Abstract
Bronchopleural fistula (BPF) is a potentially fatal complication and remains a surgical challenge. Concomitant problems, such as pulmonary infection and respiratory failure, are typically the main contributors to mortality from BPF because of improper contact between the bronchial and pleural cavity. We present the case of a 75-year-old male patient with a history of right upper lobe lung cancer resection who developed complex BPFs. Following appropriate antibiotic therapy and chest tube drainage, we treated the fistulas using endobronchial valve EBV placement and local argon gas spray stimulation. Bronchoscopic treatment is the preferred method for patients who cannot tolerate a second surgery because it can help to maximize their quality of life. Our treatment method may be a useful reference for treating complex BPF.
Keywords
Introduction
Bronchopleural fistula (BPF) is one of the most serious and rare postoperative complications. BPF is most commonly caused after pneumonectomy, with an estimated frequency of 4.5% to 20% for total pneumonectomies and 0.5% to 3% for lobectomies. The mortality rate for BPF after a pneumonectomy can be as high as 16% to 71%.1,2 The primary clinical symptoms of BPF include fever, chills, purulent cough, chest distress, and dyspnea. If left untreated, BPF can lead to recurrent lung infections, sepsis, respiratory failure, and even death. Therefore, early detection and treatment of BPF are vital to improve patients’ prognosis and quality of life. A multidisciplinary approach, including thoracic surgery and pulmonary intervention, is often required for BPF treatment.
We report a successful case of bronchoscopic treatment of complex BPFs that occurred 15 years after lung cancer surgery. Despite the various methods available for preventing BPF, buttressing the bronchial stump 3 and treating BPF, 4 no treatment is entirely effective, making the treatment of BPF a challenging and long-lasting process.
Case report
A 75-year-old male patient, with a height of 156 cm and body weight of 42 kg, had a history of right upper lung cancer radical resection 15 years previously. The lung cancer was poorly differentiated non-small cell type, and immunohistochemistry was positive for carcinoembryonic antigen, p53, thyroid transcription factor 1, and Ki-67 (45%). Immunohistochemistry was negative for epidermal growth factor receptor and neuron-specific enolase.
After exposure to cold, the patient developed a cough, white phlegm, and chest tightness. During this period, he fell and developed right chest pain, with a Numeric Pain Rating Scale score of 2 points. Chest computed tomography (CT) showed a right upper lobar bronchial stump, and encapsulated fluid pneumothorax in the middle and upper lobe of the right lung. The pneumothorax was communicating with the anterior chest wall. CT also showed infectious lesions in the right middle lobe and a fistula at the distal end (Figure 1a, b). The treatment plan and potential complications were discussed with the patient and his family, who provided written informed consent for treatment.

Radiological and bronchoscopic images taken on 7 May 2023 (a–c) and 10 May 2023 (d). (a) A bronchopleural fistula can be seen in the right middle bronchus. (b) The chest tube (green arrow) and a chest wall defect with pneumothorax (orange arrow) can be seen. (c) Purulent pleural effusion and (d) an endobronchial valve was inserted into the right middle lobe bronchus.
The patient was treated with antibiotics for phlegm reduction. Thoracic drainage for the phlegm was placed, but the thoracic drainage could not be removed. The chest tube drained some gas and a small amount of purulent pleural fluid (Figure 1c). Bronchoscopy showed the right upper lobar bronchial stump and a large amount of purulent discharge in the middle lobe of the right lung. A Cook balloon (Arndt Endobronchial Blocker Set, C-AEBS-7.0-65-SPH-AS; Cook Incorporated, Bloomington, IL, USA) was used to seal the bronchus in the right middle lobe, and verified that the BFP originated from the right middle bronchus.
The patient underwent Zephyr endobronchial valve (EBV) (EBV-TS-5.5; Emphasys Medical Inc., Redwood City, CA, USA) placement under general anesthesia (Figure 1d). After the operation, the patient’s cough and sputum were reduced, and no fluctuation in the water column of the chest tube or air leak overflow was observed.
Four days post-surgery, the patient developed a fever of 38.8°C, and the chest tube showed increased air leakage. A chest CT scan indicated a possible fistula in the right upper bronchial stump and right middle lobe atelectasis (Figure 2a). During bronchoscopy, the EBV position in the right middle lobe was found to be optimal, with purulent secretion attached to the one-way duckbill valve of the EBV. We probed the right upper lobe with a guide wire (with a soft front end), which traveled approximately 5 cm into the stump, and confirmed the presence of a small fistula. We treated the fistula with a local spray of argon (Figure 2b, c, d). After the intervention, the chest tube still showed considerable water column fluctuation, but air leakage was reduced. The patient was discharged with the leakage tube and continued oral antibiotic therapy. The tube leaked air occasionally.
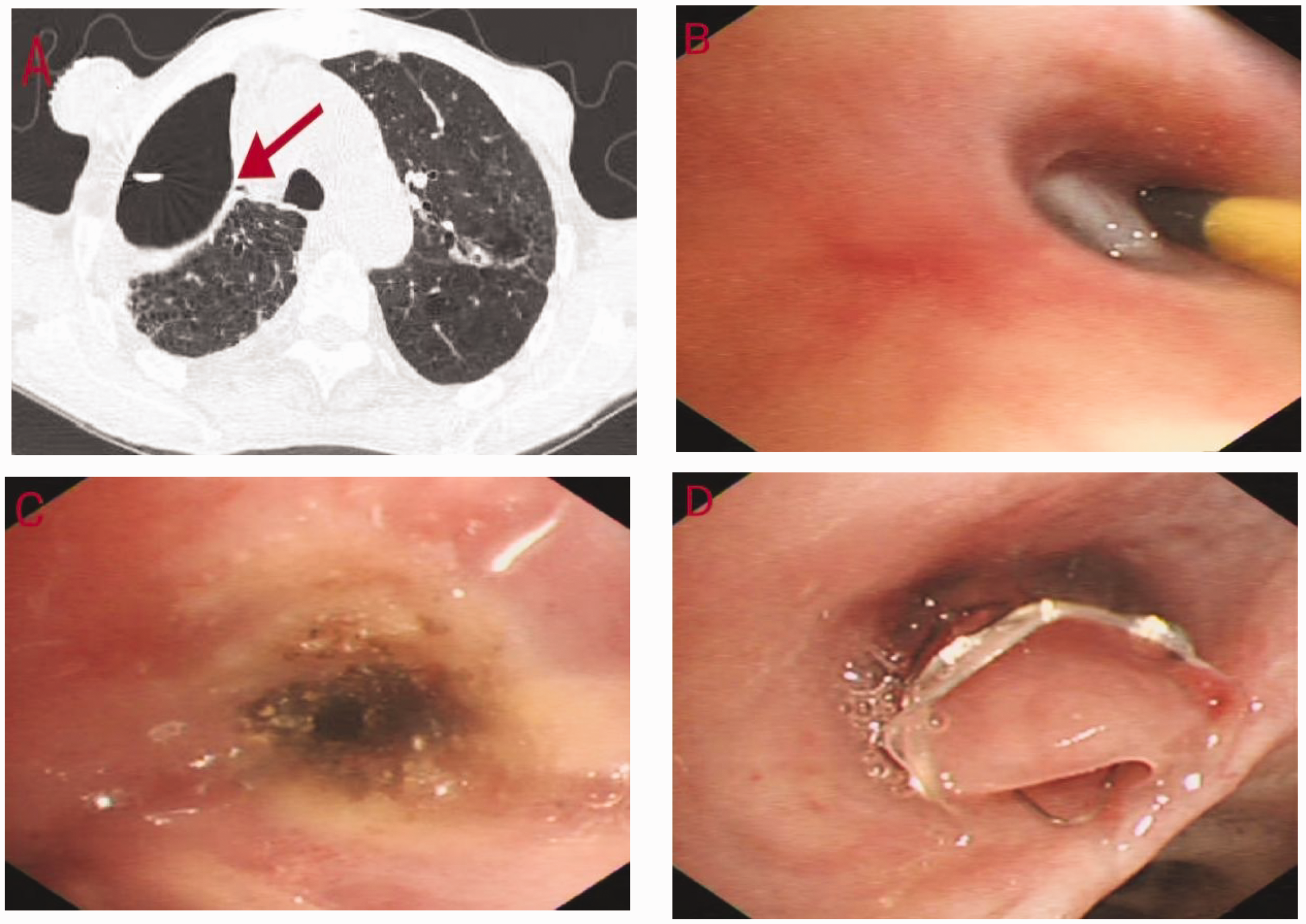
Radiological and bronchoscopic images 4 days postoperatively taken on 20 May 2023. (a) Suspected fistula in the right upper bronchial stump (red arrow). (b) Probing of the right upper lobe with a guide wire. (c) Fistula scarring after argon gas spraying and (d) position of the endobronchial valve in the right middle lobe.
On the 34th day postintervention, the patient reported less cough and sputum, lower inflammatory markers than previous measurements, and no chest tube air leakage. A chest CT scan showed a reduction in the encapsulated fluid pneumothorax and an improvement in the right middle lobe infection. Bronchoscopy showed that the tiny fistula of the right upper lobar bronchial stump had healed. The chest tube was removed and the patient was discharged with a prescription for continued oral antibiotics.
On the 58th day postoperatively, the patient reported mild coughing and sputum, no chest distress, improved exercise tolerance, and a weight gain of 2 kg. A chest CT scan showed that the right middle lobe had re-expanded and no fistula was detected (Figure 3a). Under general anesthesia, the EBV was removed with biopsy forceps (Figure 3b, c, d). The right middle lobe was unobstructed postoperatively, and there was minimal granulation tissue hyperplasia in the mucosa. A chest CT scan and bronchoscopy confirmed that the complex pleural fistula had healed. Therefore, the patient was discharged the next day after operation. At outpatient follow-up 6 months after surgery, the patient had a slight cough, no sputum, and the absence of any tightness in the chest. A flowchart of the patient’s course is shown in Figure 4.
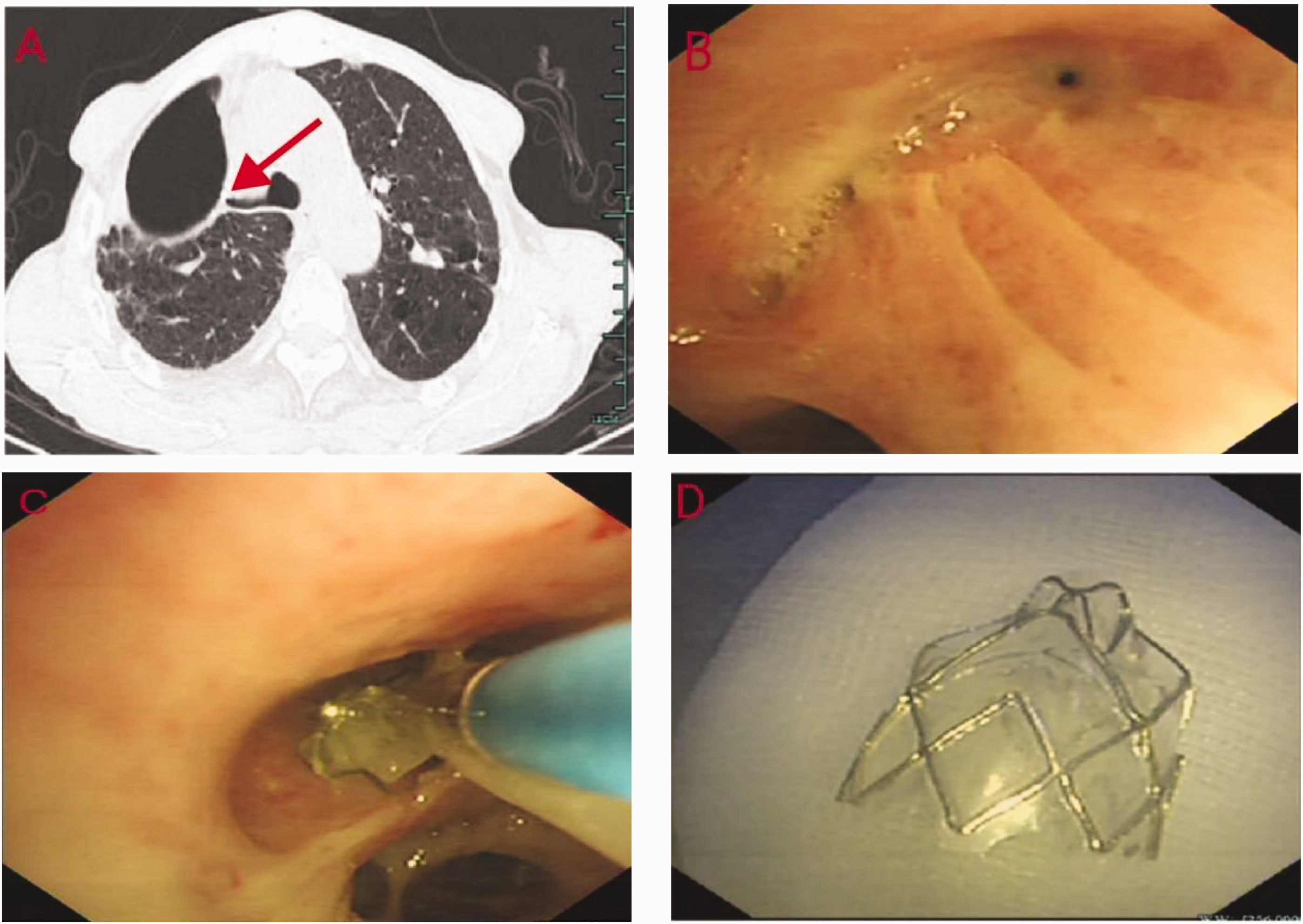
Radiological and bronchoscopic images from 58 days postoperatively taken on 11 July 2023. (a) The right upper bronchial stump can be seen. (b) The tiny fistula has healed. (c) The endobronchial valve was removed with biopsy forceps and (d) the removed endobronchial valve.
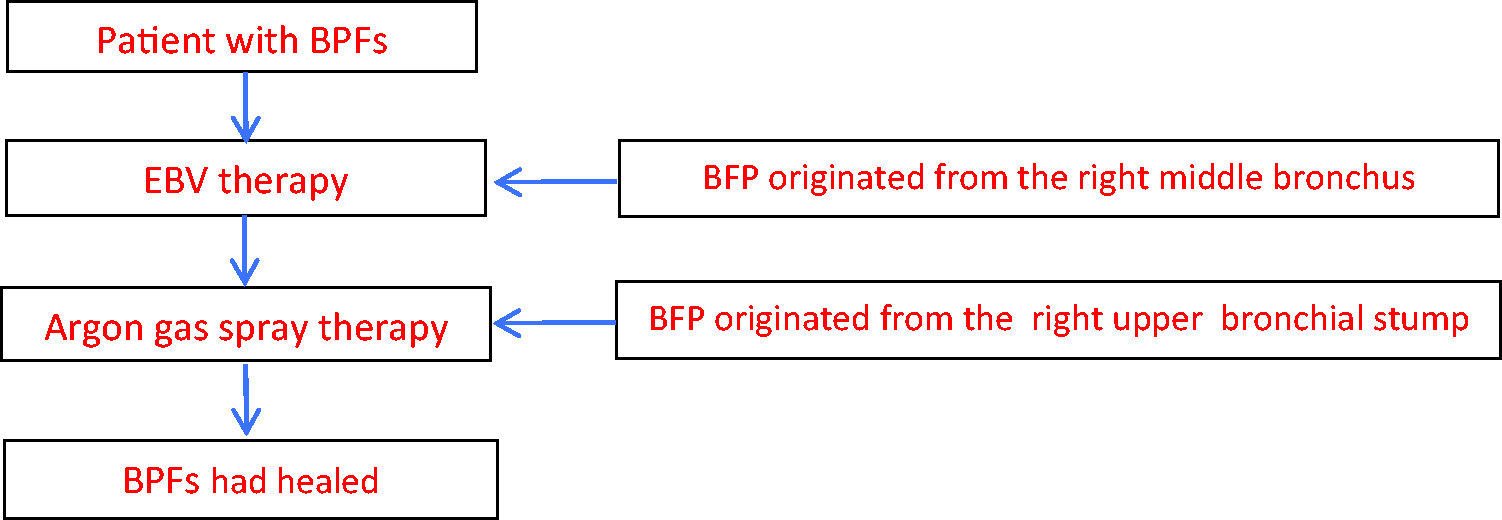
Study flowchart.
This study adheres to the CARE guidelines. 5
Discussion
EBVs are primarily used in lung volume reduction and are also used in treating BPF. 6 In a recent study by Wang et al., 33 patients with BPFs were treated with a total of 63 EBVs. 7 The success rate was 66.7%, with a mean time for successful removal of EBVs of 102.2 ± 31.3 days. Major postoperative complications included valve secretion adhesion and granulation tissue hyperplasia. Song et al. conducted a study on 26 patients, and found that approximately half of them developed BPF after surgery. 8 The effectiveness of EBV treatment was 73.1%, with minimal trauma, a short operation time, and few complications. Another study of 115 BPF cases also showed that EBV treatment was effective in >70% of patients without related complications. 9 These studies show that EBV treatment is effective for BPF with relatively mild complications. Better treatment results can be achieved with sealing agents when the patient’s fistula is <5 mm, especially when it is ≤3 mm. Electrocoagulation or laser stimulation can also be used to form scar tissue near the fistula, promoting its healing.
Our patient had a history of right upper lobe lung cancer resection 15 years previously and residual enveloped hydropneumothorax in the right middle and upper lobe. A recent cold and subsequent chest trauma led to an aggravated infection involving the chest wall and a BPF in the right middle bronchus. After treatments including antibiotics, nutritional support, and chest drainage, an EBV was placed under general anesthesia. Postoperatively, the patient experienced reduced cough and chest tightness, and air leakage in the chest tube was considerably reduced. However, fistula closure led to poor pus drainage, which worsened the disease owing to infection, and caused fever and a postoperative residual fistula of the right upper lung. Local argon gas spray was administered, and antibiotics, nutritional support, and chest drainage were continued postoperatively. The valve was eventually successfully removed. The patient’s preoperative chest tube indwelling time was 7 days, the postoperative drainage tube indwelling time was 34 days, and the time for successful removal of the EBV was 58 days, all of which were shorter than the times reported in Wang et al. 7 Mild granulation tissue hyperplasia occurred after EBV removal, but there was no fistula recanalization, which is consistent with previous reports.7,8 However, further research with a larger patient cohort is necessary to confirm these findings.
Conclusions
Uniform guidelines for the standardized treatment of BPF are not available. Bronchoscopic treatment is usually recommended for fistulas with a diameter of ≤8 mm. However, closing large fistulas is challenging. This case report emphasizes that treatment of BPF should be tailored to the fistula’s location and size, the time from onset to the time of diagnosis, and the patient’s baseline clinical condition. This recommendation is especially true for patients with multiple and complex BPFs. Selecting an appropriate endoscopic treatment method for each fistula, based on its specific situation, often leads to relatively good treatment outcomes.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241245269 - Supplemental material for Complex bronchopleural fistulas: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605241245269 for Complex bronchopleural fistulas: a case report by Miaoying Cao, Jing Yi, Hongjie Bao, Jian Sun and Yefeng Chen in Journal of International Medical Research
Footnotes
Author contributions
MYC gathered the patient’s clinical data, drafted the manuscript, and reviewed the literature. JY and HJB critically reviewed and edited the manuscript. JS and YFC collected and interpreted the clinical data. All authors read and approved the final manuscript.
Availability of data and materials
All data generated in this study are included in the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
We obtained written informed consent from the patient for publication. The Institutional Review Board of Shaoxing People’s Hospital Ethics Committee approved this case report (Approval No. 202403011738000043625).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
