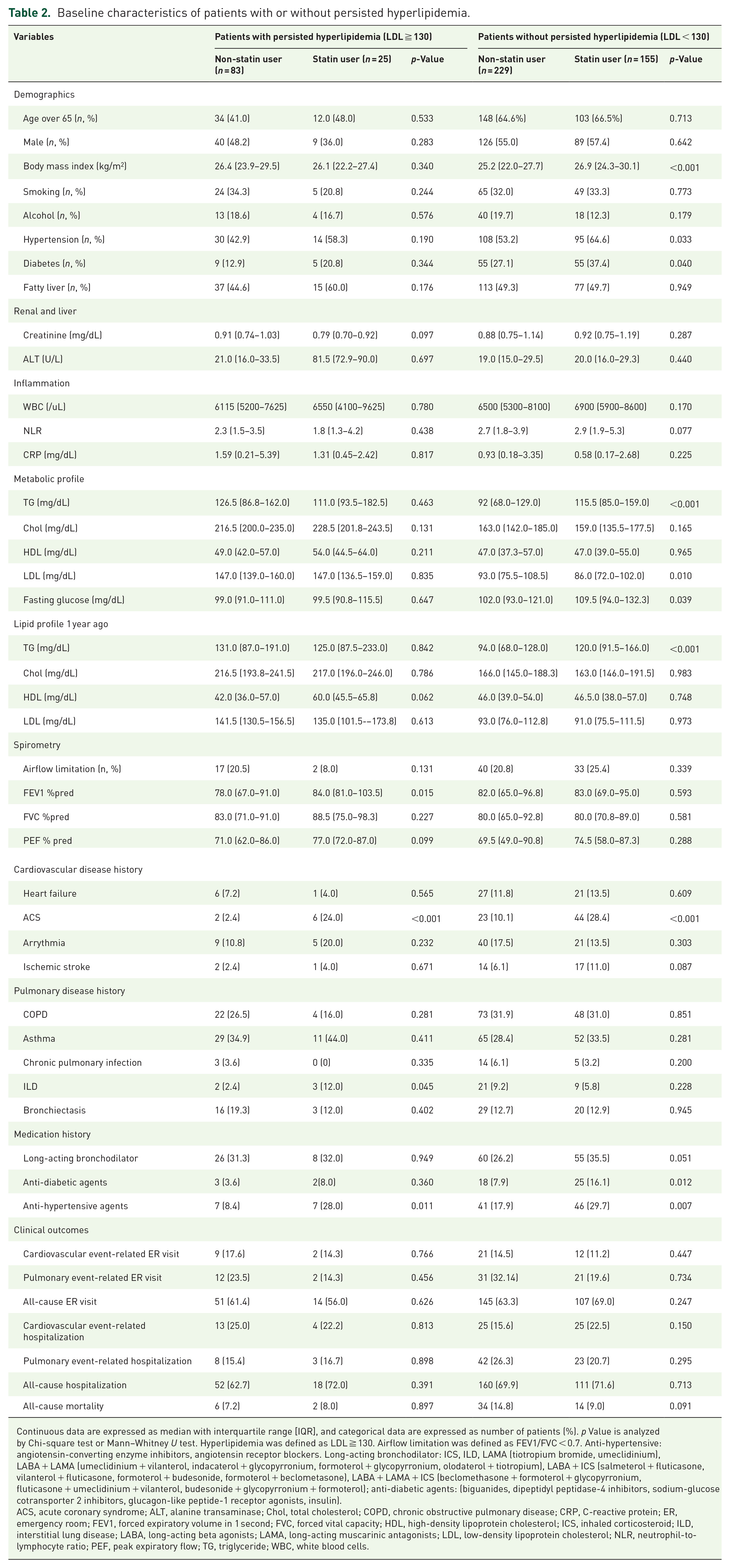
Continuous data are expressed as median with interquartile range [IQR], and categorical data are expressed as number of patients (%). p Value is analyzed by Chi-square test or Mann–Whitney U test. Hyperlipidemia was defined as LDL ≧ 130. Airflow limitation was defined as FEV1/FVC < 0.7. Anti-hypertensive: angiotensin-converting enzyme inhibitors, angiotensin receptor blockers. Long-acting bronchodilator: ICS, ILD, LAMA (tiotropium bromide, umeclidinium), LABA + LAMA (umeclidinium + vilanterol, indacaterol + glycopyrronium, formoterol + glycopyrronium, olodaterol + tiotropium), LABA + ICS (salmeterol + fluticasone, vilanterol + fluticasone, formoterol + budesonide, formoterol + beclometasone), LABA + LAMA + ICS (beclomethasone + formoterol + glycopyrronium, fluticasone + umeclidinium + vilanterol, budesonide + glycopyrronium + formoterol); anti-diabetic agents: (biguanides, dipeptidyl peptidase-4 inhibitors, sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide-1 receptor agonists, insulin).
ACS, acute coronary syndrome; ALT, alanine transaminase; Chol, total cholesterol; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; ER, emergency room; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; HDL, high-density lipoprotein cholesterol; ICS, inhaled corticosteroid; ILD, interstitial lung disease; LABA, long-acting beta agonists; LAMA, long-acting muscarinic antagonists; LDL, low-density lipoprotein cholesterol; NLR, neutrophil-to-lymphocyte ratio; PEF, peak expiratory flow; TG, triglyceride; WBC, white blood cells.
Correlation between statin use and pulmonary function in patients with persisted hyperlipidemia
It was noted that in patients with persisted hyperlipidemia, statin usage was associated with a better FEV1 predicted percentage (statins group vs without statins use group, 84.0% vs 78.0%, p = 0.015), as shown in Table 2. In Figure 2, we traced back the pulmonary function of patients from a year ago and found that in patients with persisted hyperlipidemia, statin users experienced less decline in FEV1% predicted compared to those not using statins. Subsequently, a possible dose-dependent effect of statin was identified after additional stratification to higher and lower doses according to a median of statins cDDD (Supplemental Table 1).

The trend of changes in FEV1 among persisted hyperlipidemia patients with and without statin use.
The risk factor of pulmonary function decline in patients with persisted hyperlipidemia
To better understand the risk factor associated with pulmonary function decline in patients with persisted hyperlipidemia, the population was further divided into cases with decreased FEV1 and cases without decreased FEV1 (defined by FEV1% predicted < 80%). Patients with decreased FEV1 also had a lower portion of statins use (decreased FEV1 group vs non-decreased FEV1 group, 7.3% vs 36.5%, p = 0.001), as well as higher inflammation markers including WBC (6600/µL vs 5850/µL, p = 0.013) and NLR (2.7 vs 1.9, p = 0.011). Furthermore, patients with decreased FEV1 were more likely to have a history of long-acting bronchodilator use (42.9% vs 21.6%, p = 0.027) as shown in Table 3. In Table 4, univariate logistic regression in persisted hyperlipidemia patients showed that statins usage was associated with a lower risk of decreased FEV1 (lower-dose group: cOR 0.082, 95% CI: 0.010–0.676, p = 0.020; higher-dose group: cOR 0.182, 95% CI: 0.037–0.902, p = 0.037). This association remained consistent across three adjusted models: model 1: adjusting for age over 65 and sex (lower-dose group: aOR 0.080, 95% CI: 0.010–0.672, p = 0.020; higher-dose group: aOR 0.146, 95% CI: 0.027–0.773, p = 0.024), model 2: adjusting for age over 65, sex, and smoking history (lower-dose group: aOR 0.088, 95% CI: 0.010–0.750, p = 0.026 higher-dose group: aOR 0.175, 95% CI: 0.033–0.933, p = 0.041), and model 3: adjusting for age, sex, smoking history, NLR, long-acting bronchodilator, and BMI (lower-dose group: aOR 0.036, 95% CI: 0.002–0.618, p = 0.022; higher-dose group: aOR 0.170, 95% CI: 0.019-1.552, p = 0.116). However, no protective dose effect of statins on decreased FEV1 was found in both univariate and multivariate logistic regression analyses.
Baseline characteristics of persisted hyperlipidemia patients with or without declined FEV1.

Continuous data are expressed as median with interquartile range (IQR), and categorical data are expressed as number of patients (%). p value is analyzed by the Chi-square test or Mann–Whitney U test. Decreased FEV1 was defined as FEV1 %pred < 80%. Long-acting bronchodilator: inhaled corticosteroid (ciclesonide, budesonide); LAMA (tiotropium bromide, umeclidinium), LABA + LAMA (umeclidinium + vilanterol, indacaterol + glycopyrronium, formoterol + glycopyrronium, Olodaterol + tiotropium), LABA + ICS (salmeterol + fluticasone, vilanterol + fluticasone, formoterol + budesonide, formoterol + beclometasone), LABA + LAMA + ICS (beclomethasone + formoterol + glycopyrronium, fluticasone + umeclidinium + vilanterol, budesonide + glycopyrronium + formoterol).
ALT, alanine transaminase; Chol, total cholesterol; CRP, C-reactive protein; FEV1, forced expiratory volume in 1 second; HDL, high-density lipoprotein cholesterol; ICS, inhaled corticosteroid; LABA, long-acting beta-agonists; LAMA, long-acting muscarinic antagonists; LDL, low-density lipoprotein cholesterol; NLR, neutrophil-to-lymphocyte ratio; TG, triglyceride; WBC, white blood cells.
Risk factors for decreased FEV1 among patients with persisted hyperlipidemia.

Long-acting bronchodilator: ICS (ciclesonide, budesonide), LAMA (tiotropium bromide, umeclidinium), LABA + LAMA (umeclidinium + vilanterol, indacaterol + glycopyrronium, formoterol + glycopyrronium, olodaterol + tiotropium), LABA + ICS (salmeterol + fluticasone, vilanterol + fluticasone, formoterol + budesonide, formoterol + beclometasone), LABA + LAMA + ICS (beclomethasone + formoterol + glycopyrronium, fluticasone + umeclidinium + vilanterol, budesonide + glycopyrronium + formoterol).
Force in age, gender.
Force in age, gender, smoking history.
Model with forcing in variables, including age, sex, smoking history, NLR, long-acting bronchodilator medication history, and BMI.
Statin doses are divided into lower and higher doses based on the median of cDDD (166). Decreased FEV1 defined as FEV1% predicted <80%.
aHR, adjusted odds ratio; BMI, body mass index; cHR, crude odds ratio; FEV1, forced expiratory volume in 1 second; ICS, inhaled corticosteroid; LABA, long-acting beta-agonists; LAMA, long-acting muscarinic antagonists; NLR, neutrophil-to-lymphocyte ratio.
Discussion
This study found that in patients with persisted hyperlipidemia, statins usage was correlated to better pulmonary function. Furthermore, after adjusting for confounders including age, gender, smoking history, inflammatory markers, clinical disease and medication history, statin usage independently prevents the decrease in FEV1.
Our cohort study proved that statin usage was associated with a lower risk of decreased FEV1 in patients with persistent hyperlipidemia, in line with previous publications,20,30,31 despite their greater CV risk burden. We hypothesized that statins alleviated pulmonary function decline by asserting their effect on the inhibition of chronic inflammation caused by excessive serum LDL, which is a known inflammation promoter.32 –34 This hypothesis is supported by the findings of our study: In patients with persistent hyperlipidemia and decreased FEV1, we observed an increase in inflammation-related markers, including WBC and NLR. However, this protective effect of statins did not increase with titrating up dose. This may be due to higher toxicity associated with increased statins dose, including hepatotoxicity or myopathy,8,35,36 which may affect the inflammatory status and subsequently make the dose-dependent effect less significant. In addition, patients who require higher statins dosage generally present with a greater degree of metabolic disorder, which may also affect pulmonary function and could not be completely accounted for in our study. It was noteworthy that a relatively low percentage of patients with hyperlipidemia at enrollment were noted. This could be attributed to the prior use of statins before study inclusion. In addition, statins may have been initiated based on cardiovascular (CV) risk rather than hyperlipidemia alone, as indicated by current guidelines, 37 which suggest statin therapy for individuals with relatively lower LDL levels but higher overall CV risk.
The protective effect of statins in preventing pulmonary function decline was not observed in patients without hyperlipidemia. Previous studies suggested that statins possess pleiotropic properties other than lipid-lower effect, with anti-inflammation being the most discussed. It was shown that statins could inhibit proinflammatory cytokines formation.38 –40 In an animal study, statin delivery via inhalation reduced airway inflammation. 41 However, with the oral route being the only approved route of statin administration in humans, there is no available data that could prove if the therapeutic concentration of statins reached the airway. Thus, we hypothesized that statins potentially exert their beneficial effects on the airways through their lipid-lowering properties since hyperlipidemia may cause a pro-inflammatory effect, then lead to worse pulmonary function.12 –14 However, the exact mechanism requires further confirmation through both basic and clinical research.
Limitations
Our study has several limitations. First, this is a retrospective cohort study which may not be able to precisely clarify the causal relationship between statins usage and pulmonary function, as well as other behavioral factors that may affect statins usage, including drug compliance. We included all patients who had spirometry, which may have been done due to respiratory symptoms, preexisting lung disease, health checkups, or preoperative evaluations. However, the exact indication for spirometry was not documented during enrollment and could not be analyzed due to the nature of our study design. Furthermore, due to the limited number of participants with spirometry 1 year before enrollment (18.5%, n = 91), our analysis was mainly based on a single pulmonary function test conducted at enrollment. Therefore, it is needed to design a pulmonary function follow-up study in the future to better elucidate the effects of statins. Second, the patient population was collected from a single tertiary center, raising questions about whether these results can be generalized to community patients. Despite being a small-sample-sized study, the post hoc power analysis showed sufficient power, reaching 0.829. Lastly, the underlying condition and other illnesses present during data collection may affect the legibility of the results; however, we included as many potential influencing variables as possible in our analysis and adjusted for them using statistical methods.
Strengths
Our study also has several strengths, including divided the population into different groups for analysis based on the possible mechanisms of statins on pulmonary function, along with being the first study that investigates potential dose-dependent effect of statins on pulmonary function.
Our study suggested that independent of dosage, statins could alleviate the worsening of pulmonary function in patients with persistent hyperlipidemia. According to the above result, earlier intervention with statins could be beneficial for hyperlipidemia patients who are experiencing declining pulmonary function. However, future well-designed prospective studies are necessary to help us better understand the impact of statins on pulmonary function, as well as determine the potential of dose-dependent effect.
Conclusion
In patients with persisted hyperlipidemia, our results suggested that statin usage independently associated with a lower risk of decreased FEV1. However, this potential protective effect was not dose dependent and was not observed in non-hyperlipidemic patients. Thus, early initiation of statin therapy may hold promise for individuals experiencing hyperlipidemia and declining pulmonary function.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251320875 – Supplemental material for Protective effects of statins on pulmonary function in patients with persistent hyperlipidemia: a retrospective cohort study
Supplemental material, sj-docx-1-tar-10.1177_17534666251320875 for Protective effects of statins on pulmonary function in patients with persistent hyperlipidemia: a retrospective cohort study by Hsiao-Chin Shen, Che-Hao Tseng, Yi-Hsuan Lin, Hsiao-Yun Yeh, Hung-Cheng Tsai, Shiao-Ya Hong, Tzu-Hao Li, Chien-Wei Su, Diahn-Warng Perng, Ying-Ying Yang and Ming-Chih Hou in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666251320875 – Supplemental material for Protective effects of statins on pulmonary function in patients with persistent hyperlipidemia: a retrospective cohort study
Supplemental material, sj-docx-2-tar-10.1177_17534666251320875 for Protective effects of statins on pulmonary function in patients with persistent hyperlipidemia: a retrospective cohort study by Hsiao-Chin Shen, Che-Hao Tseng, Yi-Hsuan Lin, Hsiao-Yun Yeh, Hung-Cheng Tsai, Shiao-Ya Hong, Tzu-Hao Li, Chien-Wei Su, Diahn-Warng Perng, Ying-Ying Yang and Ming-Chih Hou in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
