Abstract
Background:
Coronavirus disease 2019 (COVID-19) became pandemic in 2020 and recently, mutated coronaviruses have emerged in many countries. The aim of this study was to identify the clinical characteristics and risk factors for critical illness in hospitalized COVID-19 patients in Zhengzhou for clinical prevention and management.
Materials and methods:
A total of 70 patients hospitalized with COVID-19 were enrolled between 21 January and 29 February 2020, in Zhengzhou, China. Clinical characteristics, hematological findings, neutrophil lymphocyte ratio (NLR), platelet lymphocyte ratio (PLR), and inflammatory index on admission were obtained from medical records, COVID-19 patients with different outcomes were compared.
Results:
The median age was 55 years. Forty-three (61.0%) patients were classified as having severe or critical cases. Eighteen (25.7%) patients died in hospital and the remaining 52 were discharged. Patients who died tend to be old with expectoration and chronic obstructive pulmonary disease. Compared to survivor, non-survivor had significantly higher numbers of leucocytes and neutrophils, NLR, aspartate aminotransferase (AST), γ-glutamyl transpeptidase, total bilirubin, direct bilirubin, lactate dehydrogenase (LDH), prothrombin time, D-dimer, C-reactive protein, and decreased platelets, lymphocytes, uric acid, and albumin (ALB). Logistic regression analysis identified leucocytes, platelets, PLR, NLR, AST, and ALB as independent predictive factors for poor outcomes. The area under curve of the combination of leucocytes, PLR, NLR, and AST was 0.87, with a sensitivity of 0.83 and specificity of 0.81.
Conclusion:
Our results identified risk factors among COVID-19 patients for in-hospital mortality. Leucocytes, PLR, NLR, and AST could have important reference value for predicting prognosis, especially in low-resource countries.
Introduction
In December 2019, an pneumonia of unknown cause was detected in Wuhan, Hubei Province, and spread rapidly across the country.1,2 In early January 2020, scientists identified a novel beta coronavirus called SARS-CoV-23 and the World Health Organization officially named the disease as new coronavirus pneumonia (corona virus disease 2019, COVID-19) on February 11th. 4 At present, the second pandemic wave of COVID-19 is severe, and new cases of local transmission are being reported in north and northeast China. Globally, there have been 84,474,195 confirmed cases of COVID-19, including 1,848,704 deaths as of January 2021, according to WHO. The infection is still spreading at the time of manuscript preparation.
SARS-CoV-2 is a novel enveloped positive-stranded RNA virus. 5 It has an easily mutated RNA polymerase that makes the virus more likely to undergo gene mutation and recombination. 6 In the course of the spread, viral mutations make it difficult to identify and develop specific drugs or vaccines for treatment. Some studies indicated that the spike (S) protein of the virus binds to angiotensin-converting enzyme II (ACE II) on the host cell membrane to infect host cells. 7 Hippisley et al. showed that ACE inhibitors and angiotensin receptor blockers were associated with a significantly reduced risk of COVID-19. 8 Therefore, knowing the infectivity, clinical features, and susceptibility factors for SARS-CoV-2 is crucially important for the disease prevention. Regular blood tests are an important and easily performed way to keep track of overall physical health. Abnormal increases or decreases in cell counts as revealed in a regular blood test may indicate an underlying medical condition that needs early evaluation. 9 The neutrophil lymphocyte ratio (NLR) has been identified as a novel inflammatory marker in various clinical conditions such as ulcerative colitis, diabetic peripheral neuropathy, type 2 diabetes mellitus, and acute myocardial infarction.10–13 Similarly, the platelet-to-lymphocyte ratio (PLR) has emerged as an informative marker revealing shifts in platelet and lymphocyte counts as a result of inflammation. Furthermore, previous reports suggest there is an association between COVID-19 and both of NLR and PLR. 14 An efficient immune response against SARS-CoV-2 is considered as necessary for the treatment of COVID-19, and more attention has been paid to the identification and development of predictive inflammation biomarkers for the resolution of COVID-19.
In the past year, many published studies reported high incidence of COVID-19 in various locations, but no clinical data from Henan Province were published. Therefore, we analyzed the clinical data from 70 COVID-19 patients to investigate the clinical characteristics and relevant risk factors associated with critical illness in COVID-19 patients.
Materials and methods
Study design and participants
A retrospective, single-center study was conducted at the first affiliated hospital of Zhengzhou University from 21 January to 29 February 2020. The study was approved by the Ethics Committee of the First Affiliated Hospital of Zhengzhou University (approval number: 2021-KY-0095). Written informed consent was obtained from legally authorized representatives before the study. All procure were followed the Standards for Reporting of Diagnostic Accuracy Studies (STARD) guidelines. All hospitalized patients who were diagnosed with COVID-19 according to WHO interim guidance 15 and had clinical outcomes before 31 March, 2020 were enrolled in our study. Patients without laboratory tests or demographic data or the minimum information necessary to determine these data were excluded. Those who died from other diseases were also excluded from our study. According to the classification of the National Health Commission of China, 16 the severity of COVID-19 patients at admission was classified into four types: mild, moderate, severe, and critical types. Since the mild type was usually treated by self-isolation and not admission to the hospital, only the last three types were included in our study.
Data collection
Demographics, clinical symptoms, underlying diseases, radiological characteristics, laboratory tests at admission including complete blood count, biochemical examinations, coagulation functions and length of hospitalization were selected form electronic records. Neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and lymphocyte-to-monocyte ratio (LMR) were calculated from the absolute values of neutrophils, lymphocytes, platelets and monocytes. Laboratory confirmation of SARS-CoV-2 virus infection by real-time polymerase chain reaction (RT-PCR) assay was performed using nasopharyngeal swab samples. The end point of our study was clinical outcome (death or recovery). All patients were followed up until death or discharge after two negative the SARS-CoV-2 tests.
Statistical analysis
Continuous variables are presented as mean ± standard deviation for normal distribution and medians (interquartile range) for non-normal distribution (the Kolmogorov–Smirnov test was used to estimate the normal distribution). Categorical variables are expressed as the values and percentages in each category. Continuous variables were compared using independent group t-tests or the Mann–Whitney test. Differences in proportions between groups were compared by the chi-square test or Fisher’s exact test. We classified patients into survivor and non-survivor groups according to the clinical outcome. Variables that were significantly different between the two groups were selected for univariate binary logistic regression analysis. Next, to determine whether variables were independently associated with mortality, multivariate binary logistic regression was used. The optimal cut-off value of the continuous variables was calculated by the area under the receiver characteristic operating curve (ROC). The Hosmer–Lemeshow test for goodness-of-fit-statistics was utilized to certify the model adequacy. All statistical analyses were performed with SPSS 26.0 software and
Results
Baseline characteristics
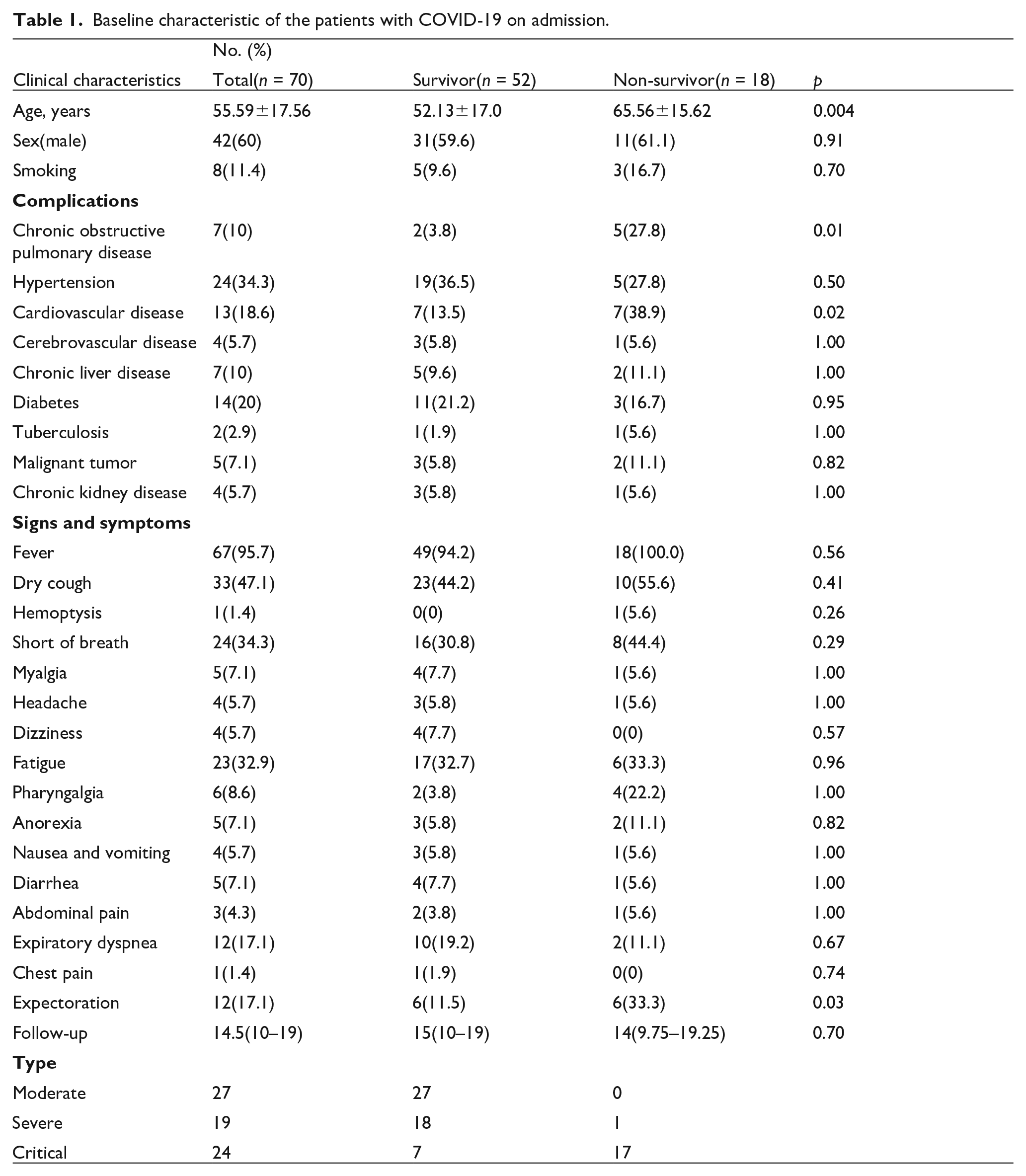
Between 21 January and 29 February 2020, data on 74 patients were primarily collected, and 4 were excluded because of aortic dissecting aneurysm or acute coronary syndrome and death within 6 h or for whom there were incomplete data. Finally, 70 hospitalized adult patients were enrolled in our study, including 52 who recovered and 18 who died. The median time from admission to discharge was 14.5 days (IQR, 10.0–19.0). There were 42 males and 28 females, with mean age of 56 years. The age of the non-survivor group was significantly greater than of survivor group (65.56 ± 15.62 vs 52.13 ± 17.0,
Baseline characteristic of the patients with COVID-19 on admission.
Laboratory tests
The laboratory tests results are summarized in Table 2. Among the variables, leucocytes, PLR, neutrophils, NLR, aspartate aminotransferase (AST), γ-glutamyl transpeptidase (GGT), alkaline phosphatase (ALP), total bilirubin (Tbil), direct bilirubin (Dbil), lactate dehydrogenase (LDH), prothrombin time (PT), D-dimer, procalcitonin (PCT), and C-reactive protein (CRP) in the non-survivor group were significantly higher than in the survivor group. In the non-survivor group, platelets (PLT), lymphocytes, uric acid (UA), and albumin (ALB), were significantly lower than in the survivor group.
Laboratory findings of patients with COVID-19.
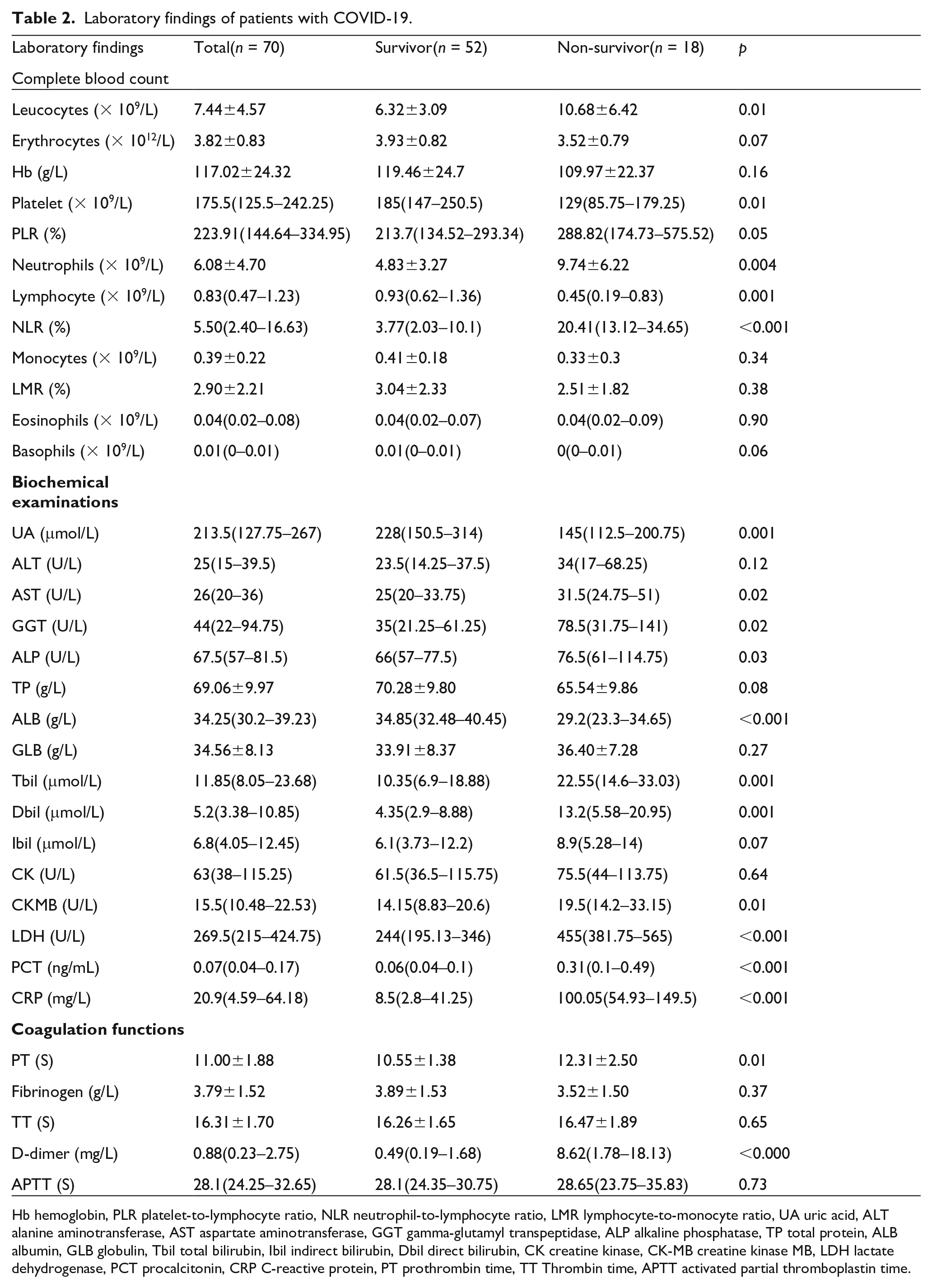
Hb hemoglobin, PLR platelet-to-lymphocyte ratio, NLR neutrophil-to-lymphocyte ratio, LMR lymphocyte-to-monocyte ratio, UA uric acid, ALT alanine aminotransferase, AST aspartate aminotransferase, GGT gamma-glutamyl transpeptidase, ALP alkaline phosphatase, TP total protein, ALB albumin, GLB globulin, Tbil total bilirubin, Ibil indirect bilirubin, Dbil direct bilirubin, CK creatine kinase, CK-MB creatine kinase MB, LDH lactate dehydrogenase, PCT procalcitonin, CRP C-reactive protein, PT prothrombin time, TT Thrombin time, APTT activated partial thromboplastin time.
Risk factors for death
The demographic and laboratory variables that were statistically different between the survivor and non-survivor group were identified, and then univariate regression analysis was used to evaluate potential risk factors for SARS-COV-2 (Table 3). The results showed that the odds ratio (OR) of 19 demographic and laboratory variables other than ALP, PCT, and creatine kinase MB (CK-MB) were predictors of in-hospital death. Considering the collinearity of the leucocytes, neutrophils, and lymphocytes, CRP, and PCT, we excluded neutrophils, lymphocytes and CRP from multivariate regression analysis. Finally, 13 laboratory variables were analyzed and the results showed that leucocytes (OR = 8.52, 95% confidence interval [CI] = 1.07–67.92,
Univariate analysis for the risk factor to mortality of patients with COVID-19.

OR Odds ratio, CI confidence intervals, PLR platelet-to-lymphocyte ratio, NLR neutrophil-to-lymphocyte ratio, UA uric acid, AST aspartate aminotransferase, GGT gamma-glutamyl transpeptidase, ALB albumin, Tbil total bilirubin, Dbil direct bilirubin, LDH lactate dehydrogenase, PT prothrombin time, APTT activated partial thromboplastin time, CRP C-reactive protein.
Multivariate analysis for the independent risk factor to mortality of patients with COVID-19.

OR Odds ratio, CI confidence intervals, PLR platelet-to-lymphocyte ratio, NLR neutrophil-to-lymphocyte ratio, AST aspartate aminotransferase, ALB albumin, NA not applicable.
Receiver operator characteristic curve for prediction death
To predict the accuracy of these risk factors in COVID-19 patients, a ROC analysis for continuous variables was conducted to identify the optimal cut-off value. A statistical model, including leucocytes, PLR, NLR, and AST was developed and the ROC curve obtained. The AUC of leucocytes, PLR, NLR, and AST were 0.72 (95% CI = 0.57–0.86,
Sensitivity and specificity for significant parameter in the prediction of COVID-19.
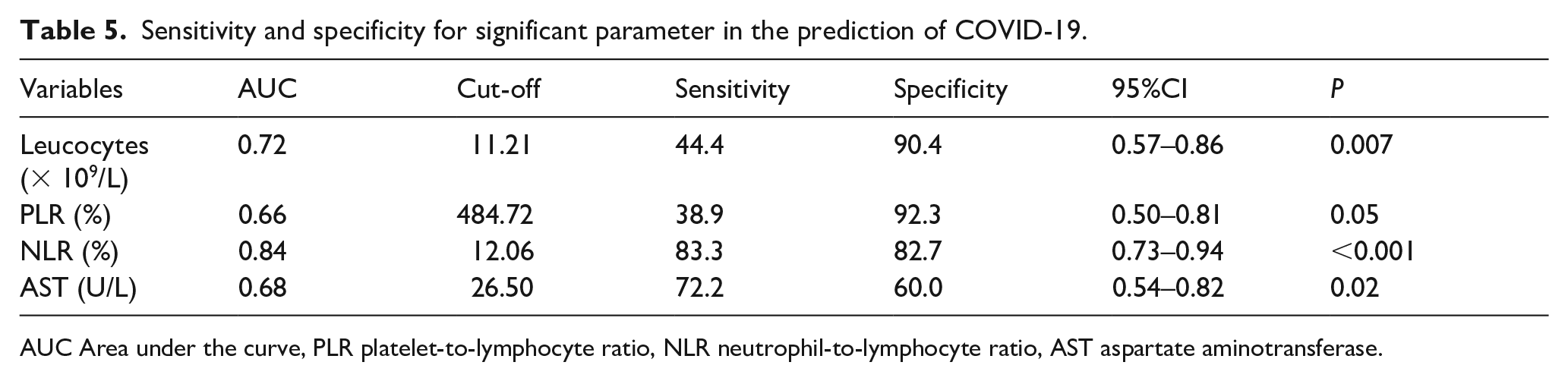
AUC Area under the curve, PLR platelet-to-lymphocyte ratio, NLR neutrophil-to-lymphocyte ratio, AST aspartate aminotransferase.

Receiver operator characteristic curve analysis for mortality prediction in COVID-19 patients.
Discussion
In December 2019, the outbreak and spread of COVID-19 pneumonia had unpredictable social ramifications. The long incubation time of this highly contagious virus and highly heterogeneous symptoms, ranging from asymptomatic and mild to severe and fatal have had a tremendous effect on economic and social development. 17 Rapid and precise identification of asymptomatic people who are in the incubation phase is urgently needed to prevent of community infection. Our study aimed to identify the potential risk factors and available laboratory tests for the early detection of SARS-CoV-2.
In our study, 70 newly diagnosed patients from a single clinical center in Zhengzhou, Henan Province were included. With respect to gastrointestinal symptoms, there were no significant differences between the survivor and non-survivor groups, unlike in the Jin et al. studies. 18 Only 4% of COVID-19 patients had a normal temperature on admission, and this percentage is much lower than in previously published studies.19–21 The clinical characteristics of COVID-19 patients indicate that advanced age and underlying diseases were crucial risk factors for death. The most common underlying diseases were hypertension, followed by diabetes, cardiovascular disease, chronic obstructive pulmonary disease, and chronic liver disease. However, the proportions of hypertension, diabetes, and chronic liver disease were not different between the survivor and non-survivor groups. The median age of the non-survivor group was significantly higher than of the survivor group, which is probably related to the decline and dysregulation of immune function. 22 The destructive effects of SARS-CoV-2 on many organs, such as heart, kidney, and liver, ultimately leading to multiple organ dysfunction syndrome (MODS), may cause the death of critically ill patients.23,24
In the laboratory examinations, we demonstrated that leukocytes, neutrophils, lymphocytes, NLR, PLR, liver enzymes, and inflammatory indicators were elevated. Serum albumin, platelets, and UA were all decreased. An elevated level of NLR and inflammatory indicators reflects the increasing secretion of cytokines such as IL-6 and IL-8, aggravating the inflammatory response caused by SARS-CoV-2. 25 These results are consistent with those of previous studies.26–28 The blood of COVID-19 patients is often in a hypercoagulable state and may have arterial thrombosis such as acute coronary syndromes. 29 Zhang et al. demonstrated that platelets expressed two major cellular components, ACE II and transmembrane serine protease 2(TMPRSS2), which are responsible for the interaction of the S protein of SARS-CoV-2 for infecting cells. 30 Therefore, SARS-CoV-2 usually shows decreased platelets, increased D-dimer and fibrinogen levels. Platelet circulation, activation, and aggravation were increased, leading to a decrease in the number of platelets. Our study provided evidence to the value of anti-thrombosis treatment for COVID-19 patients.
From the results of the logistic regression analysis, we identified six independent risk factors for death in COVID-19 patients. NLR is a widely used prognostic biomarker for the clinical outcome of infection. 31 Yang et al. 27 reported that NLR was significantly increased in patients with severe COVID-19, which is in line with our study. We also demonstrated NLR was an independent risk factor for COVID-19 patient mortality, indicating that an increase in neutrophils and decrease in lymphocytes play a crucial role in virus-related dysregulated immune regulation.
Since these biomarkers can be easily assessed and NLR and PLR were easily calculated, the ROC curves were operated to early diagnosis for patients who were at high risk of death. There were four independent risk factors that reflected inflammatory response (leucocytes and NLR), cell damage (AST), and coagulation function (PLR), respectively. The results indicate that in the early stage of SARS-COV2 infection, increased leucocytes, NLR, decreased PLR, and higher AST were prone to have adverse outcomes. In the combination of four biomarkers as measures of accuracy of the indicators, the AUC was 0.87, with sensitivity of 0.83 and specificity of 0.81, which was higher than any one of the individual indicators. In addition, the cut-off value of NLR for predicting death in COVID-19 patients in our study was 12.06, with a sensitivity of 0.83 and a specificity of 0.66, obviously higher than the other studies. Ciccullo et al. reported an NLR of higher than 4 was a predictor of admission to the ICU. 32 Halil et al. prospectively validated an optimal cut-off value of 5.94 to predict high in-hospital mortality for COVID-19 patients.33,34 Serve COVID-19 patients have a higher NLR > 3.2, compared with the non-severe group. Interpretation of NLR not only depends on the presence of infection or inflammation but is also affected by the clinical context, such as toxic shock or pulmonary embolism. NLR is normally ranges from 1 to 3, whereas 6–9 suggests mild stress and higher than 9 indicates critically illness. Our study results contribute to assessing prognosis in the early stages of COVID-19 patients and could be a supplement to a previous study that reported patients with risk factors were more likely to have an adverse outcome. Liang et al. identified a risk score (COVID-GRAM) including 10 independent factors to predict development of critical illness. 35 Among 10 predictors, values of NLR, LDH, and Dbil were in accord with our results. In addition, diagnostic and prognostic risk scores such as the National Early Warning Score 2 (NEWS 2) and 4C mortality score for COVID-19 are accessible and they all demonstrated have excellent discriminative performance.36–38 However, our predictors did not include clinical characteristics, such as dyspnea and unconsciousness, mainly because of lacking of availability in a retrospective context.
There are some limitations in our study. First, it is a single center, retrospective study and ascertain power were not evaluated. Second, more detail about the therapeutic responses and cytokine changes would be better to determine prognosis in COVID-19 patients. Third, although the risk factors for mortality were identified using the data on admission, the follow-up laboratory information for disease progression was absent. Fourth, the majority of the enrolled patients were had ordinary or serve disease, and clinical data from patients with mild disease were absent from the analysis. Finally, we only emphasized the laboratory data of adult COVID-19 patients, so the results are not generalizable to young patients.
Conclusion
In conclusion, we showed for the first time that high leucocyte levels, NLR, and AST and low PLR were risk factors associated with mortality among COVID-19 patients with laboratory-confirmed coronavirus in Henan Province for the first time. Prediction of COVID-19 mortality is significant in creating management strategies and early prevention. Further studies are needed to confirm our findings in the other cohorts to provided additional evidence for disease management.
Footnotes
Authors’ contributions
All authors contributed to the study conception and design. Conceptualization, data curation, formal analysis, drafted the manuscript were performed by Jia Li. Analysis and interpretation of data were performed by Chaoyang Hua. Project administration, supervision, review and editing were performed by Zhangsuo Liu and Yanfang Yang. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
All procedures were followed the Ethics Committee of the first affiliated hospital of Zhengzhou University (approval number: 2021-KY-0095).
Informed consent
Written informed consent was obtained from legally authorized representatives before the study.
