Abstract
Obstructive diseases of the airway are a diverse group, although they share in common airway narrowing as a sequel to inflammation, which leads to increased work of breathing. Optimal treatment strategies for this heterogeneous group of asthma, chronic bronchitis and emphysema should be multidimensional and embrace pharmacological and nonpharmacological means as well as surgery in a highly select group of patients with emphysema. We review the current status of the bronchoscopic interventions that have been in development for the past decade with the objectives of providing better symptom control in asthma and palliation in individuals with emphysema who are otherwise poor candidates for lung volume reduction surgery.
Bronchial thermoplasty
Overview
Asthma is a chronic inflammatory disorder of the airways. The airway inflammation causes recurrent episodes of wheezing, breathlessness, chest tightness and coughing. The airway hyperresponsiveness that characterizes asthma is excessive bronchoconstriction in response to a stimulus, a consequence of chronic airway inflammation that leads to mucus production, airway remodeling and smooth muscle hypertrophy.
In the last decade, treatment of asthma has focused on abating airway inflammation, and most patients with asthma derive good disease control from inhaled corticosteroids. However, 10% of cases remain symptomatic despite high-dose inhaled corticosteroids, often in combination with long-acting β2 agonists and leukotriene inhibitors [Holgate and Polosa, 2006]. Since inflammation is not the only pathophysiological mechanism of asthma, these therapies have not been found to reverse the airway remodeling and smooth muscle hypertrophy responsible for bronchoconstriction [Bergeron and Boulet, 2006]. A novel approach that targets airway remodeling by reducing airway smooth muscle mass has recently been approved by the US Food and Drug Administration to complement standard drug treatments for patients with severe persistent asthma.
Procedure
Bronchial thermoplasty (BT) uses heat to induce structural changes of the airways. With the Alair system (Boston Scientific, Maple Grove, MN, USA) controlled thermal energy is administered to the patient’s airway walls via a radiofrequency catheter during flexible bronchoscopy. The distal tip of the radiofrequency catheter measures 1.5 mm, and can be applied through a 2 mm working channel of the bronchoscope. Thermal energy at 65°C is circumferentially transmitted to the airway via a four-pronged expandable electrode array that has a 5 mm area of contact with the airway wall (Figure 1). A 10 s pulse of thermal energy is administered to the airway wall each time the footswitch is activated. All accessible airways measuring 3–10 mm in diameter are treated distal to proximal by withdrawing the radiofrequency catheter 5 mm at a time aided by markers on the catheter following delivery of each energy pulse. With the exception of the right middle lobe, due to concerns about the middle lobe syndrome (bronchiectasis from stricture), all other lung lobes are sequentially treated in three procedures lasting 45–60 min each. BT treatments are performed 3 weeks apart to ensure healing of the thermally treated airway sites, and patients are prescribed 50 mg of prednisolone for 3 days to minimize airway inflammation arising from BT.

Bronchial thermoplasty.
Studies on bronchial thermoplasty
Preclinical studies on canine airways have shown that airway recovery was complete after thermal injury and the only histological difference between treated and untreated airways 3 months after BT was the reduction of smooth muscle mass. At the treated sites smooth muscle was replaced by connective tissue, and even at 3 years there was persistent reduction of smooth muscle mass which correlated with decrease in airway hyper-responsiveness to methacholine [Danek et al. 2004; Miller et al. 2001]. The first human study involving BT to the airways of patients scheduled for lung cancer surgery confirmed a similar effect on human airway smooth muscle with resolution of inflammatory changes over 1–3 weeks.
Four key clinical studies in patients with asthma ensued (Table 1). The first was a feasibility study consisting of 16 patients with mild to moderate asthma, and the results showed significant reduction in airway hyperresponsiveness associated with symptom improvement that lasted for more than 2 years [Cox et al. 2006]. Asthma Intervention Research (AIR) was a large multicenter, prospective, randomized trial of 112 patients with moderate to severe asthma that showed objective and subjective benefits of BT as early as 3 months after treatment and lasting up to 12 months. Patients who received BT had 10 fewer mild exacerbations per year compared with controls, demonstrated improved peak flow rates, more symptom-free days, better symptom scores and Asthma Quality of Life (QoL) and Asthma Control questionnaires and less need for rescue medications. However, forced expiratory volume in 1 s (FEV1) and airway hyperresponsiveness were not significantly different [Cox et al. 2007]. In the Research in Severe Asthma (RISA) trial which recruited 32 patients with severe and refractory asthma [Pavord et al. 2008], improvements were observed in FEV1, QoL, asthma control and use of rescue medication. Although the AIR and RISA trials failed to demonstrate change in airway hyperresponsiveness, BT was superior to standard care for asthma control. The AIR2 study incorporated a sham arm to eliminate plausible placebo effects in previous studies. A total of 287 patients with severe asthma who remained symptomatic despite compliance with combination therapy (high-dose inhaled corticosteroid and long-acting β2 agonist) were recruited to participate in this randomized, double-blind, sham-controlled trial. Patients who received BT reported improvements in Asthma QoL scores that persisted for a year; significant reductions of 32% in severe exacerbations, 84% in emergency room visits and 73% in hospitalizations, as well as fewer days away from work, school or other daily activities [Pavord et al. 2008]. The efficacy of BT persisted at 2 years of follow up [Castro et al. 2011; Wu et al. 2011].
Clinical trials of bronchial thermoplasty.
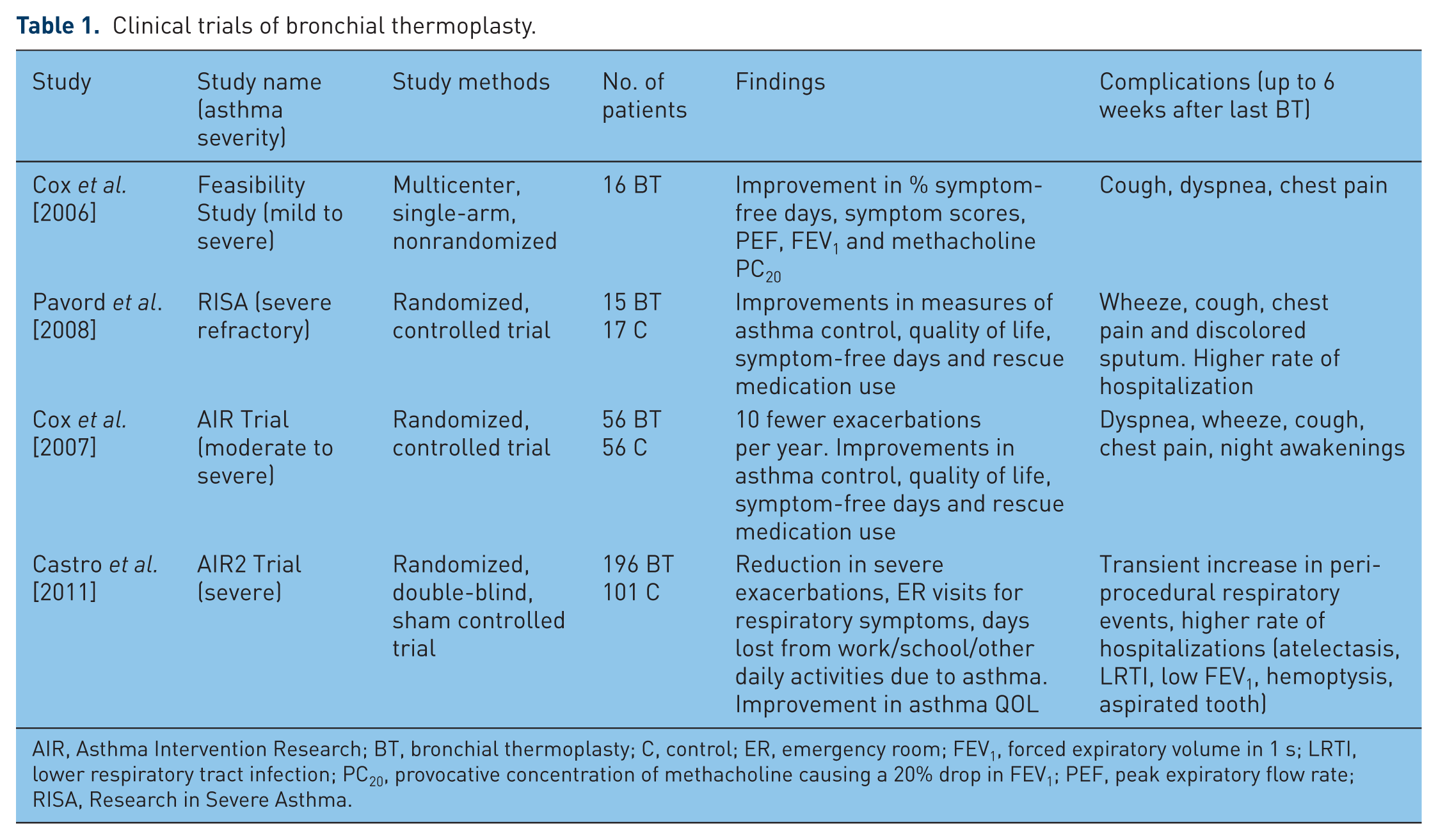
AIR, Asthma Intervention Research; BT, bronchial thermoplasty; C, control; ER, emergency room; FEV1, forced expiratory volume in 1 s; LRTI, lower respiratory tract infection; PC20, provocative concentration of methacholine causing a 20% drop in FEV1; PEF, peak expiratory flow rate; RISA, Research in Severe Asthma.
Safety of bronchial thermoplasty
The AIR2 trial documented a short-term increase in wheezing, upper and lower respiratory tract infections, atelectasis and hemoptysis only during the treatment period. Although the most significant adverse event was hemoptysis that required bronchial artery embolization, no respiratory failure necessitating mechanical ventilation, pneumothorax, cardiac arrhythmias or death were documented with BT. High-resolution CT thorax at baseline, 12 and 24 months post treatment did not reveal parenchymal abnormalities or airway stricture [Wu et al. 2011; Cox et al. 2008, 2009, 2010].
Bronchial thermoplasty: discussion
Half of patients with severe asthma do not have their symptoms controlled despite compliance with medications [Thomson et al. 2011]. BT complements anti-inflammatory therapy which is the cornerstone of asthma treatment by targeting airway smooth muscle responsible for bronchoconstriction. Notably, increased airway smooth muscle mass has been found on autopsy in patients with fatal or severe asthma, thereby revisiting the important role smooth muscle hypertrophy plays in airway remodeling of this subset of patients [Bateman et al. 2001]. It is apparent that phenotypes exhibiting different responses to treatment exist in the group with severe asthma [Pepe et al. 2005; Haldar et al. 2008], and an example is a patient whose symptoms are discordant with the degree of inflammation and does not respond to escalating doses of anti-inflammatory therapy. As therapy for asthma becomes individualized, guided by sputum analysis and radiological imaging, better outcomes may be achieved for this subgroup through the ablation of airway smooth muscle using BT in addition to anti-inflammatory treatment. The benefits of BT would outweigh the risks, particularly in those with symptom-predominant disease due to airway hyperresponsiveness and bronchoconstriction, and do not abate with repeated courses of oral steroids, high doses of inhaled corticosteroids or in combination with long-acting β2 agonists and leukotrienes (Figure 2). The role of BT in chronic obstructive pulmonary disease (COPD) has yet to be determined. It is foreseeable that with clearer characterization of asthma phenotypes, it will aid the physician in personalizing treatment [Good et al. 2012].

Individualized therapy according to asthma phenotypes.
Bronchoscopic lung volume reduction in emphysema
Overview
COPD is a major cause of mortality and morbidity worldwide, and is the fourth leading cause of death. Tobacco smoking accounts for 80–90% of COPD with more than 2 million people in the USA with emphysema [Chapman et al. 2006; Lopez et al. 2006]. Although an overall decline in tobacco smoking is apparent in the West, smoking prevalence rates in Asian countries continue to rise. It is estimated that the number of COPD cases in Asia exceeds three times the total number of COPD cases for the rest of the world [Regional COPD Working Group, 2003].
Inhaled cigarette smoke and other noxious particles cause lung inflammation, a normal response that is amplified in patients who develop COPD. Airway inflammation and loss of elastic recoil from lung parenchyma destruction cause airflow limitation in COPD [Barnes et al. 2003], resulting in premature closure of small airways, air trapping and hyperinflation [Hogg et al. 2004]. Hyperinflation impairs exercise tolerance [O’Donnell and Webb, 1993] as it forces the respiratory muscles to operate at mechanical disadvantage, and flattening of the diaphragm further affects its effectiveness for a given neural stimulus [Braun et al. 1982]. Medical treatment of emphysema offers symptom palliation but does not halt disease progression. Smoking cessation, oxygen therapy and lung volume reduction surgery (LVRS) have been shown to have an impact on mortality. LVRS improves elastic recoil by allowing the rib cage and diaphragm to function more effectively, and for the remaining less diseased lung to expand [Brantigan et al. 1959]. Although operative mortality is 16%, those who survive the surgery have derived symptomatic relief [Cooper et al. 1995]. The National Emphysema Treatment Trial (NETT), a randomized, controlled study involving 17 centers in North America [Fishman et al. 2003] showed a distinct survival advantage in the group with predominantly upper lobe disease (PULD) and low baseline exercise tolerance over medical treatment. Patients with homogenous emphysema, FEV1 less than 20% (predicted) and diffusing capacity for carbon monoxide (DLCO) less than 20% should not undergo LVRS as it leads to 28% 90-day mortality. LVRS also carries a higher risk of death for patients without PULD and preserved exercise capacity. Importantly, more than half suffered cardiopulmonary complications within 30 days of LVRS, and 90-day mortality ranged between 5 and 10% [Naunheim et al. 2006]. NETT concluded that LVRS impacted on a small group of patients, it was costly, irreversible and associated with high morbidity and mortality.
In the last decade bronchoscopic approaches have been developed to reproduce the effects of LVRS: valves that allow unidirectional airflow in exhalation to collapse target lung lobe; biological lung volume reduction with biodegradable gel into bronchi; creation of airway bypass tracts; coils; and thermal vapor ablation.
Methods and results
Bronchial valves
The intrabronchial valve (Spiration Inc., Redmond, WA, USA) has six struts made of nitinol covered by polyurethane membrane in the shape of an umbrella (Figure 3). The intrabronchial valve (IBV) limits air flow to the target lobe during inspiration but allows air and mucus to escape around its edges during expiration. IBVs are available in different sizes and can be deployed via ‘direct’ and ‘catheter’ methods. The intended airway is sized with a calibrated balloon, and the catheter containing the appropriately sized valve is passed through a 2.8 mm working channel of the flexible bronchoscope. Once the valve is deployed, it can be adjusted and removed by grasping the central rod with forceps. In a multicenter trial, 91 patients with heterogeneous emphysema and PULD underwent bilateral IBV therapy during which a median of six valves were used (range 3–11). One patient died of tension pneumothorax, another had nonfatal myocardial infarction, eight developed pneumothoraces, and seven bronchospasm. Although no valve migration, expectoration, erosion or hemoptysis occurred, removal was necessary in 16 patients due to unresolving pneumonia, persistent bronchospasm and air leak. FEV1, 6 min walk test and total lung volume did not change following IBV therapy, but better health-related QoL scores, which could be explained by reduction of lung volume (without atelectasis) on computer tomography (CT) scan, and better ventilation-perfusion matching [Sterman et al. 2010].

Intrabronchial valve (Spiration Inc.) with delivery system deployed via working channel of bronchoscope.
The Zephyr endobronchial valve (Pulmonx Inc., Palo Alto, CA, USA) is a one way valve that is mounted on a self-expanding nitinol stent. The endobronchial valve (EBV) allows unidirectional airflow mucus clearance in expiration and is available in various sizes (Figure 4). The appropriate size EBV is determined during bronchoscopy using an endoscopic gauge. The loading catheter carrying the appropriate size EBV is advanced to the target airway through the working channel of a flexible bronchoscope and deployed by means of an actuation handle. EBV can be easily removed and repositioned. Studies have demonstrated good safety and efficacy of EBV for heterogeneous emphysema [Snell et al. 2001; Yim et al. 2004]. A larger trial consisting of 98 patients in whom 396 EBV valves were deployed and followed up for 90 days showed 11% and 9% improvements in FEV1 and forced vital capacity (FVC) respectively, and a 4.9% decrease in residual volume (RV). The 6 min walk test also improved by 23%. The complication rate at 90 days was 8%, which included one death, COPD exacerbations, pneumonia and pneumothoraces. Unilateral EBV treatment, lobar targeting with resultant lobar exclusion/collapse and patients with baseline FEV1 less than 30% or RV greater than 225% were predictors of good outcome [Wan et al. 2006]. These results prompted a prospective, multicenter trial that compared unilateral EBV treatment with medical therapy for heterogeneous emphysema. High-resolution CT was used to score disease severity and distribution as well as interlobar fissue integrity before target lobe selection. Two hundred and twenty patients were randomized to receive EBV and 101 to medical therapy that included pulmonary rehabilitation. Differences between the two groups were a 6.8% increase in FEV1 and 5.8% in 6 min walk test favoring EBV. These objective improvements were more marked in the subgroup with higher heterogeneity scores (>15% difference between targeted and adjacent lobes) and complete fissures on CT. The complication rate at 12 months was lower in the medical therapy group (4.6%) compared with 10.3% in the EBV group. Complications encountered with EBV were death, massive hemoptysis, respiratory failure, pneumonia, empyema, pneumothoraces with persistent air leak, device malfunction, valve expectoration, migration and granulation tissue [Sciurba et al. 2010].

Sizing of the bronchial segment before deployment of appropriately sized one-way endobronchial valve selection (Pulmonx Inc.).
Lobar atelectasis is observed in less than 25% of patients undergoing EBV. Lobar exclusion with atelectasis is highly desirable as it leads to physiological improvements akin to LVRS, and is emerging as an important predictor of good outcome. Successful lobar exclusion depends on the presence of complete fissure on CT, which suggests that atelectasis is hindered by ventilation through low-resistance collateral channels across incomplete fissures [Terry et al. 1978]. Incomplete fissures affect 21–30% of oblique and 88% of right horizontal fissures, and are more common in emphysema [Rosenberg and Lyons, 1979]. Collateral ventilation to the target lobe can be measured by a balloon catheter (Chartis System, Pulmonx Inc.) inserted through a 2.8 mm working channel of a flexible bronchoscope. The balloon is first inflated to seal the airway. This prevents air from entering the target lobe but allows air to escape through the central lumen of the catheter. Airflow resistance is calculated and represented in a graphic format. Higher values were found to correlate with lobar atelectasis with EBV. This device appears to be the only sensitive method of measuring collateral ventilation that is currently available [Aljuri and Freitag, 2009].
Bronchoscopic lung volume reduction has also been used as a bridge to lung transplantation, in reducing hyperinflation in the native lung of patients with single lung transplantation, as well as in the treatment of giant emphysematous bullae [Venuta et al. 2011; Pato et al. 2010; Cresp et al. 2007; Santini et al. 2011; Noppen et al. 2006].
Biological lung volume reduction
A biodegradable gel is instilled into subsegmental bronchi leading to emphysematous areas to cause resorption atelectasis by airway occlusion and inflammatory response that scars and contracts the targeted lung. A multicenter trial of 50 patients with PULD who underwent bilateral therapy showed that those who received 20 ml fibrinogen and thrombin suspension per subsegment had localized lung scarring detected on CT, which correlated with better FEV1, FVC and lower RV compared with those who received 10 ml per subsegment [Criner et al. 2009]. Similar improvements were achieved in patients with homogenous emphysema in favor of higher doses. Targeting five to eight subsegments of one upper lobe yielded better results than both upper lobes of four subsegments each. A majority experienced fever and leukocytosis postoperatively, some had COPD exacerbations but no fatality was observed [Rafaely et al. 2010]. Polymeric lung volume reduction (Aeris Therapeutics Inc., Woburn, MA, USA) based on the same concept is under evaluation. Hydrogel foam instead of gel is instilled into the bronchial subsegments, which adheres to the alveoli, absorbs the gas and collapses the target lobe. Results of phase II trials are awaited.
Bronchoscopic thermal vapor ablation
This technique works on the principle that administration of steam at controlled thermal energy causes acute tissue injury, scarring and contraction of the target lung akin to LVRS. The impact of collateral ventilation is negated, and respiratory mechanics improve. The vapor catheter is introduced to the segmental airway, the balloon is inflated to create a seal followed by administration of a predetermined vapor dose. Eleven patients with severe heterogeneous emphysema underwent unilateral upper lobe bronchoscopic thermal vapor ablation. Complications included bacterial pneumonia and COPD exacerbation. Although there was no change in FEV1 or RV at 6 months, gas transfer, Medical Research Council Dyspnea and QoL scores improved [Snell et al. 2009].
Lung volume reduction coil
The lung volume reduction coil (PneumRx Inc., Mountain View, CA, USA) made of nitinol is deployed with a guidewire and catheter delivery system via bronchoscopy. Commonly used coils measure 100 mm and 125 mm in length. The airway leading to the target lobe is identified and a guidewire is advanced with the aid of fluoroscopy. The length of the airway is measured and the appropriate size coil is selected. The catheter is passed over the guidewire and the coil is deployed. The coil assumes its shape and pulls the airway and attached parenchyma with it, thereby mechanically compressing the lung segment. It negates the impact of collateral ventilation, but unlike gel and steam when the lung is irreversibly scarred, these coils can be removed or repositioned. In a safety trial 11 patients underwent coil implantation and 10 required second treatment. Dyspnea, cough, COPD exacerbations and chest pain were adverse events reported. No pneumothorax was observed and patients with heterogeneous emphysema benefited from this treatment [Herth et al. 2010]. Results of ongoing trials are awaited.
Airway bypass tracts
The rationale for creating extra-anatomic fenestrations between the lung and bronchial tree is to facilitate gaseous emptying to decompress the hyperinflated lung. Conceivably airway bypass tracts (ABTs) would confer benefit to patients with homogenous emphysema. This technique involves identification of blood vessels in the vicinity of the planned sites using a Doppler probe so that they are avoided before puncturing and dilating the bronchus–lung tract with needle balloon catheter. The extra-anatomic tract is then kept patent by a paclitaxel eluting stent (Figure 5). In a multicenter trial of 35 patients with severe homogenous emphysema, a median of eight ABTs (range 2–12) per patient were created in both lungs. Patients with baseline RV/TLC greater than 0.67 had significant improvements in FVC, TLC, RV and health-related QoL scores while those with RV/TLC less than 0.67 did not. FEV1 and 6 min walk test remained unchanged in both groups. Although bleeding could be controlled with topical epinephrine, one patient died of massive hemorrhage. Other adverse events included COPD exacerbations, pneumonia and pneumomediastinum. At 6 months, 69% stented fenestrations remained patent on bronchoscopy which could be detected by multidetector CT [Cardoso et al. 2007]. A recent randomized, double-blind and sham-controlled study involving 38 respiratory centers worldwide showed that ABTs could be created safely; patients enjoyed improvements of symptoms and spirometry, but unfortunately these were not sustained at 6 months [Shah et al. 2011].

Stented airway bypass tract.
Discussion
Bronchoscopic techniques currently employed to effect lung volume reduction range from experimental to investigative in multicenter, randomized, sham-controlled trials (Table 2). Minimally invasive bronchoscopic techniques are associated with less morbidity and mortality than LVRS and will remain attractive alternatives even though many theoretical benefits remain unproven (Table 3).
Interventions for bronchoscopic lung volume reduction.

COPD, chronic obstructive pulmonary disease; LVR, lung volume reduction.
Key randomized studies in bronchoscopic lung volume reduction.

6MW, 6 min walk; AB, airway bypass; C, control; CC, complications composite; COPD, chronic obstructive pulmonary disease; CPS, composite primary safety; EASE, exhale airway stents for emphysema; EBV, endobronchial valve; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; HRQL: health related quality of life; IBV, intrabronchial valve; MRC, Medical Research Council; PULD, predominantly upper lobe disease; TLC, total lung capacity ; VENT: endobronchial valve for emphysema palliation trial; V/Q: ventilation perfusion matching in non-UL: upper lobe, Sterman et al., 2010.
Conclusion
Novel bronchoscopic approaches are being developed as therapies for obstructive airway diseases and careful patient selection is pivotal in assuring good outcome. Precise definition of clinical phenotypes individualizes asthma treatment so that BT may complement standard anti-inflammatory treatment. In patients with severe heterogeneous emphysema, endobronchial valves facilitate lobar atelectasis while ABTs reduce air trapping in individuals with homogeneous emphysema. Methods that target emphysema of variable severity and distribution, and possibly to be used in combination in the future, strive to reduce hyperinflation to provide the symptomatic patient better respiratory mechanics akin to LVRS, but without its attendant peri- and postoperative morbidity and mortality.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
