Abstract
Background:
Pneumonia is one of the most common complications after lung resection. However, there are currently no reports of postoperative pneumonia in patients with bronchiectasis.
Objectives:
Our study aims to construct a new nomogram to predict the risk of postoperative pneumonia in patients with localized bronchiectasis.
Design:
The clinical data of patients with localized bronchiectasis from April 2012 to August 2022 were retrospectively analyzed.
Methods:
Independent risk factors were identified through simple linear regression and multiple linear regression analysis, and a new nomogram was constructed based on independent risk factors. The validity of the nomogram was evaluated using the consistency index (C-index), receiver operating characteristic curve, calibration chart, and decision curve analysis chart.
Results:
The new nomogram prediction model included five independent risk factors: tuberculosis history, smoking history, platelet–lymphocyte ratio (PLR), diffusing capacity of the lung for carbon monoxide, and controlled nutritional status score. The area under the curve of the prediction model is 0.870 (95% CI: 0.750–0.892), showing good discrimination ability, and the probability threshold was set at 0.2013. In addition, the calibration curve shows that the nomogram has good calibration. In the decision curve, the nomogram model showed good clinical net benefit.
Conclusion:
This study is the first to construct a nomogram prediction model for postoperative pneumonia of localized bronchiectasis, which can more accurately and directly assess the risk probability of postoperative pneumonia, and provide certain help for clinicians in prevention and treatment decisions.
Keywords
Introduction
Bronchiectasis (BE) is a clinical syndrome caused by a variety of reasons. The main clinical symptoms of patients are cough, sputum, and repeated chest infections. The pathophysiological mechanism of the disease is that bronchiectasis leads to impaired mucociliary clearance of the lower airway, retention of bacteria and mucus, leading to persistent infection and further damage to the airway structure, forming a vicious cycle of repeated infections, seriously affecting the quality of life, and ultimately leading to respiratory failure and death.1 –4 Although medical treatment is currently considered the main treatment for bronchiectasis, surgical interventions play an irreplaceable role in improving the prognosis of bronchiectasis and the quality of life. 5 As the effectiveness of surgery in the treatment of localized bronchiectasis becomes increasingly apparent. However, it cannot be ignored that the complication rate after lung resection in these patients is as high as 9.4%–53%, among which the incidence of postoperative pneumonia is relatively high.6 –9 Previous studies have shown that postoperative pneumonia may lead to adverse outcomes such as reintubation or tracheostomy, prolonged hospitalization, increased medical costs, and unexpected readmission rates, and is closely related to high mortality.10,11 Therefore, it is crucial to accurately identify patients at high risk for postoperative pneumonia in bronchiectasis.
Postoperative pneumonia (POP) is one of the most common complications after thoracic surgery. Previous studies have shown that smoking, age, resection range, body mass index (BMI), diabetes, neurological diseases, etc. are independent risk factors for pneumonia after lung cancer surgery.12 –14 In addition, Jin et al. established a prediction model for pneumonia after esophageal cancer surgery, in which age, gender, abdominal surgery method, chest tube insertion time, anastomotic leakage, and recurrent laryngeal nerve paralysis are independent risk factors for pneumonia. 15 Previous studies have confirmed that preoperative controlled nutritional status score (CONUT) is an independent risk factor for overall postoperative complications in localized bronchiectasis. 8 However, there have been no studies reporting the risk factors for postoperative pneumonia in bronchiectasis. Considering the disease characteristics of bronchiectasis, these patients are a high-risk group for POP. With the development of precision medicine, it is urgent to build a prediction model to reduce the incidence of postoperative pneumonia.
The nomogram is an intuitive and visual prediction tool that can accurately predict the risk probability of clinical events for each patient. It makes the model more targeted by adding potential biomarkers, thereby achieving precise and personalized predictions.16,17 At present, nomograms have been widely used in the study of the prognosis of various malignant tumors and have achieved good results.18 –21 Therefore, this study aims to identify independent risk factors for POP in localized bronchiectasis and construct a new nomogram to help clinicians accurately screen out high-risk patients with POP.
Methods
Study population
We retrospectively analyzed the clinical data of 260 patients with localized bronchiectasis who underwent consecutive lung resections in our hospital from April 2012 to August 2022. The surgically resected specimens of all patients were confirmed by histopathological examination. Inclusion criteria: (1) patients with localized bronchiectasis undergoing surgical treatment; (2) no nutritional supplementation during the perioperative period; (3) clinical data are complete. Exclusion criteria: (1) diffuse bronchiectasis or bronchiectasis disease incomplete resection; (2) refuse surgical treatment; (3) inability to tolerate surgery due to various reasons such as cardiopulmonary function; (4) patients with malignant tumors; (5) patients requiring perioperative nutrition supplementation.
The reporting of this study conforms to the strengthening the reporting of cohort, cross-sectional and case-control studies in surgery (STROCSS) statement 22 (Supplemental File).
Data collection
We collect patient information through the electronic medical record system: basic information includes gender, age, BMI, symptoms of diagnosis, duration of illness, and preoperative comorbidities (history of tuberculosis, hypertension, diabetes, smoking history, drinking history, coronary heart disease). Preoperative laboratory tests include: white blood cells, neutrophils, total lymphocytes, albumin, total cholesterol, red blood cells, platelets, hemoglobin, activated partial thromboplastin time, prothrombin time, and D-dimer. Preoperative pulmonary function includes forced expiratory volume in 1 second (FEV1), the proportion of FEV1 to the predicted value, diffusing capacity of the lung for carbon monoxide (DLCO), and maximum ventilation volume per minute (MVV). Surgery-related information includes surgical method, extent of resection, duration of surgery, intraoperative blood loss, and postoperative pneumonia.
Blood samples were collected from all patients to complete preoperative evaluation. Patients undergoing emergency surgery had their blood drawn and sent for testing on the day of surgery, and the remaining patients had their blood drawn and sent for testing on the first day of hospitalization. Follow-up ceased at the time of patient discharge.
Definition of POP, localized bronchiectasis, and calculation of CONUT, NLR, PLR, SII
Postoperative pneumonia refers to new lung infiltration, consolidation, and other imaging abnormalities found on postoperative chest X-ray or CT examination. And there is at least one of the following criteria: (1) Postoperative respiratory symptoms (cough, purulent sputum or shortness of breath, etc.) are new or continue to worsen; (2) fever > 38°C, excluding other causes (3) white blood cell count is >10 × 109/L or <4 × 109/L or C-reactive protein is elevated. Sputum culture and blood culture confirmation were performed when necessary but not essential. According to the Clavien-Dindo classification system, 23 the severity of postoperative pneumonia in this study was Grade ⩾ II.
Localized bronchiectasis was defined as: the presence of the following signs on high-resolution computed tomography and the extent of the lesion was only limited to a single lung segment, lobe, or two adjacent lobes, and bronchiectasis was confirmed by pathology after surgery.
(1) The ratio of the inner diameter of the bronchus to the diameter of the accompanying pulmonary artery is >1; (2) the bronchus does not gradually become thinner from the center to the periphery; (3) the bronchial shadow can be seen within 1 cm of the peripheral pleura or close to the mediastinal pleura. 24
The Controlled Nutritional Status Score (CONUT) is a new scoring system used to evaluate preoperative nutritional status. 25 It includes serum albumin level, peripheral blood lymphocyte count, and cholesterol level, respectively reflecting the patient’s nutritional status, lipid profile, and metabolism and immune function, the total score is calculated based on the scores of various laboratory test results (Table 1), and according to the total score, it is divided into normal nutrition (0–1), mild abnormality (2–4), moderate abnormality (5–8), and severe abnormalities (9–12), according to which patients were divided into two groups in this study.
Assessment of nutrition status by the CONUT score.

CONUT, Controlled Nutritional Status Score.
The NLR is one of the effective indicators of systemic inflammatory response, reflecting the balance between neutrophils and lymphocytes, and can reflect the patient’s immune status. The platelet–lymphocyte ratio (PLR) can reflect the balance of platelet and lymphocyte levels in the body, is a sign of systemic inflammatory response, and plays an important role in the judgment of many diseases and prognosis. The systemic immune-inflammation index (SII) was first defined in 2014 as platelet count × neutrophil count/lymphocyte count, which reflects the body’s inflammatory state more comprehensively than a single inflammatory index. NLR, 26 PLR, 27 and SII 28 are new inflammatory markers derived in recent years. They are often used together to judge the disease situation. The determination method is simple, easy, cheap, and convenient.
Construction and evaluation of nomogram
Simple linear regression was used to analyze potential risk factors, and variables with p < 0.05 were included in multifactor logistic regression. All predictors with p values <0.05 in the multiple linear regression analysis model were included in the prediction model. The “rms” R package was used to construct a nomogram. Use the “pROC” R package to draw the receiver operating characteristic (ROC) curve, and confidence intervals of area under the curve (AUC) values were calculated by resampling using the default bootstrap method in “pROC.” Use the “rms” R package’s bootstrap method of 1000 resampling to draw the calibration chart. The accuracy of nomogram in distinguishing POP is judged by the AUC, and the threshold probability was calculated by the optimal cutoff value of ROC. The consistency between the predicted probability and the actual probability is described by the calibration curve. The closer the two lines are, the closer the predicted incidence rate is to the actual incidence rate, indicating that the consistency of the prediction model is better. Finally, decision curve analysis (DCA) was drawn using the “rmda” package to evaluate the net benefit to patients and clinical utility by quantifying the net benefit at different threshold probabilities.
Statistics
Normally distributed continuous variables are represented by mean ± SD, and Student’s t test is used to compare differences between groups. Non-normally distributed continuous variables are represented by median (Q1, Q3), and the Mann–Whitney U test is used to analyze differences between groups. Categorical variables are expressed as numbers (%), and the χ2 test or Fisher’s exact test was used. All analyses were performed using IBM SPSS Statistics 26.0 and R 4.3.2. SPSS (Version 26.0; IBM, Armonk, NY, USA); RStudio (Boston, Massachusetts, United States). A two-sided p value <0.05 was considered statistically significant.
Results
Patient characteristics
A total of 260 patients with bronchiectasis were included in this study (Table 2), of which 111 were males (42.7%) and 149 females (57.3%), with an average age of 49.72 ± 13.41 years and 32 (12.3%) patients developed postoperative pneumonia. The median time from surgery to the development of pneumonia was 2 days. According to the CONUT score, the patients were divided into a normal nutrition group and a nutritional abnormality group. There was a significant difference in BMI between the two groups of patients. The average BMI of patients in the high group was significantly lower than that in the low group. Comparing the laboratory test data of the two groups, as shown in Table 2, the total lymphocytes, red blood cells, hemoglobin of patients, and albumin (and total cholesterol in the low group were all significantly higher than the high group). However, the neutrophils NLR, PLR, and SII were significantly lower than the high group. In addition, there was a significant correlation among the CONUT score, the surgical method, surgical time, and intraoperative blood loss. However, there were no significant statistical differences between the two groups in terms of gender, age, symptoms, comorbidities, pulmonary function, and extent of resection. Comparing the incidence of postoperative pneumonia between the two groups, the incidence of patients in the low group was significantly lower than that in the high group.
Baseline characteristics of the patients sorted by the CONUT score.

n (%).
x ± s.
(Q1, Q3).
Bold values indicate statistically significant differences (P<0.05).
APTT, activated partial thromboplastin time; BMI, body mass index; CHD, coronary heart disease; DLCO, diffusing capacity of the lung for carbon monoxide; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; Hb, hemoglobin; Lobe, lobectomy; MVV, maximum ventilation volume per minute; NLR, neutrophil to lymphocyte ratio; PLR, platelet to lymphocyte ratio; PT, prothrombin time; RBC, red blood cells; SII, systemic inflammation index; S/W, segmentectomy/wedge resection; VATS, video-assisted thoracoscopic surgery; WBC, white blood cells.
Independent risk factors for postoperative pneumonia
The results of the single-factor analysis are shown in Table 3. BMI, tuberculosis history, smoking history, preoperative hemoglobin content (Hb), albumin, PLR, CONUT ⩾ 2 points, DLCO, surgical method, resection range, surgical time, and intraoperative blood loss are all significantly related to postoperative pneumonia (p < 0.05).
Univariate and multivariate analyses of risk factors associated with postoperative pneumonia.

Bold values indicate statistically significant differences (P<0.05).
APTT, activated partial thromboplastin time; BMI, body mass indewx; CHD, coronary heart disease; DLCO, diffusing capacity of the lung for carbon monoxide; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; Hb, hemoglobin; Lobe, lobectomy; NLR, neutrophil to lymphocyte ratio; PLR, platelet to lymphocyte ratio; PT, prothrombin time; RBC, red blood cells; SII, Systemic Inflammation Index; S/W, segmentectomy/wedge resection; VATS, video-assisted thoracoscopic surgery; WBC, white blood cells.
The above variables (p < 0.05) were included in multifactor logistic regression analysis. The results showed that history of tuberculosis (OR = 12.051, 95% CI: 1.384–35.142, p = 0.029), smoking history (OR = 8.735, 95%CI: 1.053–33.292, p = 0.046), PLR (OR = 1.016, 95% CI: 1.002–1.029, p = 0.023), DLCO (OR = 0.538, 95%CI: 0.318–0.910, p = 0.021) and CONUT ⩾ 2 (OR = 7.257, 95% CI: 1.387–37.981, p = 0.034) is independent risk factors for postoperative pneumonia in patients with bronchiectasis.
Construction and evaluation of nomogram
A nomogram for predicting postoperative pneumonia in patients with bronchiectasis was constructed using tuberculosis history, smoking history, PLR, DLCO, and CONUT (Figure 1). In the nomogram, each parameter of each factor is assigned a score on the model. By accumulating the total score and drawing a straight line downward, the probability of postoperative pneumonia for each patient can be intuitively estimated. In the ROC curve, as shown in Figure 2, the AUC is 0.870, indicating that the nomogram has excellent discrimination ability. We set the threshold probability to 0.2013, and for patients exceeding this probability, clinicians can consider early intervention.

Nomogram for predicting the probability of postoperative pneumonia in patients with localized bronchiectasis.
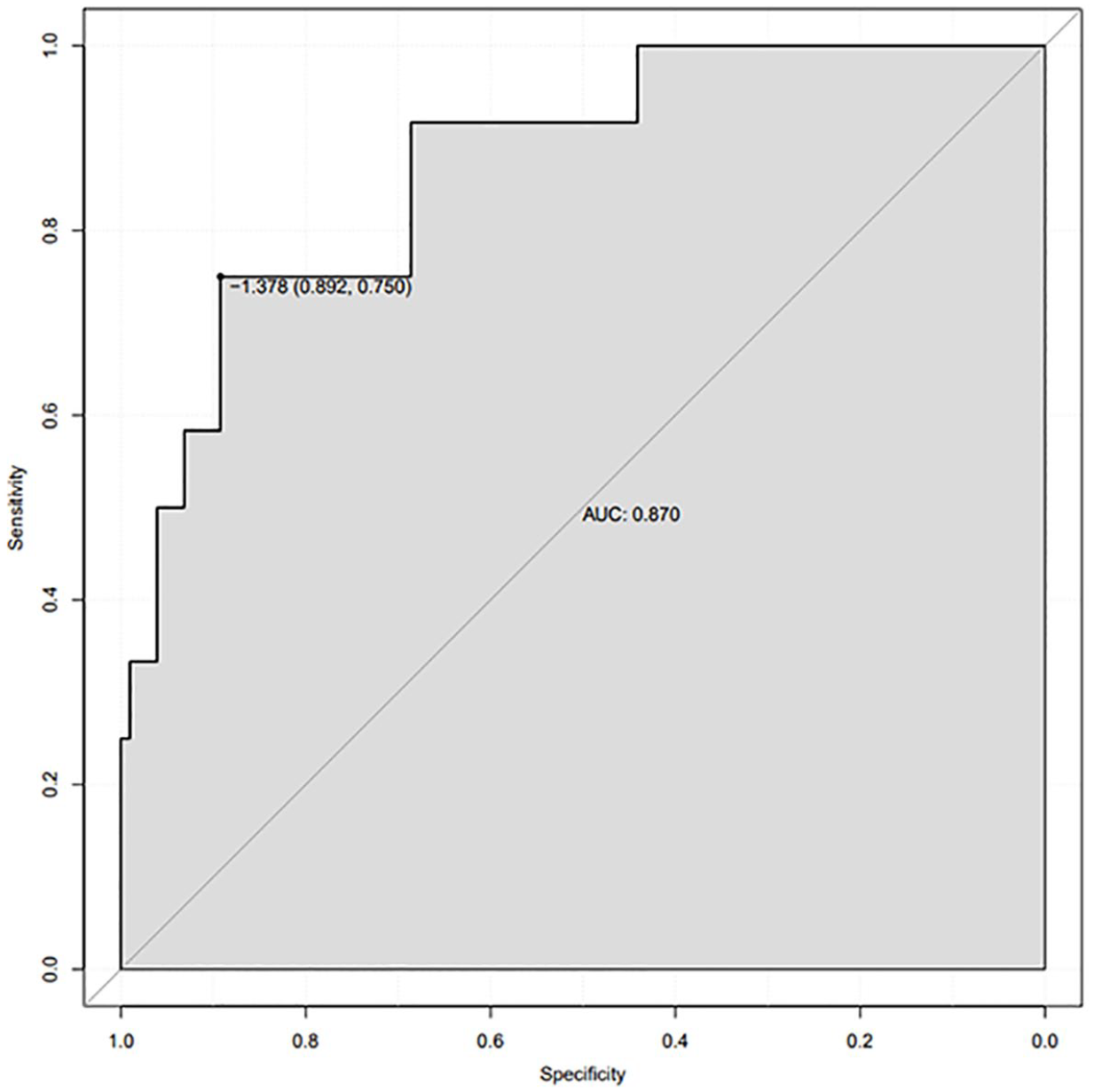
ROC curve for distinguishing postoperative pneumonia with AUC of 0.870 (95% CI: 0.750–0.892).
The application of the calibration curve shows that the nomogram-predicted probability and the true probability have relatively good consistency (Figure 3). The DCA curve also shows (Figure 4) that nomogram has good clinical application value and can bring benefits to patients.

Calibration curves of the novel nomogram. The x-a axis shows the model’s predicted probabilities, and the y-a axis shows the actual probabilities.

Decision curve analysis of the novel nomogram.
Discussion
In this study, the incidence rate of postoperative pneumonia was as high as 12.3%. We identified five independent risk factors through logistic regression analysis, including tuberculosis history, smoking history, PLR, DLCO, and CONUT. Based on this result, a new nomogram was constructed to predict the risk of POP in patients with localized bronchiectasis. This model has accurate predictive ability, with a C-index of 0.870, and has good clinical application value.
Currently, an increasing number of patients with localized bronchiectasis undergo surgical intervention because of its ability to significantly improve prognosis. However, postoperative complications significantly affect the effect of surgical treatment, among which postoperative pneumonia is relatively common,29,30 which deserves more effort from clinicians. It has been reported that approximately 1000 people per 100,000 people have bronchiectasis in Western countries over the past decade, and the number of affected individuals is increasing at a rate of approximately 8% per year. 31 In China, patients with bronchiectasis are more common than in the West. Current research shows that among people over 40 years old, the incidence rate of patients with bronchiectasis is about 1.2%, and the incidence rate is on an increasing trend. 32 In addition, postoperative pneumonia is one of the most common complications after lung resection and can cause respiratory failure and even death.33,34 Therefore, it is very important to construct a mature prediction model to reduce the occurrence of postoperative pneumonia in patients with bronchiectasis.
Nomogram is a method that accurately predicts the probability of each individual outcome event by integrating multiple predictive indicators. For the first time, we developed a nomogram to predict the probability of developing pneumonia after lung resection in patients with localized bronchiectasis. Previous studies have confirmed that nomogram can accurately predict the risk of postoperative pneumonia in lung cancer and esophageal cancer.15,29 Jin et al. constructed and verified a prediction model for postoperative pneumonia on 1225 patients with lung cancer. 29 The model included smoking, diabetes, history of preoperative chemotherapy, thoracotomy, The American Society of Anesthesiologists classification, and operation time, and its C-index was 0.717. In addition, Mao et al. constructed a new nomogram for predicting minimally invasive postoperative pneumonia using 609 patients with esophageal cancer, 15 including age, gender, abdominal surgery method, chest tube insertion time, anastomotic leakage, and recurrent laryngeal nerve palsy. The C-index of the model is 0.769. The above two studies have achieved the purpose of individualized and accurate prediction.
The results of this study show that patients with localized bronchiectasis who have a history of tuberculosis have a significantly increased probability of developing postoperative pneumonia. Few studies have reported the relationship between tuberculosis history and postoperative pneumonia. A previous study showed 35 that approximately 3.3% of patients with postoperative pneumonia had a history of tuberculosis. The possible mechanisms are as follows: First, in China, pulmonary tuberculosis is one of the common causes of bronchiectasis, 24 and the number of patients with a history of tuberculosis may be larger; in addition, pulmonary tuberculosis can cause anatomical changes and functional impairment of the lungs, Although the condition has been stabilized or even cured, local structural damage is irreversible, making patients more susceptible to postoperative pulmonary infection.36,37
Smoking can lead to airway hyperresponsiveness and long-term inflammation, which can easily lead to various postoperative complications. The correlation between smoking and postoperative pneumonia has been widely reported. The results of our study showed that smoking patients with bronchiectasis were eight times more likely to develop postoperative pneumonia than non-smokers, which is consistent with previous research results.38,39 Schussler et al.’s study showed 38 that smokers have a significantly higher risk of postoperative pneumonia than never-smokers (OR = 9.43); Mariko et al. reported the importance of smoking cessation on the outcome of primary lung cancer surgery. 39 Among them, smokers are 2.8 times more likely to suffer complications after lung resection than non-smokers. Therefore, preoperative education on smoking cessation can significantly reduce the incidence of postoperative pneumonia.
Both PLR and CONUT are independent risk factors for postoperative pneumonia in localized bronchiectasis. PLR and CONUT include four laboratory parameters: platelets, lymphocytes, albumin, and cholesterol, which represent inflammatory status, immune function, nutritional status, and lipid metabolism respectively. Fitting together provides a more comprehensive assessment of patient inflammation-nutrition-immunity status, in which elevated PLR values and CONUT scores indicate that patients are in a state of inflammation and nutritional abnormalities, with increased susceptibility to infection, delayed wound healing, and significantly increased risks of various postoperative complications. 40 Malnutrition and high inflammatory status have been widely confirmed to be risk factors for poor patient prognosis.8,41 –45 Sasahara et al. used a study of 320 patients with thoracic esophageal squamous cell carcinoma 41 to show that compared with patients with low preoperative PLR (<166), patients with high preoperative PLR (⩾166) had a higher incidence of postoperative pneumonia. Lee et al. reported the correlation between CONUT and postoperative complications of non-small cell lung cancer. 45 The results showed that patients in the high CONUT group (⩾2 points) had twice as many types of postoperative pneumonia as those in the low CONUT group. Therefore, clinicians should pay attention to the laboratory tests of preoperative patients and evaluate patients with bronchiectasis by calculating PLR and CONUT scores based on corresponding parameters to reduce the occurrence of postoperative pneumonia.
The results of this study show that patients with lower DLCO values in localized bronchiectasis also have a significantly higher incidence of postoperative pneumonia. Pulmonary function test is one of the routine assessment methods before patients undergo lung resection, including FEV1, FEV1/FVC, DLCO, MVV, and PEF, especially FEV1 and DLCO, which are often used to determine the patient’s perioperative risk. 46 It is well known that poor lung function is significantly related to poor prognosis. Previous studies have shown that FEV1 <60% of the predicted value is an independent risk factor for postoperative complications of bronchiectasis.6,8,47 However, there are few studies on the correlation between DLCO and postoperative pneumonia. The results of Yao et al.’s study showed that compared with higher DLCO (⩾80%), lung cancer patients with DLCO (40%–59%) and DLCO (<40%), the probability of postoperative pneumonia increased by 4.328 times and 4.725 times respectively. Similarly, Wang et al. reported that DLCO is an independent risk factor for pneumonia after neoadjuvant immunochemotherapy for esophageal cancer. Our study reports for the first time the important role of DLCO in postoperative pneumonia in bronchiectasis.
We recognize that the limitations of this study are as follows: first, the single-center, retrospective study reduces the generalizability of the model; second, the sample size is limited, and the next step is to improve and verify the model with multicenter and large samples; finally, this study only looked at patients during hospitalization, and a more accurate model would require longer follow-up.
Conclusion
We established a new and effective nomogram that can predict the risk of postoperative pneumonia in patients with localized bronchiectasis. This model has excellent recognition ability and provides an effective method for preoperatively assessing patients’ risk of postoperative pneumonia, which can help clinicians develop appropriate personalized prevention and treatment strategies in advance.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251320471 – Supplemental material for A novel nomogram for predicting postoperative pneumonia risk in patients with localized bronchiectasis
Supplemental material, sj-docx-1-tar-10.1177_17534666251320471 for A novel nomogram for predicting postoperative pneumonia risk in patients with localized bronchiectasis by Cai Yongsheng, Ke Lihui, Hao Xuefeng, Qiao Anbang, Yang Xiaoxiao, Chen Wenhui, Li Weiqing, Yang Zeng and Wei Bo in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
