Abstract
Background:
Bronchoscopic lung volume reduction (BLVR) can be an effective treatment for highly selected patients with severe emphysema but only half of carefully selected patients derive clinical benefit. Two commercially available platforms exist to help determine candidacy for BLVR via quantitative analysis of computed tomography (CT) scans.
Objectives:
To determine if the two commercially available quantitative platforms identified the same patient population that may benefit from BLVR.
Design:
A multicenter, retrospective cohort study.
Methods:
Consecutive patients referred for BLVR between January 1, 2022 and March 31, 2023 at three medical centers in the United States with the same CT scan submitted for quantitative analysis to two commercially available platforms to determine BLVR candidacy were analyzed. The primary outcome of interest was whether quantitative analysis provided different recommendations for individual patients. The recommendation to proceed with BLVR was based on a prespecified algorithm using criteria established in clinical trials for each quantitative platform, respectively.
Results:
A total of 83 patients referred for BLVR across three centers were included; patients were a median 67 years old, had a median post bronchodilator FEV1 of 30% predicted (IQR: 25, 38), a median residual volume of 220% predicted (IQR: 185, 268), and 29 (34.9%) received endobronchial valves. A total of 26 patients (31.3%) received different recommendations from the two quantitative platforms.
Conclusion:
In this cohort of patients evaluated for BLVR across multiple medical centers, nearly a third of patients received different recommendations based on the platform utilized for valve assessment. This suggests that the selection process for BLVR may warrant refinement.
Introduction
In carefully selected patients with severe emphysema and hyperinflated lungs, bronchoscopic lung volume reduction (BLVR) can improve exercise tolerance, decrease lung volume, and may even decrease mortality.1 –9Accordingly, BLVR is an approved treatment for patients with advanced emphysema and is now recommended with Level A evidence by the Global Initiative for Chronic Obstructive Lung Disease (GOLD). 10 Yet, only approximately half of patients in clinical trials for BLVR derived clinical benefit.6 –8
Successful BLVR is thought to require both severe emphysema and lack of collateral ventilation between lobes of the lung.6,8,9,11 If collateral ventilation exists, the targeted lobe will not collapse. Likewise, without significant parenchymal destruction from emphysema, patients will not derive benefit from the procedure.6,8,9,11 While fissure integrity and emphysema is assessed via qualitative review of CT scans, a key part of the patient selection process is quantitative analysis of CT scans to ensure fissure integrity and quantify emphysema destruction to identify the target lobe.12,13 There are currently two different proprietary commercial platforms (StratX quantitative platform, PulmonX, Redwood City, CA, USA; SeleCT quantitative platform, Olympus Corporation, Center Valley, PA, USA) that aim to provide an unbiased measure of fissure integrity and emphysema destruction.14,15 However, it is unclear if these platforms provide concordant estimates of fissure integrity and emphysema destruction and if discrepancies between these platforms may therefore result in different patient selection for BLVR.
In this multicenter, retrospective cohort of patients with severe emphysema undergoing evaluation for BLVR, we compare patient selection for BLVR using the two different commercially available platforms in the United States. We hypothesize that the different platforms would identify different patients that meet criteria for BLVR and that there would be large differences in fissure integrity scores between platforms.
Patients and methods
Study setting
This was a multicenter, retrospective observational cohort study of patients with severe emphysema referred for evaluation for BLVR between January 1, 2022 and March 31, 2023 at CHI Creighton University Medical Center, University of North Carolina Medical Center, and Washington University in St. Louis. The study was approved with a waiver of informed consent by the Washington University in St. Louis Institutional Review Board. This study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline for observational studies. 16
Quantitative platforms
There are currently two commercially available quantitative platforms on the market in the United States to help determine candidacy for BLVR: (1) the StratX quantitative platform (PulmonX, Redwood City, CA, USA), which is designed for use with the Zephyr Valve and (2) the SeleCT quantitative platform (Olympus Corporation, Center Valley, PA, USA), which is designed for use with Spiration Valve System. Both of these systems are cloud-based platforms that use proprietary software to provide an unbiased measure of fissure integrity and emphysema destruction through analysis of a high-resolution CT chest.14,15
Cohort identification and patient selection
Patients with severe emphysema referred at each participating site for evaluation for BLVR were included if they had a high-resolution CT chest scan analyzed by both of the commercially available quantitative platforms (StratX and SeleCT). If the CT scan was not compatible with both software platforms, patients were excluded from analysis.
Primary exposure and outcome definition
The primary exposure of interest was the recommendation to proceed with BLVR based on an evidence-based approach.9,13 We defined an evidence-based approach according to the algorithm depicted in Figure 1. This strategy was prospectively defined using existing best practice recommendations, modeled after the pivotal study for each platform/valve (EMPROVE 9 for SeleCT platform/Spiration valve, LIBERATE 6 for StratX platform / Zephyr valve). For the SeleCT platform, patients were determined to be candidates for BLVR if there was (1) ⩾40% emphysema destruction in the target lobe (at −920 HU), (2) ⩾90% fissure integrity score between the target lobe and ipsilateral lobes, (3) and ⩾10% difference in emphysema destruction score between the target lobe and ipsilateral lobe. 9 For the StratX platform, patients were determined to be candidates for BLVR if (1) there was ⩾50% emphysema destruction in the target lobe (at −910 HU), (2) ⩾95% fissure integrity score between the target lobe and ipsilateral lobes, and (3) ⩾15% difference in emphysema destruction score between the target lobe and ipsilateral lobe. If the fissure integrity score was <95% but ⩾80%, then the recommendation was to proceed with functional assessment for collateral ventilation (using the Chartis system).6 –8 In this case where the fissure integrity score was <95% but ⩾80%, and the patient met other criteria, they were categorized as a possible candidate for BLVR pending functional assessment. For all patients, the target lobe was the most destroyed lobe. For the primary analysis, we only considered patients with heterogenous disease.

Evidence-based approach for BLVR.
We performed a sensitivity analysis to include patients with homogenous emphysema only if the residual volume was > 200% predicted (Supplemental Figure 1). 8 For this analysis if there was < 10% difference in emphysema destruction score between the target lobe and ipsilateral lobe with the SeleCT platform, or < 15% difference in emphysema destruction score between the target lobe and ipsilateral lobe with the StratX platform, then they were considered for BLVR.
The primary outcome of interest was the recommendation to proceed or not to proceed with BLVR based on analysis from each platform with the caveat that the ultimate decision to proceed with BLVR is dependent on physician evaluation. Secondary outcomes of interest included if the platforms agreed upon the target lobe, a comparison of fissure completeness scores between the platforms for each fissure (right major, right minor, left), and the percentage of patients with > 10% difference in estimated fissure score.
Data collection
For each patient with quantitative analysis from both platforms, review of electronic medical record was performed. Collected data included patient demographics, pulmonary function testing, symptom scores, emphysema destruction scores (−920 HU for SeleCT and −910 HU for StratX), fissure integrity scores, and if the patient received endobronchial valves (EBV).
Statistical analysis
Descriptive statistics were used to describe baseline demographics, pulmonary function testing, symptom scores, and outcome of BLVR. Discrete variables were described by number and percentage, while continuous variables were described by median and interquartile range. We did not perform a sample size calculation so the limited number of samples may affect the statistical significance of the results. Statistical analyses were performed using STATA/MP version 18.0 (StataCorp, College Station, TX, USA). We considered p < 0.05 (two-sided) to be significant.
Results
A total of 83 patients were referred for BLVR evaluation across the 3 centers and had quantitative analysis submitted and successfully analyzed by both platforms (Table 1). The median age was 67 (IQR: 61, 72), 56.7% of patients were female, the vast majority (90.4%) were white, and the median pack-year of smoking was 45 (IQR: 34, 60). Patients had severe COPD: 50.6% were Gold Stage III and 47.0% were Gold Stage IV with a median FEV1 of 0.86L (IQR: 0.62, 1.06), median FEV1 percent predicted of 30% (IQR: 25, 38), and median DLCO percent predicted of 44 (IQR: 36, 53). Patients had significant air trapping and hyperinflation with residual volume of 220% (IQR: 185, 268) predicted and a total lung capacity of 129% (IQR: 119, 143) predicted. A total of 29 patients (34.9%) received endobronchial valves for bronchoscopic lung volume reduction. Of the patients receiving valves, 15 (51.7%) received Spiration valves and 14 (48.3%) received Zephyr valves. Among the 20 patients with data for lobar collapse at time of hospital discharge, 14 patients received Spiration valves and 6 patients received Zephyr valves. Among these patients, 11 (55%) had lobar collapse at time of hospital discharge. There was no association between valve type and lobar collapse (p = 0.10).
Patient characteristics (n = 83).

DLCO, diffusion capacity for carbon monoxide; FEV1,forced expiratory volume in 1 second; L, liters; RV, residual volume; TLC, total lung capacity.
Among all patients, 26 (31.3%) received a different recommendation for BLVR based on the quantitative platform used (Figure 2 and Table 2), and this was consistent across each of the three sites (range 25.0%–35.5%, p =0.73). For 18 patients (21.7%), both platforms recommended that the patients were candidates for BLVR and for another 39 patients (47.0%), both platforms agreed that patients were not candidates for BLVR. For 13 patients (15.6%), the StratX platform recommended that the patients were potential candidates but required functional assessment (using Chartis system) to determine if BLVR was feasible. However, in these same patients, the SeleCT platform advised that 7 (8.4%) were not candidates due to incomplete fissures. For 10 (12.1%) patients, SeleCT recommended that the patients were candidates for BLVR, whereas the StratX recommended that the patients were not candidates for BLVR or even for functional assessment of collateral ventilation. For 3 (3.6%) patients, SeleCT recommended that the patients were not candidates for BLVR, whereas the StratX platform suggested they could proceed directly to valve placement for BLVR. The recommendation to proceed or not proceed with BLVR differed significantly between the two platforms (Table 2; p < 0.001).
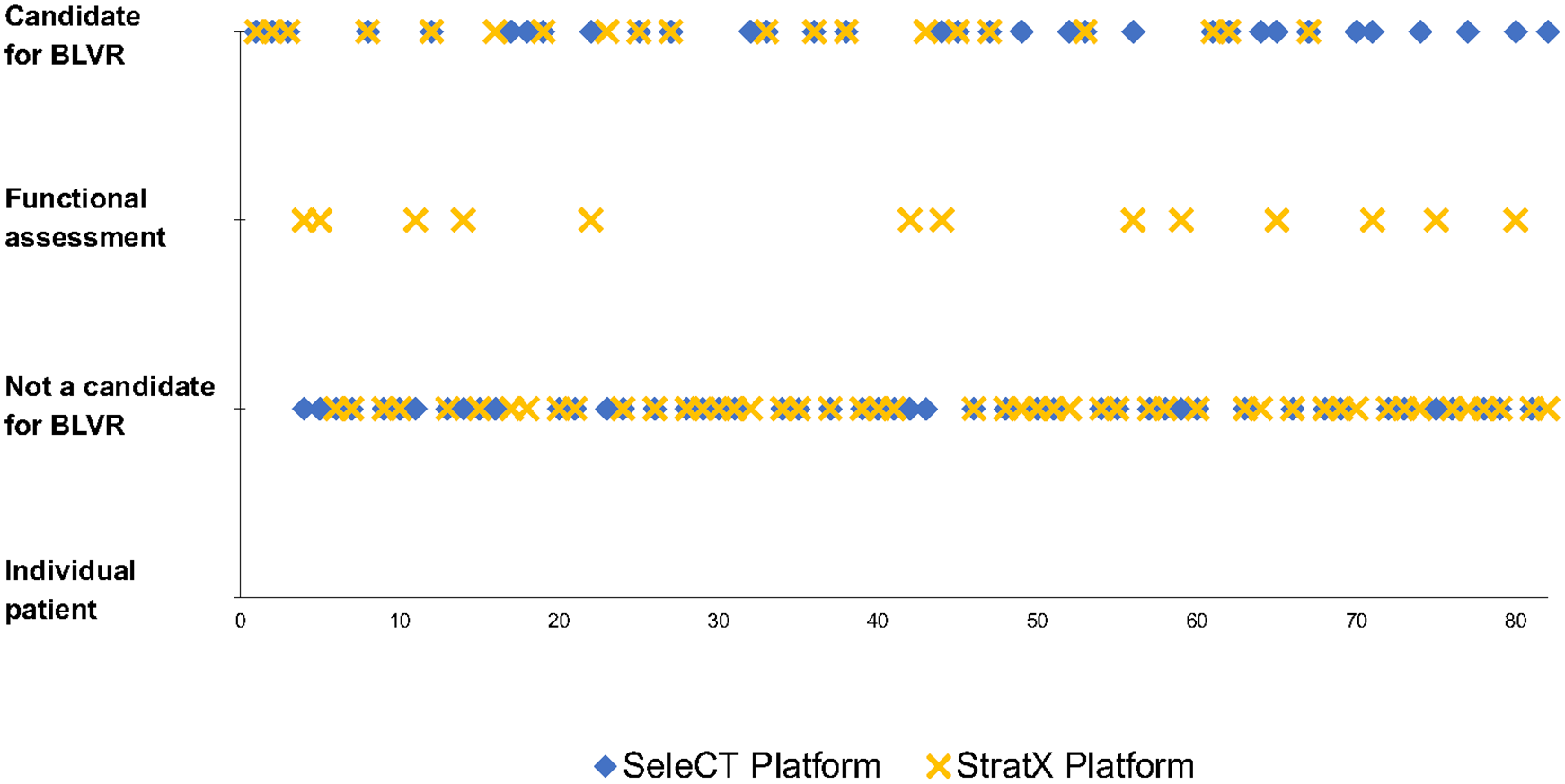
BLVR recommendation based on quantitative platform.
Recommendation for BLVR based on quantitative platform (n = 83).
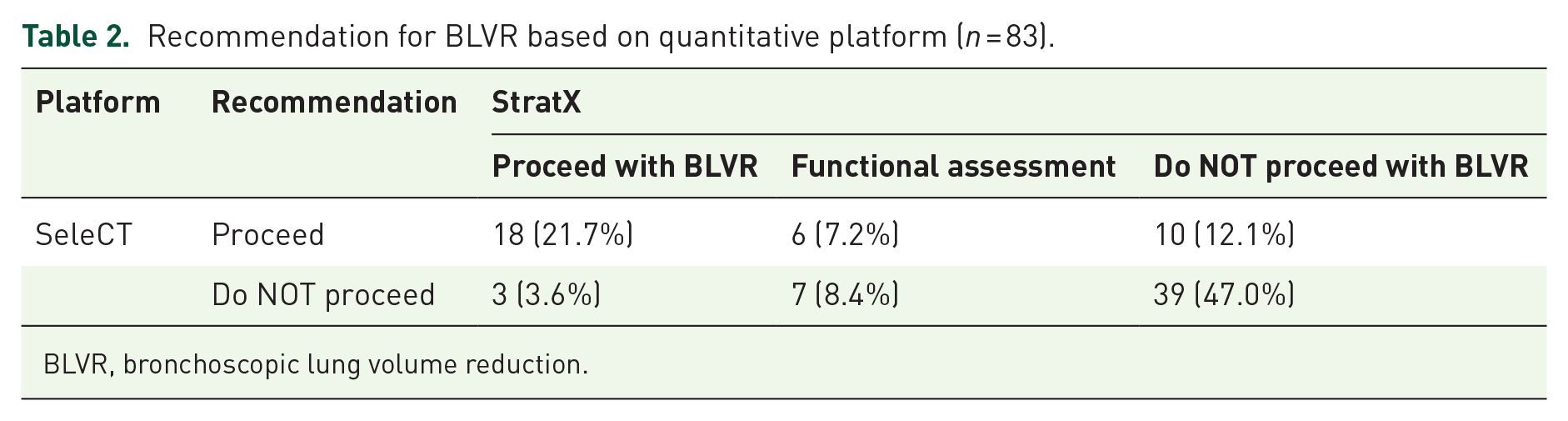
BLVR, bronchoscopic lung volume reduction.
Amongst patients (n = 24) where both platforms suggested that the patient was a potential candidate (i.e., SeleCT recommended that patient was a candidate, StratX recommended either functional assessment or proceed directly to BLVR), 16.7% (n = 4) received different recommendations for the best target lobe. Amongst the patients (n = 18) where both platforms recommended to proceed with BLVR, one patient received a recommendation to proceed with BLVR, but the two platforms recommended different lobes as the best target.
Among the 29 patients that ultimately received valves, 19 (65.5%) received the same recommendation across platforms and 10 (34.4%) received discordant recommendations. Among the 10 patients that ultimately received valves and had different recommendations between the platforms, 6 were deemed candidates for BLVR by the SeleCT platform but were not a candidate for BLVR (either to proceed with valves or for functional assessment) according to the StratX platform, 2 of the patients were deemed not a candidate by the SeleCT platform but were candidates for functional assessment by the StratX platform, and 2 patients were candidates for BLVR according to the SeleCT platform but required functional assessment based on the StratX analysis. Accordingly, among these patients receiving valves, 8 (27.6%) would have different outcomes (i.e., whether they received BLVR or not) depending upon which platform was used to determine candidacy.
The two platforms provided significantly different values for fissure estimates (right major p < 0.001, left major p < 0.001, right minor p = 0.02 difference in fissure; Figure 3). The median difference between estimates for the right major fissure was 4 (IQR: 2, 8) but 20 (24.1%) had at least a 10% point or more difference in fissure estimate and 4 (4.8%) had at least a 20% difference in fissure estimate. The median difference between estimates for the right minor fissure was 4 (IQR: 1, 7), 15 (18.1%) had at least a 10% point or more difference in fissure estimate, and 5 (6.0%) had at least a 20% difference in fissure estimate. The median difference between estimates for the left fissure was 4 (IQR: 1, 7), 15 (18.1%) had at least a 10% point or more difference in fissure estimate, and 5 (6.0%) had at least a 20% difference in fissure estimate.

Percentage of patients with ⩾10% difference in estimated fissure score between platforms.
In sensitivity analysis in which patients were evaluated for BLVR for heterogenous disease and then, if not a candidate, for homogenous disease (assuming RV > 200%), 32 (38.6%) patients received different recommendations based on the platform used (Supplemental Figure 2 and Table 1), and this was consistent across the three sites (range 35.0–43.8%, p = 0.74). For 40 patients (48.2%), both platforms recommended that the patients were candidates for BLVR and for another 11 patients (13.3%), both platforms agreed that patients were not candidates for BLVR. For 27 patients (32.7%), the StratX platform recommended that the patients were potential candidates but required functional assessment (using Chartis system) to determine if collateral ventilation was present. However, in these same patients, the SeleCT platform advised that 14 (16.9%) were not candidates due to incomplete fissures. For 2 (2.4%) patients, SeleCT recommended that the patients were candidates for BLVR, whereas the StratX recommended that the patients were not candidates for BLVR. For 3 (3.6%) patients, SeleCT recommended that the patients were not candidates for BLVR, whereas the StratX platform suggested they were candidates for BLVR. The recommendation to proceed or not proceed with BLVR differed significantly between the two platforms (Supplemental Table 1; p < 0.001).
Discussion
Patient selection for BLVR is critical for procedural success. The difference in quantitative analysis between the two commercially available platforms that we identified for the same patients had a substantial impact on patient candidacy in our cohort. We found that among over 80 patients referred for BLVR at three different medical centers, the two quantitative platforms provided different recommendations for BLVR candidacy in almost a third of patients. In addition, among the patients in this cohort that received valves, nearly 30% received different recommendations depending upon which platform was utilized to determine candidacy for BLVR. Likewise, there were significant differences in fissure integrity scores between platforms, with nearly 1 in 5 patients receiving at least 10% difference estimate in fissure completeness.
Earlier work has shown poor inter-rater reliability of radiologists interpreting fissure completeness.17,18 Additionally, there is significant differences in identifying fissure completeness when comparing CT scans to intra-operative assessment by surgeons.19,20 Likewise, earlier work comparing a functional assessment of fissure integrity to quantitative analysis of CT scans found discordance in just under a third of patients, which matches our discordance of 31%.21,22
However, as both the SeleCT and StratX platforms use a quantitative computer-based algorithm, the human element of inter-observer variability should not be present. Additionally, these platforms use the same input for their analysis as opposed to comparing imaging characteristics with a functional assessment. To our knowledge, this is the first report that compares the two different commercially available quantitative platforms methods for helping to determine candidacy for BLVR and identifies a substantial difference between the two platforms. Given that bias has been described in other computer-based applications,23,24 our finding that the two proprietary algorithms provided discordant results may not be so surprising.
In the clinical trials for BLVR, response rates varied, but in general, about 50% of patients that received valves derived clinical benefits.6 –9 For example, only 37%–56% of patients had a 15% improvement in FEV1, 54%–62% had a clinically meaningful improvement in St. George’s Respiratory Questionnaire, 44%–50% had a 1+ point improvement in the modified Medical Research Council Dyspnea score, and 32%–56% had a 25+ meter improvement in 6-minute walk distance following BLVR.6 –9 Given the stringent inclusion and exclusion criteria applied within these trials, it is likely that the real-world clinical benefit of BLVR is less than that seen in trials. Optimizing patient selection to identify those most likely to derive benefit is critical given the high rate of complication and the expense involved in these procedures. 11
The discrepancies in BLVR candidacy between platforms that we identify suggest that the patient selection process may benefit from further investigation and clarification to best identify the targeted lobes and the patients who might benefit most from the procedure. It may be that we are not selecting appropriate patients for BLVR. Some patients may be selected for the procedure who will never benefit but accept all of the potential risks because of ambiguous quantitative CT scans, while we may be excluding others who may actually derive benefit. This may explain why only 50% of patients undergoing BLVR in the clinical trials had clinically meaningful improvement.
Limitations
Our study should be interpreted in light of several limitations. This was a retrospective project with a limited number of sites and patients. Yet, the findings were consistent across the sites. Second, the clinical impact of these findings is unclear. Given the small sample size and retrospective nature of this project, we are unable to assess if differences in candidacy had any association with clinical outcomes. Moving forward, combining the two quantitative platforms and seeing if patients with concordant recommendations were more likely to derive benefit would be one possible way to test this hypothesis. Third, given the proprietary nature of each platform, we are unable to ascertain why the fissure estimates differed between platforms. As the platforms use different thresholds for emphysema destruction, it is possible that some of the differences in recommendations may be related to this. However, this does not explain the substantial difference in fissure estimates that we identify. In addition, while a functional assessment of fissure integrity may address this bias of fissure scores, it still does not address the discordance in lobe selection that we identified, which is of substantial clinical concern. Finally, the decision to proceed with BLVR ultimately requires a careful assessment by a trained physician.
Our study also has several strengths. Importantly, we used best practices as outlined in clinical trials to define our algorithm for BLVR selection. Additionally, we defined our exposure a priori to minimize bias. By including multiple sites, we were able to increase our sample size and reduce bias and our findings were consistent across each site.
Conclusion
In this cohort of patients evaluated for BLVR across multiple medical centers, nearly a third of patients received different recommendations based on the quantitative platform utilized for assessment. Additionally, there were substantial differences in fissure integrity scores across quantitative platforms. This may affect clinical outcomes of BLVR and may suggest the need to refine the patient selection process.
Supplemental Material
sj-docx-1-tar-10.1177_17534666251314724 – Supplemental material for The impact of quantitative platform on candidacy for bronchoscopic lung volume reduction: a multi-center retrospective cohort study
Supplemental material, sj-docx-1-tar-10.1177_17534666251314724 for The impact of quantitative platform on candidacy for bronchoscopic lung volume reduction: a multi-center retrospective cohort study by Max Wayne, Suchitra Pilli, Hee Jae Choi, Nathaniel Moulton, Praveen Chenna, Allen Cole Burks and Alexander Chen in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
