Abstract
Introduction:
A twice-daily single inhaler triple therapy consisting of budesonide/glycopyrrolate/formoterol fumarate (BGF) was approved by the US Food and Drug Administration (FDA) in July 2020 as a maintenance treatment for patients with chronic obstructive pulmonary disease (COPD). The objective of this AURA study is to describe patient characteristics, exacerbation and treatment history, and healthcare resource utilization (HCRU) before BGF initiation to better inform treatment decisions for prescribers.
Methods:
This retrospective cohort study leveraged data of all payer types from IQVIA’s Longitudinal Prescription Data (LRx) linked to Medical Data (Dx). Patients with COPD who had ⩾1 LRx claim for BGF between 1 October 2020 and 30 September 2021 were included. The date of first BGF claim was the index date. Patient demographic and clinical characteristics, history of COPD exacerbation or related event, treatment history, and HCRU were assessed during the 12 months before index (baseline).
Results:
We identified 30,339 patients with COPD initiating BGF (mean age: 68.2 years; 57.1% female; 67.6% Medicare). Unspecified COPD (J44.9; 74.0%) was the most commonly coded COPD phenotype. The most prevalent respiratory conditions/symptoms were dyspnea (50.8%), lower respiratory tract infection (25.3%), and sleep apnea (19.0%). Uncomplicated hypertension (58.8%), dyslipidemia (43.9%), cardiovascular disease (41.4%), and heart failure (19.9%) were the most prevalent nonrespiratory conditions. During the 12-month baseline, 57.9% of patients had evidence of a COPD exacerbation or related event, and 14.9% had ⩾1 COPD-related emergency department (ED) visit; 21.0% of patients had evidence of prior triple therapy use, while 54.3% had ⩾1 oral corticosteroid (OCS) fill. Among OCS users, 29.9% had cumulative exposures >1000 mg [median [Q1–Q3] exposure: 520 (260–1183) mg].
Conclusion:
This real-world data analysis indicates that BGF is being initiated in patients with COPD experiencing symptoms and exacerbations despite current therapy, and among patients who have various chronic comorbidities, most often cardiopulmonary-related.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD), including emphysema and chronic bronchitis, is a heterogeneous lung condition that is characterized by chronic respiratory symptoms (e.g. dyspnea) due to abnormalities of the airways and/or alveoli that cause persistent, often progressive, airflow obstruction. 1 COPD is among the leading causes of death in the United States 2 and is associated with significant economic burden. It is estimated that US$800.9 billion direct medical costs and US$101.3 billion indirect costs from missed work days can be attributable to COPD in the next 20 years, 3 with exacerbations from uncontrolled disease accounting for a large proportion of the total COPD burden. 4 The burden of COPD is expected to increase as the population ages. 5
While COPD poses a significant burden on patients and society, various pharmacological treatments are available to manage COPD symptoms and improve the functional status of patients. The main goals for the pharmacological treatment of COPD are to manage symptoms, prevent exacerbations, increase exercise capacity, improve quality of life, and reduce risk of mortality. 6 Triple therapy (TT) consisting of an inhaled corticosteroid (ICS), a long-acting muscarinic antagonist (LAMA), and a long-acting beta2 agonist (LABA) is recommended as the initial pharmacological treatment for patients with COPD who have eosinophil count ⩾300 cells/μl and those with ⩾2 moderate exacerbations or ⩾1 exacerbation leading to hospitalization. 1 TT is also recommended as a step-up treatment for patients who develop further exacerbations on dual therapy of a LABA plus a LAMA or an ICS and a LABA and with eosinophil counts ⩾100 cells/μl. 1 Two single-inhaler TT (SITT) consisting of fluticasone furoate/umeclidinium/vilanterol (FUV) in a drug powder inhaler (DPI) and budesonide/glycopyrrolate/formoterol fumarate (BGF) in a pressured metered dose inhaler (pMDI) were approved by the US Food and Drug Administration (FDA) in September 2017 7 and July 2020, 8 respectively, as maintenance treatment for patients with COPD. The introduction of SITT to the US market has provided viable options for people living with COPD to improve treatment persistence and adherence, 9 which could further improve treatment outcomes. 10
As new therapies become available to patients, understanding the characteristics of patients receiving these therapies could further inform prescriber treatment choices. While characteristics of patients with COPD initiating MITT and FUV have been recently assessed,11,12 to our knowledge, this is the first study to describe patients who initiated BGF, the most recently approved SITT, in the real-world setting in the United States. This retrospective database study aimed to evaluate patient characteristics, exacerbation and treatment history, and healthcare resource utilization (HCRU) prior to initiation of BGF among patients with COPD in the United States.
Methods
Study design and data source
This was a retrospective cohort study leveraging data from IQVIA’s Longitudinal Prescription Data (LRx) linked to Medical Data (Dx) databases. The LRx database captures information on dispensed prescriptions with 92% coverage of prescriptions from the retail channel, 72% coverage of standard mail service, and 76% coverage of long-term care facilities in the United States. The Dx database captures over one billion pre-adjudicated claims obtained annually from approximately 800,000 office-based physicians and specialists, covering 75% of American Medical Association providers. Medical claims from ambulatory and general health care sites as well as outpatient clinics associated with hospitals (e.g. rehabilitation, same-day surgery, and chemotherapy centers) are also included in the Dx database. All data were de-identified and compliant to the Health Insurance Portability and Accountability Act (HIPAA), thus Institutional Review Board approval or waiver was not required for this study. Both databases have been previously discussed in the literature.13,14
Study population
As detailed in Figure 1, patients with ⩾1 LRx claim for BGF between 1 October 2020 and 30 September 2021 and ⩾1 COPD diagnosis [International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) codes of J41.X, J42, J43.X, J44.X] between 1 October 2019 and 30 September 2021 (i.e. study period) were selected into the study. The date of the first observed LRx claim for BGF was the index date. Patients were required to have ⩾12 months of LRx and Dx data before the index date (i.e. baseline). The overall BGF cohort was further classified into subgroups of patients who initiated BGF in 2020 and those who initiated BGF in 2021 for a sensitivity analysis to explore the potential impact of COVID-19 pandemic. A subgroup of patients with ⩾24 months of LRx and Dx baseline data was also identified from the overall BGF cohort to examine patient exacerbation history.

Flow chart of patient selection.
Study measures
All study measures were assessed during the 12-month baseline period. Patient demographic and baseline clinical characteristics were described, including nonrespiratory Elixhauser Comorbidity Index (ECI) score 15 (van Walraven adaption, excluding Elixhauser concept of chronic pulmonary disease, which includes asthma and COPD), individual ECs, selected non-EC comorbidities/symptoms, and COPD phenotype based on ICD-10-CM diagnosis codes. As COPD exacerbations may be miscoded or under-recorded in administrative claims, three claims-based algorithms were used to identify moderate COPD exacerbations and related events, including moderate COPD exacerbations, potential acute COPD exacerbations, and lower respiratory tract infection (LRTI). A moderate COPD exacerbation episode was defined as having an outpatient visit for COPD (COPD diagnosis in any position) in Dx and an LRx claim for an oral corticosteroid (OCS) or antibiotic with supply ⩽14 days within ±7 days of the visit; 16 an LRTI episode was defined similarly based on an outpatient visit for LRTI and usage of an OCS or antibiotic. The episode started on the day of the first qualified outpatient visit and was considered lasting for 14 days. All outpatient visits within the 14-day period were considered part of the same episode. 16 A potential acute COPD exacerbation was defined as having LRx claims for both an OCS and an antibiotic on the same day with supply ⩽14 days for each. 17 COPD treatment history, all-cause and COPD-related HCRU, and index prescriber specialty were also assessed. COPD-related HCRU was defined by claims with diagnosis of COPD in any position. Among patients with ⩾1 OCS fill, the cumulative exposure in prednisone equivalent doses (Supplemental Table 1) was reported.
Statistical analyses
Descriptive statistics were produced for all study measures for the overall study cohort. To explore the potential impact of the COVID-19 pandemic which began affecting the US healthcare resources in early 2020, a sensitivity analysis was conducted to describe selected baseline clinical characteristics separately for patients who initiated BGF in 2020 (i.e. most patients with partial baseline prior to the COVID-19 pandemic) and those who initiated BGF in 2021 (i.e. most patients with baseline spanning the COVID-19 pandemic). Statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patient demographic and clinical characteristics
There were 30,339 patients with COPD who initiated BGF during the selection window (Figure 1) and who met the study criteria for data availability. The demographic and baseline clinical characteristics of patients are shown in Tables 1 and 2. Briefly, the mean [standard deviation (SD)] and median [interquartile range (IQR)] age was 68.2 (10.5) and 69.0 (62.0–76.0) years, respectively. The majority of patients were female (57.1%), located in the southern United States (55.6%), and Medicare beneficiaries (67.6%) (Table 1). The most commonly observed COPD coded-phenotype was unspecified COPD (J44.9; 74.0%), followed by COPD with exacerbation (J44.1; 13.2%) and emphysema (J43.x; 12.6%). During the 12-month baseline period, the mean (SD) nonpulmonary EC index score was 5.1 (7.9); 43.9% of patients had an ECI score of 5 or greater. The most prevalent EC was uncomplicated hypertension (58.8%), followed by cardiac arrhythmias (23.8%), and uncomplicated diabetes (19.9%). Of non-EC comorbidities/symptoms examined, dyspnea (50.8%), LRTI (25.3%), sleep apnea (19.0%), and asthma (18.1%) were the most commonly observed respiratory conditions/symptoms. Dyslipidemia (43.9%), cardiovascular disease (41.4%), and heart failure (19.9%) were the most commonly observed nonrespiratory conditions and 7.9% of the patients had evidence of COVID-19 infection. In terms of prescriber specialty, approximately half of patients (49.5%) had their first BGF prescribed by a pulmonologist, with the next most common prescriber specialty being primary care physician (17.2%) or nurse practitioner (20.2%) (Table 2).
Patient demographic characteristics.
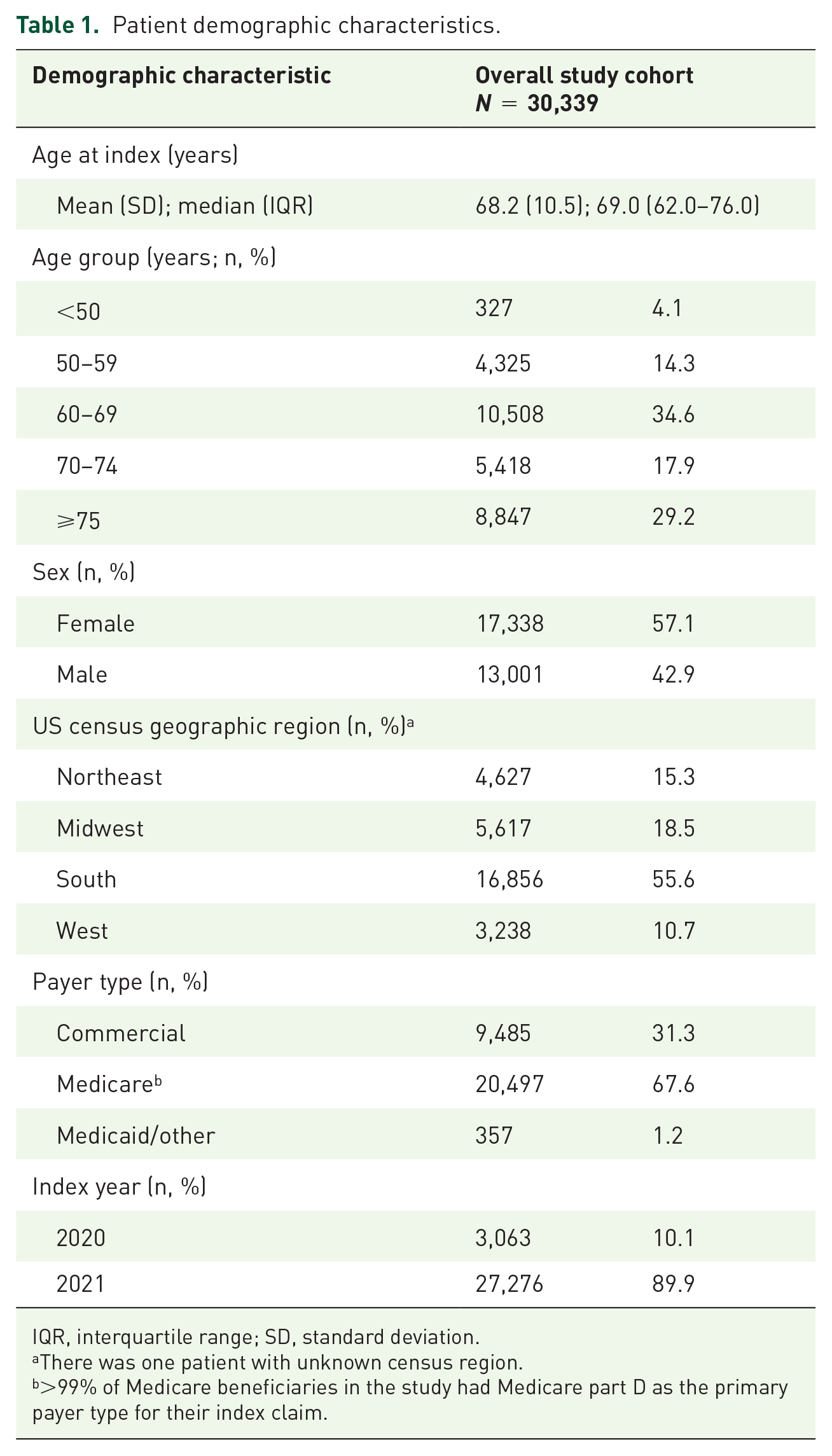
IQR, interquartile range; SD, standard deviation.
There was one patient with unknown census region.
>99% of Medicare beneficiaries in the study had Medicare part D as the primary payer type for their index claim.
Patient clinical characteristics during the 12-month baseline period.
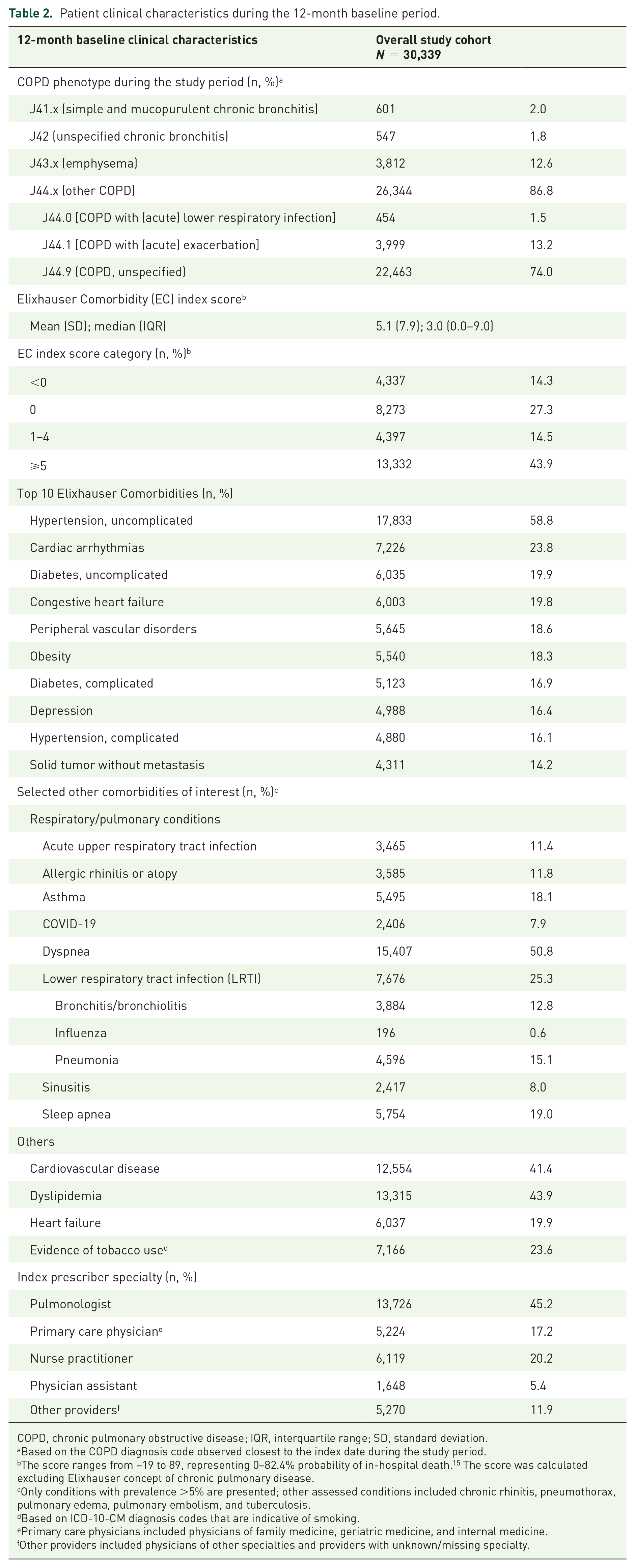
COPD, chronic pulmonary obstructive disease; IQR, interquartile range; SD, standard deviation.
Based on the COPD diagnosis code observed closest to the index date during the study period.
The score ranges from −19 to 89, representing 0–82.4% probability of in-hospital death. 15 The score was calculated excluding Elixhauser concept of chronic pulmonary disease.
Only conditions with prevalence >5% are presented; other assessed conditions included chronic rhinitis, pneumothorax, pulmonary edema, pulmonary embolism, and tuberculosis.
Based on ICD-10-CM diagnosis codes that are indicative of smoking.
Primary care physicians included physicians of family medicine, geriatric medicine, and internal medicine.
Other providers included physicians of other specialties and providers with unknown/missing specialty.
COPD exacerbation history
During the 12-month baseline period, 57.9% of patients had evidence of ⩾1 moderate COPD exacerbation or related event (Figure 2): 41.4% of patients had evidence of ⩾1 moderate COPD exacerbation episode and 11.4% had ⩾2 episodes. Based on the algorithm of potential acute COPD exacerbation, 38.4% and 17.2% of patients had evidence of ⩾1 episode and ⩾2 episodes, respectively. Evidence of LRTI was less commonly observed, with 15.5% and 2.3% of patients having ⩾1 or ⩾2 LRTI episodes, respectively. In the subgroup of patients with ⩾24 months of baseline data (n = 27,671), 65.6% had evidence of ⩾1 moderate COPD exacerbation or related event (Supplemental Figure 1).

Patients with evidence of moderate COPD exacerbation or related event during the 12-month baseline period.
Treatment history and costs
During the 12 months prior to BGF initiation, 71.4% of patients had exposure to other COPD maintenance treatments, with 21.0% having prior TT (FUV or MITT) and 47.4% having mono or dual therapies (Figure 3). Among patients who had prior TT, 61.7% switched from FUV to BGF. The median (IQR) time from the end of previous maintenance treatment to BGF initiation was 47 (21–105) days, and the median (IQR) proportion of days (PDC) covered with maintenance treatment was 56.9% (25.0–81.9%). Among all patients initiating BGF, the most commonly observed maintenance treatment class during baseline was LABA/ICS (35.8%), followed by LAMA (23.2%), LAMA/LABA (17.2%) and FUV (17.1%). Among patients with at least one fill of the respective treatment class, the median number of fills per patient ranged from 2 to 4 during the 12-month baseline.

Treatment history during the 12-month baseline period.
Most patients (86.3%) had evidence of short-term treatment, and the short-term treatment class observed most frequently was short-acting beta agonist (SABA; 73.7%) followed by OCS (54.3%). The median (IQR) number of fills was 4 (2–7) among SABA users and 2 (1–4) among OCS users. Furthermore, for those with OCS use, the median (IQR) cumulative OCS dose in prednisone equivalents during the 12-month baseline was 520 (260–1183) mg, with 51.5% of patients having a cumulative dose >500 mg, 29.9% having dose >1000 mg, and 20.0% having dose >1500 mg (Supplemental Figure 2).
Almost one-third of patients (31.6%) did not have evidence of any inhaled maintenance therapy (mono, dual, or triple) and 6.8% of patients did not have any maintenance or short-term treatment for COPD during the 12-month baseline period (Figure 3). Among patients with 24 months of baseline data (n = 27,671), almost one-quarter of patients (21.4%) did not have any evidence of maintenance therapy for COPD.
Regarding other relevant medical procedures or supportive therapies for COPD during the 12 months prior to BGF initiation (Supplemental Figure 3), less than one-third of patients (27.2%) had evidence of spirometry, 25.8% of patients received oxygen therapy, 12.8% had mechanical ventilation, and 1.5% had pulmonary rehabilitation. While anticipated to be underreported due to a lack of population vaccination records in the United States, evidence of COVID-19 vaccination was observed through claims in about one-fifth of patients (20.5%).
All-cause and COPD-related HCRU
As depicted in Figure 4, during the 12 months prior to BGF initiation, 70.6% of patients had ⩾1 all-cause primary care physician visit and 45.2% of patients had ⩾1 COPD-related primary care visit. In terms of specialist visits, 43.7% of patients had ⩾1 all-cause pulmonologist visit and 39.0% had ⩾1 all-cause cardiologist visit; 35.7% of patients had ⩾1 COPD-related pulmonologist visit. All-cause emergency department (ED) visits were observed in just less than half (42.8%) of patients, with COPD-related visits observed in 14.9%. Among patients with ⩾1 respective COPD-related visit, the median (Q1–Q3) number of visits within the 12 months prior to BGF initiation was two (1–5) visits to primary care physicians, two (1–4) visits to pulmonologists, and one (1–2) visit to ED.

All-cause and COPD-related HCRU during the 12-month baseline period.
COVID-19 sensitivity analysis stratified by index year
Compared with patients who initiated BGF in 2021, those who initiated BGF in 2020 appeared to have higher prevalence of many respiratory/pulmonary conditions, including LRTI (28.9% and 24.9%), acute upper respiratory tract infection (15.8% and 10.9%), and allergic rhinitis or atopy (14.4% and 11.5%) during the 12-month baseline. Patients who initiated BGF in 2020 also had higher prevalence of COPD exacerbation or related event during baseline (62.1%) compared with those who initiated BGF in 2021 (57.4%), likely reflective of lower HCRU during pandemic. However, all-cause (41.8% and 42.9%) and COPD-related (15.8% and 14.8%) baseline ED visits were similar between the two groups (Supplemental Table 2).
Discussion
This AURA study describes patient characteristics, exacerbation history, prior COPD medication use, and baseline HCRU among patients with COPD who initiated BGF, which was the most recently approved SITT for COPD in the United States. To our knowledge, this is the first study to characterize this patient population using a large geographically diverse database that includes all payer types. The 2022 Global Initiative for Chronic Obstructive Lung Disease (GOLD) report recognizes that escalation to TT can occur by various approaches for patients who develop further exacerbations or who have persistent symptoms. 6 Overall, the study findings suggest that most patients escalated to BGF after receiving other maintenance treatments, having exacerbations and/or other respiratory comorbidities/symptoms, which is in line with the 2022 GOLD criteria that were available at the study conduct. 6 The latest 2023 GOLD report also recommends using blood eosinophil counts to predict ICS effects in addition to assessing exacerbation risk for the decision of escalating to TT and using TT as the initial pharmacological treatment. 1 Eosinophil levels are not available in the study data and therefore were not examined in the study. Future studies are needed to demonstrate how eosinophil levels are leveraged in the real-world prescribing decision of TT.
In general, the clinical characteristics of patients with COPD who initiated BGF in this AURA study were similar to patients who initiated FUV or MITT, as reported in previous studies,12,18 with the majority being 65 years or older, female, and Medicare beneficiaries. Consistent with findings of these two previous studies describing patients initiating FUV and MITT,11,12 a high proportion of patients initiating BGF in our study had cardiopulmonary-related comorbidities, indicating that the presence of such comorbidities may also play a role in prescribing decisions of TT in real-world practice. Dyspnea and other pulmonary/respiratory conditions (e.g. LRTI, asthma) were also common symptoms/comorbidities observed among patients with COPD in the 12 months prior to BGF initiation, suggesting that the presence of COPD symptoms (e.g. dyspnea) that could be similar or masked by other pulmonary/respiratory conditions may have prompted providers to initiate BGF. About 18% and 15% of patients with COPD in this study had asthma and pneumonia prior to BGF initiation, respectively, while higher prevalence was reported in the two previous studies describing patients initiating FUV (39.7% and 26.4%) and MITT (25.4% and 24.4%).11,12 This may be related to reduced access to care during the COVID-19 pandemic, 19 as comorbidities were identified from administrative claims from health service encounters. Furthermore, it is plausible that the lower prevalence of respiratory conditions was due to reduced exposures to common viral respiratory infections or other environmental triggers for respiratory diseases following pandemic responses such as mask-wearing and social distancing. 20
The prevalence of moderate COPD exacerbations prior to BGF initiation observed in this AURA study (41.4%) was in line with recently published claims-based studies, where 37.3–45.2% of patients initiating FUV or MITT had evidence of moderate COPD exacerbations during the 12-month baseline.11,12 While the 2022 GOLD report classifies exacerbations as mild, moderate, or severe, 6 our study was not able to assess severe exacerbations due to lack of inpatient claims from the study databases. However, in addition to moderate COPD exacerbations, potential acute COPD exacerbations (38.4%) were examined in the study as some patients may get OCS fills without corresponding office visits, especially during early phases of the COVID-19 pandemic when telehealth utilization soared. 21 We also examined patients with evidence of LRTI (15.5%) and COPD-related ED visit (14.9%), which is often associated with exacerbations and may in fact be a missed opportunity to identify COPD exacerbation in retrospective database studies. 17 Even with broader definitions for potential exacerbation and related events, exacerbations are expected to be underestimated in claims-based studies. Such underestimation could have been further heightened during the early phases of the pandemic.
Use of OCS could also be seen as an indicator of exacerbations and underlying symptoms among COPD patients. 22 The use of OCS should be closely monitored as it has been associated with various adverse events (e.g. fluid retention, hypertension).6,23–25 The latest GOLD report recommends up to 280 mg of OCS for an exacerbation. This AURA study found that almost one-third of patients had >500 mg cumulative OCS exposure in prednisone equivalents and 16% had exposure exceeding 1000 mg over the 12 months prior to BGF initiation, indicating an unmet need in this patient population. Furthermore, the majority of patients were found to be nonadherent (PDC <80%) to their maintenance treatment during baseline, which may have prompted escalation to an SITT. Notably, we observed about 13% of patients switching from FUV to BGF. While the reasons of switching cannot be examined in the study data and cannot be fully understood from claims data, the choice of inhaler device (DPI versus pMDI) and potential risk of pneumonia associated with fluticasone-based compared with budesonide-based treatments are known as important considerations for managing COPD and therefore may contribute to the observed switches among other unobservable factors. Taken together, various factors, such as prior exacerbation, comorbidities, adherence to prior medications, choice of inhaler device, and risk of adverse events, may have driven BGF initiation among patients with COPD.
The most commonly visited specialists observed in our study prior to BGF initiation were pulmonologists and cardiologists. In agreement with the comorbidity findings described previously, this indicates that many patients who initiated BGF were those with a significant comorbidity burden and requiring care from multiple specialties. Future studies comparing treatment pathways and outcomes between patients who receive specialist care and those who do not may help better understand the role of both primary care and specialist care in the management of COPD, particularly the role nurse practitioners may have in prescribing decisions. Notably, about one-fifth of patients had their first BGF prescribed by a nurse practitioner in our study, suggesting that nurse practitioners are an important and emerging provider for COPD. As COPD progresses, a lack of patient knowledge about medications and/or delivery device can lead to poor treatment adherence and patient outcomes. 26 Our findings highlight the need of empowering nurse practitioners and other healthcare providers (e.g. physician assistants, clinical pharmacists) in the role of COPD management by providing patient education and assisting treatment monitoring. 27
Although the GOLD guidelines recommend annual spirometry for patients with COPD, this AURA study found that less than one-third of patients had evidence of spirometry during the 12 months prior to BGF initiation. This highlights the need for more routine spirometry for better disease assessment and treatment adjustment, especially among patients who are at high risk of exacerbation and/or with complex comorbidities. 6
A challenge of this study is that BGF launched during the COVID-19 pandemic, which likely affected care access and correspondingly the findings of this study. To address this, we conducted a sensitivity analysis to explore the impact of the pandemic and COVID-19 vaccination on BGF initiators by comparing patient comorbidities and exacerbation history among patients indexed in 2020 (baseline available prior to pandemic) versus 2021. Only small differences were observed between the two subgroups. Patients indexed in 2021 had a slightly lower prevalence of moderate COPD exacerbations or LRTI during baseline relative to patients indexed in 2020, which may be due to patients not seeking or unable to access care because of the pandemic and social distancing. Future studies with two matched groups of patients identified from pre- and post-COVID-19 periods are warranted to help better understand the impact of COVID-19 pandemic on patients initiating TTs.
In addition to the challenging study period, this study has several limitations inherent to retrospective database analyses of claims data. Due to the ‘open’ nature of the study databases, continuous capture of patient medical encounters could not be confirmed. To that end, services received from providers and medications filled at pharmacies that are out of the network of data suppliers of the databases were not captured. However, consistent with other studies leveraging the study databases,28–30 standard approaches of ensuring stable data supply and patient activities in the study databases were used as proxies for continuous follow-up. The study data also do not capture inpatient utilization, which may lead to underestimation of COPD exacerbations and prevalence of comorbidities (e.g. COVID-19). Clinical data that can better represent COPD severity and phenotypes (e.g. % predicted FEV1, blood eosinophil count) were not available from the study databases. Inherent to studies using claims database, billing codes were used to identify COPD and comorbidities, and therefore misclassification of diagnosis is possible.
Conclusion
This real-world data analysis indicates that initiation of BGF occurred predominantly in patients experiencing exacerbations and who likely remained symptomatic while on prior maintenance therapy. To that end, BGF was used most often in accordance with guideline recommendations, as a treatment escalation in patients who have persistent symptoms and/or exacerbations, despite treatment with maintenance therapy. Furthermore, in addition to exacerbation history, other factors such as comorbidities and adherence to prior medications may have also driven BGF initiation among patients with COPD. Notably, among patients on prior TT, switches to BGF may have been influenced by patient device choice, risks associated with pneumonia, or other unobservable factors. BGF is being initiated among patients with COPD who have various chronic comorbidities, most often cardiopulmonary-related conditions. Finally, findings of this AURA study highlight the need of empowering nurse practitioners and other providers such as physician assistants in their role of COPD management by providing patient education and helping monitor treatments.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231164534 – Supplemental material for Characteristics of initiators of budesonide/glycopyrrolate/formoterol for treatment of chronic obstructive pulmonary disease (COPD) in the United States: the AURA study
Supplemental material, sj-docx-1-tar-10.1177_17534666231164534 for Characteristics of initiators of budesonide/glycopyrrolate/formoterol for treatment of chronic obstructive pulmonary disease (COPD) in the United States: the AURA study by Edward C. Portillo, Michael Pollack, Inyoung Lee, Kainan Sun, Xiaohui Zhao, Lisa Kruse, Norbert Feigler, Sushma Patel and Aimee M. Near in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
