Abstract
In the last decade several promising bronchoscopic lung volume reduction (BLVR) treatments were developed and investigated. One of these treatments is BLVR treatment with coils. The advantage of this specific treatment is that it works independently of collateral flow, and also shows promise for patients with a more homogeneous emphysema disease distribution. Seven years ago, the very first patients were treated with BLVR coil treatment and currently large randomized, controlled trials are underway. The aim of this article is to review the available literature and provide an update on the current knowledge on the efficacy and safety of BLVR treatment with coils.
Keywords
Background
Chronic obstructive pulmonary disease (COPD) is one of the main chronic diseases, contributing a huge burden to the population worldwide [Viegi et al. 2007]. COPD is incurable and pharmacologic and nonpharmacologic treatments are aimed at reducing symptoms and risks of future events and disease progression [Vestbo et al. 2013]. Treatment options are limited for patients with more advanced stage of COPD, who have significant daily respiratory symptoms. For a small selection of patients, two effective surgical treatment options are available, lung transplantation and lung volume reduction surgery (LVRS). Lung transplantation is only available for a small group of patients due to vigorous patient selection, shortage of donor organs and high costs [Whitson and Hayes, 2014]. Although effective in expertise centres, LVRS is associated with high morbidity in this frail patient population [Criner et al. 2011] and therefore less invasive bronchoscopic lung volume reduction (BLVR) techniques have been developed.
In the last decade several BLVR treatments were developed and investigated. At this moment, the most efficacious BLVR treatment is using one-way endobronchial valves [Mineshita and Slebos, 2014]. The aim of this treatment is to fully occlude an entire lobe and to induce a complete atelectasis of the most diseased and hyperinflated lobe. Unfortunately, only a selected group of patients have the potential to benefit from this treatment [Shah and Herth, 2014]. The responders to valve therapy are particularly patients with absence of interlobar collateral flow (measured by the CHARTIS system) between the target treatment lobe and the adjacent lobe. Other important indicators of success are greater heterogeneity in disease destruction between target and non-target lobes [Shah and Herth, 2014]. One study showed that in a random severe emphysema population with a heterogeneous disease distribution, only 37% of patients had complete lobar fissures [Herth et al. 2012] and thus only a subset of patients with emphysema can be selected for this treatment. A beneficial BLVR treatment that works independently of collateral flow, and also shows promise for patients with a more homogeneous disease distribution is BLVR treatment with coils. Seven years ago, the very first patients were treated with BLVR coil treatment [Herth et al. 2010] and currently large randomized, controlled trials are underway. The aim of this article is to review the available literature and provide an update on the current knowledge on the efficacy and safety of BLVR treatment with coils.
BLVR treatment with coils
The lung volume reduction (LVR) coil system was extensively tested in animal models and an acceptable performance of the system was demonstrated (data not published) before the first treatment in patients took place in 2008. The device consists of two components: the coil and the delivery system, which is composed of a loading cartridge, delivery catheter and forceps. An example of a coil in its predetermined shape and the other components of the LVR coil system are shown in Figure 1. The coils (RePneu, PneumRx Inc., a BTG Inter-national group company, Mountain View, USA) are made from shape-memory nitinol wire, ranging in lengths from 70–200 mm in the initial studies to the currently available 100, 125 and 150 mm sizes to accommodate airways of different sizes. During the treatment procedure, which is performed almost entirely under fluoroscopic guidance, first the guide wire is advanced into the selected airway. After this, the delivery catheter is passed over the guide wire, and the length of the airway is measured using dedicated radio opaque markers on the guide wire to choose the appropriate length of the coil, after which the guide wire is removed. Next, a straightened LVR coil preloaded in the loading cartridge and connected to the biopsy forceps is introduced into the distal end of the catheter and pushed forward. Once the coil reaches the distal end of the loading catheter, the catheter is removed while the coil is held in place. As the catheter is pulled back, the LVR coil returns to its predetermined shape inside the lung, bending the airway and attached parenchyma with it. Finally, the coil is released from the biopsy forceps. On average, 10–12 coils are placed per upper lobe and 10–14 per lower lobe treatment. Generally, both sides of the lung are treated, and this is performed in two separate bronchoscopic procedures with in general 1–2 months in between [Klooster et al. 2014b]. An example of a chest X-ray after bilateral coil treatment is shown in Figure 2. The procedure is performed using a flexible bronchoscope with a 2.8 mm working channel or bigger under fluoroscopic guidance using a C arm. The procedure can be performed either under deep conscious sedation or general anaesthesia (using a laryngeal mask, flexible endotracheal tube or rigid bronchoscope). The average procedure time per lung is around 20–40 min with 10–20 min of fluoroscopy time [Klooster et al. 2014b].

RePneu lung volume reduction coil system. Reproduced with permission of PneumRx Inc., a BTG International group company.

Example of a chest X-ray after bilateral coil treatment.
The coil is designed to compress lung parenchyma most damaged by emphysema. The proposed mechanism of action is that the compression of the parenchyma reduces airflow to the treated parts of the lung and as a consequence increases airflow to healthier untreated parts of the lung. This compression could also potentially lead to less lung hyperinflation combined with an improvement of the function of the diaphragm and other inspiratory muscles. Furthermore, the coil could restore elastic recoil of the lung, which could improve expiratory flow rates and prevent airway collapse with air trapping. However, the exact coil mechanisms still need to be further investigated.
Feasibility, safety and efficacy of LVR coil treatment
To review the feasibility, safety and efficacy of the LVR coil treatment, we included all studies containing original trial data that were published before April 2015. In total, six studies investigated the LVR coil treatment (Table 1). The studies were performed in hospitals in Germany, the Netherlands, the UK and France. In January 2008, the first pilot study started [Herth et al. 2010], followed in April 2009 by a second pilot study with optimized treatment protocol and use of second-generation coils [Slebos et al. 2012]. At the end of 2009 a larger multicentre, prospective cohort study started [Deslee et al. 2014]. Only one randomized, controlled trial (with crossover) has been published so far, which started in January 2010 [Shah et al. 2013; Zoumot et al. 2015]. In 2011, two uncontrolled studies started specifically focusing on patients with homogeneous emphysema [Klooster et al. 2014a] and patients with incomplete fissures [Kontogianni et al. 2014].
Published literature on trials investigating lung volume reduction coil treatment.

RCT, randomized, controlled trial.
Feasibility
In the six studies, a total of 168 patients were treated, of whom 122 were treated bilaterally. The reasons for unilateral treatment were: 26 per protocol, one patient died, three patients had only one eligible lung, two patients already had a large improvement after the first lung, two patients were lost to follow up, in five patients the physician decided to treat only one lung, two patients withdrew and in five patients the reason was not mentioned in the article. At baseline the average forced expiratory volume in 1 s (FEV1) ranged from 27% to 33% predicted and the average residual volume (RV) from 225% to 279% predicted. Except for the first pilot study, the median number of coils placed per procedure was approximately 10 (range 5–15). The median procedure time was approximately 40 min per procedure (range 20–135). In general, the studies reported that the procedure was feasible and well tolerated by the patients.
Safety
Periprocedural safety
All six studies reported that no periprocedural adverse events occurred and no coils needed to be removed, demonstrating procedural safety.
Recovery period
Four studies divided the reported adverse events into a so-called ‘post treatment recovery period’ (⩽30 days after each LVR coil procedure) and the follow-up period (1–6 months of follow up). We summarize the results of these studies in Table 2. In the recovery period the reported adverse events were comparable between studies. In total, 11 patients (6.5% in total or 3.5% per procedure risk) had a pneumothorax after the procedure. Furthermore, after the procedure, COPD exacerbations and pneumonia requiring hospitalization were reported. Furthermore, Deslee and colleagues [Deslee et al. 2014] concluded that the rates of post-procedure exacerbations and pneumonia were comparable to reported events after BLVR treatment with valves and also did not exceed the rates reported in a sham bronchoscopy control group without intervention in the same population of patients with severe emphysema [Shah et al. 2011]. Other LVR coil specific procedure-induced events that occurred were very mild, self-limiting haemoptysis (approximately 50% of patients) and transient chest discomfort (approximately 33% of patients), both during the initial days after the procedure and requiring no intervention.
Reported (serious) adverse events in the recovery and follow-up period.
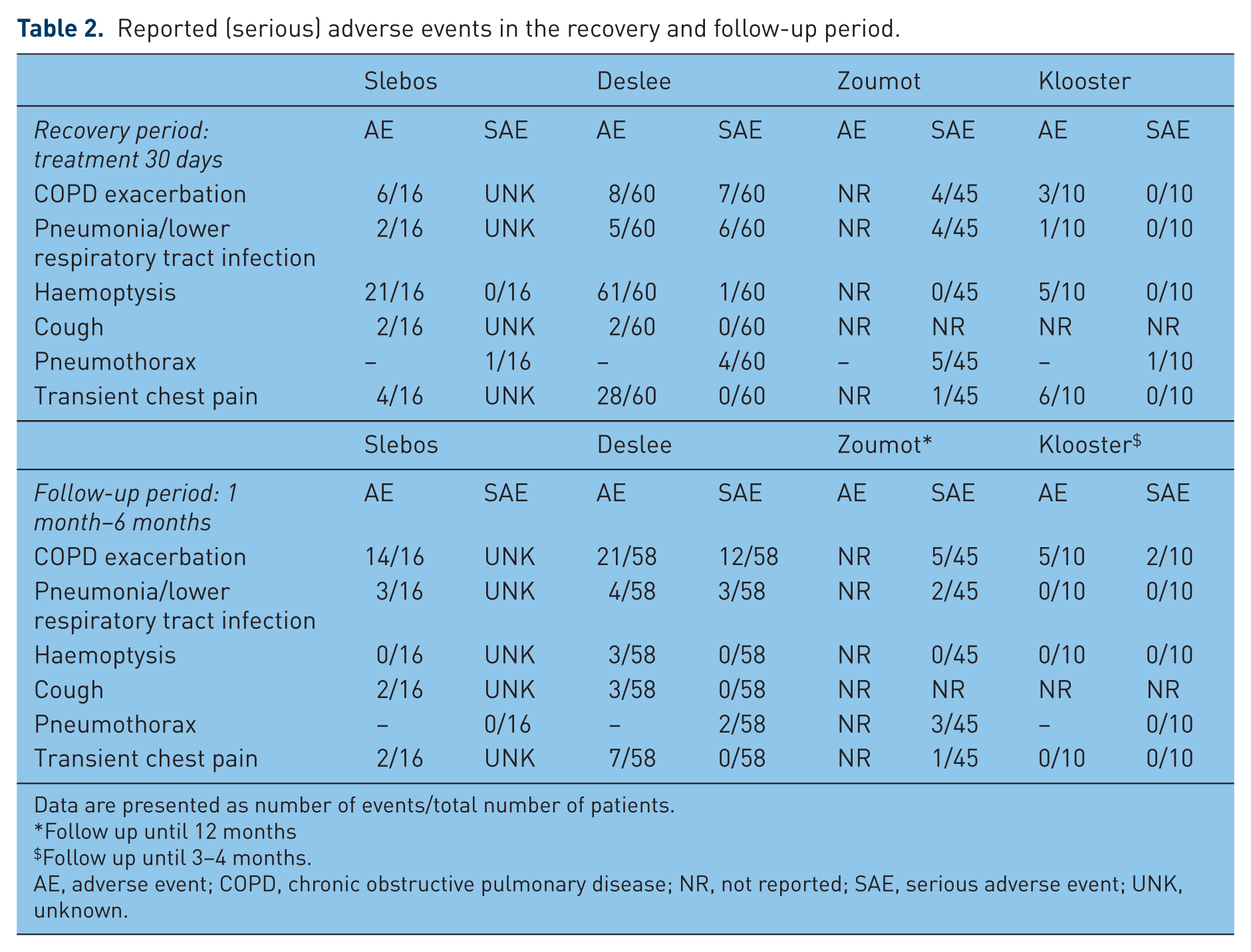
Data are presented as number of events/total number of patients.
Follow up until 12 months
Follow up until 3–4 months.
AE, adverse event; COPD, chronic obstructive pulmonary disease; NR, not reported; SAE, serious adverse event; UNK, unknown.
Follow-up period (1–6 months after the treatment)
In the follow-up period up to 6 months, no unexpected adverse events were reported (Table 2). Furthermore, the randomized, controlled trial did not detect between-group differences in serious adverse events between treatment group and the usual care group [Shah et al. 2013].
Efficacy
Lung function [forced vital capacity (FVC), FEV1, RV), exercise capacity [6 min walk distance (6MWD)], quality of life (St George’s Respiratory Questionnaire (SGRQ)] and dyspnoea severity [modified Medical Research Council (mMRC)] were measured in most of the studies. An overview of these results is summarized in Table 3.
Efficacy results of lung volume reduction coil treatment in the published studies.
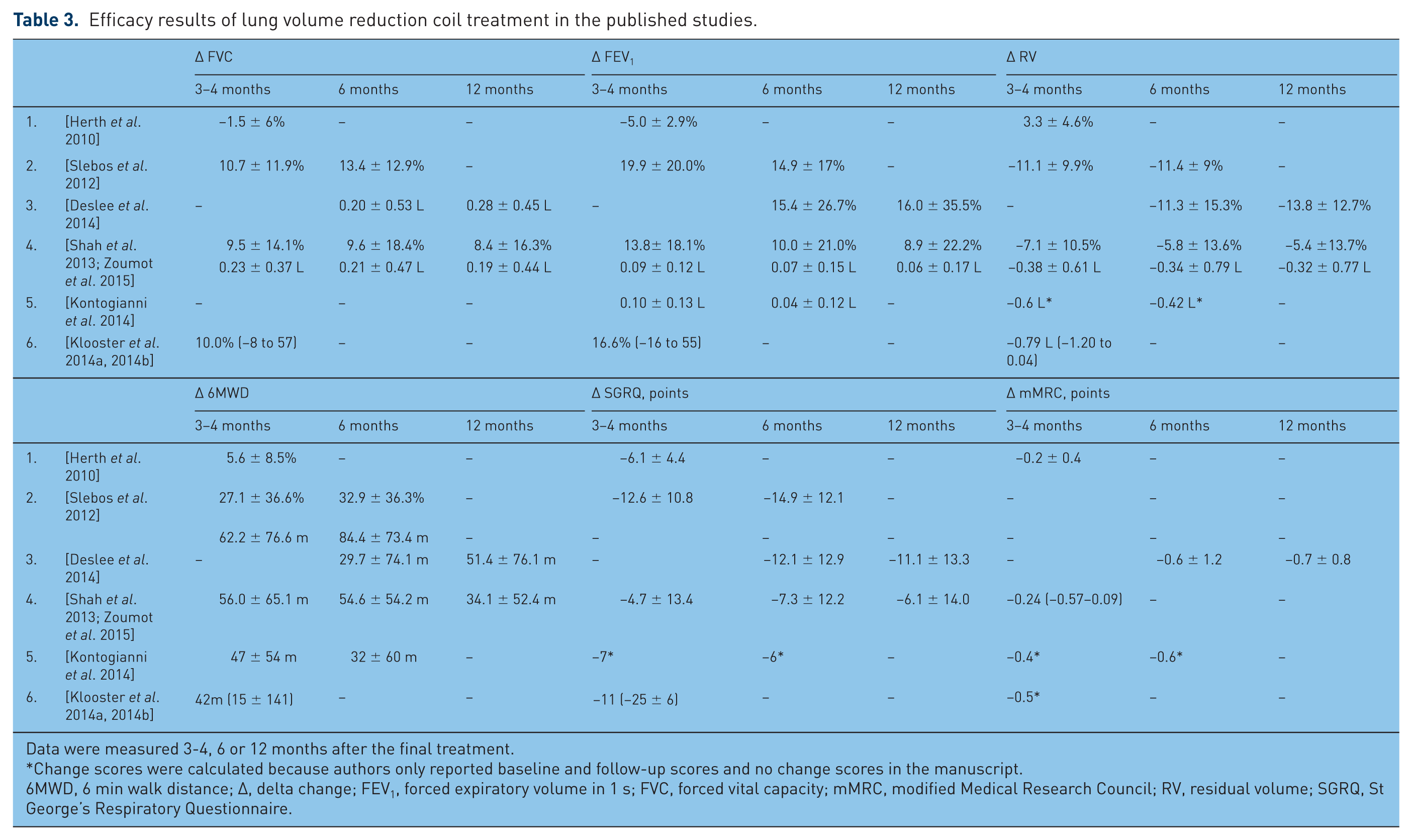
Data were measured 3-4, 6 or 12 months after the final treatment.
Change scores were calculated because authors only reported baseline and follow-up scores and no change scores in the manuscript.
6MWD, 6 min walk distance; Δ, delta change; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; mMRC, modified Medical Research Council; RV, residual volume; SGRQ, St George’s Respiratory Questionnaire.
Pulmonary function
In general, the efficacy results of the first pilot study were less distinct. The reason for this could be that this pilot study with fewer coils being placed per lobe was not intended or powered to analyse effectiveness. Furthermore, the treatment was optimized after this first study using both a second-generation coil design and a larger number of coils placed per lobe. Therefore, the following results do not include the results of the first pilot study. At the available 3–4-month follow-up time point, the studies reported an average increase in FEV1 of 14–20% and after 6 months of approximately 13%. After 3–4 months, RV decreased on average by −0.4 to −0.8 liter, and after 6 months by approximately −0.4 liter.
Exercise capacity
At the 3–4-month follow up, the studies reported an average increase in 6MWD of 42–62 m and after 6 months of 30–84 m.
Quality of life
At the 3–4-month follow up, the studies reported an average decrease in SGRQ total score of 7–13 points and after 6 months of 6–15 points.
Dyspnoea severity
At the 3–4-month follow up, the studies reported an average decrease in mMRC dyspnoea score of 0.2–0.5 points and after 6 months of 0.6 points.
Other measurements
Besides the variables described above, only some limited additional results on computed tomography (CT) scan data, airway resistance and physical activity level were reported up to the time of writing. Two studies evaluated CT scan data. In 10 patients with homogeneous emphysema, the lung volumes in the treated upper lobes measured on the CT scan significantly decreased from 3204 to 2941 ml [Klooster et al. 2014a], indicating that the coils do reduce lung volume. In unilaterally treated patients with heterogeneous emphysema, the lung volumes and emphysema index significantly decreased but the emphysema volume did not [Kontogianni et al. 2014]. One study, including only patients with homogeneous emphysema, showed that the airway resistance significantly improved 4 months after the final treatment [Klooster et al. 2014a]. Physical activity was measured during a pilot study that included 14 patients from one centre who participated in one of two of the published studies [Slebos et al. 2012; Deslee et al. 2014]. Six months after the treatment, physical activity did not significantly improve despite significant changes in static lung hyperinflation [Hartman et al. 2012], indicating that a post-treatment physical therapy program might be of use in this disabled patient group.
Responders to the treatment
In Table 4 the reported responder rates are summarized based on the number of patients who reached the minimal important difference for RV, FEV1, 6MWD or SGRQ. In the short term up to 6 months after the coil treatment, the reported responder rate for RV was approximately 64% (range 57–70%), for the FEV1 approximately 56% (range 43–66%), for the 6MWD approximately 70% (range 53–86%) and for SGRQ approximately 65% (range 46–79%). The chosen minimal important differences (MIDs) for RV and FEV1 slightly differed between studies, making them more difficult to compare.
Reported responder rates based on number of patients who reached the MID.

Data are presented as percentage of patients who reached a minimal important difference (MID).
6MWD, 6 min walk distance; FEV1, forced expiratory volume in 1 s; RV, residual volume; SGRQ, St George’s Respiratory Questionnaire.
At this time, little is known about more exact predictors of response for LVR coil treatment. The first pilot study indicated that patients with heterogeneous emphysema might experience more pronounced benefits from the treatment [Herthet al. 2010]. However, later studies showed that patients with homogeneous emphysema benefit from the treatment to the same extent [Desleeet al. 2014; Zoumot et al. 2015; Klooster et al. 2014a]. Furthermore, upper lobe treatment compared with lower lobe treatment was only significantly different for change in FEV1 but not for change in RV, 6MWD or SGRQ [Deslee et al. 2014]. Two studies performed multivariate regression analysis to investigate independent responders of beneficial change in patient outcomes. The first study reported that none of the input variables (RV% predicted, RV/total lung capacity, FEV1% predicted, FVC, age, carbon monoxide lung transfer factor(TLCO), emphysema type) were useful in associating patient outcomes at 6 months [Deslee et al. 2014]. In the other study, only worse baseline 6MWD performance was independently associated with a greater improvement in 6MWD after 3 months [Kontogianni et al. 2014].
Long-term follow up (⩾12 months)
Until now, only three published studies investigated the LVR coil treatment after 6-month follow up. Deslee et al. [Deslee et al. 2014] followed a subset of their study population and Zoumot et al [Zoumot et al. 2015] their complete study population [including cross-over] up to 12 months after final treatment. Followed a subset of their study population up to and …’] Furthermore, results are reported of patients who were treated in one single centre in two of the LVR coil trials [Slebos et al. 2012; Deslee et al. 2014] and were invited for a voluntary annual follow-up visit up to 3 years after the treatment [Hartman et al. 2015].
Safety
In the long term, up to 3 years after treatment, the authors witnessed no late pneumothoraces, coil migrations, no major haemoptysis, no infectious complications or unexpected adverse device events and no treatment-related deaths [Hartman et al. 2015].
Efficacy
The two studies demonstrated a sustained response at 12-month follow up in static lung hyperinflation, exercise capacity, quality of life and dyspnoea severity, and one of the two studies in lung function as well [Deslee et al. 2014; Hartman et al. 2015]. After 12 months, the clinical benefit gradually declined over time and returned to baseline values after 3-year follow up. After 3 years, still approximately 50% of the patients maintained improvements above the MID for 6MWD, SGRQ and mMRC [Hartman et al. 2015]. However, the follow up in this study was voluntary with no control data being available.
Conclusion and future perspectives
Until now, six studies investigating LVR coil treatment have been published reporting the results of 168 treated patients up to 12 months after coil treatment. The advantage of this specific coil treatment is that it works independently of collateral flow, and besides patients with heterogeneous emphysema, patients with homogeneous emphysema can benefit as well. The published studies showed that the treatment is feasible and has an acceptable safety profile. Furthermore, clinical benefits of the treatment in terms of lung function, static hyperinflation, exercise capacity, quality of life and dyspnoea severity were reported up to 12 months after the treatment.
Unfortunately, until now only one randomized, controlled trial has been published, so clearly more are needed. Currently, two large randomized, controlled trials are underway, investigating 315 patients (RENEW study) [ClinicalTrials.gov identifier: NCT01608490] and 100 patients (STICREVOLENS) [ClinicalTrials.gov identifier: NCT01822795] [Deslee et al. 2012]. These important studies will increase knowledge of the treatment’s efficacy. Furthermore, the STICREVOLENS trial will also investigate the cost effectiveness of the treatment and will provide insight into this aspect of the treatment as well [Deslee et al. 2012].
In the six published studies, the chosen outcome parameters were almost identical, which is important for the comparison of different studies. However, to gain more insight into the exact mechanism of action of LVR coil treatment it would be useful to measure other clinical aspects as well, like diaphragm function, lung compliance and efficiency of ventilation during exercise testing. Assessing other CT scan parameters such as the emphysema destruction scores or emphysema phenotyping could also be useful for more insight into the best response profile of the treatment. Unfortunately, up to this point no clear predictors for treatment success could be identified. Including other variables and measurements could increase our knowledge on this. For example, Deslee and colleagues [Deslee et al. 2014] recommended also investigating the following potential variables with regard to response to treatment: nuanced emphysema phenotypes beyond heterogeneous or homogeneous classification, such as more or less small airway disease, centrilobular versus panlobular emphysema and variability in placement strategies, including proximal versus distal placement within the subsegmental airways and the number and size of coils deployed.
Until now, most studies only investigated the efficacy of the treatment in the short term up to 6 months after treatment. Only two studies followed a subgroup of patients up to 1 year per protocol [Deslee et al. 2014]. Furthermore, one centre published results of patients who participated in two of the published trials who were invited for a voluntary yearly follow-up visit to the hospital [Hartman et al. 2015]. These results suggest that the treatment is still beneficial after 12 months but that clinical benefit declines over time, although a significant group of patients still benefit 3 years after the treatment. The follow up was voluntary and therefore the results should be interpreted with caution and more research is needed investigating the treatment in the long term. In the current ongoing RENEW study, a 5-year follow up is included in the protocol.
To summarize, the published studies investigating LVR coil treatment show that the treatment is feasible, has an acceptable safety profile and is clinically effective in the short term. The treatment is a valid option for patients with severe emphysema with presence or absence of collateral flow and a homogeneous or heterogeneous disease distribution. More insight is needed into efficacy in the long term, the exact mechanism of action and the best-responder profile of the treatment. Large randomized, controlled trials are underway and will provide more knowledge on the potential of the treatment.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
PneumRx Inc., a BTG International group company (PneumRx/BTG), was not involved in the writing of this review. KK received travel grants from PneumRx/BTG and financial support from PneumRx/BTG as a consultant not related to this manuscript. DJS is a physician advisor to PneumRx/BTG, is the principal investigator of three published coil trials, and received travel grants and speaker’s fees for presentations at scientific and educational meetings from PneumRx/BTG.
